Abstract
Background
Diabetes mellitus prevalence has reached epidemic levels despite the existence of contemporary treatments. People thus started looking at the possible therapeutic value of natural therapies. Crushed shoot tips of Crinum abyssinicum (Amaryllidaceae) are mixed with water in Ethiopia to treat diabetes, yet this practice is not well supported by science.
Objective
In this experiment, mice models were used to verify the blood sugar and lipid-lowering benefits of solvent fractions of C. abyssinicum shoot tips.
Materials and Methods
In a single-dose treated Streptozotocin (STZ)-induced diabetic model, mice were randomly grouped into eleven categories which include diabetic negative control, diabetic positive control, and 9 diabetic treatment groups. In repeated daily doses treated STZ-induced model, Mice were divided into 6 groups which included normal and diabetic negative control (TW80), diabetic positive control (5 mg/kg glibenclamide), and three diabetic treatment groups 100, 200, and 400 mg/kg). Finally, blood glucose, lipid level, and body weight were examined.
Results
In the single-dose treated diabetic model, there was a significant blood glucose reduction at 200 and 400 mg/kg doses of aqueous fraction and glibenclamide starting from the sixth-hour post-administration unlike ethyl acetate and chloroform fraction compared to baseline and negative control. In repeated daily dose-treated diabetic mice, all three doses (100, 200, and 400 mg/kg of aqueous fraction) resulted in a substantial reduction (P < .001) in blood glucose compared to baseline and negative control on the seventh day and 14th day. Besides the AQF shows improvement in lipid levels and body weight parameters.
Conclusion
The results of the study demonstrated that C. abyssinicum shoot tip fractions have the greatest potential to lower blood sugar and lipid levels, supporting conventional claims for the treatment of diabetes.
Introduction
Diabetes mellitus (DM) is a condition in which the body produces insufficient insulin or does not use it properly, leading to abnormally high blood sugar (glucose) levels. 1 Long-term DM can have serious complications, some of which are fatal. It is linked to significant changes in cholesterol, triglycerides, and lipoprotein composition in the blood. Patients with diabetes are on the rise around the world, reaching epidemic proportions. As a result, it has an impact on people’s lives on a global scale in one or more ways. 2
When compared to conventional medication, herbal therapy has a long history of usage in the treatment and prevention of disorders, including DM. Up to four billion people, or 80% of the world's population, are thought to rely on herbal medicine to improve their fundamental health, generate income, and sustain their way of life.3-5
Medicinal herbs can create a variety of Phyto-constituents, or organic chemicals, that can be employed to perform critical biological tasks such as antioxidation and free radical scavenging with little toxicity or side effects. Due to their excellent antioxidant and pharmacological relevance, medicinal herbs are being extensively explored as a therapy technique of choice against diabetes. They also have anti-hyperglycaemic effects via increasing insulin production, decreasing glucose absorption in the gut, or restoring pancreatic tissue function, which allows for metabolite transfer in insulin-dependent activities.6,7
Crinum abyssinicum hochst. ex a. rich (Amaryllidaceae) is ingested orally for the treatment of diabetes in Ethiopia, according to ethnomedicinal surveys and qualitative investigation research.8-11 Additionally, hydro-alcoholic crude extract of C. abyssinicum shoot tips demonstrated anti-diabetic, anti-hyperlipidemic, and body weight recovery in diabetic mice. 12 This follow-up study was conducted on two different animal models (single and repeated dose STZ-induced diabetic mice models) to compare the results of the crude extract with those of the solvent fraction, determine which solvent fraction(s) has(have) the best anti-hyperlipidaemic and antidiabetic activity, and inspire the future isolation of the primary bioactive compounds from the studied plant materials. The current study’s findings will be used as a model for future research into the use of this experimental plant in Ethiopian traditional medicine for a variety of other ailments. Preliminary phytochemical experiments in laboratory animals are one of the first phases in the medication development process. As a result, the current study could be viewed as a critical step in developing a novel therapeutic agent that is inexpensive, effective, and has the fewest side effects feasible when compared to conventional therapy.
