Abstract
Introduction:
While each of the 44 National Health Service commissioned artificial limb clinics in the United Kingdom record information about their own prosthetic limb users, these are not collated to give a national picture of amputee epidemiology. The requirement to respond to Freedom of Information (FoI) requests within 20 working days offers another way of extracting data from all centres, and this study describes a first attempt to use this method to update national epidemiological data.
Methods:
Questions were sent to the FoI email addresses of all 44 centres, requesting numbers of adult unilateral below-knee amputees, adult unilateral above or through-knee amputees and child unilateral above or through-knee amputees (all of K2 level mobility), numbers of people consistently using a prosthesis with a single axis myoelectric hand, and access to an occupational therapist with skills to teach someone to use a myoelectric hand. A FoI request was sent to NHS England seeking release of data that they collect every month from all the prosthetic services which they commission in England.
Results:
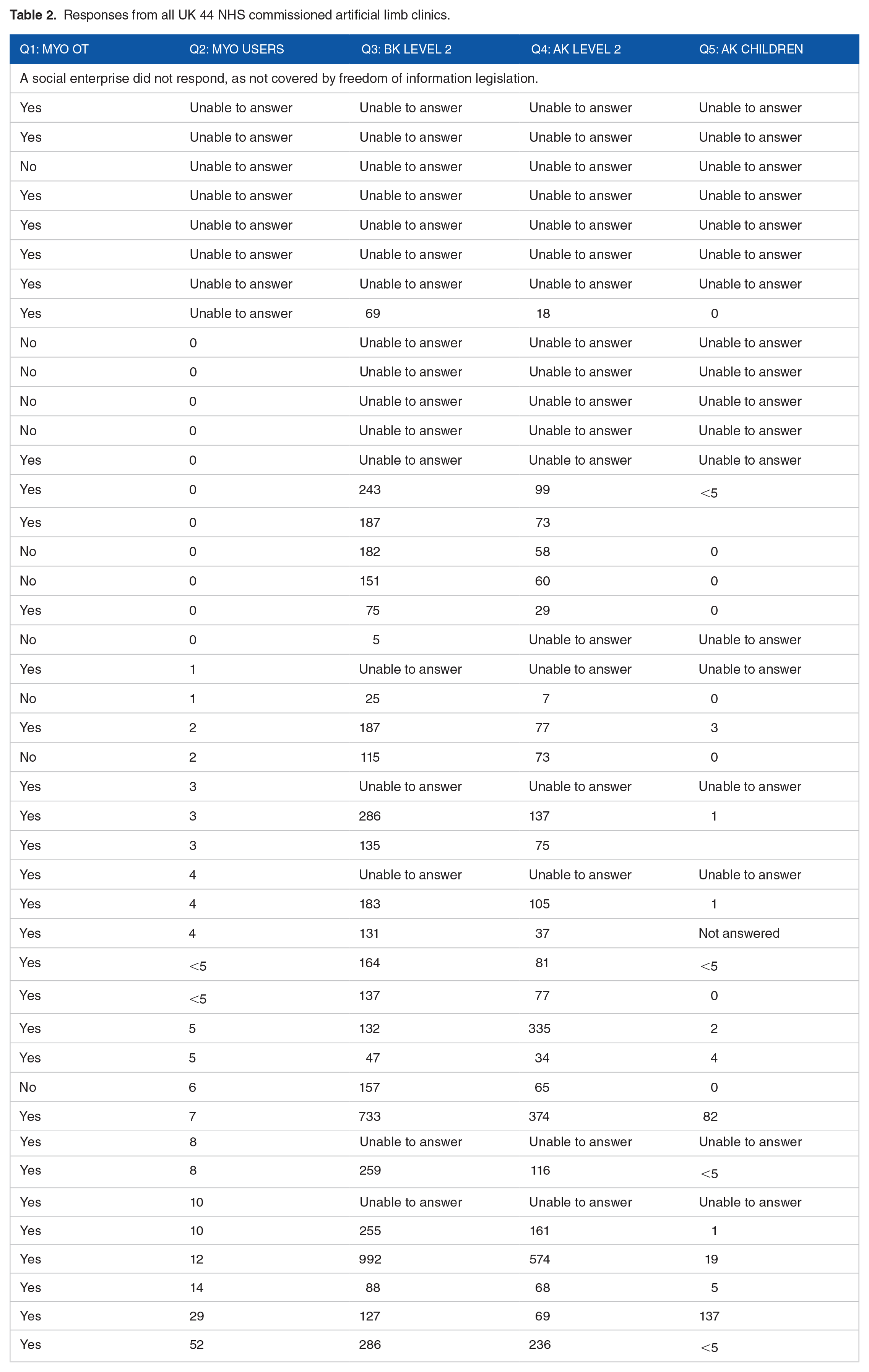
All but one of the UK centres responded, the great majority within 31 days. Incomplete results were generally due to centres finding the questions ambiguous and many did not record mobility levels consistently. While 33 centres had access to skilled occupational therapy, only 4 reported more than 10 patients who constantly used a single axis myoelectric hand. Eighteen centres were unable to provide complete data, and the remainder reported a ranges of 5 to 992 below-knee amputees, 7 to 574 adult above-knee amputees and 0 to 137 child above-knee amputees, suggesting different approaches to managing missing mobility level data.
