Abstract
Sepsis is an increasingly common presentation to which ambulance resources are dispatched. Whilst early administration of antibiotics (AB) has been associated with improved outcomes, the impact of prehospital administration in an ambulance service context appears uncertain. This systematic review aimed to compare the effect of prehospital administration of antibiotics together with usual care (oxygen and intravenous fluids), to usual care alone, on mortality for patients with sepsis. A systematic review was conducted adherent to JBI methodology. Studies were eligible for inclusion if they were published after 2000; conducted in the prehospital setting; compared AB plus usual care to usual care alone in the prehospital phase; and reported an outcome of mortality at any time point. Systematic searches of Medline, CINAHL, EMBASE and Google Scholar were conducted, with included articles subjected to quality assessment using JBI appraisal tools. Each stage was completed by two authors, with a third engaged to resolve conflicts. A narrative synthesis was conducted and reported, and certainty of evidence was assessed. Of 587 studies identified from the searches, five satisfied the inclusion criteria and were included in the data synthesis. Two were randomized controlled trials, and three used observational comparative designs assessed as being at low-to-moderate risk of bias. Regarding the primary outcome of mortality, there was no evidence from high-quality studies with a low risk of bias that prehospital administration of ABs decreased mortality when measured at 28, 30 or 90 days. Regarding secondary outcomes, there was no evidence from high-quality studies with a low risk of bias that prehospital ABs reduce the length of stay in the hospital generally or the intensive care unit. The certainty of findings was low for mortality at 90 days, and very low for measurement at 28 and 30 days. There was insufficient evidence from high-quality studies with a low risk of bias indicating prehospital administration of ABs in addition to usual care, compared to usual care alone, reduces mortality at 28, 30 or 90 days, or length of stay in hospital or ICU, for adult patients with sepsis. There is insufficient evidence to enable the recommendation of routine administration of antibiotics to patients with sepsis presenting to ambulance service clinicians in the prehospital setting. Investigation of administration to more severe sepsis presentations in settings where prolonged prehospital intervals are inherent is warranted.
Introduction
Sepsis is a multifaceted condition that contributes significantly to the global burden of disease. 1 Defined as a ‘life-threatening organ dysfunction caused by a dysregulated host response to infection’, 2 sepsis accounts for an estimated 48.9 million cases and 11 million sepsis-related deaths worldwide, representing almost 20% of all global deaths. 3 Those who survive sepsis are at risk of complex ongoing cognitive and physical disability, further adding to the overall impact on survivors and the health systems in which they receive ongoing care and rehabilitation. 4
The 2021 Surviving Sepsis Campaign International Guidelines emphasize screening and early treatment among the core principles of care. 5 While the focus has traditionally been on the implementation of these in the emergency department (ED), there remains interest in the prehospital phase of the continuum of care on the assumption that earlier AB administration must inherently be better. A growing body of research has investigated the effectiveness of screening by prehospital clinicians to improve early recognition and detection, and this has in most instances become the focus of prehospital care for sepsis.6–11 However, more recently early pharmacological intervention, specifically the administration of antibiotics, has been proposed as a logical evolution of strategy aimed at reducing time to definitive care. Antibiotics are a core component of the sepsis ‘bundle of care’ and facilitating administration as early as possible has been argued to be the optimal approach. The International Guidelines recommend administering antimicrobials within an hour of recognition, and for patients with shock, administering at least 30 mL/kg of IV crystalloids within 3 h of recognition. 5 Exactly what constitutes ‘early’ though continues to be unclear and subject to ongoing debate. Research by Kumar et al. from 2016 suggested a 7.6% increase in mortality for every 1 h delay in administration. 12 While often cited, few other studies have found such dramatic results, though it seems clear that early administration is important. More recently, a systematic review by Sterling et al. 13 and a critical analysis of time to administration by Weinberger et al. 14 suggest earlier may not necessarily translate to improved outcomes.
In the context of this present systematic review, ‘prehospital’ refers to care provided in an ambulance service context regardless of the type of clinician providing the care. Ambulance service clinical staffing models are not homogenous internationally; Australia, New Zealand, South Africa and the United Kingdom (UK) are almost exclusively paramedic-based systems, whereas European countries frequently have nurses or physicians staffing ambulances. 15 The type of clinician involved in the early administration of antibiotics for sepsis is important to note, as each may have different accuracy for identifying sepsis in the field and subsequent implications for appropriateness of administration.10,16
Prehospital administration of antibiotics has been described in a number of largely observational studies generally describing case series or non-comparative cohort studies, summarised in two systematic reviews in 2016 and 2022.17,18 The 2016 review by Smyth et al. 7 examining prehospital care interventions for sepsis broadly, reported no evidence to support prehospital administration of antibiotics, with only one of 19 included papers presenting mortality-based outcome data. The 2022 review of nineteen studies by Varney et al., 18 which included a meta-analysis of four studies of varying methodology, concluded prehospital administration can reduce mortality compared to usual care (oxygen and intravenous fluids). The conflict in findings between these existing reviews, publication of new research, 19 concerns regarding the methodology used by Varney et al. 18 regarding their mortality meta-analysis, inclusion of unreferenced studies, and lack of certainty of evidence assessment, collectively serve as justification for the conduct of this current review that sought to analyse studies presenting comparative data.
The aim of this systematic review therefore was to investigate whether prehospital administration of antibiotics in addition to usual care (oxygen plus IV fluids), compared to usual care alone, reduces mortality and hospital length of stay in adult patients with sepsis of any severity.
Methods
Methodology, reporting and registration
This review was conducted using the JBI methodology for systematic reviews 20 and reported in adherence to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (‘PRISMA’). 21 The protocol for the review was registered with the National Institute for Health Research (NIHR) International Prospective Register of Systematic Reviews (‘PROSPERO’) as Protocol CRD42021272768.
Eligibility
The inclusion criteria were:
Care context must have been ‘out-of-hospital’ or ‘prehospital’ care for suspected sepsis in an ambulance service context, ensuring the question remained focused on the setting and administration earlier than in an ED. This was important given the type of clinician providing emergency care in the ambulance service context differs internationally and may involve paramedics, nurses or physicians. Any comparative study design. Must compare ‘usual care’ (fluid therapy plus oxygen) to ‘usual care plus intervention’ (usual care + antibiotic administered prior to arrival at ED). The volume or type of fluid was not specified, nor the dose or type of antibiotic. Published in a language other than English. The research team did not have the capacity for translation of research articles but determined there was a low likelihood of English-language bias. Unavailability of a full-text article, as this would prevent appraisal and assessment of risk of bias, and prevent determination of certainty of evidence. Conference abstract only, as the lack of information would prevent appraisal and assessment of the risk of bias and prevent determination of certainty of evidence. Non-peer-reviewed article. Studies involving ‘secondary’ or ‘crossover’ data from other included studies. Non-comparative designs (case study, case report, case series and single cohort study). Reviews, commentaries or similar.
