Abstract
Introduction:
Secondary craniosynostosis can occur in patients with metabolic and hematologic disorders. However, current studies have been limited due to the rarity of these pathologies. The current study uses a large nationwide multi-institution database to evaluate patient demographics, suture patterns, and associated comorbidities.
Methods:
A retrospective analysis of the TriNetX database was performed for patients diagnosed with craniosynostosis and X-linked hypophosphatemia (XLH), vitamin D-resistant rickets (VDRR), pseudohypoparathyroidism, glycosaminoglycan disorder, osteopetrosis, sickle cell, thalassemia, and polycythemia vera. Demographics, suture patterns, and craniofacial, orthopedic, and systemic congenital malformations were evaluated.
Results:
About 1,902 patients with secondary craniosynostosis with metabolic or hematologic disorders were identified. Two hundred sixty six patients with XLH, 236 with VDRR, 136 with pseudohypoparathyroidism, 283 with hyperthyroidism, 103 with glycosaminoglycan disorder, 27 with osteopetrosis, 509 with sickle cell, 302 with thalassemia, and 40 with polycythemia vera. The most common suture involvement was sagittal in XLH (68.18%), VDRR (55.56%), hyperthyroidism (52.94%), thalassemia (43.18%), and sickle cell (36.78%). Multiple suture involvement also commonly occurred, especially in osteopetrosis (66.67%), pseudohypoparathyroidism (40.00%), and glycosaminoglycan disorder (30.00%). Chiari malformations and hydrocephalus were common in XLD, VDRR, and osteopetrosis. Orthopedic comorbidities occurred frequently in osteopetrosis, glycosaminoglycan disorder, VDRR, and XLH. Congenital cardiac malformation was common in all groups. Elevated rates of all congenital systemic malformations were seen in XLH, VDRR, and pseudohypoparathyroidism.
Conclusions:
Metabolic and hematologic disorders with secondary craniosynostosis are associated with sagittal and multi-suture closure patterns. Multiple groups showed increased rates of other craniofacial, orthopedic, and congenital systemic malformations.
Keywords
Introduction
Secondary craniosynostosis is characterized by premature cranial suture fusion resulting from an abnormal physiologic environment. 1 It may arise in the setting of metabolic or hematologic disorders, or as a complication of certain medications or procedural interventions.1-9 In contrast, primary craniosynostosis results from intrinsic genetic abnormalities leading to cranial suture closure.
The most common metabolic diseases associated with craniosynostosis involve phosphate and calcium regulation.1,3,10 These include X-linked hypophosphatemia (XLH), vitamin D-resistant rickets (VDRR), and pseudohypoparathyroidism. Other disorders, such as mucopolysaccharidosis, result in metabolite accumulation within the cranial sutures, contributing to pathological suture fusion and craniosynostosis. 11 Disorders such as hyperthyroidism and osteopetrosis affect osteogenic activity of the cranial sutures.3,12-14 Hematologic disorders such as sickle cell disease, thalassemia, and polycythemia vera lead to craniosynostosis through bone marrow hyperplasia, causing expansion and altered skeletal growth.15-18
The severity of secondary craniosynostosis primarily correlates with the extent and severity of the underlying metabolic disease and the age of onset. 1 These patients typically present at significantly older ages than non-syndromic cases and those with known craniosynostosis-associated genetic syndromes. 19 While the sagittal suture is the most commonly affected, multi-suture or pan synostosis can also occur.1,3,10,11,15,19-22 Additionally, signs of increased intracranial pressure, such as Chiari malformation, have been reported to be as high as 43%.23,24 Therefore, frequent screening for craniosynostosis in these patients with metabolic and hematologic disorders is essential to detect and address increased intracranial pressure and its sequela.
TriNetX is a global research network that aggregates de-identified clinical data drawn directly from electronic health records, billing systems, and pharmacy data, encompassing demographics, diagnoses, procedures, laboratory values, medications, and longitudinal clinical encounters. Contributing institutions include academic medical centers, community hospitals, and health systems, allowing researchers to access large patient populations while maintaining privacy protections. Due to the rarity of secondary craniosynostosis associated with these disorders, evaluating epidemiological characteristics of these patient cohorts has been challenging, with only limited case reports and series in the literature. With the advent of large nationwide multi-institution patient databases, these rare pathologies have become possible to study with adequate power. This study evaluates the demographics, suture patterns, and associated comorbidities in patients with secondary craniosynostosis and metabolic and hematologic disorders utilizing the TriNetX database. A better understanding of these patient features can improve screening, diagnosis, and arrangement of proper multidisciplinary care.