Materials and Methods
Chemical, Diagnostic Kits, Drugs, and Instruments Utilized
Chemical, diagnostic kits, drugs, instruments utilized at the time of investigation were Streptozotocin (Fisco Research laboratories), ethanol absolute (Nice Chemical, India), citric acid monohydrate (Lab Tech Chemicals, Mumbai, India), tri-sodium citrate di-hydrate (Blulux Laboratories, Faridaban, ethyl acetate (Blulux Laboratories, Faridaban, India), chloroform (BDH Chemical, Uk), glibenclamide (Julphar Pharmaceuticals, Ras Al Khaimah, UAE), halothane (Primal Enterprises, India), rotatory evaporator (Hamato, Japan) and desiccators, muslin, Whatman filter paper (number1), beakers, funnels, measuring cylinder, glass rods, lyophiliyzer (Labfreez, China), gavage (oral feeding syringe), Cages, syringe with needle, capillary tubes, electronic balance, Gluco meter with strips (careSens N, Korea), 40% glucose solution (Reyoung Pharmaceuticals, Shandong, China), distilled water, Digital electronic balance (EPH-400 Abron Exports), pH meter (Banteinstruments,UK), deep freezer (Labfreez Instrument Groups, Germany), Chemistry analyzer (Shenzhen Midray Biomedical Electronics Co., Ltd,China), hot air oven (Medit-MedizinTechnik,Germany).
Plant Sample Collections and Deposition of a Voucher Specimen
The fresh shoot tips of C. abyssinicum were collected at Sinan, East Gojjam Zone, Amhara Reginal State in December 2019. Botanical identification and authentication were done by a botanist (Mr.Abiyu Enyew) at the Department of Biology, College of Natural and Computational Science, University of Gondar, Ethiopia. Then, a voucher specimen (001BAT/2019) was deposited in the herbarium of the University (Figure 1). Crinum abyssinicum hochst. ex a. rich shoot tips (Photo By Bantayehu Addis).
Solvent Fractionation Protocol
Selecting the appropriate solvent is crucial for solvent extraction and fractionation processes. Selectivity, solubility, cost, and safety were all considered when selecting a solvent for this purpose of fractionation. Other considerations included market availability, solvents previously used for extraction or fractionation on different parts of this material, the patient's traditional mode of use, and polarity order (the least polar to water with the highest polarity).
13
To separate the chloroform, ethyl acetate, and aqueous fraction, sequential solvent partitioning under increasing solvent polarity index was carried out after the hydro-alcoholic crude extract of C. abyssinicum shoot tips was prepared (Figure 2). In a separatory funnel, a defined amount of dried crude extract was soaked in distilled water to form a suspension. Chloroform was then added to the aqueous layer and gently shaken. The mixture was left to sit for 2 h until two distinct layers had developed. The procedure was repeated three times, with the lowest chloroform layer being separated each time. The ethyl acetate fraction was then obtained by three sequential separations of the aqueous residue in the same manner. The water-based mixture (aqueous fractions) was gathered as the third fraction. The ethyl acetate and chloroform fractions were concentrated in a hot air oven set at 40°C. The aqueous fraction was frozen (−2°C) overnight in the refrigerator before being dried in a lyophilizer. The dried fractions' percentage yield was calculated. The fractions were kept in sealed bottles in a refrigerator at a temperature of −4°C until they were used.
14
A schematic representation of the Solvent Fractionation Process.
