Abstract
Cellulosic ethanol, a biofuel product derived from microbial fermentation of cellulose-rich agricultural waste biomass, holds the potential to significantly contribute to sustainable development, energy security, and environmental friendliness as a green and renewable alternative energy source. Cellulosic ethanol offers remarkable energy benefits and carbon emission reductions. However, the biorefining process of cellulosic ethanol faces challenges and complexities. This article revolves around five aspects: feedstock, composition, pretreatment, cellulase hydrolysis, and ethanol fermentation processes in the biorefining of cellulosic ethanol. It provides an overview of the process flow and characteristics of cellulosic ethanol biorefining, emphasizing its significance. The main technological bottlenecks in cellulosic ethanol production are analyzed. Recent research advancements in cellulosic ethanol biorefining are summarized, followed by an outlook on the research focus and future prospects in the field of cellulosic ethanol.
Introduction
Bioethanol, a renewable energy source produced from biomass, was already in widespread use in Europe and the United States as early as the early 1900s, making it not a novel energy option. However, due to its higher production costs compared to gasoline, it was largely overlooked. It was only after the first oil crisis of the 1970s that people began to pay attention to bioethanol production (Ji et al., 2012).
In the chemical industry, bioethanol serves as a solvent and a raw material for synthesizing various organic compounds, such as ethylene, polyethylene, 1,3-butadiene, and ethyl acetate (Azadi et al., 2012). Additionally, bioethanol is a renewable and sustainable liquid fuel that can be used either independently or blended with gasoline to form ethanol-gasoline as automotive fuel. The incorporation of ethanol into gasoline serves two purposes: first, ethanol's high-octane rating of up to 120 allows it to replace lead additives that pollute the environment, thereby improving the anti-knock properties of gasoline (Dirrenberger et al., 2014); second, ethanol's high oxygen content enhances combustion, reducing incomplete combustion pollutants such as carbon deposits and carbon monoxide emissions within the engine. Comparatively, bioethanol-gasoline, on a volumetric basis, has approximately 30% lower calorific value than pure gasoline. However, since only a 10% blend is typically used, the reduction in calorific value is not significant, and no engine modifications are required for its utilization (Hsieh et al., 2002; Mohammed et al., 2021).
Bioethanol can be derived from the fermentation of sugars extracted from cellulose and hemicellulose. Based on the different feedstocks used for preparation, bioethanol can be categorized into first-generation bioethanol, produced from sugary and starchy crops, second-generation bioethanol, obtained from lignocellulosic biomass such as wood fibers, and third- and fourth-generation bioethanol, primarily sourced from aquatic algae.
First-generation bioethanol production technology is mature and exhibits high efficiency and yield (Bertrand et al., 2016). Second-generation bioethanol, utilizing lignocellulosic biomass, benefits from a wide range of feedstock sources, higher yields, and avoids competition with human food resources. However, wood-derived cellulose ethanol has been identified to contain 29 types of impurities, including high concentrations of acetic acid, acetaldehyde, methanol, and furan-related compounds. In contrast, first-generation bioethanol derived from sugars or starches contains only 16 types of impurities (Habe et al., 2013). Third and fourth-generation bioethanol rely on aquatic algae as the primary feedstock, such as microalgae and macroalgae, with the fourth generation utilizing genetically modified algae with high lipid content. These feedstocks contain lipids, proteins, carbohydrates (polysaccharides), and possess thin cellulose walls, making them favorable for bioethanol production. However, the pretreatment costs for algae are comparatively higher than for lignocellulosic biomass (Anto et al., 2020; Baeyens et al., 2015).
In addition to producing feedstock for bioethanol, a number of researchers have also focused on improving the production process (Li et al., 2018). Bifunctional solid catalysts enable efficient, low-energy, and environmentally-friendly one-pot transformations of biomass into biofuels and related chemicals, reducing or eliminating purification steps (Li et al., 2016; Wu et al., 2021). Therefore, discussing the current development of second-generation bioethanol is of great significance for the improvement research and industrialization of bioethanol.
Raw materials
The disadvantages of bioethanol include its low energy density, strong corrosiveness, lower flame luminosity, low vapor pressure (making cold starts difficult), and poor miscibility with water (Balat et al., 2008). One major issue with bioethanol production is the availability of feedstock.
First-generation bioethanol is produced from two types of food crops: sugary and starchy. Sugary feedstocks include sugarcane, sugar beets, and sweet sorghum, while starchy feedstocks primarily comprise wheat, corn, barley, cassava, potatoes, sweet potatoes, and yams (Phuong et al., 2017). Sugary feedstocks can be directly fermented to produce bioethanol, while starchy feedstocks need to be hydrolyzed into sugars before undergoing fermentation to produce bioethanol, as shown in Figure 1 (Ren et al., 2018). However, this practice leads to a reduction in food supply and is not conducive to mitigating global carbon emissions and greenhouse effects (Ayodele et al., 2020).

The first-generation bioethanol production process (Ren et al., 2018).