Methods
The TriNetX database (TriNetX, LLC; MA, USA) was queried for patients with secondary craniosynostosis following institutional review board (IRB) exemption from the University of Texas Southwestern Medical Center. The following international classification of disease (ICD) 10 codes were utilized to identify secondary craniosynostosis patients: XLH (E83.31), VDRR (E83.32), osteopetrosis (Q78.2), osteogenesis imperfecta (Q78.0), pseudohypoparathyroidism (E20.1), hyperparathyroidism (E21.0), glycosaminoglycan metabolism disorder (E76), sickle cell disease (D57), thalassemia (D56), and polycythemia vera (D45). The identified patients were then evaluated for co-diagnosis of craniosynostosis (Q75.0). Patient demographics, including gender, ethnicity, and race, were collected.
A subgroup of patients with specified craniosynostosis diagnoses was evaluated to determine suture involvement. They were separated into sagittal (Q75.01), unilateral coronal (Q75.021), metopic (Q75.03), unilateral lambdoid (Q75.041), and multi-suture craniosynostosis (Q75.05). Associated craniofacial, orthopedic, and systemic deformities were also collected. Craniofacial comorbidities included Chiari malformation (Q07.0), syringomyelia (G95.0), hydrocephalus (G91), optic atrophy (H47.2), and cleft lip & palate (Q35-37). Orthopedic comorbidities included spinal deformity (Q76), hip deformity (Q65), limb deformity (Q74), hand deformity (Q68.1), and foot deformity (Q66). The systemic comorbidities included congenital cardiac (Q20-28), congenital renal (Q60-64), congenital respiratory (Q30-34), congenital digestive (Q38-45), and congenital genital (Q50-56) malformations. Statistical analysis was performed to determine totals and percentages of patient demographics, suture involvement, and associated comorbidities.
Results
A total of 1902 patients with secondary craniosynostosis with metabolic or hematologic disorders were identified (Table 1). Disorder-specific totals included 266 patients with XLH, 236 with VDRR, 136 with pseudohypoparathyroidism, 283 with hyperthyroidism, 103 with glycosaminoglycan disorder, 27 with osteopetrosis, 509 with sickle cell, 302 with thalassemia, and 40 with polycythemia vera. All groups were a majority male except for hyperthyroidism, which had a female predominance. Most patients were of white race except for sickle cell and thalassemia, which were primarily of black race.
Secondary Craniosynostosis in Metabolic and Hematologic Disorder Patients.
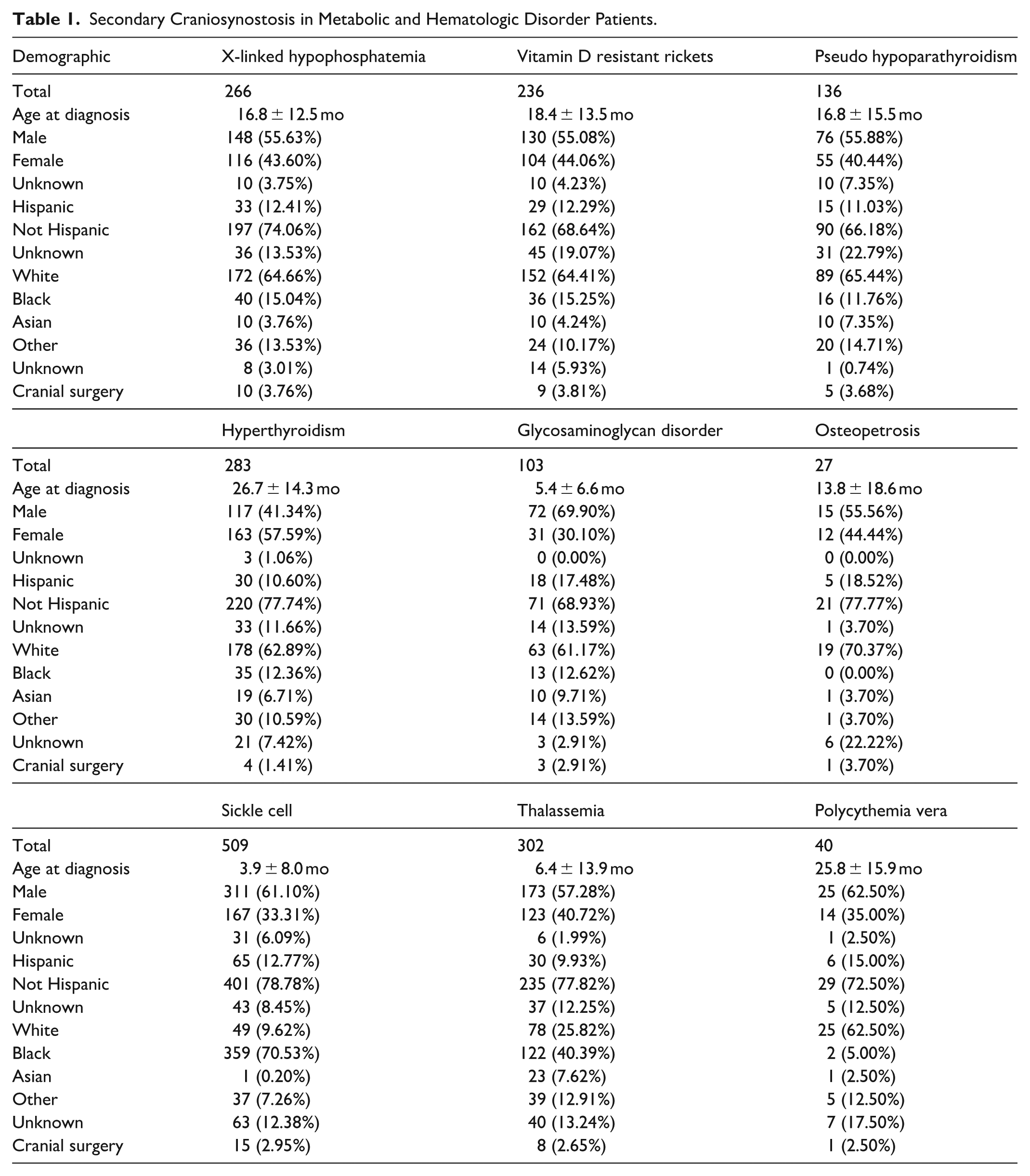
Subgroup analysis of 198 patients with specified suture involvement was evaluated (Table 2). Overall, the most common single-suture involvement was sagittal synostosis. Sagittal suture closure was common in patients with XLH, VDRR, pseudohypoparathyroidism, hyperthyroidism, glycosaminoglycan disorder, sickle cell, and thalassemia. Multiple suture involvement also commonly occurs, especially in pseudohypoparathyroidism, glycosaminoglycan disorder, and osteopetrosis.
Secondary Craniosynostosis Suture Type Involvement.