Experimental Animals
Swiss albino male mice weighing 25-30 g (8-12 weeks) bred in the animal house of the Department of Pharmacology, School of Pharmacy, College of Medicine and Health Sciences, University of Gondar were used in the investigation. Six mice were housed in a cage with a 12 h light/12 h dark cycle, and they had unrestricted access to normal pellets and water. They were also handled as per the international criteria for the use and maintenance of experimental animals. The mice were given a week to acclimate to laboratory surroundings. At the end of experiment, all mice were sacrificed via cervical dislocation and then buried in the college's waste disposal area at the end of each experiment. 15
Ethical Consideration
Before the study began, the ethical review committee of the University of Gondar's Department of Pharmacology, School of Pharmacy, College of Medicine and Health Sciences reviewed and approved the study proposal with reference number Sop/285/12 on March 16, 2020. Moreover, all protocols were performed based on the International Animal Care and Welfare Guidelines (the OECD guidelines for testing of chemicals). 16
Experimental Design and Dosing of Animals
Figure 3 below shows the animal dosage and experimental design. In a single dose, STZ-induced diabetic animal model, there were 11 different groups (six mice/group), namely: the diabetic negative control group (group I) which was treated with vehicle (2% Tween 80); the diabetic positive control group (group II) which was treated with standard drug (GLC5 mg/kg); The chloroform, ethyl acetate, and aqueous fractions of the shoot tips of C. abyssinicum were administered at three different dose levels to nine diabetic test groups (group III-XI). Dosage selection was based on acute toxicity studies. A lower dose level of 100 mg/kg, or half of the middle, a middle dose level of 200 mg/kg, or one-tenth of the amount used for acute toxicity (2000 mg/kg of body weight), and a higher dose level of 400 mg/kg, or twice the middle dose, were chosen.
17
A schematic representation of the animal grouping and dosing.
Mice were fasted overnight for 14 hours and divided into six groups in the STZ-induced experiment with repeated daily doses. Group I: diabetic negative control treated with 2% tween 80; Group II diabetic positive control treated with the reference drug (GLC5mg/kg); Group III-V diabetic mice treated with chloroform, ethyl acetate, and aqueous fractions (100, 200, and 400 mg/kg, respectively); Group VI normal/non-diabetic negative control treated with 2% tween 80.
Measurement of Experimental Parameters
Blood samples were taken aseptically by snipping the tip of the tail in all animal models to determine the blood glucose level (BGL). BGL was measured with an electronic glucometer and test strips. The average values were taken into account after triplicate measurement of fasting BGL. Moreover, the measurement of serum lipids using a chemistry analyzer and body weight using an electronic balance was carried out. 18
Induction of Experimental Diabetes
To induce diabetes, a newly prepared solution of STZ dissolved in .1 M cold citrate buffer with pH = 4.5 was used. To determine the best dose range, pre-tests were performed at 60, 90, and 120 mg/kg body weight. Depending on the results of the pre-test, a single dose of 150 mg/kg of the newly generated solution was administered intraperitoneally to overnight fasted (14 h) mice. The animals were given full access to food and water 30 minutes after receiving STZ. To prevent hypoglycemia shock caused by hyperinsulinemia, the animals were given a 5% glucose solution for 24 h. After 72 h of STZ administration, the animals were examined for diabetes induction. Mice with a fasting BGL of >200 mg/dL were recruited for the study.19,20
The Anti-Hyperglycaemic Activity of Single Doses of Solvent Fractions in Streptozotocin-Induced Diabetic Mice
After an overnight fast (14 h), STZ-induced diabetic mice were randomly assigned to 11 separate groups (six mice/group), and mice were then given 2% TW 80, GLC5 mg/kg, and solvent fractions of C. abyssinicum according to their respective groups, as mentioned above. BGL was measured before therapy (at 0 h) and then again at 2, 4, 6, and 8 h after treatment. 21
The Anti-Hyperglycaemic Activity and Effect on Body Weight of Repeated Doses of Fractions in Stz-Induced Diabetic Mice
The experimental animals were separated into six groups after effectively establishing STZ-induced diabetes (6 mice/group). As per the protocol, the vehicle, standard medication, and solvent fractions were given once daily for 14 days. The fast BGL and body weights were measured on day 0, the seventh day, and the 14th day.22,23
The Effects of Solvent Fractions on Serum Lipid Level of STZ-Induced-Diabetic Mice
On day 15, blood was drawn from 14 h fasted diabetic mice using a sterile tube after cardiac puncture under halothane anesthesia. After two hours at room temperature, the blood samples were centrifuged at 2500 rpm at 30°C for 15 minutes. The supernatant was extracted immediately from the pellet to create serum samples for automated chemistry analyzer analysis of serum triglyceride (STG), serum total cholesterol (STC), high-density lipoprotein (HDL), and low-density lipoprotein (LDL). 12
Statistical Analysis
Statistical software for Social Science (SPSS) version 23 was used to conduct all statistical analyses. For six mice in each group, the data were reported as mean ± standard error of the mean (S.E.M.). One-way analysis of variance (ANOVA) and Tukey's post hoc multiple comparison tests were used to compare the means of all parameters across groups and within each group. Statistical significance was defined as a P-value <.05.