From the perspective of feedstock availability, the second-generation bioethanol, represented by lignocellulosic biomass, particularly agricultural waste, holds tremendous potential. It is rich in monosaccharide components such as glucose, xylose, and arabinose, which can be utilized and converted into ethanol by microorganisms (Ganguly et al., 2020; Toor et al., 2020). These feedstocks include corn stover, wheat straw, rice straw, corn cobs, bagasse, palm empty fruit bunches, distillers’ grains, and agricultural processing residues (Fitria et al., 2019; Mikulski et al., 2019; Ntimbani et al., 2021; Zheng et al., 2019; Zhu et al., 2019). Utilizing these materials to produce cellulose ethanol can enhance the comprehensive benefits of waste resource utilization and energy production while contributing to carbon emission reduction (Li and Chen, 2020; Yang et al., 2019).
However, the production cost of cellulose ethanol is twice that of fossil fuels. The preparation process involves pretreatment, hydrolysis, fermentation, and distillation (Peng et al., 2021), as depicted in Figure 2.
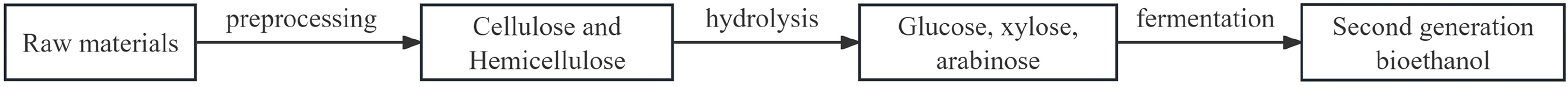
Second generation bioethanol production process (Peng et al., 2021).
The feedstock for third-generation bioethanol is algae, which can be categorized into microalgae and macroalgae. Macroalgae, due to their high polysaccharide and protein content, The feedstock for third-generation bioethanol is algae, which can be categorized into microalgae and macroalgae. Macroalgae, due to their high polysaccharide and protein content, are primarily used for human consumption. Microalgae are particularly suitable for bioethanol production because of their high carbohydrate and lipid content, and they exhibit rapid growth under biological or non-biological stress conditions. Utilizing carbohydrates and lipids from algae as raw materials requires a pretreatment process. The primary objective of pretreatment is to facilitate the extraction or chemical conversion of compounds within the cells, such as proteins, carbohydrates, lipids, oligosaccharides, pigments, etc (Anto et al., 2020). The specific preparation process for third-generation bioethanol is illustrated in Figure 3.

The third generation bioethanol production process (Anto et al., 2020).
Currently, the most promising and commercially viable option is the second-generation bioethanol derived from lignocellulosic biomass. It provides an opportunity to replace fossil fuels with cleaner, low-carbon biofuels. By combining the production processes of second-generation bioethanol with established first-generation bioethanol facilities, and leveraging synergistic effects, it is possible to maximize the integration of process technologies and enhance both economic and environmental benefits. This approach allows for the utilization of the advantages of both production methods, promoting a more sustainable and efficient bioethanol production industry (Mutenure et al., 2018).
Chemical composition
The ability of biomass to be compressed and molded into fuel at relatively low temperatures is attributed to its unique chemical properties. Biomass is primarily composed of three high molecular weight compounds: cellulose, hemicellulose, and lignin, which together form the cell walls and intercellular layers of biomass. The chemical composition of common biomass feedstocks is presented in Table 1.
Chemical composition of common biomass feedstocks.

As observed from Table 1, the relative proportions of these major chemical constituents in biomass vary depending on the specific type of biomass (Ayodele et al., 2020; Mani et al., 2006; Okwudili et al., 2021; Vassilev et al., 2012; Zabed et al., 2017). Therefore, the chemical composition of biomass, as well as the patterns of its changes and the mechanisms involved during the shaping process, form the foundation for understanding the quality-enhancing and modification effects of pretreatment processes on biomass-based solid fuels.
Cellulose
Cellulose is a linear high molecular weight compound composed of D-glucose units linked together through β-1,4-glycosidic bonds, with a molecular formula of (C6H10O5)n. Each glucose unit in cellulose contains three active hydroxyl groups (-OH), which can form hydrogen bonds between -OH groups or with O-, N-, and S- groups, exhibiting bond energy stronger than van der Waals forces (Xun et al., 2007). Within the cell wall, cellulose arranges into orderly microfibril bundles through molecular chains. During the compression process, the interconnected microfibrils formed by hydrogen bonds act as a structural framework within the cohesive matrix, contributing to the enhancement of strength in the formed fuel. Furthermore, cellulose possesses both extensive non-crystalline and crystalline regions, accompanied by hydrogen bonding, leading to a highly robust crystal structure (Haibin et al., 2012).
Hemicellulose
Hemicellulose and cellulose both fall under the category of carbohydrates. However, in contrast to cellulose, hemicellulose is a heterogeneous polysaccharide composed of two or more types of monosaccharides. Due to its chemical structural heterogeneity, natural hemicellulose is in an amorphous state and consists of relatively low molecular weight, branched polymers, with a degree of polymerization ranging from 80 to 100 (Lawther et al., 1995). Hemicellulose possesses an amorphous structure, making it susceptible to hydrolysis, and its structural strength is lower compared to cellulose. Hemicellulose has a complex structure; it connects with cellulose through hydrogen bonds, forms covalent bonds (primarily α-aryl ether bonds) with lignin, and establishes ester bonds, acetyl groups, and hydroxyl groups linked to cinnamic acid. Hemicellulose infiltrates the cellulose “skeleton” in an amorphous state, enhancing the rigidity of the cell wall. It is referred to as the matrix material. Hemicellulose's main chain and side chains contain numerous hydrophilic groups like hydroxyl and carboxyl groups. It is a hygroscopic component in biomass. Under the combined action of pressure and hydrolysis, hemicellulose can transform into lignin, thus serving as a certain adhesive agent (Haibin et al., 2012).