The exclusion criteria were:
Information sources
Data were sourced from the following databases: Medline via OVID; Cumulative Index for Nursing and Allied Health Literature (‘CINAHL’) via EBSCOhost; Excerpta Medica Database (‘Embase’) via OVID, and Google Scholar. To assist in identifying completed clinical trials, data were additionally sourced from the following clinical trial registries: World Health Organization – International Clinical Trials Registry Platform, ClinicalTrials.gov, European Union Clinical Trial Register, Cochrane Register of Controlled Trials (‘CENTRAL’), and Australian and New Zealand Clinical Trials Registry (‘ANZCTR’). Given the majority of paramedicine-focused peer-reviewed journals are not currently indexed in the aforementioned databases, manual searches were conducted of the following journal websites for completeness: International Journal of Emergency Services, Irish Journal of Paramedicine, British Paramedic Journal, Journal of Paramedic Practice, International Paramedic Practice, South African Journal of Prehospital and Emergency Care, Journal of EMS Medicine, Journal of High Threat and Austere Medicine, Australasian Journal of Paramedicine, International Journal of Paramedicine, and Journal of Emergency Medical Services. The searches were originally conducted in March 2022 and then updated in January 2023 as part of a routine pre-submission search.
Search strategy
Searches were conducted electronically (databases) and manually (reference lists and non-indexed journal websites). Electronic searches were conducted independently by two authors, using Boolean search strategies. The original Medline search incorporated a validated prehospital search filter, 22 which was then modified on an engine-specific basis for the other electronic sources. Proposed search terms were explored using ‘explosion’ functions, and examination of MeSH term scope descriptions to increase specificity and reduce duplication. The full search strategy for Medline is provided in Table 1 and Supplemental Appendix 1. Manual searches were conducted of the reference lists of articles that passed full-text screening and of known paramedicine-specific non-indexed journal websites.
Medline search terms (setting terms from Olaussen et al. 22 ).
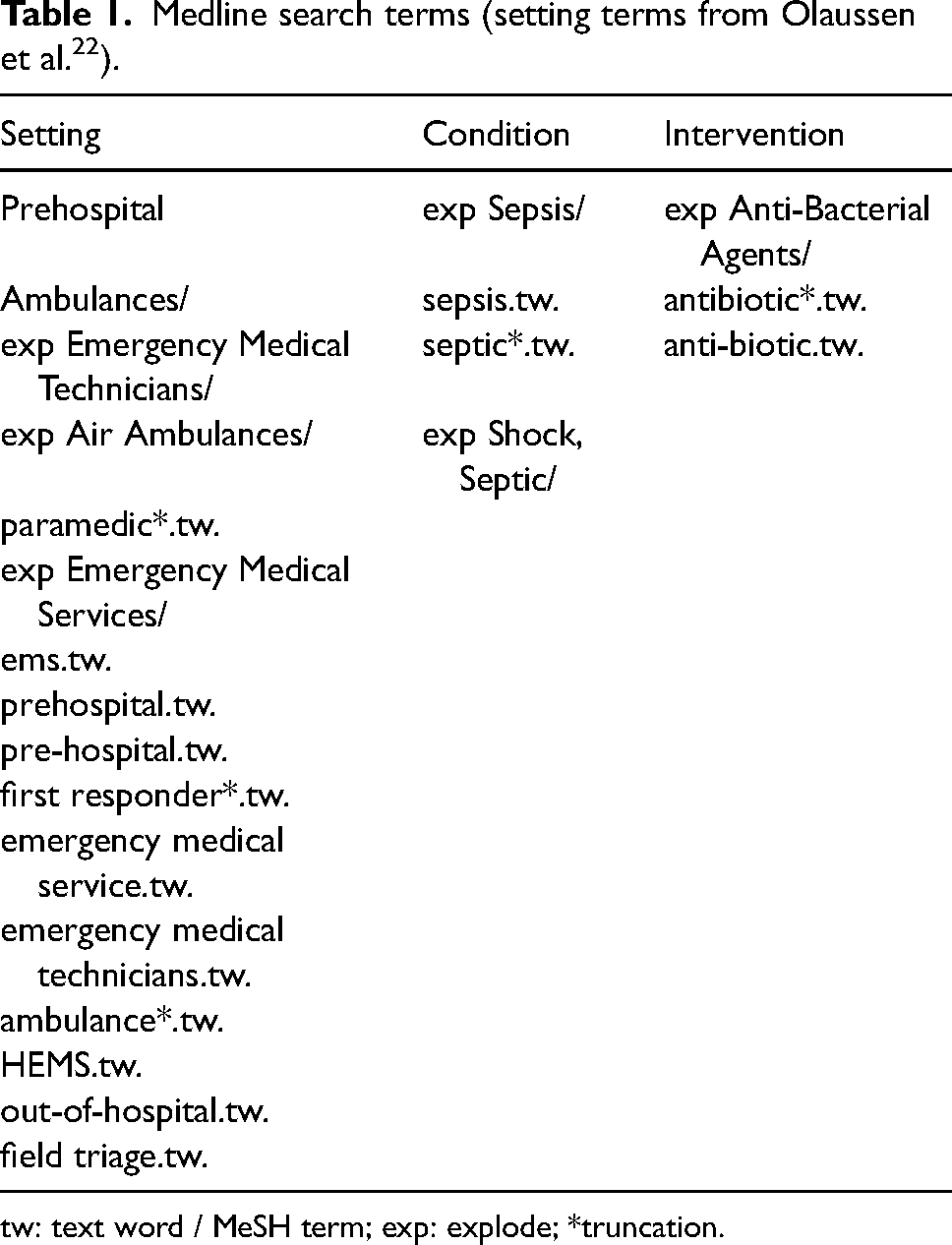
tw: text word / MeSH term; exp: explode; *truncation.
Data selection process
Data selection was conducted using Covidence (Veritas Health Innovation, Melbourne, Australia). The results from the search of each data source were imported into Covidence, and duplicate citations were examined and removed once confirmed. Where only an abstract was available, attempts were made to contact the respective lead author to identify full-text publications. Two authors undertook title and abstract screening independently, with conflicts resolved by a third. Two authors then undertook full-text screening independently, again with conflicts resolved by a third.
Data extraction
Data extraction was performed independently by two authors, with discrepancies resolved by a third. The extracted variables were author name, year of publication, study setting (country), number of study sites, study design, usual care, intervention (type of antibiotic), sepsis identification instrument, mortality, length of stay and hospital admission.
