Abstract
Background
Attention deficit hyperactivity disorder is generally widely under-recognised and undertreated in adults. Real world evidence is needed to characterise healthcare resource utilisation and economic burden of adult attention deficit hyperactivity disorder in England.
Methods
This retrospective cohort study used primary care medical records (Clinical Practice Research Datalink – Aurum) linked to secondary care data (hospital episode statistics). Patients were indexed on their first-ever record of an attention deficit hyperactivity disorder diagnosis, made during adulthood, between 1 January 2016 and 30 June 2018. Healthcare resource utilisation and associated costs were compared 18-months before and after diagnosis.
Results
2236 patients were included; mood disorders and anxiety were recorded in 47.1% and 46.2%, respectively. In 2153 patients with available data for both periods, mean total healthcare resource utilisation cost per patient was £408.6 (95% CI £264.4–£552.8) higher in the post- versus pre-index period (p < 0.001). The mean prescriptions cost per patient doubled (£322.5 versus £614.5) and represented the largest proportional cost increase post-diagnosis compared to other healthcare resource utilisation costs measured.
Discussion
An attention deficit hyperactivity disorder diagnosis in adulthood ensures patients receive appropriate treatment for attention deficit hyperactivity disorder and comorbid anxiety and depression. Total healthcare resource utilisation costs appeared to increase post attention deficit hyperactivity disorder diagnosis; however, further studies are required to confirm this. This cost increase should be set in the context of multiple societal benefits likely to accrue from the effective management of adult attention deficit hyperactivity disorder.
Keywords
Introduction
Attention deficit hyperactivity disorder (ADHD) is a lifelong disorder characterised by the presence of developmentally inappropriate and impairing levels of inattention, hyperactivity, and impulsivity (American Psychiatric Association, 2013; Asherson et al., 2016; Faraone et al., 2021; Schmidt & Petermann, 2009). It is classified as a neurodevelopmental disorder but often managed within psychiatric or psychosocial services due to its behavioural manifestations. Prevalence estimates of ADHD in adults range from 2.5% to 3.4% (Fayyad et al., 2007; Simon et al., 2009). Co-occurrence of ADHD with psychiatric conditions, including anxiety and depression, is frequent and represents a challenge for accurate diagnosis (Agarwal et al., 2012; Bushe et al., 2015). Adults with ADHD experience multiple negative consequences, such as disability, worse quality of life, poor marital relationships, reduced employment status and work performance, and premature death due to accidents (Adamou et al., 2013; Agarwal et al., 2012; Dalsgaard et al., 2015; de Graaf et al., 2008; Eakin et al., 2004; Fayyad et al., 2007; Halmøy et al., 2009). Despite this, ADHD remains widely under-recognised and undertreated (Rivas-Vazquez et al., 2023).
ADHD is diagnosed clinically based on the presence and duration of symptoms, degree of psychosocial distress or impairment, and exclusion of other conditions which may produce a similar clinical profile (Rivas-Vazquez et al., 2023). The United Kingdom (UK) National Institute for Health and Care Excellence (NICE) Guideline 87 (NG87), published in March 2018 updating the Clinical Guideline 72 (CG72) published in September 2008, covers recognising, diagnosing, and managing ADHD and aims to improve recognition, diagnosis, and quality of care and support for people with ADHD (NICE, 2018). Several classes of pharmacological agents and various psychotherapeutic treatment modalities help reduce symptoms and enhance functional outcomes to varying degrees, emphasising the importance of accurate diagnosis (Boland et al., 2020; Fullen et al., 2020; Rivas-Vazquez et al., 2023). In the UK, healthcare resource utilisation (HCRU) costs for patients with ADHD have been estimated to be more than four times higher than for patients without ADHD (Holden et al., 2013). However, to our knowledge, no study has evaluated HCRU before and after ADHD diagnosis in adults.