Result
The Comparison of the Yields of the Solvent Fraction of Crude Extract.

Blood Glucose Lowering Activity of Single Doses of Solvent Fractions in STZ-Induced Diabetic Mice
Blood Glucose Lowering Activity of Single Doses of Solvent Fractions in Streptozotocin-Induced Diabetic Mice.
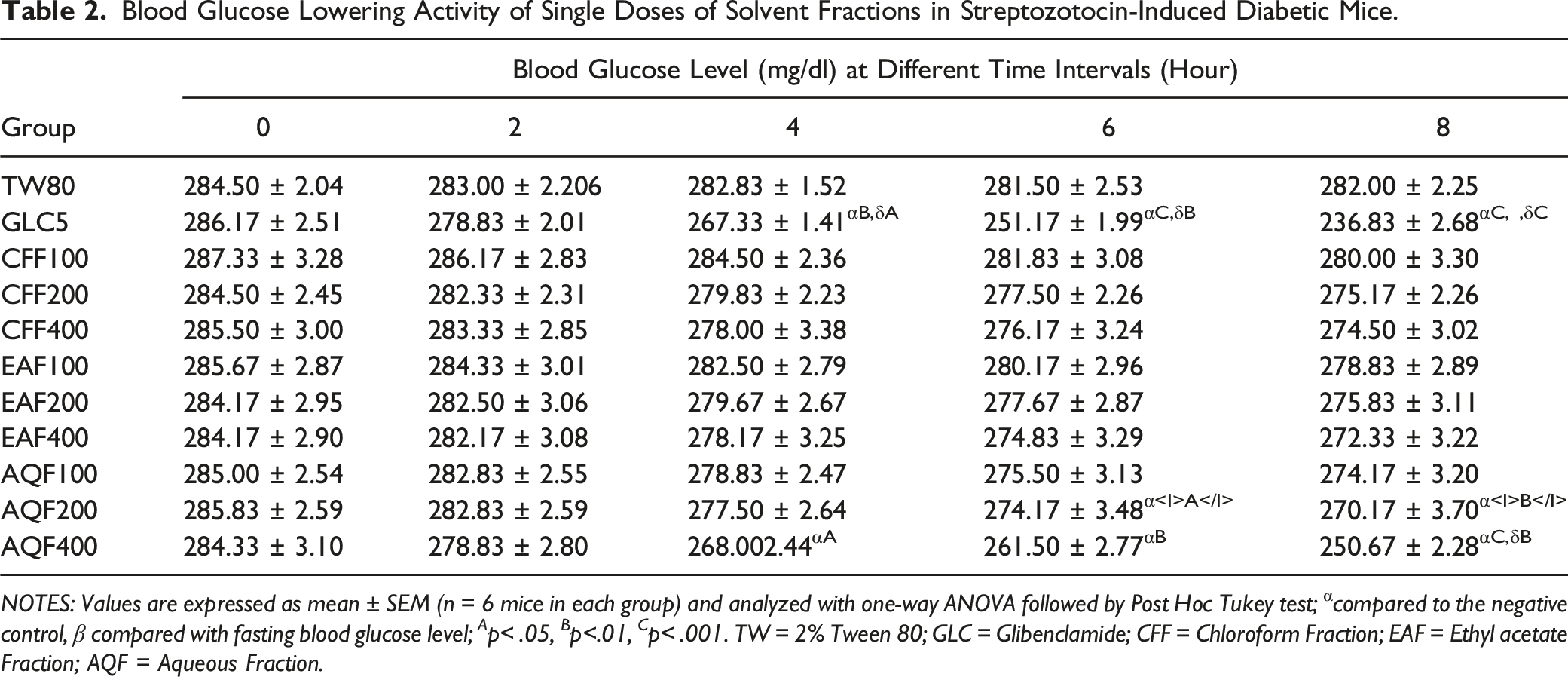
NOTES: Values are expressed as mean ± SEM (n = 6 mice in each group) and analyzed with one-way ANOVA followed by Post Hoc Tukey test; α compared to the negative control, β compared with fasting blood glucose level; A p< .05, B p<.01, C p< .001. TW = 2% Tween 80; GLC = Glibenclamide; CFF = Chloroform Fraction; EAF = Ethyl acetate Fraction; AQF = Aqueous Fraction.