Risk of bias assessment
A risk of bias assessment was performed in adherence to the JBI approach using JBI appraisal instruments to produce a qualitative result. 23 As per contemporary JBI recommendations, numerical scoring of risk of bias was not performed, nor was assessment used to exclude studies from inclusion in the narrative synthesis. Randomized controlled trials (RCTs) were assessed using the updated JBI RCT risk of bias instrument, enabling examination of four domains relating to internal validity bias as well as statistical conclusion validity. 24 The other studies were assessed using the JBI assessment checklist for cohort studies.
Study outcomes
The primary outcome for this review was all-cause mortality, reported at any time point. Results are described for each time point identified. The secondary outcomes of interest were length of stay in an intensive care unit, and length of stay in hospital, both measured in days and reported as means or medians as per the original study reporting.
Data synthesis
Data were initially subjected to narrative synthesis, including examination of clinical and methodological homogeneity. Subsequently, the a-priori plan to conduct a meta-analysis for the primary outcome of mortality was abandoned due to concerns relating to unreconcilable heterogeneity in study design and methodology.
Assessment of certainty
Assessment of certainty in evidence was conducted using the Grading of Recommendations, Assessment, Development and Evaluation (GRADE) approach, 25 and a Summary of Findings (SoF) was created using GRADEpro GDT (McMaster University, ON, Canada). The SoF presents only findings related to the primary outcome of mortality and is stratified by time of measurement (28, 30 and 90 days). As meta-analysis was not performed, the SoF is reported narratively as per guidance from Murad et al. 26
Results
Study selection
The results of the electronic search strategy revealed 587 potentially relevant studies. After 53 duplicates were removed, 534 studies remained. The title and abstract of 534 studies were reviewed, resulting in the exclusion of 505 studies that did not address the clinical question or meet inclusion criteria. One abstract reporting what was described as a prehospital randomized controlled trial of antibiotics from Lebanon was not included as no associated full-text paper could be identified. 27 The remaining 28 texts underwent full-text screening for eligibility, with 24 texts excluded, leaving four for inclusion.28–31 A final updated search was conducted on 21 January 2023, in which a further study 19 was identified and deemed eligible for inclusion. The flow of study identification and selection of the five final included articles is illustrated in Figure 1.
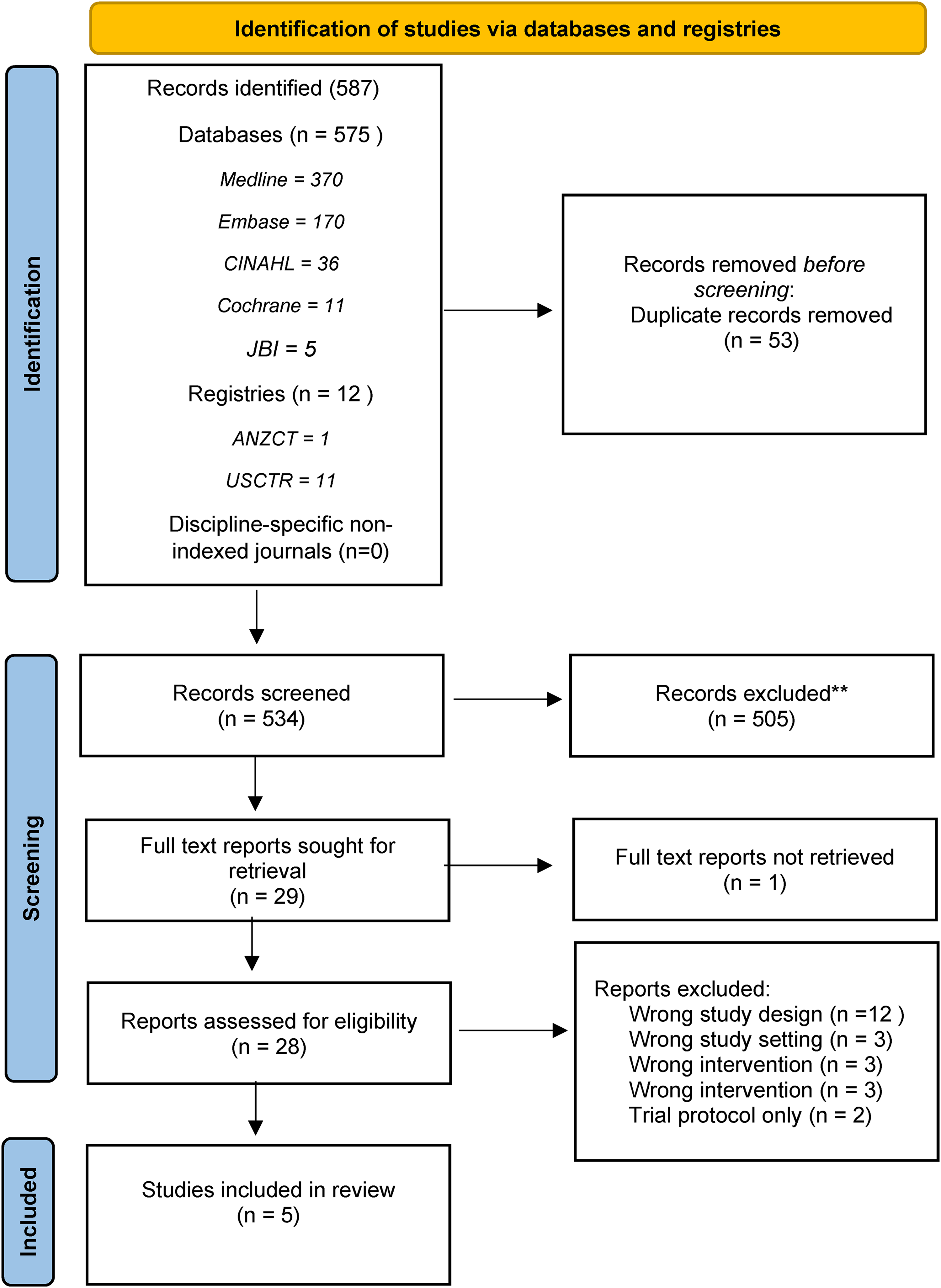
Study identification and selection.
Characteristics of included studies
The characteristics of each of the included studies have been summarised in Table 2. There was substantial heterogeneity in study design in terms of methodology and healthcare setting. Two were RCTs,19,30 the other three were observational comparative cohort studies. Martel et al. 28 employed a before and after sequential comparative cohort study, whilst Jouffroy et al. 29 reported a retrospective comparative cohort study using routinely collected ambulance records. Cunningham et al. 31 used a prospective cohort study with a historical comparison via medical record chart review. There was variation regarding the type of clinician providing care. Martel et al. 28 and Cunningham et al. 31 reported paramedics performing the identification of sepsis and providing care; Jouffroy et al. 29 reported physician-centred care delivered in a mobile intensive care ambulance; Alam et al. 30 nurses with emergency care training, and Jones et al. 19 paramedics.
