Abstract
Purpose:
X-linked hypophosphatemia is characterized by skeletal abnormalities, particularly lower limb angular deformities. Although burosumab has demonstrated short-term clinical improvements, its mid- to long-term effects on skeletal alignment remain underexplored. This study evaluated skeletal outcomes of burosumab therapy over 2 years, focusing on lower limb deformities.
Methods:
We retrospectively analyzed 20 pediatric X-linked hypophosphatemia patients (10 boys and 10 girls) who initiated burosumab at a mean age of 7.5 ± 2.4 years. Rickets severity score, mechanical axis deviation, mechanical lateral distal femoral angle, medial proximal tibial angle, lateral distal tibial angle, and standing height were assessed at baseline, 12 months, and 24 months. Outcomes were analyzed using age-standardized z-scores.
Results:
Rickets severity improved from the first year, with Rickets Severity Score decreasing from 3.5 ± 1.2 to 0.6 ± 0.5 at 24 months (mean change 2.9; 95% confidence interval 2.4–3.4). Lower limb alignment also improved: |z|-mechanical axis deviation decreased from 2.3 ± 1.6 to 1.0 ± 1.0 (change 1.3; 95% confidence interval 0.9–1.7), showing progressive correction from the first year. |z|-mechanical lateral distal femoral angle improved mainly at 24 months, decreasing from 2.7 ± 2.1 to 1.3 ± 1.2 (change 1.4; 95% confidence interval 0.8–2.0). |z|-medial proximal tibial angle and |z|-lateral distal tibial angle showed smaller overall changes (0.8 and 0.9, respectively), indicating modest tibial correction. Standing-height z-scores remained stable (−1.5 ± 0.8 to −1.3 ± 0.7), with no measurable change in growth over 2 years.
Conclusions:
Burosumab therapy may improve skeletal deformities in pediatric X-linked hypophosphatemia. Early improvements in rickets severity were followed by gains in alignment, particularly mechanical axis deviation and mechanical lateral distal femoral angle, though standing height remained unaffected. Longer-term follow-up is required to confirm sustained skeletal benefits.
Level of Evidence:
Level IV
Introduction
Hypophosphatemic rickets is a rare hereditary disorder causing impaired bone metabolism from early childhood.1–4 The most common form, X-linked hypophosphatemia (XLH), results from loss-of-function mutations in the phosphate-regulating endopeptidase homolog, X-linked (PHEX) gene, leading to elevated fibroblast growth factor 23 (FGF23).1,4 Excess FGF23 reduces renal phosphate reabsorption and suppresses 1,25-dihydroxyvitamin D synthesis, resulting in persistent hypophosphatemia. Consequently, defective growth plate mineralization leads to skeletal abnormalities, including lower limb angular deformities, short stature, and gait disturbances.1,5
Conventional therapy of XLH combines oral phosphate supplements and active vitamin D analogs. 3 More recently, burosumab (Crysvita®; Kyowa Kirin Co., Ltd., Takasaki, Japan), a fully human monoclonal antibody targeting FGF23, 2 has emerged as a targeted therapeutic option. Approved in 2018, 2 burosumab has shown efficacy in improving metabolic profiles and rickets severity.4–8 However, its short-term effects on lower limb alignment may be limited.5–8 Despite improvements in metabolic parameters, surgical intervention by pediatric orthopedic surgeons is often still required. 2 Thus, mid- to long-term follow-up is needed to assess the sustained impact of burosumab on skeletal deformities.1,2 The present study aimed to evaluate the effects of burosumab therapy on lower limb alignment, with particular focus on frontal deformities, over a 2-year follow-up period.
Materials and methods
A retrospective analysis was conducted of medical and radiographic records from pediatric patients diagnosed with XLH who initiated burosumab therapy between June and August 2023 at a single tertiary children’s hospital. Ethics approval for this study was granted by the Institutional Review Board (IRB No. H-2505-140-1643). Given the retrospective design and the use of anonymous clinical data, the requirement for informed consent was waived. This study was designed and reported in accordance with the STROBE (Strengthening the Reporting of Observational Studies in Epidemiology) guidelines.