Conclusions:
Freedom of Information requests are an inexpensive way of gathering data from NHS prosthetic clinics, which can identify age, sex, level of amputation but not activity levels or use of a prosthesis and hence only gives limited demographic information of the amputee cohort.
Introduction
National epidemiological statistics about limbless people referred to National Health Service (NHS) artificial limb clinics throughout the United Kingdom were collated and published by the National Amputee Statistical Database (NASDAB) between 1997/1998 and 2006/2007, and the most recent report available on line is from 2004 to 2005 (https://web.archive.org/web/20060514063930/http://www.nasdab.co.uk:80/pdf.pl?file=nasdab/news/2004-05_annual_report.pdf). Dr Martin Twiste took over responsibility for collating the annual data in 2010, and annual reports are available on the Limbless Statistics website (http://www.limbless-statistics.org/) for 2007/2008–2011/2012. National data has not been published since then.
Following reorganisation of NHS commissioning of prosthetic services in 2013, NHSEngland has required each (English) centre to submit data about each patient appointment since 2019 (https://www.england.nhs.uk/publication/limb-prosthetics-reporting-specification-technical-detail-specific-data-requirements-2021-22/). This includes individual patient identifiers, limb clinic identifier, date of birth, date of appointment, primary or established phase of treatment, level of amputation for each limb (above or below knee, above or below elbow) and cause of amputation (trauma, dysvascularity without diabetes, dysvascularity with diabetes, infection, neurological disorder, neoplasia, congenital without amputation, congenital with amputation). Reports of this data have not been published.
The Freedom of Information Act 2000 (https://www.legislation.gov.uk/ukpga/2000/36/contents) and similar legislation in Scotland, Wales and Northern Ireland gives people the right to ask to see recorded information held by public authorities. They are expected to provide this within 20 working days, without charge, unless the data is not stored or available in a form which can be easily searched, for example, old handwritten medical records. They can decline to release information if it may be so specific that it could be linked to individual people.
This paper describes use of Freedom of Information requests to gather nationwide epidemiological data about people with limb loss attending NHS prosthetic clinics.
Method
Freedom of Information email addresses were identified from the host trust website for each NHS prosthetic clinic in the UK. To see what sort of information was available, questions were phrased to identify how many people might fit the NHSEngland criteria for supply of newly commissioned multi-grip myoelectric hands (https://www.england.nhs.uk/wp-content/uploads/2022/09/2009-clinicial-comm-policy-multi-grip-prosthetic-hand-all-ages-1.pdf), how many people might be eligible for K2 level hydraulic ankles, and the population of children using prosthetic knees. The NHSEngland criteria of “consistently” using a single grip myoelectric hand for 1 year, is not further defined in that policy.
The prosthetic service in Cambridge is provided by OpCare, who require their prosthetists to document a patient’s mobility level or upper limb function, at each appointment, using the same definitions as K 0-4 levels devised for Medicare in USA in 1995, and the task more clearly defined by Gailey et al in 2002, 1 see Table 1.
K levels of lower limb amputee mobility.

However OpCare record these as A0-4, as originally developed by NHS management in 2004 (personal communication, Dominic Hannett). This survey revealed that the terms A0-4 are not generally accepted outside OpCare, although the K levels themselves are widely understood.
Five questions were asked:
Do they have access to an occupational therapist with skills to teach an amputee how to use a myoelectric hand?
2) How many amputees do they have who have consistently used a single axis myoelectric hand for the past 12 months?
3) How many unilateral below knee amputees, classified as A2 level mobility, born before 1/10/2004, who have attended the clinic or had a job done after 1/10/2020?
4) How many unilateral above or through knee amputees, classified as A2 level mobility, born before 1/10/2004, who have attended the clinic or had a job done after 1/10/2020?