Characteristics of included studies (n = 5).
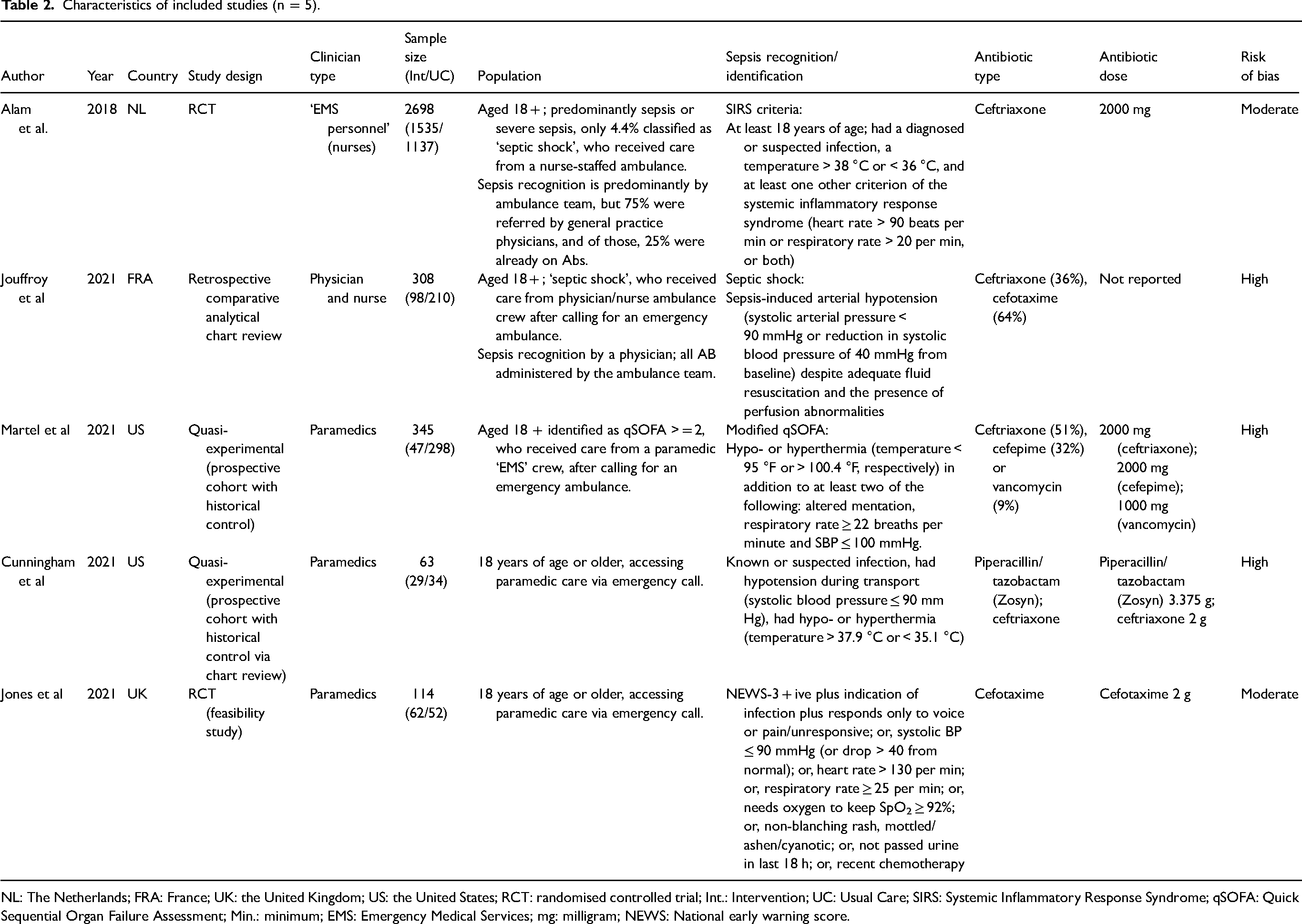
NL: The Netherlands; FRA: France; UK: the United Kingdom; US: the United States; RCT: randomised controlled trial; Int.: Intervention; UC: Usual Care; SIRS: Systemic Inflammatory Response Syndrome; qSOFA: Quick Sequential Organ Failure Assessment; Min.: minimum; EMS: Emergency Medical Services; mg: milligram; NEWS: National early warning score.
All included studies had similar inherent patient populations, that being adults with suspected sepsis in the prehospital setting, though criteria for identifying sepsis and determining eligibility for inclusion varied. Martel et al., 28 Jouffroy et al., 29 Jones et al. 19 and Cunningham et al. 31 included populations that were predominantly more unwell and more commonly in septic shock or ‘severe sepsis’, whereas Alam's RCT enrolled a population of whom only 4% were identified as being in septic shock. 30
Each study compared patient outcomes when an ambulance personnel regardless of clinician type administered intravenous broad-spectrum antibiotics in addition to the usual care of supplemental oxygen and fluid resuscitation. Ceftriaxone was the most administered antibiotic.
There was heterogeneity regarding the timepoint at which the mortality was measured, occurring at 28, 30 and/or 90 days. Only one study was appropriately statistically powered with an a-priori sample size to detect a difference in mortality. 30 No studies were statistically powered to the secondary outcomes of length of stay (hospital generally or ICU).
Risk of bias
The results of the risk of bias assessment are described for randomised controlled trials and observational studies in Tables 3 and 4, respectively. Alam et al. 30 and Jones et al. 19 were at moderate risk of bias, despite their RCT designs. Alam et al.30,32 was at least risk of bias; selection and allocation were sound, and an intention-to-treat analysis was performed, though internal validity was compromised due to no blinding of participants or clinicians. The study was powered appropriately to detect a difference in mortality outcome, and overall statistical conclusion validity was high. Jones et al. 19 demonstrated sound selection and allocation but also had a greater risk of bias due to lack of blinding. It was unclear regarding whether ITT analysis was performed and was weakened by being a feasibility study not powered to detect a difference in mortality.
Risk of bias assessment for randomised controlled trials (n = 2).