Blood Glucose Lowering Activities of Repeated Daily Doses of the Aqueous Fraction on STZ-Induced Diabetic Mice
Blood Glucose Lowering Activities of Repeated Daily Doses of the Aqueous Fraction on Streptozotocin-Induced Diabetic Mice.
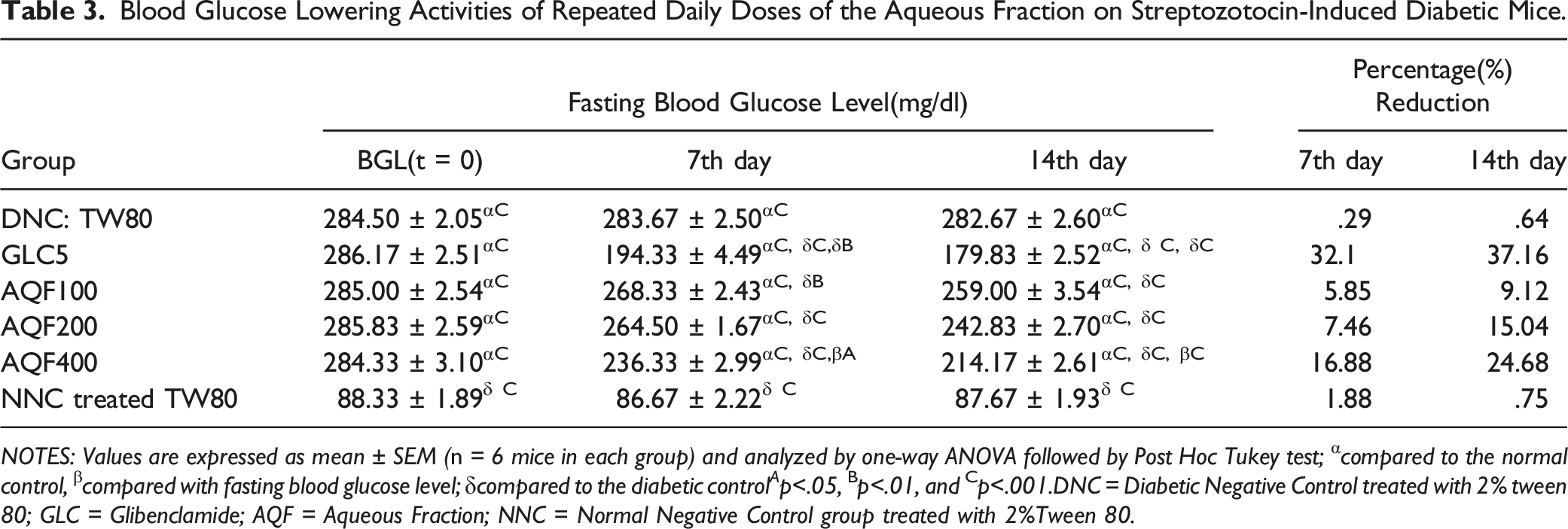
NOTES: Values are expressed as mean ± SEM (n = 6 mice in each group) and analyzed by one-way ANOVA followed by Post Hoc Tukey test; αcompared to the normal control, βcompared with fasting blood glucose level; δcompared to the diabetic control A p<.05, Bp<.01, and Cp<.001.DNC = Diabetic Negative Control treated with 2% tween 80; GLC = Glibenclamide; AQF = Aqueous Fraction; NNC = Normal Negative Control group treated with 2%Tween 80.
