Abstract
Despite rehabilitation, stroke patients continue to have impaired function and reduced health-related quality of life (HRQoL) even in a chronic stage. However, no clear information is available on long-term variations in HRQoL. In this study, we aimed to report the short- and long-term changes in HRQoL in a subacute stroke sample that was enrolled in a clinical trial on arm rehabilitation. Thirty-nine stroke survivors (62% male, mean age 68 years) were assessed using the Stroke Impact Scale Version 2.0 (SIS 2.0) pre and post rehabilitation and at 6 months and 6 years follow-up. Long-term changes in physical function were explored through clinically meaningful changes in the Stroke Impact Scale-16 (SIS-16). After rehabilitation (P < .001), an overall improvement was found in all SIS domains except the memory and thinking, emotions, and communication domains. The baseline SIS-16 score (P < .001), the presence of a sensory deficit, and rehabilitation setting (P < .05) were factors related to the SIS-16 domain scores at the end of rehabilitation and at 6 months follow-up. Patients showed the most deterioration in the mobility (P < .001), strength (P < .003), and hand function (P < .05) domains 6 years after stroke. Stroke severity, male gender, and age ⩾65 years are related with a long-term HRQoL reduction after stroke.
Introduction
The increased survival rate after stroke and the aging population contribute to the high prevalence of individuals living with disability. 1 After stroke, a wide range of motor and cognitive impairments with a broad spectrum of severity can be found. Specifically, mobility impairment, increased dependence in activities of daily living, and cognitive, communication, and emotional deficits with mood alterations and social alienation have been reported even at a chronic stage.2,3 Among motor impairments, one of the most common consequences is upper limb paresis, which impacts up to 85% of stroke survivors. 4 Moreover, it is well known that arm and hand function are closely associated with health-related quality of life (HRQoL) at discharge from inpatient rehabilitation. 5 The exploration of changes in HRQoL domains after stroke is a timely clinical research need, considering how HRQoL reflects a meaningful aspect of human functioning that includes physical and psychological health, social relationships, and environmental factors, as suggested by the World Health Organization (WHO). 6 Indeed, stroke survivors’ HRQoL can be affected in multiple ways, even in those with mild strokes4,5 and at the chronic stage. 7 HRQoL is usually poorly investigated in stroke rehabilitation clinical trials and when it is considered, generic and non-specific questionnaires, such as the SF-36 8 or EQ-5D, 9 are used.
Within this framework, the Stroke Impact Scale (SIS) can be used to measure HRQoL after stroke. This self-reported, stroke-specific instrument evaluates the domains of physical and emotional states, activities, and participation in everyday life.10,11 To date, several studies have revealed negative impacts on an individual’s hand function, strength,2,12,13 activities of daily living (ADLs), and social participation2,14 after stroke. However, there is a paucity of studies on the perceived impact of stroke at different time points following a stroke.15 -18 Therefore, a better understanding of the perceived change in HRQoL over time is needed to design and provide timely and proper rehabilitation interventions. 19
Generally, rehabilitation therapy is delivered within the first few months after stroke.20,21 However, several studies highlighted the need for rehabilitation even in a chronic stage,22,23 especially in patients with moderate or severe stroke. 22 After specific stroke rehabilitation, several factors, such as the severity of paresis, 24 spasticity, 25 general deconditioning, 26 or social isolation, 27 can independently contribute to the worsening of motor and cognitive functioning achieved during recovery.
With the aim of exploring temporal variations in HRQoL in a sample of stroke survivors, we conducted a secondary analysis of a randomized controlled trial (RCT) in which we failed to prove the superiority of technology-assisted arm rehabilitation, namely, unilateral proximal arm robot-assisted therapy combined with hand functional electrical stimulation (RAT+FES), to intensive conventional therapy (ICT) for arm motor recovery and disability. 28 The aim of this study was to examine global and domain-specific HRQoL in subacute stroke patients after rehabilitation and at a short- (6 months) and long-term follow-up (6 years). We hypothesized that HRQoL would improve with rehabilitation and reach a plateau in the chronic phase. However, given specific factors, such as age, severity, deconditioning, or social isolation, HRQoL can deteriorate years after stroke.