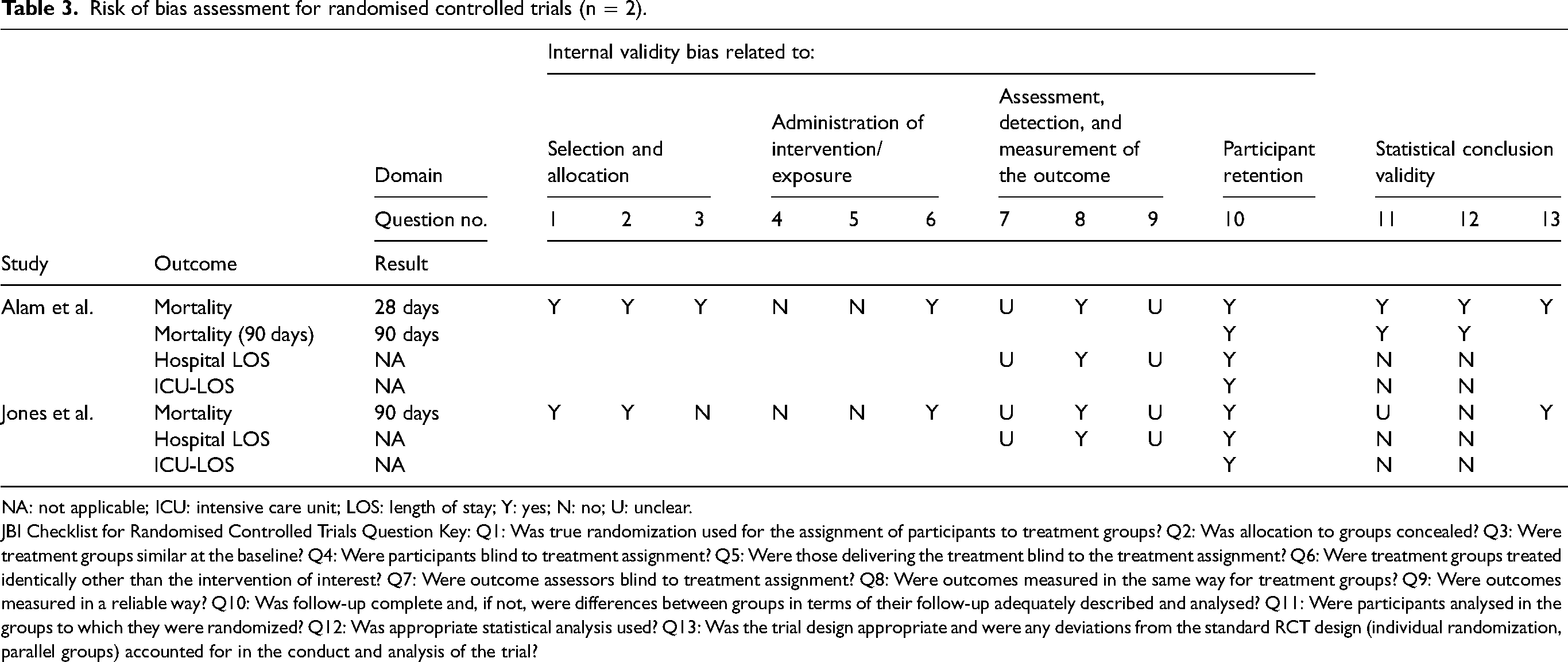
NA: not applicable; ICU: intensive care unit; LOS: length of stay; Y: yes; N: no; U: unclear.
JBI Checklist for Randomised Controlled Trials Question Key: Q1: Was true randomization used for the assignment of participants to treatment groups? Q2: Was allocation to groups concealed? Q3: Were treatment groups similar at the baseline? Q4: Were participants blind to treatment assignment? Q5: Were those delivering the treatment blind to the treatment assignment? Q6: Were treatment groups treated identically other than the intervention of interest? Q7: Were outcome assessors blind to treatment assignment? Q8: Were outcomes measured in the same way for treatment groups? Q9: Were outcomes measured in a reliable way? Q10: Was follow-up complete and, if not, were differences between groups in terms of their follow-up adequately described and analysed? Q11: Were participants analysed in the groups to which they were randomized? Q12: Was appropriate statistical analysis used? Q13: Was the trial design appropriate and were any deviations from the standard RCT design (individual randomization, parallel groups) accounted for in the conduct and analysis of the trial?
Risk of bias assessment for cohort studies (n = 3).
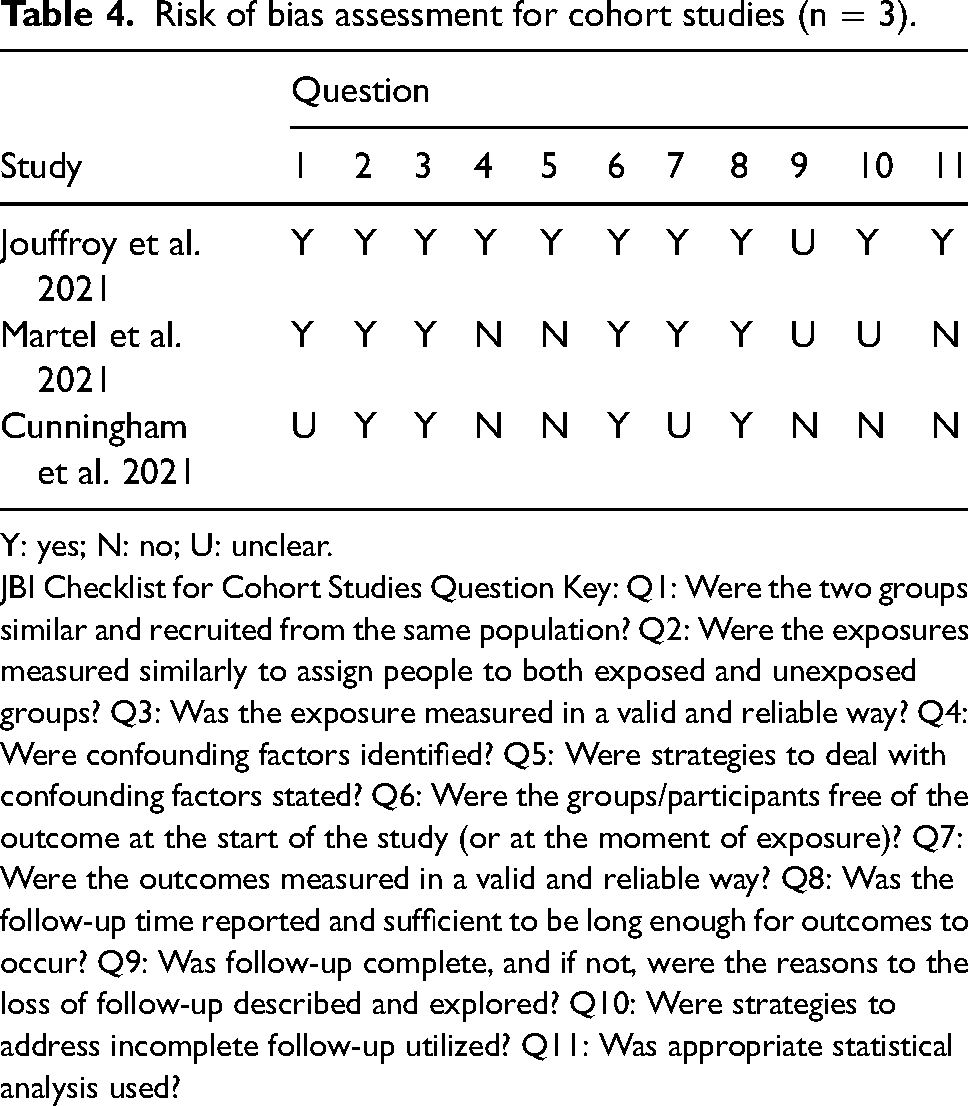
Y: yes; N: no; U: unclear.