Given the complex pathway to diagnosis and treatment, there is a need to generate real world evidence to characterise the treatment pathway, HCRU, and associated costs of ADHD in adult patients in England. We aimed to evaluate the economic burden of adult-onset ADHD following diagnosis and compare it to that during the period leading up to the diagnosis.
Methods
Study design and data sources
A retrospective observational cohort study of adults with ADHD in England was conducted using primary care records extracted from the Clinical Practice Research Datalink – Aurum (CPRD Aurum) database with linked hospital episode statistics (HES). This study included an 18-month pre-index period and 18-month post-index follow-up period, where the index date was defined as the date of a first ever record of adult ADHD diagnosis in CPRD or HES between the 1 January 2016 and 30 June 2018, using applicable codes from CPRD or HES (see Table e1 in Supplemental material for code list). CPRD Aurum is a large dataset with 13.4 million patients currently registered (20% of the population) from 1373 primary care practices dispersed throughout England (CPRD, 2023; Wolf et al., 2019). These data contain anonymised information on patient demographics, medical diagnoses, drug exposure, clinic visits, procedures, laboratory and pathology testing, and referrals to secondary and tertiary care. Clinical data are coded using a combination of Read Version 2, Systematized Nomenclature of Medicine – Clinical Terms (SNOMED-CT), and local EMIS Web® codes (Wolf et al., 2019). Drug prescriptions are coded using medical drug codes, which are linked to the British National Formulary (BNF, 2021). HES data include all in-patient admissions, outpatient appointments, and accident & emergency (A&E) visits to National Health Service hospitals in England, and diagnoses are coded using International Classification of Diseases 10 Revision codes. Approximately 99% of practices within CPRD Aurum were eligible for HES linkage during the study period.
Study population
Patients aged ≥18 years at the beginning of the pre-index period, with a first record of an adult ADHD diagnosis between 1 January 2016 and 30 June 2018, a CPRD record linked with HES, and ≥18 months lookback and follow-up in CPRD pre- and post-index date were included in the study. Patients diagnosed with ADHD beyond June 2018 were not included to limit any potential bias (which may result in underestimation of HCRU) as they may have had limited access to healthcare during the post-index period as a result of the coronavirus disease 2019 (COVID-19) pandemic. Anyone with a history of an ADHD diagnosis during childhood or adolescence prior to index date, incomplete information for key demographics (i.e., date of birth or gender), or prescription of a medication for ADHD (i.e., methylphenidate, lisdexamfetamine, dexamfetamine, atomoxetine, bupropion, clonidine, and guanfacine) during the pre-index period was excluded from the study.
During the study period NICE guidelines on the diagnosis and management of ADHD were changed. Subgroup analyses for patients whose period of evaluation was entirely covered by the original (CG72) and by the revised (NG87 – published March 2018) guidelines was performed as follows:
CG72 subgroup – index date between 1 January 2016 until 1 of September 2016 with a follow-up window until 28 February 2018 NG87 subgroup – index date between 1 March 2018 until 30 of June 2018 with follow-up window until 31 December 2019.
Study variables and outcomes
Data on gender, ethnicity, and comorbidities were extracted any time prior to or at index date. Age, general practice (GP), region of England, and index of multiple deprivation (IMD) was extracted at index date. An IMD decile was assigned based on the patient's actual deprivation score relative to the geographical location of their registered practice (i.e., decile 1 = most deprived, 10 = least deprived).
Outcome measures included:
Description of primary and secondary care HCRU (primary care consultations, investigations, prescriptions, hospital inpatient visits, length of inpatient stay, attended and unattended outpatient visits, and A&E visits) and their total costs per patient in the pre- and post-index periods. Cost comparison of total HCRU in pre- versus post-index period. Description of patients’ prescribed medication (ADHD and non-ADHD) in the pre- and post-index periods.