Study population
Pediatric patients diagnosed with XLH and receiving burosumab therapy with a follow-up period of at least 2 years were included in the analysis (Figure 1). Patients with inadequate radiographic data or insufficient follow-up period duration were not included in this study. The exclusion criteria also included patients without a confirmed genetic diagnosis of XLH based on PHEX gene mutation analysis and those who continued conventional medical therapy without receiving burosumab treatment. In addition, patients who underwent surgical correction after initiating burosumab therapy were excluded. Either derotation osteotomy or guided growth was performed for persistent torsional deformities or for pronounced frontal malalignment that had been severe at baseline.
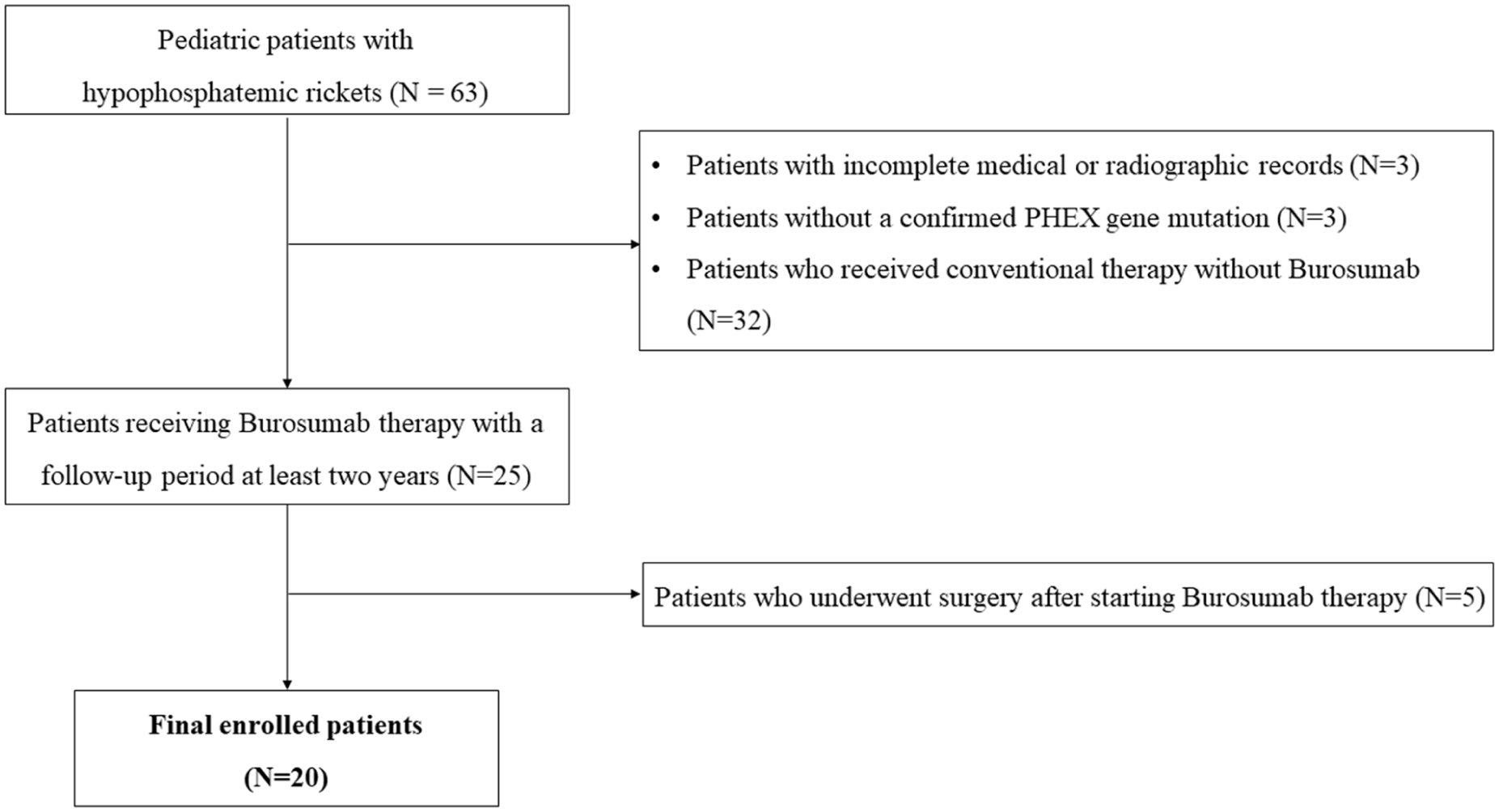
Patient selection and exclusion criteria.