Lignin
Lignin is a complex, amorphous, non-crystalline polymer with a three-dimensional network structure. It consists of guaiacyl (G), syringyl (S), and p-hydroxyphenyl (H) structural units (Pu et al., 2013). Lignin is formed in the final stages of cell differentiation and infiltrates the cell wall's framework, contributing to its hardness. It is often referred to as the matrix material or rigid material.
Under natural conditions, lignin is almost insoluble in water and organic solvents. It softens at 100 °C and forms a colloidal substance that starts melting around 160 °C (Sun et al., 2015). During the compression process of biomass, under the combined influence of pressure and moisture, the large lignin molecules become fragmented, leading to condensation and degradation. This results in significant changes in solubility properties, generating soluble lignin and insoluble lignin.
Moreover, the presence of phenolic and alcoholic hydroxyl groups facilitates the dissolution of alkali lignin. Lignin sulfonates dissolved in water can form colloidal solutions, acting as adhesives, which enhance the binding strength and durability of formed fuels (Haibin et al., 2012).
Preprocessing
The primary challenge in producing bioethanol from lignocellulosic biomass lies in feedstock pretreatment. Prior to second-generation bioethanol production, effective pretreatment of lignocellulosic biomass is crucial. The schematic representation of biomass cell wall structure before and after pretreatment is depicted in Figure 4.

Schematic diagram of biomass cell wall structure before and after pretreatment (Kumar et al., 2009).
As shown in Figure 4, the three major components of biomass cell walls (cellulose, hemicellulose, and lignin) are closely interconnected, forming a physical and chemical barrier against degradation. This results in the lower mechanical strength and energy density of biomass-formed fuels. These characteristics significantly hinder the further enhancement of biomass-formed fuel quality (Kumar et al., 2009). Therefore, it is necessary to employ suitable pretreatment techniques to break down this barrier, modify the structure and composition of biomass, and enhance the quality of biomass-formed fuels.
Similar to other lignocellulosic biomasses, the direct enzymatic hydrolysis using cellulase and hemicellulase is hindered by the complex structure and crystallinity of lignocellulose. Therefore, to achieve complete conversion of hemicellulose and cellulose into monosaccharides, physical, chemical, and enzymatic hydrolysis pretreatments are necessary (Nie et al., 2019). The purpose of pretreatment is to disrupt the cellulose crystalline regions and the 3D structure of lignin, minimizing sugar losses, enhancing enzyme conversion efficiency, and reducing inhibition of enzymes or microorganisms (Kazemi Shariat Panahi et al., 2020). A successful pretreatment process should be versatile across various feedstocks, utilize cost-effective and recyclable reagents, and exhibit characteristics such as low energy consumption, minimal investment, and low maintenance costs. Biomass pretreatment techniques can be categorized into four main classes: physical methods, physico-chemical methods, chemical methods, and biological methods. Examples include mechanical milling, acid treatment, alkali treatment, microbial treatment, microwave treatment, steam explosion treatment, low-temperature pyrolysis treatment, and hydrothermal treatment, among others (Hassan et al., 2018).
Physical pretreatment
Physical methods aim to treat lignocellulose using non-chemical means, utilizing forces or other forms of energy to disrupt the structure of lignocellulose. This category includes mechanical methods, hydrothermal methods, and steam explosion methods (Sankaran et al., 2020).
Mechanical methods involve breaking down materials with high cellulose content using machinery to increase surface area and enhance enzymatic hydrolysis (Ma et al., 2020). Qu et al. employed a vibration ball mill with zirconia as the filling medium for dry ball milling of corn stover, followed by pretreatment using NaOH solution. After enzymatic hydrolysis for over 72 hours, the glucose conversion rate reached 100% (Qu et al., 2017).
The steam explosion technique was initially invented by American scholar Mason in 1928 and was used for pulping, transforming waste wood into construction paper pulp (Mason, 1926). The main principle of steam explosion involves subjecting plant fiber raw materials to high-temperature, high-pressure steam, which leads to the degradation of hemicellulose, softening of lignin, weakening of the lateral connections between fibers, and the rapid release of high-pressure steam within a short period. The steam vapor within the pores of the raw material rapidly expands, resulting in an explosive effect that tears the material into small fibrous fragments. This achieves the separation of raw material components and structural changes (Smichi et al., 2020). Steam explosion pretreatment has garnered significant attention from researchers due to its low cost, minimal energy consumption, and lack of pollution. Lam et al. (2008) conducted steam explosion treatment on fir bark and observed that the cellulose molecular chains in the treated bark experienced fracture, internal hydrogen bonds were partially disrupted, and the mobility of cellulose chains increased, favoring the transition of cellulose towards a disordered structure. Consequently, the strength of the formed fuel after steam explosion treatment was found to be 1.4 to 3.3 times higher than before treatment, and the calorific value also exhibited a significant increase. Huang et al. (2015) conducted steam explosion treatment on acid-pretreated cotton stalks at 225 °C for 3 minutes, achieving a yield of xylose of 0.16 g/g. De Barros et al. (2019) subjected pretreated spelt wheat to high-temperature steam treatment at 180 °C for 10 minutes after undergoing dilute acid pretreatment, resulting in a glucose yield of 0.28 g/g and a furfural yield of only 3.35 mg/g.