Data analyses
Data analysis was carried out using SAS version 9.4 (2013; SAS Institute Inc. Cary, North Carolina, United States). Descriptive statistics were used to describe patient demographic and clinical characteristics, comorbidities, and HCRU variables in the overall cohort and by NICE subgroup. Continuous variables were described using means, standard deviation (SD) or 95% confidence intervals (95% CIs) where applicable, medians, interquartile range (IQR), and minimum and maximum values. Categorical variables were described using frequencies and proportions. Chi square test was used to assess differences in clinical characteristics between NICE subgroups. A p-value of <0.05 was deemed statistically significant. The proportion increase or decrease in total prescriptions/visits/consultations between the pre- and post-index was calculated. HCRU-associated costs (in the overall cohort and by NICE subgroup) were estimated following the strategy used by Holden et al. (Table e2 in Supplemental material) for the 18-month pre- and post-index period separately and for the following time intervals: 12–18 months, 6–12 months, and ≤6 months pre- or post-index date (Holden et al., 2013). All costs were mapped to one national tariff reference year (2020/2021) or by using the National Health Service (NHS) cost inflation index to inflate prices collected from other earlier tariff works to this same 2020/2021 reference year to control for differences in costs due to inflation between the pre- and post-index periods. Primary care consultation costs were for non-administrative visits only, and prescription costs included drugs prescribed in primary care only, as hospital prescriptions and dispensation are not recorded in the HES database. The total HCRU cost per ADHD patient was defined as the sum of prescription, primary care consultation, inpatient hospitalisation, attended outpatients visit, and A&E attendance costs. The crude within-patient difference in total HCRU cost (total HCRU costs in the post-index period minus total HCRU costs in the pre-index period) was reported using continuous descriptive statistics. Formal comparison of within-patient pre-and post-index period total HCRU costs (excluding index date) was performed using paired student's t-test or Wilcoxon rank-sum test as appropriate. The latter was limited to patients with available cost data (relevant codes to enable accurate costings were missing from some records).
The number/proportion of patients who received psychiatric and non-psychiatric medication for chronic conditions in the pre- and post-index period was calculated as well as those who received medicines recommended by NICE for the treatment of adult ADHD (methylphenidate, lisdexamfetamine, dexamfetamine, atomoxetine, and guanfacine) and those who received medications out of scope of current NICE recommendations for the treatment of adult ADHD (bupropion, clonidine) in the post-index period. All code lists used to identify relevant prescriptions are available from study authors.
Results
Patient demographic and clinical characteristics
A total of 31,310 patients had a first record of ADHD diagnosis in CPRD or HES between 1 January 2016 and 30 June 2018. After excluding patients with <18 months of data pre- and post-index date and those aged <18 years, 2236 patients fulfilled the eligibility criteria and were included in the study (Figure 1). The mean age at index date in the overall cohort was 35.0 (SD 11.8) years and 61.1% (n = 1366) were male (Table 1). Over 80% (n = 1827, 81.7%) of patients were White and the mean IMD value was 5.9 (SD 2.9). Patients based in London accounted for almost a quarter (n = 500, 22.4%) of the study population. Mood disorders and anxiety were recorded in 47.1% (n = 1052) and 46.2% (n = 1034) of patients, respectively (Table 1). Half (n = 1118) of patients had at least one accident requiring attendance to A&E in the 18 months prior to ADHD diagnosis. A total of 477 (21.3%) and 385 (17.2%) patients met the criteria for the CG72 and NG87 subgroups, respectively. Demographic and clinical characteristics for the NICE subgroups are displayed in Table 1. A higher proportion of patients in the NG87 subgroup (n = 196, 50.9%) had anxiety-related disorders compared to the CG72 subgroup (n = 200, 41.9%; p = 0.01).