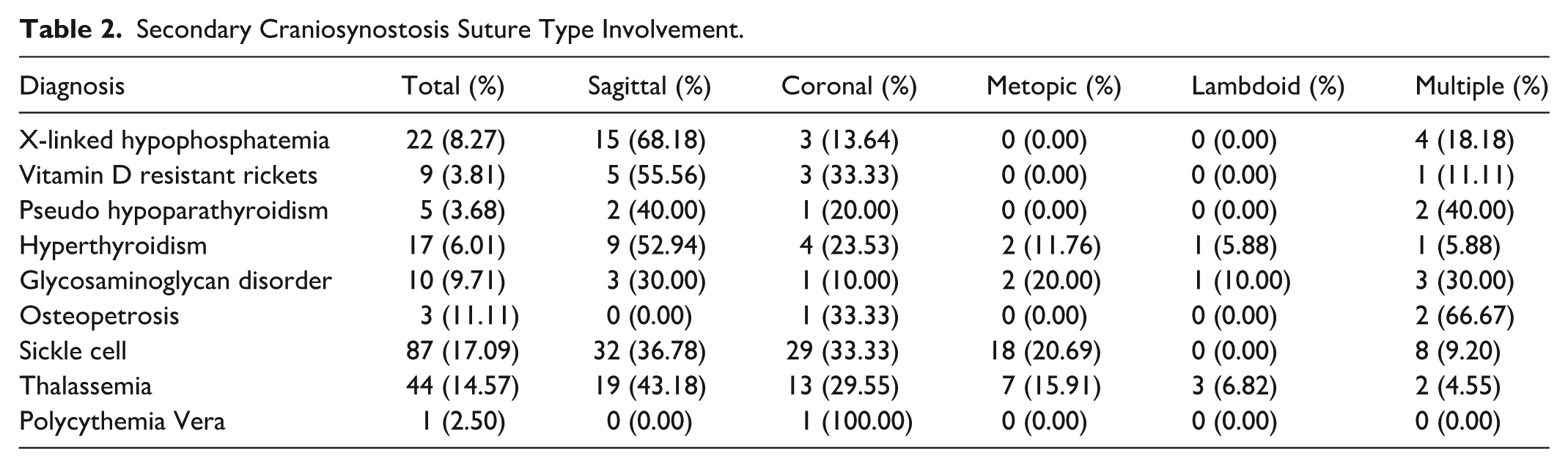
Chiari malformations were most common in osteopetrosis, XLH, and VDRR (Table 3). The rate of syringomyelia was relatively low for all secondary craniosynostosis metabolic and hematologic disorders. Hydrocephalus was relatively common in multiple disorders, including VDRR, glycosaminoglycan disorders, XLH, pseudohypoparathyroidism, and osteopetrosis. Osteopetrosis had the highest rate of optic atrophy, at 14.81%. The cleft lip and palate rate was increased in VDRR, osteopetrosis, XLH, and polycythemia vera.
Secondary Craniosynostosis Associated Craniofacial Comorbidities.
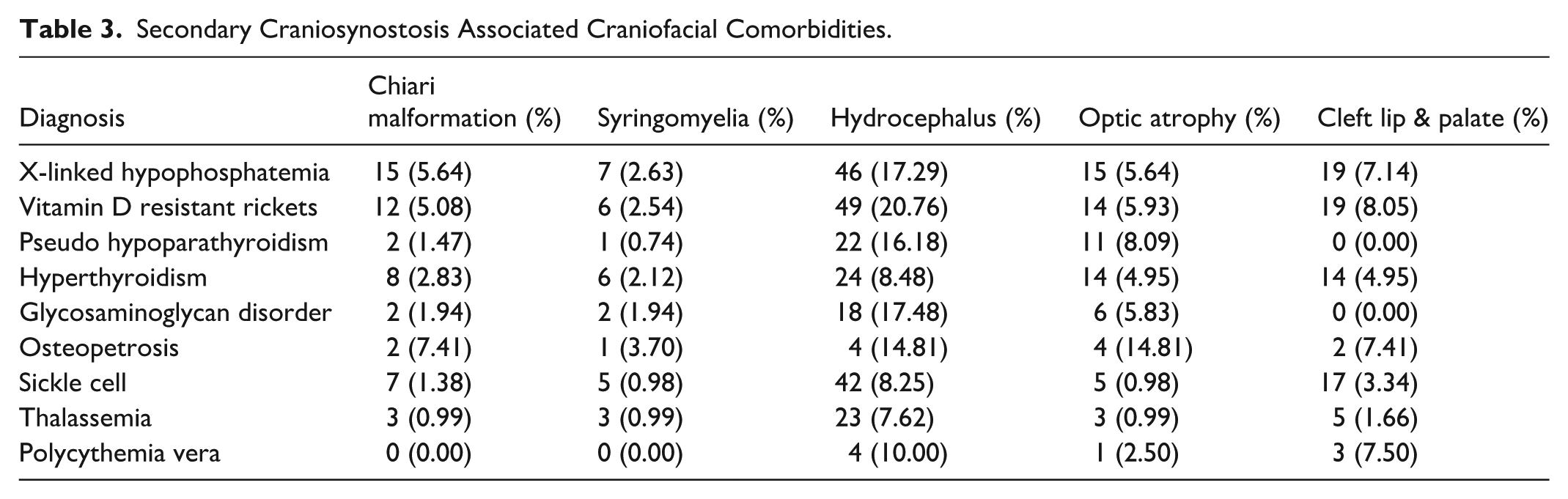
Overall, orthopedic comorbidities were commonly found in osteopetrosis, glycosaminoglycan disorder, VDRR, and XLH (Table 4). Spinal deformities were the most common comorbidity within these groups, with glycosaminoglycan disorders having the highest rate at 24.27%. Hip, limb, and foot deformities were also common across these groups. Hand deformities were less often seen, with the most common being associated with osteopetrosis at 7.41%.