5) How many unilateral above or through knee amputees, classified as A2 level mobility, born after 1/10/2004, who have attended the clinic or had a job done after 1/10/2020?
Influenced by the preferred terminology mentioned in responses from individual centres, a FoI request was sent to NHS England asking for :
Number of individual people with unilateral transtibial amputation for any cause who have attended any NHS England commissioned centre in the past 2 years?
Number of individual people with unilateral transtibial amputation for any cause who have attended each individual NHS England commissioned centre in the past 2 years?
Number of individual people with unilateral transtibial amputation for any cause who have attended any NHS England commissioned centre in the past 2 years with date of birth before 1/1/1942
Number of individual people with unilateral transtibial amputation due to cancer or malignancy who have attended any NHS England commissioned centre in the past 2 years?
Results
Thirty five centres responded within 31 days, 7 in 43 to 59 days and 1 after 270 days. One did not respond, explaining that FoI legislation does not apply to social enterprise companies.
Incomplete results were generally due to centres not recognising A code mobility codes, as they use K codes, or not recording any mobility level in their database, but some felt ‘had a job done’, ‘below knee’ and ‘single axis’ myoelectic hand were unclear.
Results from all centres are listed in Table 2, in order of number of myoelectric limb users, as this was the most consistently supplied data. NHS England was unable supply data for any of my requests, saying they could not identify unilateral trans-tibial amputations, or link these with date of birth.
Responses from all UK 44 NHS commissioned artificial limb clinics.
Discussion
Cambridge University Hospital’s Research Governance department do not regard Freedom of Information requests as research and therefore these surveys do not need to be registered as such, require ethical committee approval or any consent procedure, which makes this method quick and inexpensive to the person seeking the data. Where the contact details of the relevant department cannot be identified on public websites, the FoI officer in the receiving trust is in a good position to find the appropriate person. While formal ethics committee review would complicate and delay this simple method of data collection, review by a colleague (or second author) and informal testing of questions at a few centres would have identified the ambiguities at an early stage.
There has been some concern that using Freedom of Information legislation to gather data to inform medical practice may become restricted due to ‘ethics creep’, 2 and it has been questioned ‘if this is not counted as research, what is the ethical position of reporting the data?’ (Personal communication, anonymous reviewer of this paper). Reviewers may be reassured if an explicit statement of how the requested information would be used, is included in future surveys. This method would allow the epidemiologist to get nationwide data for their particular remit, rather than rely on annual reports from a national registry, like NASDAB, which only reported on new referrals to NHS prosthetic centres.
In practice this method has been less successful than initially anticipated. The legislation requires public authorities to supply the (available) data within 20 working days, but in practice this has not been achieved, and the whole process has taken longer than expected. While individual centres were able to extract data from their computer records, NHSEngland was not able to do so centrally.
These returns also demonstrate the small number of clinics who maintain or supply myoelectric prostheses, which will inform discussions about provision of hardware and training to use the recently commissioned multi-gesture hands in England. This is already centralised to 1 centre in Scotland, which facilitates staff training and stock control. Thirty three centres had access to skilled occupational therapy, only 4 reported more than 10 patients who constantly used a single axis myoelectric hand.
Eighteen centres were unable to provide complete data, and the remainder reported a ranges of 5 to 992 below-knee amputees, 7 to 574 adult above-knee amputees and 0 to 137 child above-knee amputees, suggesting different approaches to managing missing mobility level date.
NHS England does not require English centres to record the activity level of their patients, so this data would need to be asked of a subset of centres who regularly document this as part of their local practice. Inclusion of a measure of mobility, such as K level or SIGAM score, 3 in NHS England’s service specification would make the national data set much more useful, although at a cost of some extra work for clinicians at each appointment. A similar code to describe upper limb prosthetic use would be of value, perhaps using the A codes introduced for this purpose by NHS many years ago. It will be interesting to see if it remains possible to gather this epidemiological information from commissioners, when this function is delegated from NHSEngland to local Integrated Care Networks
In conclusion, this method has potential for gathering useful epidemiological data from a nationwide cohort, but of a rather limited nature and hence only gives limited demographic information of the amputee cohort.
Footnotes
Acknowledgements
I am grateful to the clinicians and managers at all centres who provided this information.
Funding:
The author received no financial support for the research, authorship, and/or publication of this article. Sage waived the publication fee.
Declaration Of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Freedom of information email addresses available on request.
Ethical Approval and Informed Consent
This survey required and received no ethical approval or informed consent statements.