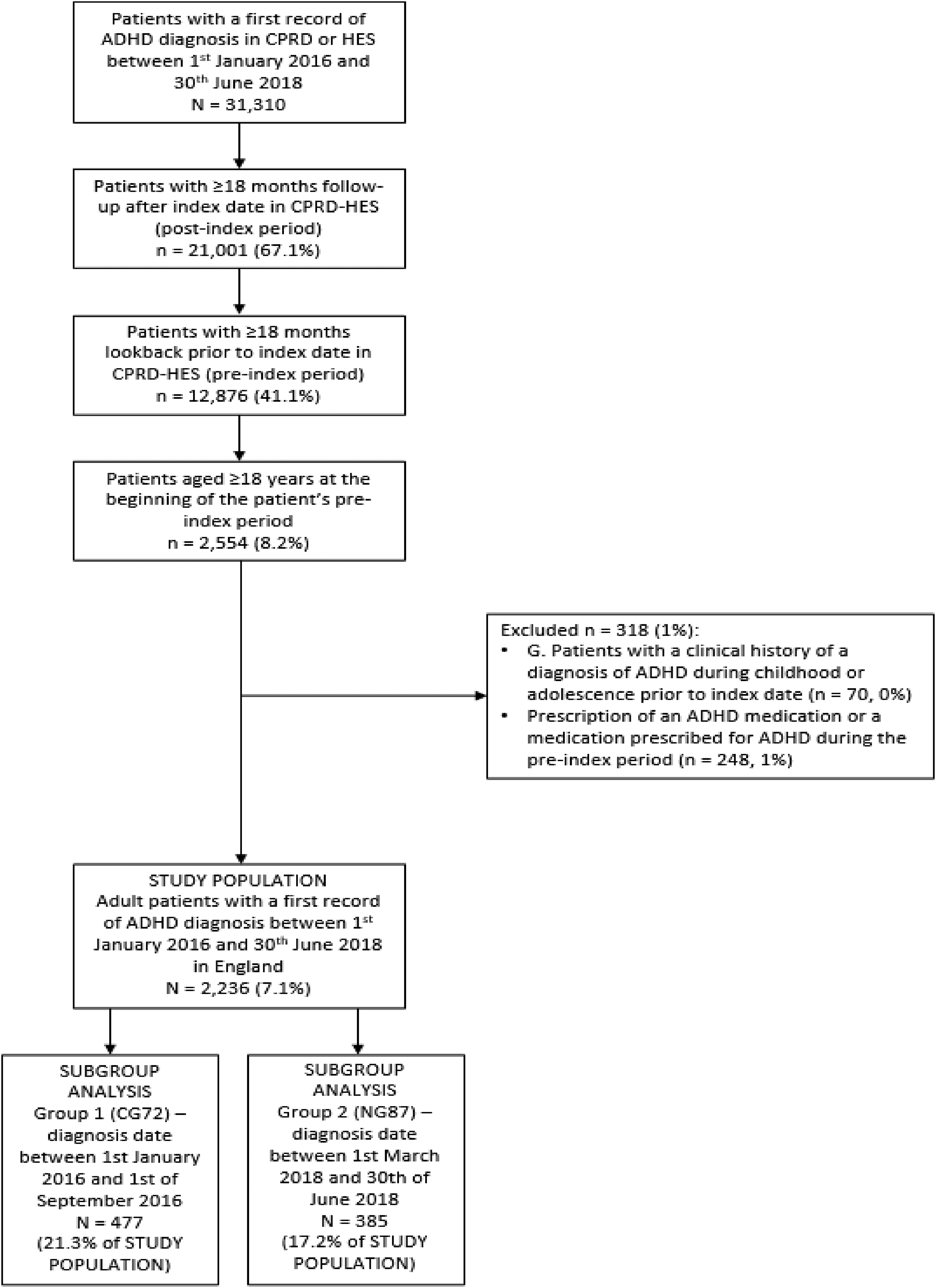
Study flow diagram.
Demographic and clinical characteristics of patients with a record of adult ADHD diagnosis in England between January 2016 and June 2018.

CPRD small number rules were applied to all study outputs to avoid identification of individual patients. This means that any number <7 (1–6) was suppressed in the results to a ‘-’, along with the next lowest number in the column (complimentary suppression), to avoid the possibility of deriving asked numbers.