JBI Checklist for Cohort Studies Question Key: Q1: Were the two groups similar and recruited from the same population? Q2: Were the exposures measured similarly to assign people to both exposed and unexposed groups? Q3: Was the exposure measured in a valid and reliable way? Q4: Were confounding factors identified? Q5: Were strategies to deal with confounding factors stated? Q6: Were the groups/participants free of the outcome at the start of the study (or at the moment of exposure)? Q7: Were the outcomes measured in a valid and reliable way? Q8: Was the follow-up time reported and sufficient to be long enough for outcomes to occur? Q9: Was follow-up complete, and if not, were the reasons to the loss of follow-up described and explored? Q10: Were strategies to address incomplete follow-up utilized? Q11: Was appropriate statistical analysis used?
The other three studies were of lesser quality given their observational nature and were deemed to be at higher risk of bias accordingly. Jouffroy et al. 33 mitigated the risk of bias to some degree through appropriate statistical adjustment for potential confounders, though the inherent limitations of retrospective data analyses remain apparent. Martel et al. 28 and Cunningham et al. 31 were open to substantial bias due to the observational sequential non-randomised design, absence of blinding, gross imbalance in enrolment in the number of participants between the two groups, and the absence of appropriate statistical adjustment for potential confounders.
Outcomes and results of individual studies
Five studies reported the primary outcome of mortality, though there was variance in the time at which that was measured, spanning 28 days,28,30 30 days31,33 and 90 days.19,30 Four studies reported the secondary length of stay outcomes (hospital and ICU).28,30,31,33 The individual study outcomes and results are detailed in Table 5.
Outcome measures and individual study results (n = 5).

RCT: randomised controlled trial; LOS: length of stay; ICU: intensive care unit; AB: antibiotic; HR: hazard ratio; p: probability.
Mortality
Five studies reported on mortality, but only one was prospectively statistically powered to this outcome. 34 Three found no difference19,30,31 whilst two reported statistically significant reductions favouring prehospital antibiotics.28,33
Regarding 28-day mortality, the RCT by Alam et al. 30 reported 8% mortality in both groups, though this time point was not the one to which the study's statistical power was calculated. Martel et al. 28 investigated 28-day mortality in their observational cohort study, reporting a large unadjusted magnitude in difference favouring ABs (8.5% vs. 25%).
Regarding 30-day mortality, neither RCT examined this timepoint, though it was reported in two observational studies albeit with conflicting results.31,33 The retrospective cohort study by Jouffroy et al. 33 which include a more shocked population reported a statistically significant difference favouring prehospital AB (24% vs. 31%), whereas Cunningham et al. 31 found no difference (27% vs. 28%) in their cohort study.
Regarding 90-day mortality, the RCTs by Alam et al. 30 and Jones et al. 19 investigated this time point and found no difference. The study by Alam et al. 34 was powered appropriately on 90-day mortality, achieving its required sample size; Jones et al. 19 was a feasibility study and whilst the proposed primary outcome is mortality at 90 days, it was not powered to that point so interpretation of their finding must be done with caution.
Overall, there was no evidence from high-quality studies at low risk of bias that prehospital administration of antibiotics in addition to usual care decreased mortality at 28, 30 or 90 days when compared to usual care alone.
Length of stay (hospital)
Four studies (two RCTs and two observational) reported the outcome of length of hospital stay.30,33 Three, including the large RCT by Alam et al. 30 with the lowest risk of bias, found no difference between prehospital antibiotics in addition to usual care compared to usual care alone.28,30,31 The observational study by Jouffroy et al. 33 reported a significant difference in hospital length of stay favouring prehospital antibiotics in addition to usual care, whereas the other three studies reporting this outcome, including the RCT by Alam et al., found no difference. None of the included studies were statistically powered to detect a difference in this outcome, hence these results are at risk of Type II error. Overall, there was insufficient evidence from high-quality studies at low risk of bias that prehospital administration of antibiotics, compared to usual care, results in a difference in length of stay in hospital.
ICU length of stay
Martel et al. 28 reported a significant difference in ICU length of stay favouring prehospital antibiotics, whereas the other three studies, including the RCT by Alam et al., found no difference. The study by Martel et al. 28 was considered at high risk of bias, casting uncertainty over that finding of difference. None of the included studies were statistically powered to detect a difference in this outcome, hence these results are at risk of Type II error. Overall there was insufficient evidence from high-quality studies at low risk of bias that prehospital administration of antibiotics, compared to usual care, results in a difference in length of stay in ICU.
Assessment of certainty of evidence
The certainty of evidence assessment results is illustrated in the summary of findings (Supplemental Appendix 2). The certainty of evidence for mortality measured at 90 days was assessed to be low. For mortality at 28 or 30 days, the certainty of evidence was assessed to be very low.
Discussion
This systematic review of five studies, which included three comparative observational studies and two clinical trials, found insufficient evidence from high-quality studies at low risk of bias to support a recommendation for routine administration of prehospital antibiotics for adult patients with sepsis. The results were inconsistent across methodologies. For the primary outcome of mortality, two RCTs showed no difference in 90-day mortality and one no difference at 28 days. Observational studies demonstrated inconsistent findings for mortality at 30 days. For the secondary outcomes of length of stay for hospital generally and ICU, there was no evidence of good quality at low risk of bias to indicate reductions favouring prehospital administration of antibiotics.