Administration of burosumab
Burosumab was administered subcutaneously at a dose of 0.8 mg/kg every 2 weeks, under the supervision of a multidisciplinary team of pediatric specialists, including nephrologists, endocrinologists, and geneticists. Dosing adjustments were made based on clinical response and biochemical markers, particularly serum phosphate levels, with a maximum single dose of 2 mg/kg. In patients previously treated with oral phosphate supplements or active vitamin D3 analogs, these were discontinued at least 1 week prior to initiation. Therapy commenced once serum phosphate concentrations had declined below the age-specific lower limit of normal.
Evaluations
All evaluations were conducted at baseline (prior to burosumab therapy) and at 12 and 24 months post-treatment. First, the Rickets Severity Score (RSS) was used to provide a quantitative assessment of radiographic abnormalities at the metaphyses and growth plates. These were evaluated on standard anteroposterior radiographs of the wrists and knees. The RSS assigns a score from 0 (no radiographic evidence of rickets) to 10 (severe rickets),9,10 based on specific pathological features, including metaphyseal margin irregularities, cupping, fraying, increased lucency, and widening of the growth plate at the distal radius and ulna, distal femur, and proximal tibia. 10 The RSS of each patient was recorded at baseline and again at 12 and 24 months following initiation of burosumab therapy as a time-point analysis to assess changes over time.
Second, lower limb alignment was assessed by measuring mechanical axis deviation (MAD) and frontal alignment angles on full-length standing radiographs, following a standardized protocol (Figure 2). Proper rotational alignment was confirmed using patellar positioning between the femoral condyles as a consistent anatomical reference point. 11 The MAD was defined as the perpendicular distance (in millimeters) from a line drawn from the center of the femoral head through the center of the talar dome, relative to the center of the femoral condyles (Figure 2).12–15 All radiographs were obtained with a 50 mm calibration marker placed at the knee level to ensure accuracy in absolute measurements. Frontal alignment angles included the mechanical lateral distal femoral angle (mLDFA), medial proximal tibial angle (MPTA), and lateral distal tibial angle (LDTA; Figure 2). The mLDFA was defined as the lateral angle between the mechanical axis of the femur and the distal femoral joint line.13,15 The MPTA was defined as the medial angle formed between the mechanical axis of the tibia and a line drawn along the tibial plateau. 13 The LDTA was defined as the lateral angle between the tibial mechanical axis and the distal tibial articular surface in the frontal plane. 15 To account for age- and sex-related differences, MAD and frontal alignment angles were converted to z-scores based on normative data from Sabharwal et al. and Popkov et al.16,17 Thresholds of z ≤ −2.0 and z ≥ +2.0 were used to define varus and valgus deformities, respectively, for z-MAD and z-MPTA. For z-mLDFA and z-LDTA, the interpretation was reversed, with z ≥ +2.0 indicating varus and z ≤ −2.0 indicating valgus deformity. For statistical analysis, absolute z-scores (|z|) were used to quantify the extent of deviation from normative values, irrespective of direction.