Identifying HRQoL changes across different rehabilitation phases and potential related factors can help to define optimal rehabilitative interventions that have a positive impact on HRQoL in stroke survivors, specifically through long-term management.29,30
Materials and Methods
This was a secondary analysis of an RCT. 28 We included adults diagnosed with first ischemic stroke within 8 weeks of onset and upper limb motor impairment (Fugl-Meyer score of 11-55). Participants were randomly allocated to one of the following arm rehabilitation interventions: RAT + FES or ICT for 30 sessions (5 times/week over 6 weeks). In addition to arm rehabilitation, the enrolled patients received multidisciplinary rehabilitation according to their specific needs (ie, mobility training, speech therapy, vocational therapy, and psychological counseling) within an inpatient or outpatient setting accordingly with their individual needs.
The local Ethics Committee approved the study, and written consent was obtained from each participant. The Barthel Index (BI) score at admission was used to determine stroke severity: BI scores of 100 to 50 signified a mild stroke; 49 to 15 signified a moderate stroke; and ⩽14 signified a severe stroke. 31
The Stroke Impact Scale (SIS) was used to assess HRQoL. It is a reliable, valid, and sensitive scale.15,32,33 The SIS Version 2.0 consists of 64 questions divided into 8 domains: strength, hand function, mobility, activities of daily living/instrumental activities of daily living, memory and thinking, communication, mood, and social participation. 15 A score between 1 and 5 is assigned to each question: higher scores reflect better quality of life. Four domains (strength, hand function, mobility, activities of daily living, and instrumental activities of daily living) can be evaluated together to form a composite physical domain called the Stroke Impact Scale-16 (SIS-16). 32 The SIS 2.0 also includes a 0 to 100 visual analog scale to evaluate a patient’s global perception of their recovery. In stroke patients with cognitive or communication impairments, their caregiver was invited to complete a proxy version of the scale. Agreement between individuals with stroke and their proxies are acceptable in most SIS domains. 34 Moreover, the SIS 2.0 can even be administered by phone, with the possibility to evaluate patients discharged from the hospital with limited mobility and those living in rural areas. 35
The SIS 2.0 was administered by an investigator blinded to the treatment group before the intervention (T0), after 3 weeks (T1), at the end of treatment (6 weeks, T2), at the 6-month follow-up (T3), and at the 6-year (T4) follow-up.
The changes in each SIS 2.0 domain over time were measured to identify the temporal profile of HRQoL variation. Moreover, to specifically address long-term changes, we categorized our sample according to the SIS-16 minimally clinically Importance Difference (MCID) (9.2 points for the strength domain, 5.9 points for the ADL/IADL domain, 4.5 points for the mobility domain, and 17.8 points for the hand function domain). 36 In line with previous studies,2,17 patients were grouped according to their clinically significant positive change (⩾MCID), clinically significant negative change (⩽MCID), and no change (if between −MCID and +MCID) between T3 and T4. Patients who were available for the T4 evaluation were included in the long-term analysis.
Data analysis
Descriptive statistics were used for clinical and demographic variables and to report SIS 2.0 data at T0 to T4. Data are reported as the mean and standard deviation and the median and interquartile range or frequency and percentage according to data characteristics and distribution, verified through a Shapiro-Wilk test. Each SIS 2.0 domain score was generated using the algorithm [(real score − lowest possible score) × 100]/possible score amplitude. 37 Correlation among the demographic characteristics (ie, age, sex, and setting), clinical factors (ie, stroke severity, cognitive/emotional deficits, and comorbidities), and SIS 2.0 domains were analyzed, using Spearman’s rank correlation coefficient (Rho). A linear regression model was used to underline variables that predicted the SIS-16 score at different time points (dependent variable).