The conclusions drawn here differ from those drawn by Varney et al. 18 in a 2022 systematic review and meta-analysis, who concluded that receiving prehospital antibiotics can significantly lower mortality in sepsis patients compared to patients who do not receive prehospital antibiotics. The authors meta-analysed four studies reporting comparisons between prehospital antibiotics and usual care (two RCTs and two observational), reporting a risk ratio (RR) favouring prehospital antibiotics (RR 0.81; 95% CI 0.68–0.97; p = 0.02). Two possible reasons for the conflicting results relate to study selection and statistical methods. Regarding study selection, Varney et al. included an RCT by Chamberlain et al. in the meta-analysis, a study that favoured prehospital antibiotics (RR 0.74; 95% CI 0.55–0.98) but of which only a conference abstract appears to exist. Whilst no clear methodological guidance exists on whether to include abstract-only data sources in systematic reviews, it has been argued the absence of a full paper prevents accurate quality appraisal, assessment of the risk of bias, and determination of certainty of evidence. 35 If included, sensitivity analysis should be conducted to examine the potential effect, a process not undertaken in that paper. The current review made the decision to exclude abstract-only resources for these reasons. Varney excluded the paper by Cunningham et al., presumably due to it being a letter (a stated exclusion in their criteria); the present study included it, as whilst it was published under the classification of a ‘research letter’, it contained what was deemed to be sufficient detail to enable assessment of quality and risk of bias, thus meeting requirements for inclusion. Overall, Varney et al. included non-comparative, single cohort studies to identify mortality rates for intervention and usual care, in contrast to the present design that included only comparative studies. Finally, the methodological and clinical heterogeneity present amongst the studies included by Varney et al. suggest that a combined meta-analysis was inappropriate. Combining observational and clinical trials in a single meta-analysis requires complex analytic modelling using meta-regression or quality effects modelling 36 ; this appears not to have been addressed in Varney et al., 18 hence the overall meta-analysis result favouring prehospital antibiotics should be interpreted with caution.
The current study's findings are consistent with the systematic review reported by Smyth et al. 17 who concluded there was ‘little robust evidence’ supporting prehospital interventions for sepsis, including administration of antibiotics. That study was from 2016, prior to the emergence of all studies included in the present analysis.
The strongest evidence within the included articles in the present study is that from Alam et al., whose RCT from the Netherlands found no difference in the primary outcome of mortality and the second outcome of LOS (hospital and ICU). Whilst assessed as being at low risk of bias and being appropriately powered to detect a difference in mortality at 90 days if such a difference existed, it has received criticism for several perceived weaknesses. 37 Alam's study population would best be described as being less seriously ill; only 4% of all enrolled patients were classified as severe, that being having ‘septic shock’, limiting the generalisability of the results to a broader population outside the auspices of a clinical trial whom may more commonly have a more severe illness. Further, 75% of the enrolled patients were referred to the ambulance service by general practitioners (GP) and 25% of those were already receiving antibiotics; this decreases the generalisability to an ambulance service emergency response context which would be more likely to see patients who have called for assistance themselves and who are not already on antibiotics. Additionally, it could be argued that a GP’s capability to recognise sepsis would most likely be different from that of paramedics or similar non-physician prehospital providers based on the likelihood of differences in education, training and clinical exposure to this population of patients. Finally, the study protocol included increased training in recognition of sepsis for the ambulance staff and the emergency department staff; the study may therefore not be a true comparison of the intervention to ‘usual care’ as the nature of usual care in the field and in the ED after patient handover was altered. 37 Despite these limitations it is overall a well-conducted RCT assessed as having a low risk of bias and would on its own constitute the best available evidence in the context of a less seriously ill population of septic patients.
The second RCT by Jones et al. 19 reported a trend towards favouring antibiotics regarding mortality at 90 days (33.1% vs. 21.2%; unadjusted OR 1.9, 95% CI 0.82–4.5, p = 0.13), however, that result was not statistically significant and being a feasibility study was driven by a small sample not powered to detect a difference in this outcome. A high proportion of patients were already taking ABs at the time of call (28% and 18% for intervention and control groups, respectively), a characteristic like that reported by Alam et al. 32 (22% and 21% for intervention and control groups, respectively). Regarding the unwellness of participants, 69% of all those enrolled by Jones et al. 19 had a qSOFA score of less than two, slightly lower than the 75% in the study by Alam et al. 30 The ‘open label’ design of both studies creates increased risk of bias in both trials, a feature that contributes significantly to a reduction in the certainty of evidence arising from each.
The observational studies presented herein were deemed to be at moderate-to-high risk of bias and low certainty of evidence, hence they offer limited value in terms of answering the question posed in this review. Observational research should not be immediately discounted because of its susceptibility to confounding and other inherent limitations; in a 2020 critical analysis of time-to-antibiotic administration and its effect on outcomes, Weinberger suggests more than 30 studies have examined early administration, the majority of which are observational in nature. 14 Newly emerging evidence will most likely continue to be dominated by observational evidence due to the feasibility of conducting appropriately powered RCTs, but it is essential that such research appropriately addresses confounding through the use of sophisticated statistical analyses that adjust for key covariates including patient presentation (signs and symptoms). Of the observational studies in the present review, two of the three (Martel et al. 28 and Cunningham et al. 31 ) present unadjusted results with no accounting for potential confounders, substantially reducing the strength of the evidence put forward.
Prehospital administration of antibiotics assumes that earlier must be better, which in turn is based on the belief that shorter time to administration improves outcomes. However, the relationship between time to administration and outcomes may not be as clear as it is perceived to be. Two meta-analyses investigating this have produced conflicting results; Johnston et al. 38 analysed 10 studies comparing those who received ABs in less than one hour in the ED compared to those more than 1 h, and found a 33% reduction in mortality with earlier administration. In contrast, Sterling et al. 13 reported no significant mortality benefit of administering antibiotics within 3 h of ED triage or within 1 h of shock recognition in severe sepsis and septic shock. These however investigated time to administration and did not include the prehospital period. The existing body of evidence, again from in-hospital research, suggests that time may not be as critical in the first 3–5 h, and that it is at 5 h and beyond that antibiotics begin to have a different, positive impact on mortality. 14
As mentioned briefly earlier, it seems that apart from time to administration, the severity of the patient's condition may be a significant determinant in what magnitude of mortality reduction occurs with early administration. Jouffroy et al. 33 examined a more severely unwell population in their observational study and found a difference favouring prehospital administration, whereas Alam et al., 30 with their less seriously unwell population did not. Large hospital-based observational studies, for example, those by Seymour et al. 39 and Liu et al., 40 which reported results stratified by severity, indicated reduced mortality with earlier antibiotic administration in cohorts with septic shock compared to those without.
In the prehospital context, it might be that most benefit could be gained from judicious use targeting sicker patients with shock and those likely to experience a prolonged prehospital interval rather than a less nuanced approach seeking to get antibiotics to all septic patients as early as possible. This could reduce over-administration of antibiotics, particularly to those unlikely to receive outcome benefits, however, a lack of evidence around an appropriate threshold for treatment makes implementation of such a protocol difficult. It is vital to emphasise that the empiric administration of antibiotics is not without potential harms; in a world where increasing antimicrobial resistance poses a real threat, an increasingly judicious approach has been advocated for. 41 Potential harms include disturbances of the gastrointestinal microbiome, colitis, acute kidney injury, and the development of antimicrobial resistance. In the context of prehospital administration, investigation of harms and/or safety of administration was beyond the parameters of this review of effectiveness.