The Effect of the Repeated Daily Doses of Aqueous Fraction on the Body Weight of Diabetic Mice
The Effect of the Repeated Daily Doses of Aqueous Fraction on the Body Weight of Streptozotocin-Induced Diabetic Mice.

NOTES: Values are expressed as mean ± SEM (n = 6 mice in each group) and analyzed by one-way ANOVA followed by Post Hoc Tukey test;, α compared to the normal control, β compared to baseline body weight; δ compared to the diabetic negative control A p<.05, B p<.01, and C p<.001. DNC = Diabetic Negative Controltreated with 2%Tween 80; GLC = Glibenclamide; AQF = Aqueous Fraction; NNC = Normal Negative Control treated with 2%Tween 80.
Lipid Activity of Repeated Daily Doses of Solvent Fractions in STZ-Induced Diabetic Mice
Lipid Activity of Repeated Daily Doses of Solvent Fractions in Strptozotocin-Induced Diabetic Mice.

NOTES: Values are expressed as mean ± SEM (n = 6 mice in each group) and analyzed by one-way ANOVA followed by Post Hoc Tukey test; α compared to the normal control; β compared to the diabetic negative control A p<.05, B p<.01, and C p<.001. DNC = Diabetic Negative Control 2%; GLC = Glibenclamide; AQF = Aqueous Fraction; NNC = Normal Negative Control treated with 2% tween 80.
The contents of STG were considerably lower (P < .05) with AQF100 mg/kg, and significantly higher (P < .001) with AQF 200 mg/kg, AQF 400 mg/kg, and GLC 5 mg/kg compared to diabetes control by the end of the experiment. When compared to diabetic control, AQF 200 mg/kg (P < .01), AQF 400 mg/kg (P < .001), and GLC 5 mg/kg (P < .001) significantly enhanced HDL content after 14 days of medication. AQF 100 mg/kg, on the other hand, considerably reduced LDL cholesterol content (P < .01) in a dose-dependent manner, whereas AQF 200 mg/kg and AQF 400 mg/kg significantly reduced LDL cholesterol content (P < .001).
Discussion
Despite the introduction of various anti-diabetic medications, diabetes has become a global public health issue. 24 Because STZ-induced diabetic mouse models closely mirror human diabetes, they are frequently used to investigate anti-diabetic properties in mice in vivo. Normally, when STZ enters pancreatic beta-cells via glucose transporter type 2 (GLUT2), it divides into glucose and methyl nitrosourea, which possesses deoxyribonucleic acid (DNA) alkylating characteristics, causing damage to Langerhans β-cells, a decrease in endogenous insulin production, and eventually T2DM. 25 Streptozotocin (i.p.) at a dose of 150 mg/kg body weight successfully (73.5%) developed DM in physiologically normal mice with elevated blood glucose levels in this study. The current study was designed to investigate the antidiabetic and anti-hyperlipidemia activities of solvent fractions of C. abyssinicum on normal and diabetic mice, based on the concept that plants have always been an excellent source of drugs for diabetes.