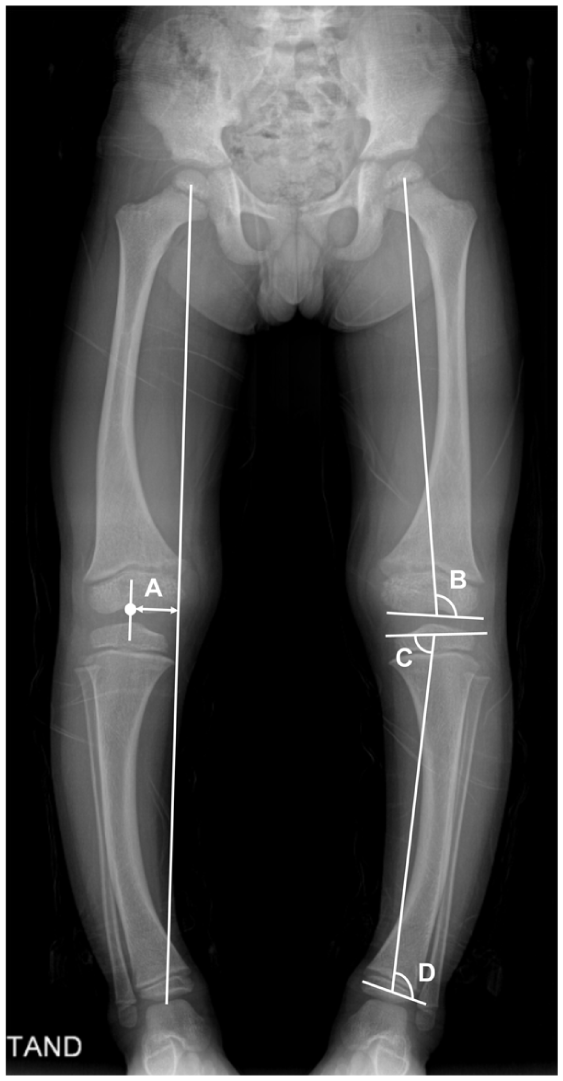
Standardized protocol for lower limb alignment assessment on full-length standing radiographs. (A) MAD was defined as the perpendicular distance (in millimeters) from a line drawn from the center of the femoral head through the center of the talar dome, relative to the center of the femoral condyles. (B) mLDFA was defined as the lateral angle between the mechanical axis of the femur and the distal femoral joint line. (C) MPTA was defined as the medial angle formed between the mechanical axis of the tibia and a line drawn along the tibial plateau. (D) LDTA was defined as the lateral angle between the tibial mechanical axis and the distal tibial articular surface in the frontal plane.
Third, standing height was assessed at routine outpatient visits using a wall-mounted stadiometer. Patients were measured barefoot, with heels together, arms resting at their sides, and the head positioned according to the Frankfort horizontal plane. 18 A trained nurse facilitated each measurement to ensure standardized posture and reduce inter-visit variability. To account for age- and sex-related differences, height measurements were converted into z-scores based on normative data from the National Growth Charts. 19 These z-scores were subsequently used for statistical analysis.
All radiographs were obtained with a 50 mm calibration marker placed at the knee level to ensure accuracy in absolute measurements.
Statistical analyses
To assess inter-rater reliability, radiographic measurements were independently performed by an experienced pediatric orthopedic surgeon and a final-year medical student. The intraclass correlation coefficient (ICC) was calculated using a two-way random-effects model with absolute agreement. ICC values were interpreted as follows: <0.5, poor; 0.5–0.75, moderate; 0.75–0.9, good; and >0.9, excellent reliability.
All statistical analyses were conducted based on the distributional characteristics and data structure of each variable. For variables measured once per patient, such as the RSS and standing height, non-parametric tests were used due to violations of parametric assumptions. Changes across the three time points (baseline, 12 months, and 24 months) were assessed using the Friedman test, followed by Wilcoxon signed-rank tests with Bonferroni correction (adjusted α = 0.017) for post hoc comparisons. For lower limb alignment parameters derived from bilateral measurements, a linear mixed-effects model was employed to account for repeated, non-independent observations (i.e., bilateral limbs per patient). Time was modeled as a fixed effect and patient as a random effect. When a significant overall time effect was observed, post hoc pairwise comparisons between time points were performed with Bonferroni correction (adjusted α = 0.017).
Continuous variables are presented as mean ± standard deviation. All statistical analyses were conducted using IBM SPSS Statistics for Windows, version 29.0 (IBM Corp., Armonk, NY, USA), and two-tailed p-values <0.05 were considered statistically significant unless otherwise specified.
Results
Patient baseline characteristics
A total of 20 patients (10 males and 10 females) were included. The mean age at the initiation of burosumab therapy was 7.5 ± 2.4 years (range, 2.7–11.7 years), with a mean follow-up duration of 24.3 ± 0.3 months. At baseline, the mean RSS was 3.5 ± 1.2, and the mean standing height z-score was −1.5 ± 0.8.
Based on the z-score thresholds (≤ −2.0 indicating varus and ≥ +2.0 indicating valgus for MAD and MPTA, with the interpretation reversed for mLDFA and LDTA), 11 limbs (27.5%) were classified as having varus alignment and 9 limbs (22.5%) as valgus based on MAD, while the remaining 20 (50%) fell within the acceptable alignment range. For mLDFA, 9 limbs (22.5%) showed varus, 8 (20%) valgus, and 23 (57.5%) were within the acceptable range. MPTA demonstrated a stronger varus tendency, with 12 limbs (30%) in varus and only 2 (5%) in valgus. The most pronounced deviation was seen in LDTA, where 17 limbs (42.5%) exhibited varus alignment, 2 (5%) valgus, and 21 (52.5%) fell within the acceptable alignment range.