Hydrothermal pretreatment, also known as wet torrefaction, refers to a process where biomass is used as the feedstock and water serves as the reaction medium in a sealed reactor. Water is maintained in a liquid state at high temperatures through pressurization. In this unique state, water's properties are utilized to decompose structurally stable biomass materials (Bach et al., 2013). The hydrothermal pretreatment process involves three stages of transformation for the feedstock: Depolymerization of precursor compounds into monomers, resulting in a decrease in the system's pH; Dehydration of monomers, inducing polymerization reactions; Aromatization reactions leading to the formation of final products (Wei and Tongxiang, 2014). The hydrothermal treatment process involves mixing the raw material with water. As a result, the process does not impose any specific requirements on the moisture content of the feedstock, and there is no need for drying the raw material beforehand. This is advantageous for biomass with high moisture content, as it eliminates the need for significant energy consumption in drying and makes it suitable for treating high-moisture sludge (Dingmei et al., 2013). Moreover, the hydrothermal pretreatment process is straightforward, conducted under mild reaction conditions without excessively high temperatures or pressures, and it does not demand advanced equipment. Consequently, the hydrothermal pretreatment process has a wide range of applications, is relatively simple to operate, and is well-suited for easy adoption and promotion.
Chemical pretreatment
Chemical pretreatment involves the use of chemical reagents such as acids, alkalis, ionic liquids, and organic solvents to disrupt the structure of lignocellulosic biomass (Qing et al., 2017). For instance, Tang et al. (2021) employed a mixture of 4% H2SO4 and humic acid at a 1:10 solid-to-liquid ratio to pretreat wheat straw for 30 minutes at 60 °C, followed by a treatment at 180 °C for 40 minutes. Under these conditions, the glucose yield after enzymatic hydrolysis reached 0.52 g/g. However, it is important to note that acid pretreatment at high temperatures can lead to the generation of toxic byproducts such as furfural and hydroxymethylfurfural, which can significantly impede ethanol fermentation (Rabemanolontsoa and Saka, 2016).
In contrast, alkali pretreatment aims to break the ester linkages within cellulose, causing its fragmentation and altering both its surface area and crystallinity (Kim et al., 2016). Mota et al. (2021) treated sugarcane bagasse with a 0.25 mol/L NaOH solution at a 1:4 solid-to-liquid ratio under 202 °C for 40 minutes. Enzymatic hydrolysis yielded 25.8 g/L of glucose with a conversion rate of 0.423 g/g.
Organic solvent pretreatment, on the other hand, dissociates cellulose and hemicellulose, thereby reducing the cellulose structure. Solvents such as methanol, acetone, and ethanol have been employed for this purpose (Min et al., 2019), but their high toxicity raises concerns about potential environmental contamination.
Ionic liquid-based pretreatment is a green and efficient method that involves using organic salts to break the β-O-4 chemical bonds in lignin, disrupting cellulose's structural stability (Usmani et al., 2020). Asim et al. (2021) utilized an ionic liquid solution made from pyridine and sulfuric acid [PyH][HSO4·(H2SO4)] to pretreat 50 g/L of wheat straw at 100 °C for 2 hours, achieving a glucose yield of 0.442 g/g after enzymatic hydrolysis. However, ionic liquids also possess certain toxicity and are challenging to recycle, which makes their industrial-scale application challenging and costly (Roy et al., 2020).
Biological pretreatment
Biological pretreatment methods primarily utilize bacteria and fungi, such as white-rot fungi, brown-rot fungi, soft-rot fungi, Trichoderma reesei, and Aspergillus niger, to initiate the initial degradation of lignocellulosic biomass (Chen et al., 2018; Hermosilla et al., 2018; Tian et al., 2018). Hermosilla et al. (2018) employed the fungus Gloeophyllum trabeum to treat wheat straw for 10 days at 25 °C, followed by treatment with Ganoderma lobatum for an additional 20 days. The final glucose yield after enzymatic hydrolysis was 0.192 g/g.
In summary, physical pretreatment methods represented by mechanical milling, hydrothermal treatment, and steam explosion have the capability to effectively increase the specific surface area and pore size of the raw material, reduce cellulose crystallinity and degree of polymerization, and generate minimal toxic byproducts. However, these methods are associated with high energy consumption, inability to remove lignin, and severe inhibition of subsequent enzymatic hydrolysis due to lignin presence. Chemical pretreatment methods using acid and alkali reagents can efficiently remove partial lignin and hemicellulose, degrading hemicellulose into monosaccharides. Consequently, they exhibit good economic viability and potential for industrial applications. Nevertheless, challenges such as intense reaction conditions, corrosion of reaction equipment, formation of complex toxic byproducts, and substantial inhibition of enzymatic hydrolysis and fermentation exist. Biological pretreatment methods employing bacteria and fungi can mildly degrade lignin and hemicellulose under gentle reaction conditions, reducing cellulose aggregation. However, the efficiency of microbial treatment is relatively low, rendering it unsuitable for large-scale industrial applications at the current stage. Thus, considering the diverse composition and structural characteristics of various lignocellulosic feedstocks, harnessing the advantages of combined physical, chemical, and biological pretreatment methods can effectively mitigate energy consumption, minimize toxic byproduct generation, enhance removal rates of hemicellulose and lignin, and maximize the retention of cellulose components. This integrated approach represents an efficient means to elevate the efficiency, applicability, and utilization of raw material pretreatment processes.