Medicinal plants are thought to have potent hypoglycaemic, anti-hyperglycaemic, and glucose-suppressive effects by inhibiting glucose absorption from the intestines, increasing pancreatic insulin secretion, increasing glucose uptake by adipose and muscle tissues, or inhibiting glucose production from hepatocytes. 26 Furthermore, natural plant-based antioxidants are particularly effective in preventing cell-damaging processes produced by oxidative stress (OS). Polyphenols, which are one of the primary classes of phytochemical ingredients characterized by the presence of one (phenolic acids) or more than one (flavonoids) phenol ring in their chemical structure, as well as alkaloids, could achieve these effects. Flavonoids are a type of antioxidant that has been shown to help with DM and inflammation management. They inhibit enzymes implicated in the propagation of Type 2 diabetes mellitus like α-amylase, α-glycosidase, and dipeptidyl peptidase-IV. Polyphenol compounds work as singlet and triplet oxygen quenchers, metal chelators, reducing agents, and hydrogen donors through an oxidation-reduction process. Furthermore, phenolic substances have been shown to efficiently control polyol enzymes involved in diabetic complications.27,28 The early phytochemical screening of a hydro-alcoholic extract of C. abyssinicum shoot tips revealed that phytochemical elements such as saponins, glycosides, cardiac glycosides, anthraquinones, alkaloids, flavonoids, phenols, tannins, terpenoids are abundant. 12 A substantial reduction in fasting BGL of all diabetic mice administered an aqueous fraction of C. abyssinicum and reference medication was seen when compared to diabetic control after a single dose of the STZ-generated diabetes model. When compared to the remainder fractions, the aqueous fraction had the highest activity. It's exciting to see that AQF400 mg/kg reduced hyper-glycemia caused by STZ by 27.04% as an equivalent dose of both CFF and EAF fractions exhibited poor activity (3.85% and 4.20%, respectively), indicating that the plants' active constituent(s) are polar. This is not in agreement with previous similar studies. 29 The antidiabetic activity correlates to GLC 5 mg/kg, as shown in Table 2, and all doses of the solvent fractions reduced blood glucose levels dose-dependently following oral treatment. This could be explained by the existence of significant concentrations of key phytochemical constituents(s) in the higher dose of 400 mg/kg of plant fraction, as opposed to the lower 100 mg/kg and medium 200 mg/kg dose levels. The anti-hyperglycaemic activity of the aqueous fraction was superior to the anti-hyperglycaemic activity of other solvent fractions in a single-dose STZ-induced mice model. This could indicate that the AQF has far more bioactive elements than CFF and EAF, both in terms of kinds and concentration. Because AQF reduces hyper-glycemia caused by STZ induction in mice more effectively, it is logical and rational to pursue additional research into its in vivo anti-hyperglycemic action in STZ-induced diabetic mice. Nardose et al. (2010) found that the aqueous fraction exhibited significant glucose-lowering efficacy, which was in keeping with this finding. 30
It appears that the glucose-suppressive and anti-hyperglycaemic activity of C. abyssinicum solvent fractions may have been activated by these secondary metabolites discovered in C. abyssinicum solvent fractions. The anti-hyperglycaemic effect of the fraction could be due to individual or synergistic effects of the phytochemical constituents (secondary metabolites), resulting in stimulation of insulin release from survived pancreatic beta-cells or insulin-mimetic effects, which stimulate glucose utilization by diabetic mice's hepatic and extra-hepatic tissues. Furthermore, the fraction may decrease glucose absorption in the gut while also enhancing the function of glyconeogenic and glycolytic enzymes.
In the anti-hyperglycaemic activity of repeated daily doses treated diabetic mice model, the highest reduction in fasting blood glucose was recorded with AQF 400 (34.68%) on the 14th day, similar to the single dose-treated diabetic model (Table 3). The comparison medicine reduced fasting BGL by 37.16% on the same day. The current result is consistent with earlier studies. 31 Although AQF 100, AQF 200, and AQF 400 all showed significant blood glucose reductions (P < .001), the size of the reductions (20.04%, 23.51%, and 34.68%, respectively) as compared to diabetic control on the 14th day varied. The fraction had a dose-dependent effect, which could be because the greater dose is more likely to have a higher concentration of the bio-active ingredients that are responsible for decreasing fasting BGL than the smaller dose. GLC 5 mg/kg pulls down fasting BGL as a result of plasma membrane depolarization, which is caused by selective blockage of adenosine triphosphate(ATP) sensitive K+(KATP) channels, activating voltage-gated Ca2+channels, cytosolic(Ca2+), and endogenous insulin secretion from pancreatic beta-cells. 32 This could indicate that STZ at a dose of 150 mg/kg intra-peritoneal was insufficient to destroy whole pancreatic beta-cells and/or that only a few cells survived to regenerate and release insulin. STZ absorption into pancreatic β-cells islets causes toxicity through a variety of pathways, including nitric oxide(NO) donation and free radical formation, resulting in a significant reduction in the intracellular insulin concentration of these cells. 33 Because of their antioxidant properties, several plant fraction phytochemical constituent(s) have been demonstrated to protect pancreas cells. C. abyssinicum fractions were found to have similar antioxidant activity and a protective effect on cells in this investigation. In other words, in STZ-induced diabetic mice, the plant extract may have an anti-hyperglycemic effect.