A repeated-measures analysis of variance (RM-ANOVA; P < .05) with time (4 levels: T0, T1, T2, and T3) as the within-subject factor and type of treatment (2 levels: ICT or RAT + FES) as the between-subject factor was performed to reveal a difference in SIS 2.0 domains within each treatment group over time. The Wilcoxon rank-sum test was used to explore changes in domain scores between the short- (T3) and long-term follow-up (T4). The Mann-Whitney U test was used to examine differences in SIS scores concerning stroke severity (mild/moderate), sex (male/female), and age (<65 or ⩾65 years) at the T4 follow-up. The categorical cutoff values were arbitrarily chosen. The significance level was set at P < .05. The software used for the analyses was STATA 13.0.
Results
Three hundred ninety-one consecutive patients with ischemic stroke were screened, and 40 were enrolled in the study (median age, 68 years; 61.5% male). 28 Participants were randomized to the 2 groups through a blocked randomization approach: 20 patients were allocated to the RAT + FES group, and 20 were allocated to the ICT group. The 2 groups did not differ in demographic or clinical characteristics. Moreover, the 2 groups were homogeneous in the SIS 2.0 domains evaluated at baseline (T0). See Table 1.
Baseline sample characteristics.
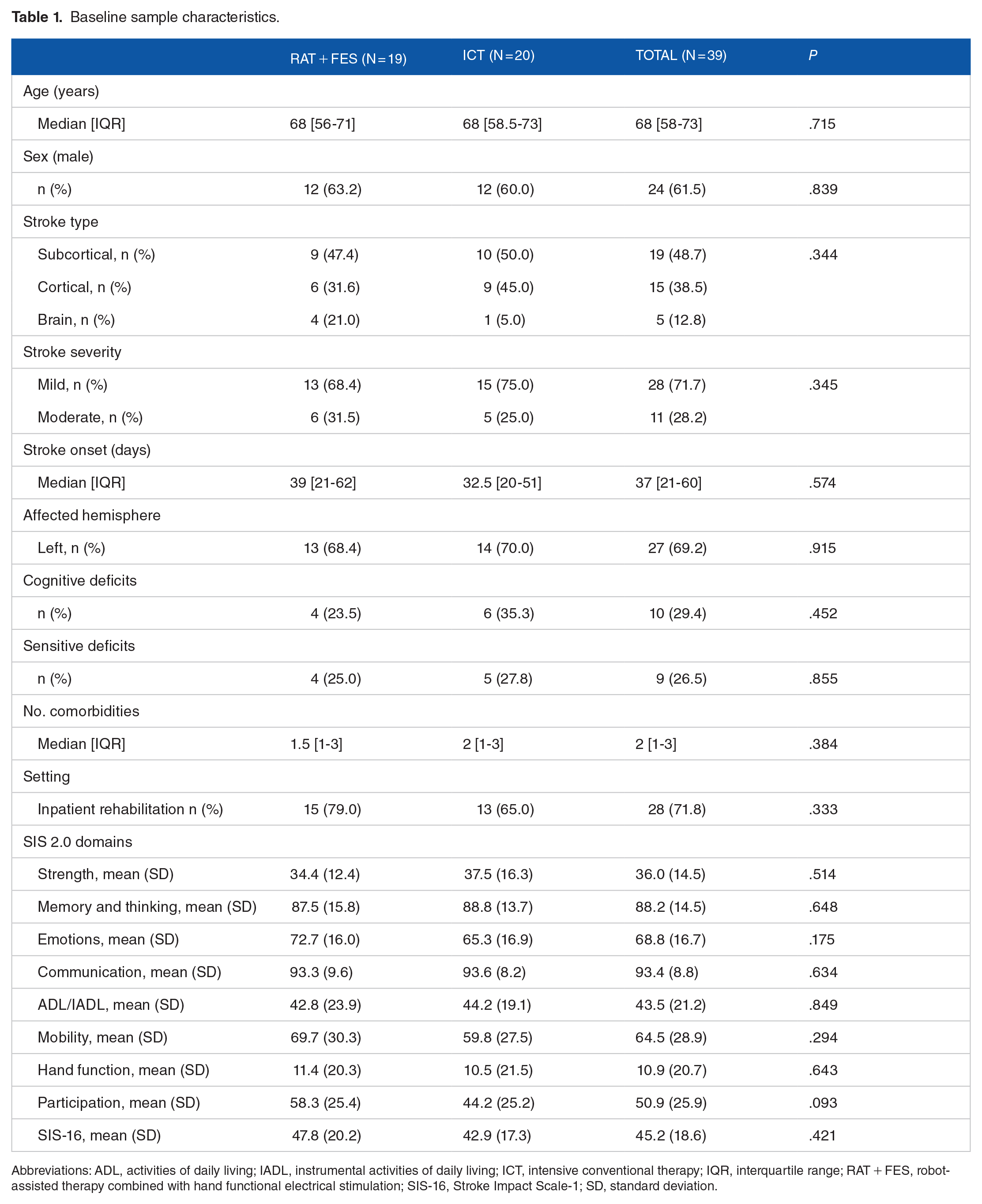
Abbreviations: ADL, activities of daily living; IADL, instrumental activities of daily living; ICT, intensive conventional therapy; IQR, interquartile range; RAT + FES, robot-assisted therapy combined with hand functional electrical stimulation; SIS-16, Stroke Impact Scale-1; SD, standard deviation.
At T4, the SIS 2.0 was completed by phone almost 6 years after stroke (mean onset = 70 months, SD = 8.78). Among the 40 enrolled patients, 17 patients did not complete the 6-year evaluation (8 were deceased or untraceable, 5 dropped-out at 6 months follow-up, and 4 refused the interview), representing 42.5% of the entire sample.