Central to judicious prehospital administration by ambulance clinicians is recognition of sepsis and application of decision making. The present review did not seek to investigate paramedics’ or other prehospital clinicians’ capability for recognising sepsis, nor the decision-making process occurring subsequent to recognition. The included studies used either SIRS or qSOFA as criteria for recognising sepsis, but conclusions as to the optimal approach to recognition are unable to be drawn based on this review. Several studies have sought to investigate paramedic screening for and identification of sepsis, summarised in a 2016 systematic review. 7 Synthesising nine studies, Smyth et al. concluded paramedic recognition of sepsis to be ‘poor’, and that the use of screening instruments appeared to result in improvement. This is an essential stream of inquiry of which pursuit in the prehospital context is recommended, as it speaks directly to the appropriateness and feasibility of a nuanced, targeted approach to prehospital antibiotic administration.
Looking to the future, adequately powered clinical trials with a low risk of bias are required to understand the true effect of prehospital administration of antibiotics, compared to usual care (oxygen plus fluids), on mortality and length of stay. Mortality benefits have not been demonstrated to date, but some have argued that mortality may not be the primary end-point of interest; in a 2018 commentary, Quinten et al. 42 argued that signs of organ failure might constitute a more appropriate endpoint in the emergency context than mortality. Several clinical trials are underway and in feasibility phases, so high-quality data may be emergent.
Limitations
There are several limitations in light of which the results presented herein should be considered. First, as in any review, there is the possibility that relevant research was not captured in the review and that such research could push the results in another direction. However, the search strategy was robust, and the data sources were broad and inclusive, resulting in a high level of confidence that all relevant data were identified and examined. Second, the study excluded papers not available beyond the abstract. This resulted in the exclusion of what appears to be an RCT by Chamberlain et al. 27 and potentially other conference abstracts not readily identifiable. The single-authored paper by Chamberlain reported a result of borderline statistical significance favouring prehospital ABs in patients with septic shock (28-day mortality rate 42.4% in the AB group vs. 56.7% who received usual care, OR = 0.56; 95% CI 0.32–1.00; p = 0.049). The uncertainty regarding the study methodology and statistical validity negatively impacts the usefulness of these data, and it is unlikely that the inclusion of the study would lead to a change in the narrative outcomes presented herein. It is essential in a systematic review that sufficient detail be available in included papers to allow quality assessment and investigation of bias. There is no clear guidance regarding the inclusion or exclusion of abstracts in systematic reviews, and the authors believe the rationale to exclude them to be sound and appropriate. Varney et al. conducted a sensitivity analysis without the abstract by Chamberlain et al. in their meta-analysis of RCTs and found it did not substantially change the result nor the conclusions drawn from it (RR 0.13; 95% CI 0.77–1.13; p = 0.049). Third, the small number of available studies, and the methodological quality of those included, makes drawing of inferences challenging, however, a review can only report on what is available. There is a clear need for high-quality prospective clinical trials at low risk of bias. Fourth, the study did not investigate health economic equations associated with the administration of prehospital antibiotics, nor safety and/or adverse events. These were beyond the scope of this review of effectiveness but deserve close investigation in future research as both speak to decision-making regarding the implementation or removal of an intervention. Fifth, the study did not include studies published in a language other than English, which may introduce a language bias. The research team did not have the capacity to investigate non-English speaking journals; it is believed that the impact of an English language bias to be minimal. Finally, unreconcilable clinical and methodological heterogeneity rendered a-priori plans for meta-analysis infeasible. Meta-analysis was explored, but the uncertainty regarding appropriateness was deemed to be of sufficient strength to cause adjustment of the analytical plan so as not to introduce unnecessary uncertainty into the results and conclusions.
Conclusion
This systematic review of five comparative studies spanning clinical trial and observational designs found insufficient evidence that prehospital administration of antibiotics in addition to usual care to patients with sepsis, compared to usual care alone (oxygen and IV fluids), makes a significant difference in mortality, length of stay in hospital, or length of stay in ICU. A recommendation for implementation of a protocol for routine empirical administration cannot be made, and future research should be sure to explore the threshold, in relation to prehospital time interval and severity of sepsis, at which prehospital antibiotics should be administered to patients with sepsis. Investigation of harms and/or safety, and health economics were beyond the scope of this review but require exploration alongside future research examining effectiveness.
Supplemental Material
sj-docx-1-pam-10.1177_27536386231207055 - Supplemental material for Prehospital administration of antibiotics in addition to usual care versus usual care alone for patients with suspected sepsis – a systematic review
Supplemental material, sj-docx-1-pam-10.1177_27536386231207055 for Prehospital administration of antibiotics in addition to usual care versus usual care alone for patients with suspected sepsis – a systematic review by Maddison Jayne Poynter, Ashley Farrugia, Elisabeth Kelly and Paul M Simpson in Paramedicine
Supplemental Material
sj-docx-2-pam-10.1177_27536386231207055 - Supplemental material for Prehospital administration of antibiotics in addition to usual care versus usual care alone for patients with suspected sepsis – a systematic review
Supplemental material, sj-docx-2-pam-10.1177_27536386231207055 for Prehospital administration of antibiotics in addition to usual care versus usual care alone for patients with suspected sepsis – a systematic review by Maddison Jayne Poynter, Ashley Farrugia, Elisabeth Kelly and Paul M Simpson in Paramedicine
Supplemental Material
sj-docx-3-pam-10.1177_27536386231207055 - Supplemental material for Prehospital administration of antibiotics in addition to usual care versus usual care alone for patients with suspected sepsis – a systematic review
Supplemental material, sj-docx-3-pam-10.1177_27536386231207055 for Prehospital administration of antibiotics in addition to usual care versus usual care alone for patients with suspected sepsis – a systematic review by Maddison Jayne Poynter, Ashley Farrugia, Elisabeth Kelly and Paul M Simpson in Paramedicine
Footnotes
Acknowledgements
This paper is an output from the Undergraduate Paramedicine Student Research Engagement Academy ('UPSTREAM'), an initiative of the Western Sydney University paramedicine program to increase engagement in research and build future paramedicine research capacity. The authors wish to acknowledge the School of Health Sciences at Western Sydney University for their support of the UPSTREAM program.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Paul Simpson is Editor in Chief of Paramedicine. He was not involved in any aspect of the submission process and editorial decision making, all of which were managed by other editors and conducted in adherence to the Journal's peer review policy and COPE best practice.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