Rickets Severity Score
Inter-rater reliability of the RSS measurement was excellent (ICC = 0.93; 95% confidence interval (95% CI) 0.88–0.96). Changes in RSS over time showed a significant difference across baseline, 12 months, and 24 months (χ²(2) = 34.563, p < 0.001; Figure 3). Post hoc pairwise comparisons demonstrated statistically significant reductions in RSS, indicating radiographic improvement, across all intervals: baseline to 12 months (p < 0.001), 12 to 24 months (p = 0.002), and baseline to 24 months (p < 0.001).
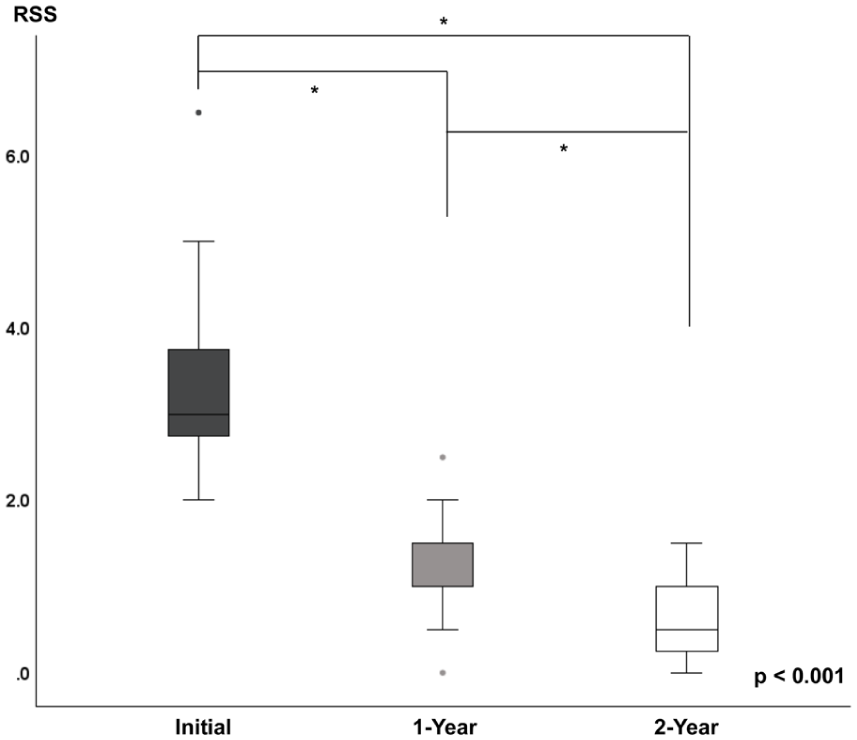
Changes in RSS at baseline, 12 months, and 24 months after initiation of burosumab therapy. Asterisks (*) indicate statistically significant differences between time points based on Bonferroni-corrected pairwise comparisons (p < 0.017).
Lower limb angular deformities
Inter-rater reliability for lower limb alignment measurements was excellent. The ICCs were 0.991 (95% CI 0.983–0.995) for MAD, 0.989 (95% CI 0.980–0.994) for mLDFA, 0.988 (95% CI 0.977–0.994) for MPTA, and 0.992 (95% CI 0.984–0.996) for LDTA.
Among the alignment parameters, |z|-MAD and |z|-mLDFA showed statistically significant changes across the three time points (Table 1 and Figure 4). Post hoc pairwise comparisons revealed that all time points differed significantly for |z|-MAD (all p < 0.017), and significant differences were also observed for |z|-mLDFA between baseline and 24 months (p < 0.001) and between 12 and 24 months (p = 0.002). While |z|-MPTA and |z|-LDTA showed overall significant changes over time (p = 0.020 and p = 0.016, respectively), pairwise comparisons did not consistently meet the Bonferroni-adjusted threshold (α = 0.017), except for |z|-LDTA, in which only the comparison between baseline and 24 months reached statistical significance (p = 0.014).
Longitudinal changes in radiographic and clinical parameters.