Thus, 6 years after stroke, the SIS 2.0 questionnaire was administered to 23 patients (61% aged > 65 years at stroke onset; 61% male). The Barthel Index (BI) was used to categorize stroke severity 31 : according to the BI score at T0, 17 patients had mild stroke and 6 had moderate stroke. Among these patients, 16 (70%) had a lesion in the right hemisphere, 6 (26%) had cognitive deficits, and 8 (35%) had anxious and depressive symptoms and sensory deficits. In the 6 previous years, 4 (17%) patients got a stroke recurrence. Other neurological (6, 26%), orthopedic (6, 26%), or not specified (8, 35%) disorders were diagnosed in 20 out of 23 (87%) patients overall.
At T4, 17 (74%) patients completed the SIS 2.0 questionnaire directly, and 6 (26%) caregivers completed the questionnaire due to severe cognitive impairment or poor patient cooperation. At the time of telephone contact, all patients were living in their homes. The study flow diagram is reported in Figure 1.

Study flow diagram.
The impact of stroke on short-term HRQoL
In the initial total sample (n = 39), an overall improvement was found in most SIS 2.0 domains between baseline (T0), after rehabilitation (T2), and the 6-month follow-up (T3). The SIS-16 composite physical domain showed more significant improvement than the other domains during assessments at multiple time points (see Figure 2a and b). Moreover, hand function and ADL still improved after the end of rehabilitation significantly (T3-T4).

Mean percentage scores versus time (T0, T1, T2, and T3) in (a) SIS-16; (b) other SIS 2.0 domains.
RM-ANOVA confirmed a significant main time effect (P < .001), except in the memory and thinking (P = .208), emotions (P = .231), and communication (P = .501) domains. Conversely, the interaction between treatment and time was never significant (P > .1).
The SIS-16 score obtained at T0 and the rehabilitation setting were positively associated with the score obtained at T2 and T3. These findings revealed how patients with higher level of HR-QoL at admission and those who received outpatient rehabilitation, reported higher levels of HR-QoL in the short-term.