Saccharification and hydrolysis
Cellulose hydrolysis, also known as saccharification, is the process of converting cellulose into glucose. Hydrolysis can be broadly categorized into two main types: acid hydrolysis and enzymatic hydrolysis. Acid hydrolysis is a well-established technique, but it is associated with strong equipment corrosion, challenging recovery and wastewater treatment, and unfavorable environmental impacts, resulting in low sugar conversion rates (Gómora-Hernández et al., 2020). Enzymatic hydrolysis, on the other hand, offers good selectivity, operates under mild reaction conditions, and is environmentally and equipment friendly. However, there is substantial room for improvement in terms of enzyme activity, reusability, and cost-effectiveness.
Cellulase hydrolysis pretreatment process facilitates the detachment of cellulose components, rendering cellulose more amenable to enzymatic degradation into fermentable monosaccharides. This enhancement benefits the utilization and conversion of cellulose-to-ethanol producing strains. Cellulase is a composite mixture comprising endoglucanases and exoglucanases that disrupt the β-1,4 glycosidic bonds of cellulose, resulting in glucose generation (Rajnish et al., 2021). In actuality, cellulase hydrolysis stands as the most cost-intensive unit operation within cellulose-to-ethanol production processes. On one hand, cellulase activity is susceptible to temperature, pH, substrate, and hydrolysis product influences. On the other hand, environmental factors stemming from residual lignin components from pretreatment can induce matrix effects, cellulase adsorption effects, water binding effects, inhibition effects, and more, impacting mass transfer, heat transfer, and reaction characteristics. This culminates in decreased cellulase activity, reduced hydrolysis efficiency, and lowered raw material utilization, necessitating heightened or supplemented cellulase dosages, thereby elevating cellulose-to-ethanol production costs.
While the yields of glucose and ethanol increase with higher cellulose content, cellulose content is not the sole determinant of glucose and ethanol production. When the lignin content of biomass reaches 7% to 9%, the yields of glucose and ethanol notably decrease, resulting in reduced ethanol yield (Raud et al., 2016). The enzymatic hydrolysis process involves the adsorption of cellulase enzymes onto the cellulose surface, cellulose hydrolysis, and desorption of cellulase enzymes (Barbosa et al., 2020; Zhao et al., 2022). Key factors influencing hydrolysis include substrate-inhibitory lignin and biomass porosity. Alkaline pretreatment has a more pronounced effect on increasing biomass porosity compared to other pretreatment methods (Alam et al., 2019). High crystallinity obstructs enzymatic cellulose breakdown, causing molecular chains to become more ordered and reducing internal fiber pores. Therefore, the removal of hemicellulose and lignin is advantageous for enhancing the accessibility of cellulases to the substrate.
Factors influencing the enzymatic saccharification process include substrate concentration, enzyme loading, temperature, and saccharification time (Mu et al., 2015). Cellulase enzymes are commonly used for hydrolyzing residual lignocellulose in second-generation bioethanol production. However, cellulase enzymes exhibit low activity towards pretreated lignocellulose. To address this challenge, researchers have explored the endoglucanase activity of Fibrobacter, Bacillus, and Microbacterium genera on coconut shell fibers at different pH values (pH 5-9) and temperatures (20–50 °C). The results demonstrate that Fibrobacter exhibits the highest activity under neutral pH and at 40 °C conditions (Fonseca et al., 2020).
Furthermore, cellulase enzymes constitute a significant portion of the production cost in bioethanol manufacturing. To reduce production costs, Baskaran et al. (2022) employed Trichoderma reesei NCIM 1186 to produce cellulase enzymes from pomegranate peels. Under optimal culture medium and process conditions, they achieved a maximum cellulase enzyme yield of 12.3 IU/mL. This approach utilized waste-derived cellulase enzymes and effectively lowered production costs. Non-productive binding of cellulase enzymes with lignin might increase at high substrate loads, particularly as hydrolysis progresses and cellulose content decreases. Rosgaard et al. (2007) implemented a “fed-batch” strategy to enzymatically hydrolyze pretreated barley straw, gradually increasing substrate concentration from 5% to 15% through substrate additions or enzyme dosing. This enabled high substrate concentration hydrolysis, resulting in elevated glucose concentrations. Shen et al. (2021) combined cellulase produced by Trichoderma reesei (30 FPU/g) with β-glucosidase produced by Aspergillus niger (50 FPU/g). Under conditions of 50 °C and 150 rpm in a citrate buffer, they enzymatically hydrolyzed NaOH-pretreated corn stover. The cellulose conversion rate exceeded 70%. This enzyme mixture exhibited enzymatic activity comparable to Novozymes’ Cellic Ctec2 at equivalent concentrations, thereby contributing to cost reduction.