ADHD: attention deficit hyperactivity disorder; CG72: Clinical Guideline 72; IQR: interquartile range; NG87: NICE Guideline 87; NICE: National Institute for Health and Care Excellence; SD: standard deviation; CPRD: Clinical Practice Research Datalink.
Including ankylosing spondylitis, colitis, and autoimmune thyroid disease only.
Including psoriasis and atopic dermatitis only.
Including asthma and allergic rhinitis only.
Significantly different between NICE CG72 and NG87 groups by Chi-square test.
Pre-index period HCRU and costs
Patients in the overall cohort had a median of 10 (IQR 5–18) primary care consultations recorded during the 18-month pre-index period; almost a third (n = 655, 29.3%) had ≥16 consultations recorded during this period (Table 2). Over half of patients (n = 1188, 53.1%) had at least one investigation in primary care; none of these investigations included an ADHD diagnostic assessment. Over half (n = 1282, 57.3%) of patients had outpatient visits, with a median of 3 (IQR 2–7) visits per patient. A total of 2349 A&E visits were recorded in 1005 (45.0%) patients and 679 day-case inpatient hospitalisations were recorded among 401 (17.9%) patients in the pre-index period. At least one overnight inpatient hospitalisation was recorded in 265 (11.9%) patients; the median length of stay for these patients was 3 days (IQR 1–7). Pre-index HCRU for each NICE subgroup is displayed in Table 2.
Healthcare resource utilisation of adult ADHD patients in 18 months pre- and post-diagnosis.

A&E: accident & emergency; ADHD: attention deficit hyperactivity disorder; CG72: Clinical Guideline 72; IQR: interquartile range; NG87: NICE Guideline 87; NICE: National Institute for Health and Care Excellence; SD: standard deviation.
The mean overall HCRU cost per patient was £1662.4 (95%CI £1525.3–£1799.5), based on 2190 (97.9%) patients. The mean costs per patient for each healthcare resource used were as follows: £322.5 for prescriptions; £442.6 for primary care consultations; £351.8 for A&E visits; £504.4 for outpatient visits; and £2468.1 for inpatient admissions (day-cases and overnight stays combined) (Table 3). The mean overall HCRU cost per patient in the CG72 subgroup was £1508.4 (95% CI £1318.8–£1697.9) during the pre-index period. In the NG87 subgroup, the mean HCRU cost per patient was £1697.5 (95% CI £1283.6–£2111.3). The pre-index costs per patient for each healthcare resource used in the CG72 and NG87 subgroups are displayed in Table 3.
Costs associated with healthcare resource utilisation of adult ADHD patients in 18 months pre- and post-diagnosis.

A&E: accident & emergency; ADHD: attention deficit hyperactivity disorder; CG72: Clinical Guideline 72; CI: confidence interval; IQR: interquartile range; NG87: NICE guideline 87; NICE: National Institute for Health and Care Excellence; SD: standard deviation.
Excluding non-healthcare professional-led consultations or administrative visits which are not costed for.
Post-index period HCRU and costs
Table 2 shows patients in the overall cohort had a median of 10 (IQR 5–17) primary care consultation recorded during the 18-month post-index period; over a quarter (n = 622, 27.8%) had ≥16 consultations recorded during this period. Approximately two-thirds (n = 1538, 68.8%) of patients had at least one investigation in primary care with a total of 4742 investigations. A total of 1365 (61.1%) patients had outpatient visits, with a median of 4 (IQR 2–9) visits per patient. A total of 2325 A&E visits were recorded in 915 (40.9%) patients and 704 day-case inpatient hospitalisations were recorded among 406 (18.2%) patients. At least one overnight inpatient hospitalisation was recorded in 257 (11.5%) patients with a median length of stay of 4 days (IQR 1–10). Post-index HCRU for each subgroup is displayed in Table 2.