Secondary Craniosynostosis Associated Orthopedic Comorbidities.
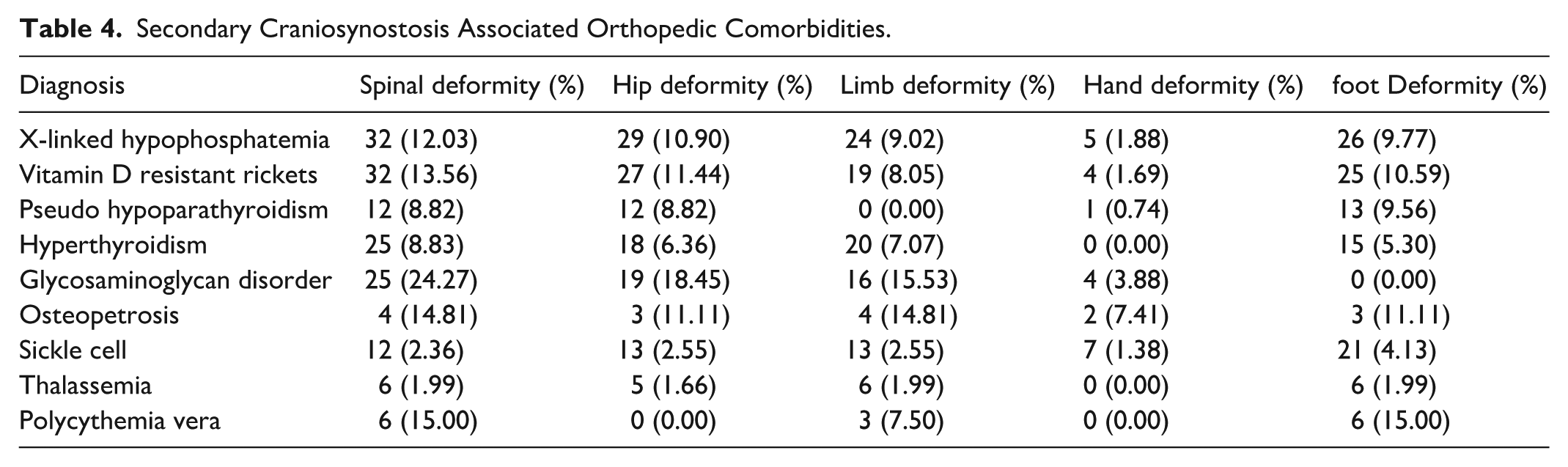
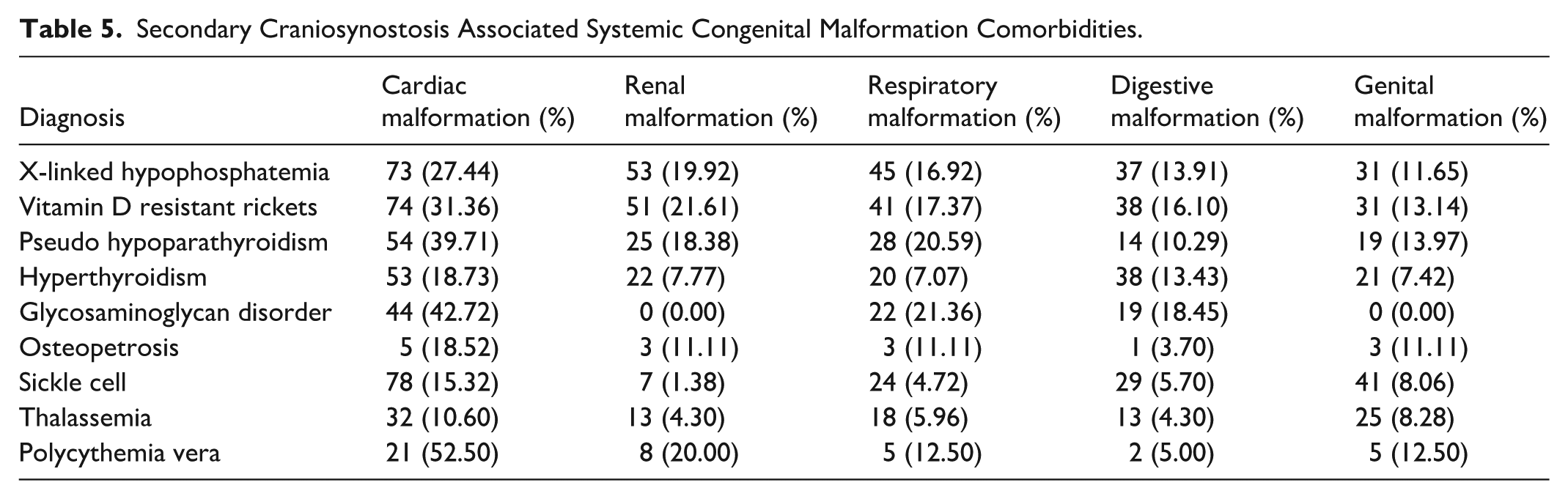
Congenital cardiac malformations were frequently seen in all secondary causes of craniosynostosis, with up to 52.50% in polycythemia vera (Table 5). XLH, VDRR, and pseudohypoparathyroidism had elevated rates of all systemic congenital malformations. Elevated rates were also seen across congenital malformations in hyperthyroidism, osteopetrosis, and polycythemia vera.
Secondary Craniosynostosis Associated Systemic Congenital Malformation Comorbidities.
Discussion
Secondary craniosynostosis is contingent on primary disturbance from various physiologic and interventional causes.1-9 Diagnosis can be delayed or missed due to the rarity of the diagnosis, and identification often relies on non-surgeons caring for the associated medical conditions. Additionally, the suture closure leading to craniosynostosis tends to occur at older ages outside the typical window where the head shape is closely monitored.1,19,23 Due to the infrequency of secondary causes of craniosynostosis, few surgeons have extensive diagnostic experience, with current literature remain limited by small cohort size. Utilizing an extensive nationwide database, we stratified patients based on secondary pathophysiology to create a demographic picture to assist with diagnosis and comprehensive craniosynostosis management.
X-Linked Hypophosphatemia (XLH) and Vitamin D Resistant Rickets (VDRR)