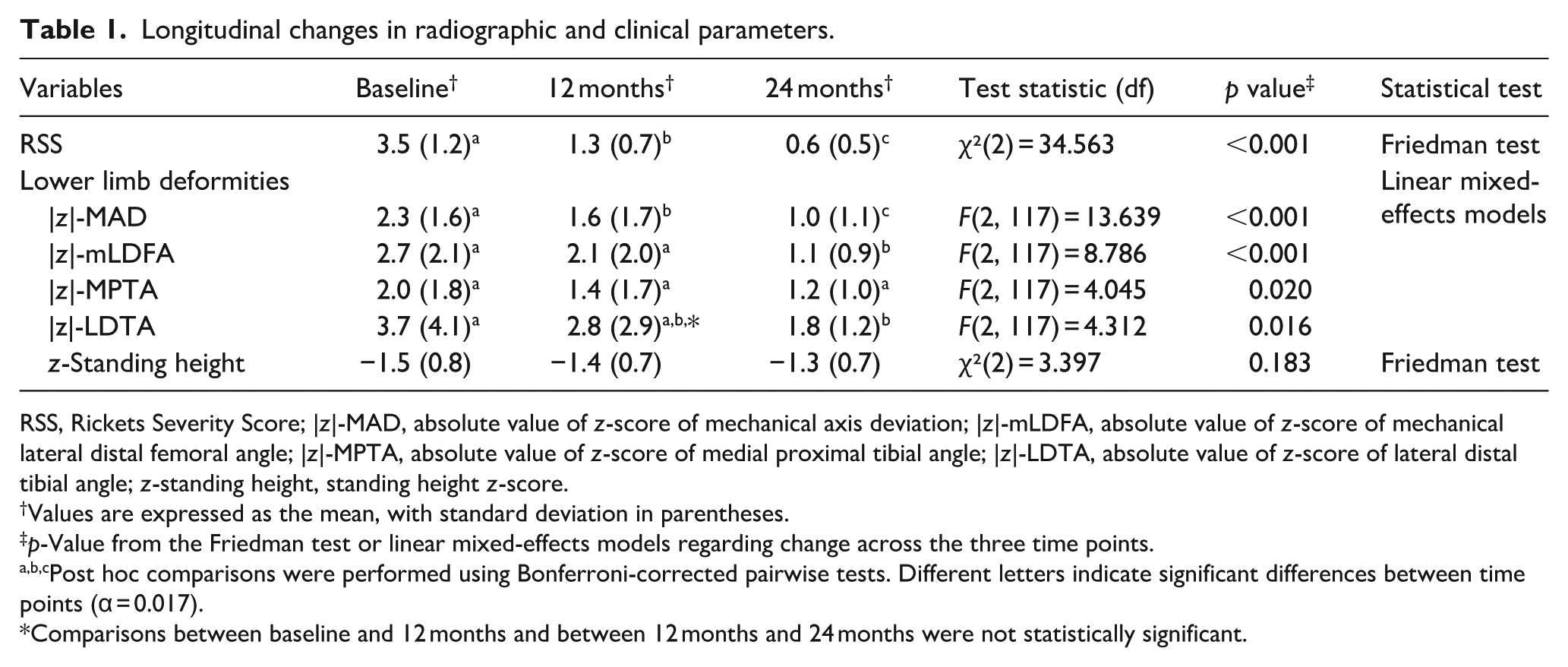
RSS, Rickets Severity Score; |z|-MAD, absolute value of z-score of mechanical axis deviation; |z|-mLDFA, absolute value of z-score of mechanical lateral distal femoral angle; |z|-MPTA, absolute value of z-score of medial proximal tibial angle; |z|-LDTA, absolute value of z-score of lateral distal tibial angle; z-standing height, standing height z-score.
Values are expressed as the mean, with standard deviation in parentheses.
p-Value from the Friedman test or linear mixed-effects models regarding change across the three time points.
Post hoc comparisons were performed using Bonferroni-corrected pairwise tests. Different letters indicate significant differences between time points (α = 0.017).
Comparisons between baseline and 12 months and between 12 months and 24 months were not statistically significant.