The mean overall post-index HCRU cost per patient was £2065.0 (95% CI £1915.6–£2214.5) (Table 3). The mean costs per patient for each healthcare resource used were as follows: £614.5 for prescriptions; £416.9 for primary care consultations; £428.3 for A&E visits; £560.3 for outpatient visits; and £2966.1 for inpatient admissions (day-cases and overnight stays combined; Table 3). The mean overall HCRU cost per patient in the CG72 subgroup was £2069.6 (95% CI £1715.5–£2423.7) during the post-index period. In the NG87 subgroup, the mean HCRU cost per patient was £2046.6 (95% CI 1683.6–£2409.6). The post-index costs per patient for each healthcare resource used in the CG72 and NG87 subgroups are displayed in Table 3.
Comparison of 18-month pre- versus 18-month post-index HCRU and costs
A decrease of 1.0% and 6.4% in the total number of A&E visits and primary care consultations, respectively, was seen in the 18-month post-index compared to 18-month pre-index period (Table 3). The number of inpatient hospitalisations, prescriptions, and attended outpatient visits increased by 8.3% [extra 93 hospitalisations], 26.1% [extra 19,190 prescriptions], and 32.9% [extra 2763 visits], respectively. In 2153 patients (96.3% of the 2236 study patients) where cost calculations were possible, the total HCRU cost per patient was an average of £408.6 (95% CI £264.4–£552.8) higher in the 18-month post-index compared to 18-month pre-index period (p < 0.001; Figure 2). The mean costs of prescriptions per patient doubled from the 18-month pre- to post-index period (£322.5 versus £614.5) and represented the largest proportional cost increase post-diagnosis compared to other HCRU costs measured (Figure 2). When specific time intervals were considered, there was no overall difference in total HCRU cost between the 6-month pre-index period and < 6-, 6–12-, or 12–18-month post-index (Figure e1 and Table e3 in Supplemental material). A total of 460 (96.4%) patients in the CG72 subgroup and 369 (95.8%) patients in the NG87 subgroup had total HCRU costs available for both the 18-month pre- and post-index periods. The total HCRU costs in the CG72 and NG87 subgroups were £569.2 (95% CI £223.5–£915.0; p < 0.002) and £346.5 (£18.6–£674.3; p < 0.001) higher per patients in the 18-month post- compared to 18-month pre-index period, respectively.

Mean costs per adult ADHD patient in 18 months pre- and post-diagnosis by type of healthcare resource used.
Treatment
Approximately half of patients in the pre-index (n = 1118, 50%) and post-index (n = 1032, 46.2%) period received antidepressants while 13.3% (n = 297) and 11.6% (n = 259) of patients received anxiolytics in the two periods, respectively (Table 4). Approximately half (n = 1058, 47.3%) of the study population had at least one prescription recorded in primary care for any ADHD on- or off-label medication post diagnosis (Table 4). The most common NICE-recommended ADHD medication prescribed was methylphenidate (n = 733, 69.3% of treated patients/32.7% of total cohort). Prescribed medications out of scope of current NICE recommendations included bupropion (n = 10) and clonidine (n = 7). In the NG87 subgroup, 20.3% (n = 78) of patients received lisdexamfetamine compared to 8.6% (n = 41) in the CG72 subgroup (p < 0.001).
Treatments received by adult ADHD patients in 18 months pre- and post-diagnosis.

Significantly different between NICE CG72 and NG87 groups.
CG72: Clinical Guideline 72; NG87: NICE Guideline 87; NICE: National Institute for Health and Care Excellence; ADHD: attention deficit hyperactivity disorder.
Discussion
Our study describes real world HCRU burden and treatment provision in adult ADHD patients in the UK during the 18 months leading up to and 18 months post diagnosis. Our results show that adults diagnosed with ADHD have considerable HCRU both pre- and post-diagnosis. Total HCRU costs appeared to increase in the 18 months post- compared to 18 months pre-diagnosis; however, further studies are required to confirm this.