In contrast, the presence of sensory deficits was negatively related to the SIS-16 score at T3. In the adjusted linear regression model, the SIS-16 score obtained at T0 was a potential positive predictor of the SIS-16 score at T2 (β0 = .654, P ⩽ .001) and T3 (β0 = .641, P ⩽ .001). The presence of sensory deficits (β0 = −15.7, P = .025) was a potential predictor of the SIS-16 score at T3. Finally, the rehabilitation setting was identified as a possible predictor of the SIS-16 score at T2 (β0 = 14.2, P = .024) and T3 (β0 = 13.9, P = .036; Table 2).
Analysis of variance for Stroke Impact Scale (SIS) domains.

Abbreviations: ADL, activities of daily living; IADL, instrumental activities of daily living; SIS-16, Stroke Impact Scale-16.
The impact of stroke on long-term HRQoL
Overall HRQoL decreased between T3 and T4 with a critical deterioration in the strength (P = .002), mobility (P ⩽ .001), and hand function (P = .037) domains (see Table 3).
Long-term HRQoL after stroke.

Abbreviations: ADL, activities of daily living; IADL, instrumental activities of daily living; SD, standard deviation; SIS, Stroke Impact Scale; T3, 6-month follow-up; T4: 6-year follow-up.
Similarly, as shown in Figure 3, the highest proportion of negative clinically meaningful changes were reported in the mobility (86%) and strength (62%) domains at the long-term follow-up. Conversely, the ADL/IADL domain showed mixed results, given that approximately 40% of the patients had a clinically significant deterioration in independence, whereas 29% had significant improvements at the long-term follow-up. For the hand function domain, more frequently, no changes were found between T3 and T4 (53%), and a deterioration was seen in almost 40% of the sample.

Percentage of negative, positive, and no changes in SIS-16 at long-term follow-up.
The potential influence of stroke severity, sex, and age on long-term HRQoL was tested. It has been shown how moderate stroke patients reported a higher self-reported impact of stroke in the strength (P = .027) and mobility (P = .006) domains than mild stroke patients. Similarly, men reported a higher deterioration in the memory and thinking (P = .012) and hand function (P = .015) domains than women. Furthermore, persons who had a stroke at an older age (⩾65 years) reported a higher impact of stroke in the mobility domain (P = .046; see Table 4).
Factor influencing long-term HRQoL after stroke.

Abbreviations: ADL, activities of daily living; IADL, instrumental activities of daily living; SD, standard deviation; SIS, Stroke Impact Scale.
Discussion
Through multiple assessments, this study aimed to address the variation in HRQoL over time in stroke patients who participated in a clinical trial within a subacute, multidisciplinary arm rehabilitation program. 28 These stroke patients usually have high, multidisciplinary rehabilitation needs and recovery potential, for which highly qualified rehabilitative health care resources are provided.
Our findings helped us to define how stroke specific HRQoL can be modified by rehabilitation (short-term variation) and how it changes after a prolonged period of chronicity (long-term variation). After stroke, HRQoL is often reduced due to motor and cognitive impairments and reduced functioning and participation in everyday life activities. 38 Understanding how rehabilitation can influence HRQoL and how it changes over time in patients who receive multidisciplinary rehabilitation is of interest for identifying rehabilitation needs and delivering appropriate interventions, including long-term management.
Our findings supported how after rehabilitation, an improvement in HRQoL occurs without any significant differences between different arm rehabilitation protocols, as previously reported. 39 Overall, it is assumed that rehabilitation is beneficial and contributes to reducing the impact of stroke on HRQoL in the subacute phase. The most significant variation was seen in the physical domains, such as mobility, hand function, and strength, as well as in independence in ADL/IADL and social participation, as previously reported. 40
In contrast, HRQoL in the memory and thinking, communication, and emotion domains remained almost constant. However, neuropsychological and language impairments are usually contraindications for enrolling patients in clinical trials.41,42 For this reason, these data must be considered cautiously and cannot be generalized to the overall stroke population.