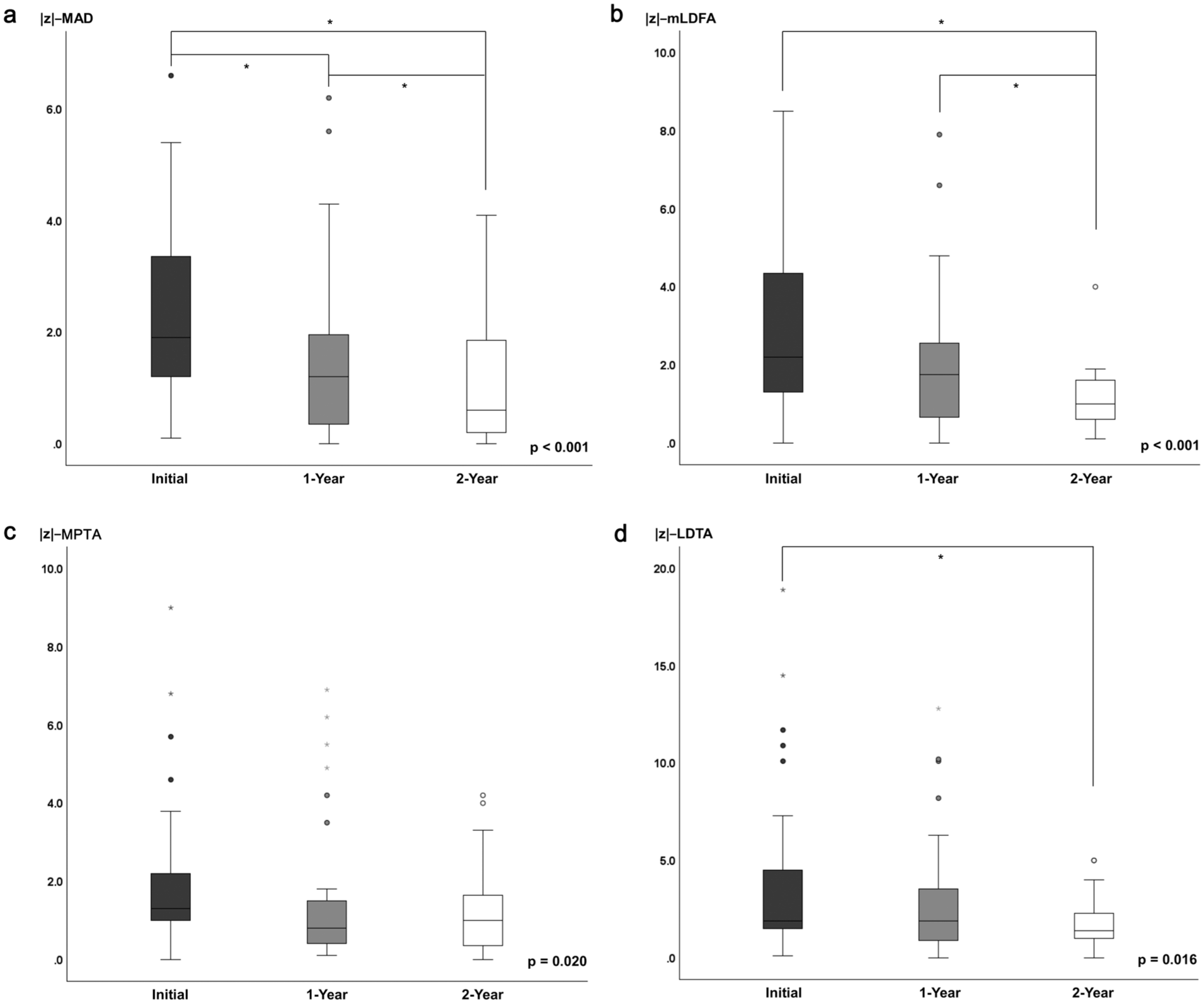
Changes in lower limb alignment parameters over the 2-year treatment period. Asterisks (*) indicate statistically significant differences between time points based on Bonferroni-corrected pairwise comparisons (p < 0.017). (a) |z|-MAD. (b) |z|-mLDFA. (c) |z|-MPTA. (d) |z|-LDTA.
A representative case is shown in Figure 5, demonstrating progressive correction of frontal plane deformities over 2 years of burosumab therapy in a 3-year-old girl.