X-linked hypophosphatemia is primarily caused by mutations in PHEX genes, resulting in aberrant osteocyte function and their associated lineage. 3 The subsequent bone mineralization failures and accumulation of inhibitors will increase circulating FGF23. Given its resemblance to FGF 2 and 3, it can bind to those receptors, causing fusion of the cranial sutures. 25 Cases of VDRR typically result from mutations in the vitamin D receptor, causing dysfunctional calcium and phosphorus homeostasis and lack of response to vitamin D supplementation. 26 However, some forms of VDRR can be seen in patients with XLH. VDRR can also be present because of mutations in the 1-alpha-hydroxylase enzyme, which activates vitamin D in the human body. 3 The result of these mutations is hypocalcemia and poor bone mineralization.
Existing literature estimates the presence of sagittal fusion being the most common.10,20-22 LaValley et al 20 reviewed patients with XLH and found that 58% had sagittal involvement. Our data corroborate this, showing that sagittal suture involvement was dominant in both diseases. Seruya et al, 19 in a series of three patients with XLH, found that two-thirds had multi-suture involvement. In our cohort, we found that 18% of patients had more than one suture involved. A cohort of 26 patients by Arenas et al 21 found sagittal suture involvement in all patients, with 73% isolated and 27% involved in multi-suture patterns. Their most common complication was cardiac malformation, as well as a significant burden of hydrocephalus. Both associations have been reported in previous literature.1,19-22,27,28 Chiari malformations have shown a wide range in the literature from 3.85% to 43.75%.21,23,24 Our findings fall at the lower end of the reported range, with approximately 5% prevalence in both conditions.