In this study, the first short-term HRQoL predictor was the SIS-16 score at baseline. Thus, greater physical health perceived at rehabilitation admission is related to higher HRQoL in the first 6 months after stroke. Similarly, patients discharged to home who received outpatient rehabilitation had higher levels of HRQoL. This finding reflects how social and familiar environments can be beneficial for patients who can participate in an outpatient intensive multidisciplinary rehabilitation program. 43 However, other factors, such as stroke severity and medical comorbidities, that are usually less represented in stroke patients discharged at home, can have influenced HRQoL as well. Finally, the presence of somatosensory deficits seemed to influence short-term HRQoL, probably because they can affect motor skills, functional performance, and social participation in the short and long term. 44
At the long-term follow-up, we observed significant deterioration in the HRQoL domains related to physical functioning, such as mobility, strength, and hand function, consistent with previous studies.2,17 Therefore, in stroke survivors, the improvement in HRQoL achieved during rehabilitation is progressively reduced in the long term. The primary determinants of this HRQoL deterioration are learned nonuse phenomena and progressive deconditioning due to limited physical activity.45,46 Moreover, the aging of the stroke population and the addition of new medical conditions, such as recurrent stroke and other neurological or orthopedical conditions can influence the deterioration of HRQoL in this population.
In this framework, it is urgent to rethink the long-term management of stroke survivors, which needs effective and sustainable health care solutions to prevent these detrimental phenomena. 47
In our sample, long-term deterioration in HRQoL was influenced by stroke severity,48,49 male sex and older age,50,51 helping us to identify potential factors affecting HRQoL in stroke patients. In literature, older adults’ HRQoL seems to be related to health characteristics, comorbidities, and perceived social support52,53; all these aspects are strongly influenced by an event like a stroke, that undermines physical and psychological aspects of QoL.
Limitations
A significant limitation of this study is the small sample size and the high drop-out rate at the long-term follow-up. We screened 391 patients with a recruitment rate of 10% and enrolled 40 subacute stroke patients with arm paresis, similar to other upper limb stroke clinical trials.54,55 This fact limits the generalizability of our findings to the entire stroke population. Moreover, several confounding factors influencing HRQoL after stroke were not controlled, such as the new medical comorbidities (ie, recurrent stroke, neurological, or orthopedical conditions), the rehabilitation received in the long-term, family support, and other environmental factors.
Conclusions
In this secondary analysis of a randomized controlled trial on arm rehabilitation in subacute stroke survivors, we explored variations in HRQoL in the short- and long-term follow-ups with the SIS 2.0. We highlighted an overall improvement in physical HRQoL domains after rehabilitation and progressive deterioration in the long-term follow-up after stroke. Reduced mobility, strength, and hand function were reported more frequently. These findings suggest the importance of tailoring meaningful interventions in the subacute phase and even in a chronic stage when the recovery process has been completed but that nonuse and deconditioning can severely affect functioning and HRQoL. This study provides insight for future research dealing with the long-term assessment of HRQoL in stroke survivors and the identification of the main determinant after stroke in a cohort-based larger observational study.
Footnotes
Author Contributions
NL, NB, FM, SL, and SS conceived the study and participated in its design. GM, AB, and LM performed the clinical data collections. SS, GM, and AB analyzed the data. SS, GM, LM, and AB interpreted the results, and drafted and revised the manuscript. All authors approved the submitted version.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The clinical trial was supported by Emilia Romagna Region (Grant Number 1786/2012). The funding body had no role in data collection or interpretation. For 6-year follow up, the study received no specific funding.
Ethical Approval
The authors assert that all procedures contributing to this work comply with the ethical standards of the relevant national and institutional committees on human experimentation and with the Helsinki Declaration of 1975, as revised in 2008. The study was approved by the Ferrara Ethics Committee.
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Data Availability Statement
The data that support the findings of this study are available on request from the corresponding author.