The presence of comorbidities may contribute to the considerable HCRU seen in adults with ADHD leading up to their diagnosis and afterwards, as the comorbidities themselves incur HCRU cost burden associated with their management. Mood disorders and anxiety were recorded in approximately half of patients in our study and is consistent with prior research (Bushe et al., 2015; van Emmerik-van Oortmerssen et al., 2012; Young et al., 2021). A study using CPRD conducted between 1998 and 2010 showed ADHD patients had a higher prevalence of psychological comorbidities than non-ADHD patients of a similar age (Holden et al., 2013). It has been suggested that ADHD may have a causal effect on depression, thus effective treatment of ADHD might help prevent subsequent development of depression in some patients (Riglin et al., 2021).
Our study showed that HCRU burden increased post diagnosis for adult ADHD patients in line with expected increases in the costs of managing and treating ADHD post diagnosis. An additional 19,190 prescriptions (26% increase) and an extra 2763 attended outpatient visits (33% increase) were recorded after diagnosis. This is in line with a similar study that found healthcare contact rates increased by 39% after ADHD diagnosis (Bushe et al., 2015). The increase in outpatient hospital visits likely reflects ADHD review appointments which can be frequent in the early months post-diagnosis as treatment is titrated. Our results indicate that between comparable 18-month periods, HCRU costs of ADHD patients in the UK increased by an average of £409 per patient post diagnosis. The greatest increase came from prescription costs (£286 per patient) which on average doubled post diagnosis. Despite this relative increase in HCRU burden, studies have shown consistent evidence that pharmacotherapies are more cost effective to healthcare providers long-term compared to ADHD patients who remain untreated (Wu et al., 2021). Treatment of ADHD not only results in improved symptoms and functional outcomes but also helps accompanying problems including criminality, occupational problems, substance use, obesity, and suicidal behaviour, which might lead to lower overall societal costs associated with ADHD (Dijk et al., 2021; Lichtenstein et al., 2012). In addition, obtaining a diagnosis is important to patients as it provides reassurance that their situation is not unique and that a body of knowledge and experience exists to help them (Craddock & Mynors-Wallis, 2014). It is important to note that we were only able to measure available costs through HES and CPRD. Thus, it is possible that what appeared to be an increase in costs in our study could possibly represent an actual decrease following discharge from secondary mental health care coordination and prescribing. Further research is needed, possibly utilising other available mental health databases. More research is also needed on the actual cost benefit of treating adult ADHD in the UK. Future studies need to explore both HCRU costs and quality of life post ADHD diagnosis and treatment. We could not assess quality of life as these measures are not recorded within the CPRD-HES linked data used for this study. It is also important to note that we could only capture prescriptions, but not assess adherence to treatment or whether patients’ medications were optimised.
The proportion of patients recorded in primary care without a prescription for an on- or off-label ADHD medication in the 18-month post-index period (53%) is consistent with proportions found in studies by Price et al. (2020) (38%) and Bushe et al. (2015) (54%) who researched both adolescent and adult ADHD prescribing in primary care. This is likely an underestimate of the actual number treated as only primary care prescriptions were captured in our study and as per NICE guidelines, GPs should not initiate prescription of ADHD medication without specialist input (NICE, 2018). Further prescriptions are likely initiated in secondary care but may not be linked back to CPRD.
NICE subgroups
There are differences in treatment practices and costs based on NICE guidelines (pre and post new guidelines). An increase in anxiety-related comorbidity recording was seen in the NG87 subgroup compared to CG72 subgroup which could be an indication that clinician recognition of ADHD symptoms and diagnosis of complex comorbid ADHD has improved alongside the expansion of the ADHD phenotype in the NG87 guidelines (NICE, 2018). Also, a greater proportion of patients in the NG87 subgroup received lisdexamfetamine which could suggest adherence to NG87 guidelines. However, NICE NG87 updated diagnostic and treatment guidelines were not published until 14 March 2018 and our study period contained patients diagnosed up to 30 July 2018 and followed-up to 31 December 2019. Thus, the study period post NG87 publication may have been insufficient to observe the full effect of the new guidelines on patient HCRU, particularly if practitioner uptake or ‘buy in’ to new guidelines is slow (Pitts et al., 2015).