Pseudohypoparathyroidism
Part of a larger group of inborn errors of metabolism, pseudohypoparathyroidism functionally results from resistance to the effects of parathyroid hormone (PTH). 29 These patients have elevated levels of PTH leading to hypocalcemia. Those abnormalities often cause the presenting symptoms of paresthesia, numbness, or cramping. Many growth plates in patients’ bones will fuse prematurely, including the cranial sutures. Previous literature has been limited to case reports and patients with craniosynostosis and documented associated complications with hydrocephalus.30-34 Our data indicate a relatively low comorbidity rate outside congenital organ malformations, with hydrocephalus being the most common. However, cardiac malformation was prevalent, seen in 40% of patients. While other work has documented cardiomyopathy with pseudohypoparathyroidism likely due to prolonged electrolyte abnormalities, congenital cardiac issues are not as well known. 35
Hyperthyroidism
Unlike the previously discussed diagnoses, hyperthyroidism can be a cause of secondary craniosynostosis if a pregnant mother is affected.12,13 This is also the most prevalent disease studied, present in roughly 1% of the American population. 36 Its widespread nature is owed to the many different causes of hyperthyroidism, from autoimmunity to congenital mutations. Despite such prominence, not much research has covered craniosynostosis as a complication, though animal studies have shown that excesses of thyroid hormone can narrow cranial sutures. 14 This is the only associated disease studied evaluated in where the majority of patients were female. The patients were largely non-Hispanic and white. Sagittal suture involvement was dominant, as were cardiac and digestive malformations. Given its commonality in the general population, appreciating that craniosynostosis is associated with hyperthyroidism is key to early diagnosis and correction.
Glycosaminoglycan
Issues metabolizing glycosaminoglycan compounds are also known as mucopolysaccharidoses. They are a group of inherited disorders that stem from poor or absent processing of glycosaminoglycan compounds in the lysosome. 37 These diseases are of varying severity in the patient’s life. Previous studies have found that many patients can experience cranial suture fusion, leading to its presence in up to 20% of cases. 38 In the current data, these patients are primarily male, non-Hispanic, and white. As compared to most other subsets, the sutural involvement in glycosaminoglycan cases was more diverse, including sagittal (30%), multiple (30%), and metopic (20%). The most common complications in this population were cardiac and respiratory malformations (43% and 21%), as well as spinal and hip deformities (24% and 18%).
Osteopetrosis
Osteopetrosis is a varied group of bone disorders primarily caused by osteoclast dysfunction, usually inherited. Its prevalence varies with the genetic pattern. The dominant form is seen in 1 in 20 000 people, and the recessive form is seen in 1 in 250 000 people. 39 While the presentations are variable, the recessive form tends to be more severe than the dominantly inherited form. Osteopetrosis was the least common condition in our cohort, with 27 identified cases. Broader literature describing craniosynostosis in osteopetrosis is mainly limited to case reports demonstrating the need for more extensive multi-center studies.40-44 Existing case reports have found either multi-suture fusion or sagittal suture fusion. This is the only condition in our cohort with no documented cases of isolated sagittal fusion. Instead, there were 2 multi-suture patients and 1 coronal suture patient. The complications of these patients were also spread across several organ systems at similar frequencies, with patients experiencing cardiac malformations, hydrocephalus, and deformities of spines and limbs. This was also the patient population in which Chiari malformations were most common.