Fermentation
At present, cellulose ethanol fermentation processes primarily include Separate Hydrolysis and Fermentation (SHF), Simultaneous Saccharification and Fermentation (SSF), Simultaneous Saccharification and Co-Fermentation (SSCF), and Consolidated Bioprocessing (CBP) processes (Nagarajan et al., 2019; Phwan et al., 2018).
SHF (separate hydrolysis and fermentation)
The SHF process operates by treating enzymatic hydrolysis and fermentation as separate units. In this approach, cellulose substrate undergoes cellulase hydrolysis to degrade into fermentable glucose monomers. Subsequently, yeast fermentation converts glucose into ethanol, with each step occurring under optimized temperature and conditions (Kawaguchi et al., 2016).
Song et al. (2020) utilized 30% H2O2 and acetic acid for pretreating bamboo at 85 °C for 2 hours, followed by thorough washing. Employing a cellulase dosage of 30 FPU/g, hydrolysis was performed at 45 °C and 200 r/min, resulting in a hydrolysate. Saccharomyces cerevisiae KCTC 7906 was employed for fermentation, yielding a maximum ethanol concentration of 13.2 g/L, with ethanol productivity and yield at 0.135 g/(L·h) and 0.144 g/g, respectively.
Chen et al. (2021) applied subcritical water pretreatment to wheat straw, followed by thorough enzymatic hydrolysis using 30 FPU/g cellulase and 1 U/g β-glucosidase at 50 °C and 120 r/min. This process yielded a final ethanol concentration of 37 g/L during fermentation with yeast NX11424, with ethanol productivity and yield of 0.192 g/(L·h) and 0.193 g/g, respectively.
Phi Trinh et al. (2016) employed a mixture of 0.25% HCl and 70% glycerol to pretreat rice straw at 190 °C for 3 hours. After detoxification and enzymatic hydrolysis through thorough washing, SHF was conducted using Candida utilis CBS 6054. The resulting ethanol concentration, productivity, and yield were 9.66 g/L, 0.101 g/(L·h), and 0.193 g/g, respectively.
SSF (simultaneous saccharification and fermentation)
The SSF process involves the concurrent operation of enzymatic hydrolysis and fermentation within a single vessel, allowing the glucose derived from cellulose substrate hydrolysis to be immediately metabolized by fermentative microorganisms into ethanol (Hans et al., 2019).
Huang et al. (2020) first utilized an alkaline H2O2 solution for a 60-min pretreatment of bamboo, followed by thorough washing. Using a solid-to-liquid ratio of 300 g/L, cellulase dosage of 25 FPU/g, and xylanase dosage of 150 U/g, SSF was carried out with yeast inoculation at 36 °C and 150 r/min, while maintaining pH at 4.8 throughout the 228-h fermentation. This process yielded 81.47 g/L of ethanol, with a productivity and yield of 0.36 g/(L·h) and 0.272 g/g, respectively.
Sewsynker-Sukai and Gueguim Kana (2018) employed a combination of salt and dilute acid for a 15-min pretreatment of maize cobs at 121 °C, followed by thorough washing and drying. Using a solid-to-liquid ratio of 200 g/L and cellulase dosage of 30 FPU/g, hydrolysis was performed at 50 °C in citrate buffer, followed by yeast inoculation at 35 °C. This resulted in a final ethanol concentration of 42.24 g/L, with a productivity of 0.59 g/(L·h) and a yield of 0.212 g/g.
Qiu et al. (2018) conducted a pretreatment of corn stover using an 85% H3PO4 and 30% H2O2 mixture, followed by ethanol and distilled water washing and thorough enzymatic hydrolysis. The subsequent yeast fermentation led to an ethanol concentration, productivity, and yield of 69.9 g/L, 0.569 g/(L·h), and 0.155 g/g, respectively.
SSCF (simultaneous saccharification and co-fermentation)
The SSCF process employs fermentative microorganisms capable of metabolizing pentoses, such as xylose and arabinose, generated from the degradation of hemicellulose in lignocellulosic feedstock, along with glucose from cellulose degradation, to concurrently produce ethanol within the same reaction system (Sharma et al., 2021).
Du et al. (2020) conducted a pretreatment of maize cobs using H2SO4-H3PO4 at 128 °C for 1 hour. They then introduced Maxkrest yeast and cellulase at 20 FPU/g into a 12.5% toxic mixed liquor, performing SSCF at 40 °C and 200 r/min. The resulting fermentation achieved a maximum ethanol yield, productivity, and yield of 12.23 g/L, 0.1 g/(L·h), and 0.098 g/g, respectively.
Bu et al. (2019) employed a 175 mmol/L potassium monopersulfate (PMS) solution for a 10-h pretreatment of sugarcane bagasse, followed by thorough washing and drying. Using a solid-to-liquid ratio of 1:3 and a cellulase dosage of 15 FPU/g, SSCF was conducted with the yeast strain SHY071 at 30 °C. After a 216-h fermentation, the ethanol concentration reached 120.77 g/L, with a productivity and yield of 0.53 g/(L·h) and 0.36 g/g, respectively.