Because body weight is the best measure of excellent health and efficient metabolic balance, STZ-induced diabetic mice were given a daily dose of hydro-alcoholic shoot tips fraction of C. abyssinicum (Table 4). Throughout the investigation, the body weight of normal control mice gradually increased. In contrast, increasing the period of treatment resulted in a considerable reduction in the body weight of untreated diabetic mice. This conclusion is consistent with a previous study by Jayaprasad et al. (2016), who found that STZ-induced diabetic mice similarly lost body weight. In STZ-induced diabetic control mice, significant body weight loss (P < .001) was detected. Dehydration, protein breakdown, and muscle withering due to the lack of carbs as an energy source and fat catabolism are thought to be the causes of body weight loss in diabetic mice. 34 These data revealed that after 14 days of therapy, GLC 5 mg/kg and all doses of fraction-treated mice gained considerable weight (P < .001, P < .01) in comparison to the diabetes control group and baseline body weight, respectively. The fraction’s ability to reduce hyperglycemia may be linked to its beneficial effect on body weight loss. In this study, the phytochemical contents of C. abyssinicum may help by suppressing free radicals produced by hyperglycemia, boosting glucose consumption, preventing muscle wasting, and sparing protein catabolism in fraction-treated diabetic mice, resulting in body weight gain.
Increased levels of STC, STG, LDL, and HDL, as well as a decrease in HDL, are frequently related to diabetes, which leads to the development of cardiovascular illnesses. The accelerated breakdown of lipid and free fatty acids from peripheral deposits in insulin insufficiency is recognized as a consequence of diabetes mellitus. Hyperglycemia is also associated with higher levels of STC, STG, and LDL in lipid profiles, as well as lower levels of HDL.35,36 The present study consistently found a positive correlation between hyperglycemia and hyperlipidemia. This explains why the levels of STC, STG, and LDL in diabetic mice treated without treatment were significantly greater than the levels of HDL. Compared to the treatment groups, STZ-induced diabetic control mice exhibited significantly greater STC, STG, and LDL content as well as decreased HDL. When compared to a diabetic negative control treated with tween 80, repeated treatment with AQF at three dose levels for 14 days significantly decreased STC, STG, and LDL content, but increased HDL content in a dose-dependent manner (Table 5). This is in line with STZ-induced diabetic rats, Centratherum anthelminticum ethanolic seeds extract exhibited a significant reduction in BGL, STC, STG, and LDL, as well as an increase in HDL. 37 However, it is unclear whether the fractions from the shoot tips were involved directly in lipid metabolism or whether the anti-hyperlipidemic effect was produced only as a result of controlled hyperglycemia.
Conclusion
According to these results, the aqueous fraction from the shoot tips of C. abyssinicum significantly reduced hyperlipidemia in diabetic mice and had significant antidiabetic effects. Although species diversity may limit the results' application to people, this investigation supported the traditional usage of C. abyssinicum in the treatment of diabetes. More research is necessary to determine how the plant material restores normalcy to blood glucose and lipid levels, as well as whether injection of the C. abyssinicum shoot tips fraction results in insulin secretion or sensitization. More study on histopathology and important biomarkers is needed to accurately pinpoint the mechanism(s) by which solvent fractions administered once or more to mice with STZ-induced diabetes are anti-hyperlipidemia and anti-diabetic.
Footnotes
Acknowledgments
The authors would like to acknowledge the University of Gondar for material support and for allowing them to use the laboratory facility.
Author Contributions
All authors made a significant contribution to the work reported, whether that is in the conception, study design, execution, acquisition of data, analysis, and interpretation, or all these areas; took part in drafting, revising, or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Data Sharing Statement
The manuscript contains all pertinent facts. On request, the data used to support the findings of this study can be obtained from the corresponding author.