Sickle Cell Disease
Sickle cell disease was the most common concurrent diagnosis in the recorded cases, which reflects its substantial presence in the general population, as 1 in every 365 black children is born with the disease. 45 Inherited in an autosomal recessive manner, patients will show symptoms like pain, anemia, and swelling before their first birthday. While the mechanistic cause of cranial suture fusion in sickle cell disease is unclear, work that has attempted to screen for fusion by Manrique et al in sickle cell patients found it in 19% of their samples, with all cases involving the sagittal suture. 15 While our data confirm the dominance of sagittal involvement in 37% of patients, coronal fusion was present in 33% of patients, thus demonstrating gaps in the characterization of secondary craniosynostosis. Sickle cell is one of the 2 diseases studied, and the patient population was predominantly black (71%). This aligns with the substantial racial burden of sickle cell in the general population, as previously described. Compared to the other diagnoses, patients with sickle cell disease experienced lower rates of orthopedic comorbidities, with spine and hip deformities observed in only 2 and 3% of patients, respectively. Cardiac malformations were the most common complication (15%), which corroborates existing literature that has found cardiopulmonary complications are the leading cause of morbidity for these patients. 46
Thalassemia
Similar to sickle cell disease, thalassemia is an inherited set of blood disorders that can cause anemia. They are typically classified based on which component of the patient’s hemoglobin is dysfunctional, alpha or beta. 47 There are multiple subgroups within both alpha and beta-thalassemia. Presenting symptoms fall within an extensive range from anemia leading to fatigue, splenomegaly, and bone deformation due to altered hematopoiesis. In the current dataset, these patients were primarily male, and the plurality was black (40%). Sagittal suture involvement was dominant (43%), with coronal involvement being the second most common (30%). Like sickle cell disease, orthopedic complications were rare (<2%), and cardiac malformations were the most common, seen in 11% of patients. Cardiac complications are also a leading cause of death for thalassemia patients. 48
Polycythemia Vera
Polycythemia vera is the only disease studied that is neoplastic in origin. It is usually the result of a mutation in the JAK2 gene, which leads to an overproduction of red blood cells. 49 A patient’s blood will become excessively viscous, leading to symptoms like bursitis and arthritis. The mechanism by which a patient’s cranial sutures fuse in cases of polycythemia vera is not understood. Unlike the other 2 hematologic disorders, most polycythemia patients in this data were white and non-Hispanic. Sutural involvement was only recorded for 1 patient, where the coronal suture was fused. These patients had the highest rate of cardiac malformation (53%), followed by renal malformations (20%). These findings highlight the need for further research into congenital malformations associated with polycythemia vera, as there is documentation that these patients suffer from substantial cardiovascular morbidity. 50
Due to patients with secondary craniosynostosis commonly presenting at later ages, surgical intervention requires cranial vault remodeling procedures for reconstruction. 19 The prognosis and progression of the individual disease should be considered, but surgical intervention is indicated in symptomatic patients to reduce the associated risks of increased intracranial pressure. In the limited reports on surgical intervention, patients with metabolic disorders tolerated various cranial vault remodeling techniques based on the affected sutures with low rates of complications.1,20 In asymptomatic patients, clinical monitoring is often performed, as demonstrated by the low operative rate across all secondary craniosynostosis patients in our study. Thus, it is vital to conduct an extensive multidisciplinary evaluation and discussion with the family to determine individualized care based on symptomatology and associated metabolic or hematologic disorders.
This study has several limitations inherent to the TriNetX platform. Because TriNetX suppresses patient counts below 10 to protect patient privacy, the database is not well-suited for identifying or analyzing extremely rare disorders with very small patient populations. As a result, metabolic and hematologic disorders with very low prevalence are not feasible. Also, statistical analysis between cohorts was not performed due to the low power of the small cohorts. Additionally, TriNetX provides information on a patient’s current age but does not supply the age at diagnosis, which limits the ability to characterize disease onset, assess diagnostic delays, or study age-specific patterns in presentation.
Conclusion
This study, using an extensive multi-center database, collated cases of secondary craniosynostosis to provide a demographic profile of patients affected by these conditions for diagnosis and comprehensive care. Given the broad nature of this dataset, a wide net was also cast in evaluating comorbidities. In doing so, not only were more commonly expected ones, such as Chiari malformation and hydrocephalus found, others, including organ system malformations and limb anomalies, were documented. This study should be helpful to physicians of all specialties in the diagnosis and treatment of secondary craniosynostosis and its comorbidities.
Footnotes
Acknowledgements
N/A.
Ethical Considerations
N/A.
Consent to Participate
Requirement for informed consent to participate has been waived by the Univeristy of Texas Southwestern Medical Center Institutional Review Board.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Presented at (If Applicable)
N/A.