CBP (consolidated bioprocessing)
The CBP process integrates the complete sequence of cellulase production, lignocellulosic hydrolysis, and ethanol fermentation within a single bioreactor, utilizing one or multiple microorganisms. This process has given rise to branch processes such as Co-processing Technology (CT) and Consolidated Bioprocessing with Simultaneous Saccharification (CBS) (Liu et al., 2020).
Khatun et al. (2017) first pretreated Jerusalem artichoke stalks with a 20 g/L NaOH solution and then inoculated with cellulase-producing wine yeast MNII AZEP. After a 60-h fermentation at 37 °C, the highest ethanol yield and productivity reached 28.2 g/L and 0.392 g/(L·h), respectively, with an ethanol yield of 0.141 g/g.
Xiao et al. (2019) employed a nitric acid-ethanol mixture to pretreat corn stover for 3 hours at 76 °C. Cultivating the wine yeast INVSc1 CBH TS at 28 °C, the fermentation endpoint yielded the highest ethanol concentration, productivity, and yield of 1.9 g/L, 0.015 g/(L·h), and 0.076 g/g (per gram of stover), respectively.
A comparison of different cellulose ethanol production processes and their performance is presented in Table 2.
Production process and performance comparison of different cellulose ethanol fermentation.
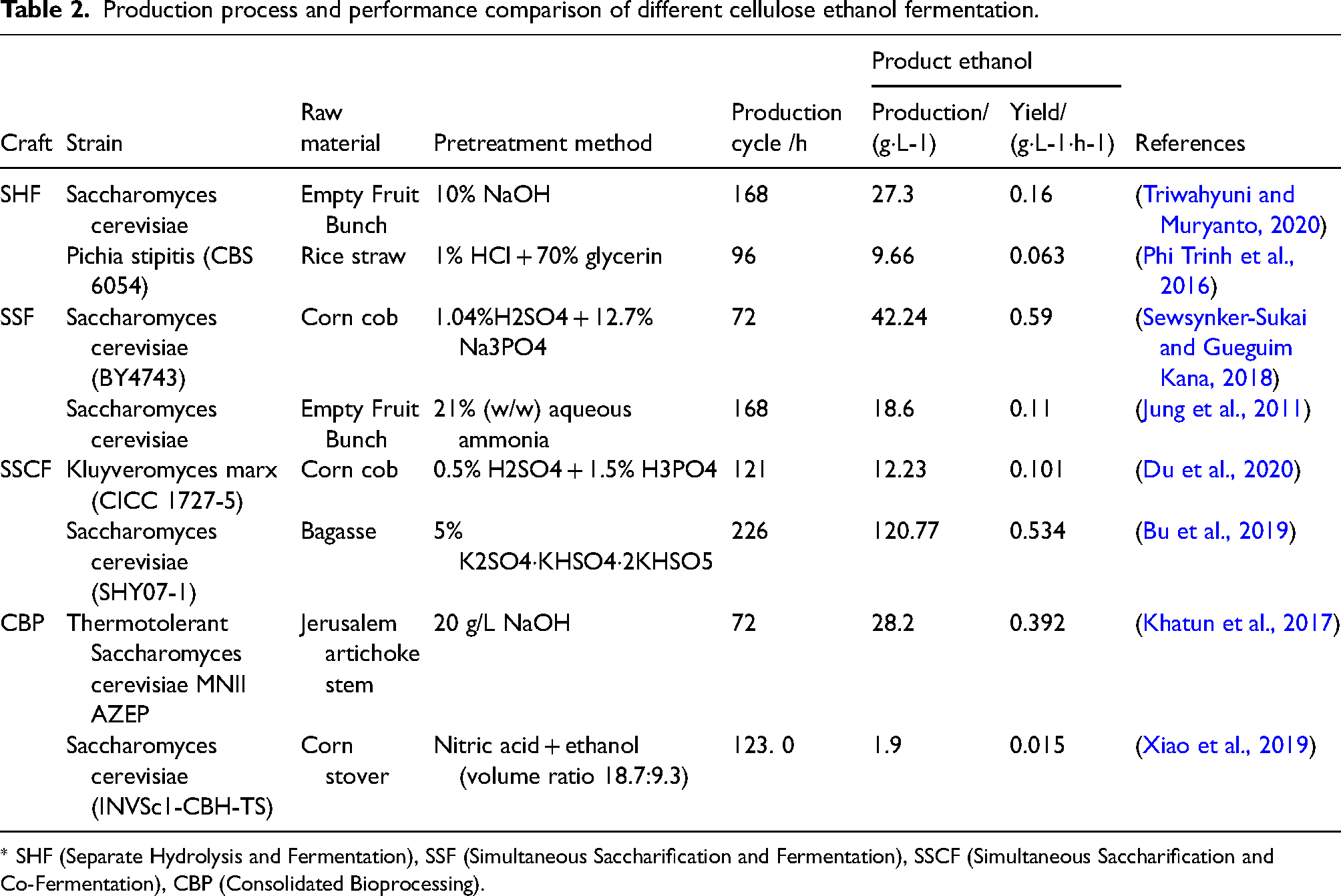
* SHF (Separate Hydrolysis and Fermentation), SSF (Simultaneous Saccharification and Fermentation), SSCF (Simultaneous Saccharification and Co-Fermentation), CBP (Consolidated Bioprocessing).