Strengths and limitations
This study has many strengths, including the large sample size and use of CPRD data which is broadly representative of the general population in the UK. Some limitations, however, need to be considered. First, certain electronic patient healthcare data, for example HES, are collected primarily for the management, billing and/or reimbursement of patient care thus may be limited by the completeness and accuracy of the underlying data. However, a large number of studies have used HES for research purposes. CPRD data are primarily collected for the purposes of patient care and management. Second, it is important to note that the patients who are diagnosed may have more severe ADHD symptoms. As healthcare services for ADHD improve and begin to diagnose patients with milder symptoms, the findings of this study related to HCRU and costs may not be applicable to this broader, less severely impacted ADHD population. The patients in our study were predominantly White; however, the demographic makeup of our sample is reflective of the national demographic composition at the time the study was conducted. Our findings may primarily apply to this group, but there is a need for further research to understand any disparities in ADHD diagnosis across different communities. Under- and misdiagnosis when symptoms do not align with stereotypes may occur. Fourth, information on prescriptions administered outside of the primary care setting is not recorded in CPRD-HES linked data, meaning that this study may have substantially under-reported actual levels of prescribing ADHD treatments. Systems at Mental Health Trusts (i.e., Mental Health Minimum Data set or Clinical Record Interactive Search) should be investigated for further research but these sources could not be accessed for this study as linkage to CPRD is not available. Fifth, our analysis of HCRU costs included direct medical resource use only. Other costs outside the NHS such as out-of-pocket and private expenditures by patients or community care were not captured. Sixth, HCRU and cost was assessed for only 18 months post-diagnosis, thus we could not determine whether the increase in HCRU was short-term and possibly a result of time taken to find the right intervention. Finally, we were not able to evaluate the quality of life, societal benefits pre- and post-diagnosis, or the true efficacy of ADHD treatment prescribed via primary care.
Conclusion
In conclusion, this novel study provides key information on HCRU and costs in the period leading up to and following an adult ADHD diagnosis in the UK. An ADHD diagnosis in adulthood ensures patients receive the appropriate treatment for ADHD and comorbid anxiety and depression. Total HCRU costs appeared to increase post-ADHD diagnosis; however, further studies are required to confirm this. This cost increase should be set in the context of multiple widely reported societal benefits likely to accrue from the effective management of adult ADHD. Further research is required into the resource burden of confirmed treated-ADHD patients only to be able to better determine the current costs of management of adult ADHD for healthcare providers in the UK.
Supplemental Material
sj-docx-1-ndy-10.1177_27546330241242873 - Supplemental material for Healthcare resource utilisation and associated costs of adult attention deficit hyperactivity disorder in England
Supplemental material, sj-docx-1-ndy-10.1177_27546330241242873 for Healthcare resource utilisation and associated costs of adult attention deficit hyperactivity disorder in England by Marios Adamou, Sophia Abner, Peter Egger, Alessandra Venerus, Gianluca Lucrezi, Mariana Mota, Jeremy Charlton, Minesh Unadkat, Simon Meadowcroft and Christine Eksteen-Ramsden in Neurodiversity
Footnotes
Acknowledgments
Medical writing support was provided by Dr Julia Granerod from Dr JGW Consulting Ltd.
Data access
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by the Independent Scientific Advisory Committee of the Medicines and Healthcare Products Regulatory Authority (ISAC protocol 21_000559) and registered with ClinicalTrials.gov (NCT05137704).
Funding
The authors received the following financial support for the research, authorship, and/or publication of this article: Sponsorship for this work was funded by Takeda UK Ltd. IQVIA received funding from Takeda to conduct this study.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