The SHF process has the advantage of allowing enzymatic hydrolysis and fermentation to occur at their respective optimal temperatures. This enables ideal enzymatic hydrolysis efficiency and ethanol fermentation efficiency, reduces viscosity at high substrate loads, and results in higher ethanol yields. However, a major drawback of the SHF process is the feedback inhibition of cellulase by the hydrolysis products, glucose and cellobiose, which affects subsequent enzymatic hydrolysis efficiency and substrate utilization. Consequently, supplemental cellulase enzymes are required. Additionally, the SHF process lacks continuity in its steps, involves relatively complex operations, and can impact ethanol fermentation yields.
The SSF process builds upon the SHF process and has the advantage of reducing the feedback inhibition of cellulase by hydrolysis products, resulting in decreased cellulase usage, shortened production cycles, fewer bioreactors needed, and reduced input costs. Nevertheless, one main drawback of SSF is the non-coordinated temperature requirements for hydrolysis and fermentation, which cannot simultaneously meet the optimal temperature conditions for both reactions. This affects hydrolysis efficiency and substrate utilization and requires additional cellulase enzyme supplementation, subsequently affecting the economic feasibility of fermentation.
The SSCF process's advantage lies in reducing the feedback inhibition of cellulase by hydrolysis products, effectively increasing the utilization of hemicellulose-derived pentoses and enhancing ethanol fermentation yield. However, like the SSF process, the SSCF process faces the challenge of non-coordinated temperature requirements for hydrolysis and fermentation, and extensive cellulase enzyme supplementation, impacting process economics.
In comparison, the CBP process is characterized by its simplicity and ease of operation, enabling the conversion of substrate to ethanol in a single step. This process represents an effective way to produce and utilize low-cost cellulase and reduce the production cost of cellulose ethanol. However, current research challenges in the CBP process lie in developing suitable microorganisms or microbial consortia through genetic and metabolic engineering strategies. On one hand, this involves introducing ethanol synthesis pathways into cellulase-producing strains, allowing them to directly ferment cellulose-derived carbon sources into ethanol after cellulose degradation. On the other hand, it involves introducing cellulase biosynthesis pathways into ethanol-producing strains, equipping them with the ability to secrete cellulases for cellulose degradation.
It is clear from the current study that investing more time into the production process will have a positive impact on the final yield. However, longer production times also mean that more production resources are invested, and the final production efficiency is not necessarily the highest. Moreover, the fermentation method may not be the same for different types of biomass feedstocks, and SSCF is the most promising method for industrial production due to the fact that the production process can be partially reduced compared to the other methods.
The first-generation bioethanol production process has reached a high level of maturity. Building upon this foundation, the preparation of second-generation bioethanol necessitates further research, particularly in the domain of pretreatment. Furthermore, the cultivation of novel yeast strains or the combination of yeast strains capable of co-fermenting pentose and hexose sugars derived from semi-cellulosic hemicellulose remains an ongoing research imperative. Although third-generation bioethanol production has witnessed considerable investigation, offering advantages such as not requiring arable land and mitigating CO2 emissions, the expensive preprocessing of algae necessitates continued exploration and refinement over time.
At present, the focal points of research in bioethanol production continue to be the pretreatment methods for second-generation bioethanol and the cultivation and selection of yeast strains for the fermentation process. As projected by the International Energy Agency (IEA), cellulose-based ethanol's annual consumption is anticipated to escalate to 1.1 billion tons by 2050, replacing 27% of transportation fuels. In comparison to fossil gasoline, cellulose-based ethanol can achieve a substantial reduction in carbon emissions, ranging from 76% to 124%. This equates to a carbon dioxide reduction of 2.67 tons per ton of cellulose ethanol produced. Consequently, cellulose-based ethanol offers significant energy benefits and carbon emission reductions, indicating a highly promising outlook for future development.
Conclusions
The preparation process of the first generation of bioethanol is very mature. Hence, the research on pretreatment needs to be strengthened to prepare the second generation of bioethanol, and it is necessary to cultivate new yeast further or combine yeast to co-ferment pentose and hexose. Although the preparation of the third-generation bioethanol has been studied a lot, and it has the advantages of not occupying cultivated land, absorbing CO2 in the air, and reducing environmental pollution, the pretreatment of algae is expensive and needs time to be further excavated and improved. At present, the pretreatment method of the second-generation bioethanol and the cultivation and selection of yeast in the fermentation process are still the current research hotspots. It can be seen that the research focus of cellulosic ethanol bio-refining in the future is to develop integrated technologies, including low-consumption, high-efficiency, and clean raw material pretreatment technology, which can reduce the consumption of neutralization and detoxification chemicals and water sources, and reduce the production of solid waste and liquid waste; Low-cost and high-efficiency cellulase production and hydrolysis technology can improve the yield of cellulose sugar and save the cost of raw materials; Matching high-efficiency production strains and engineering transformation technology can improve the tolerance of recombinant strain inhibitors and high-temperature stress, the (synchronous) transport and transformation ability of pentose and hexose, and the metabolic synthesis ability of product ethanol.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by The Ministry of Higher Education (MoHE) through the Fundamental Research Grant Scheme (FRGS) (203/PTEKIND/6711702).
Data availability
Data will be made available on request.