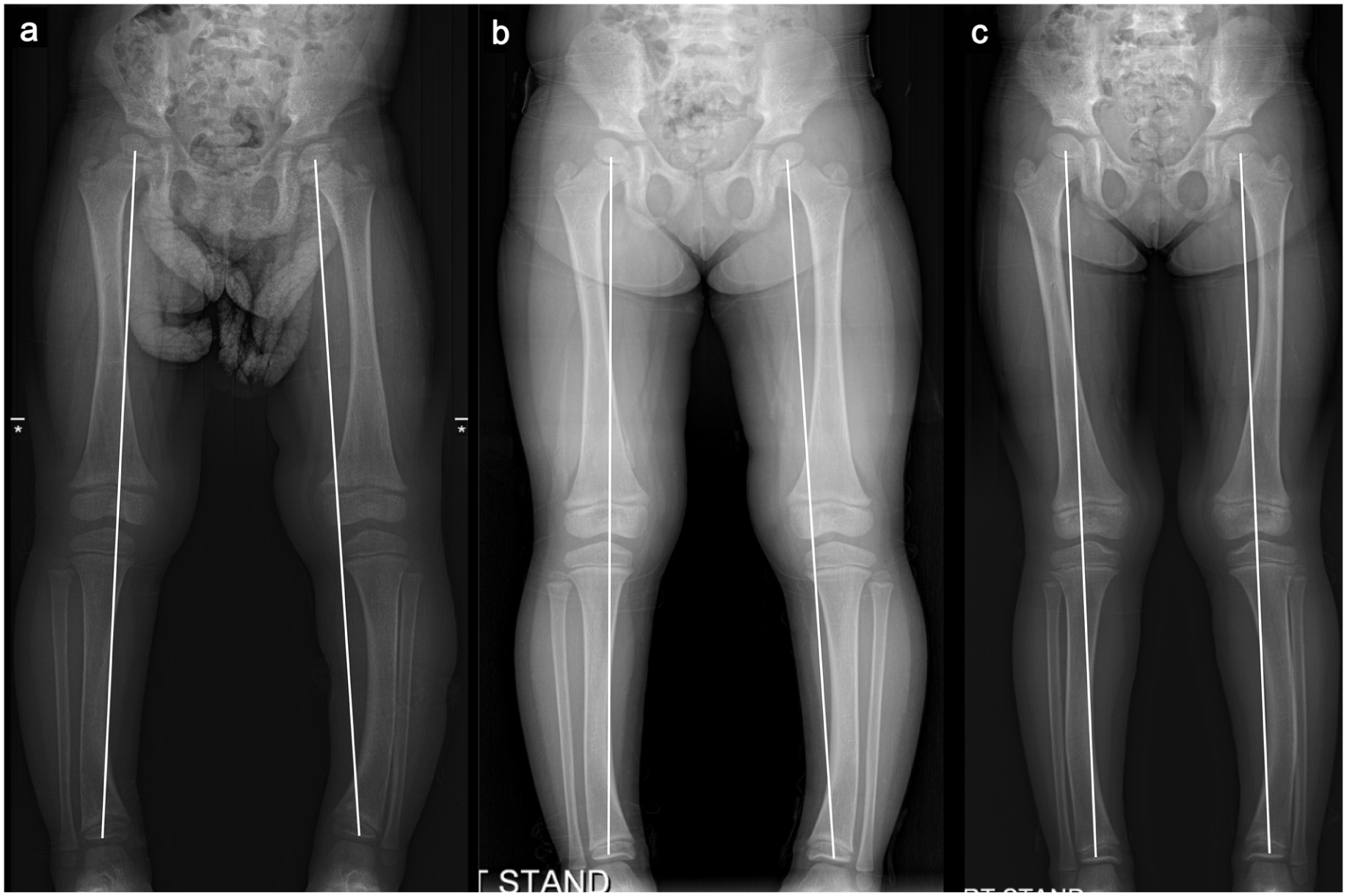
Serial full-length standing radiographs of a 3-year-old girl with X-linked hypophosphatemia at (a) baseline, (b) 1 year, and (c) 2 years after initiating burosumab therapy, demonstrating progressive correction of frontal plane deformities in both lower limbs, most notably on the left side from marked varus (−17 mm) to neutral.
Standing height z-scores
There were no significant differences in standing height z-scores across the three time points (χ²(2) = 3.397, p = 0.183; Figure 6). Accordingly, no statistically significant changes in standing height were observed over 24 months following burosumab initiation.
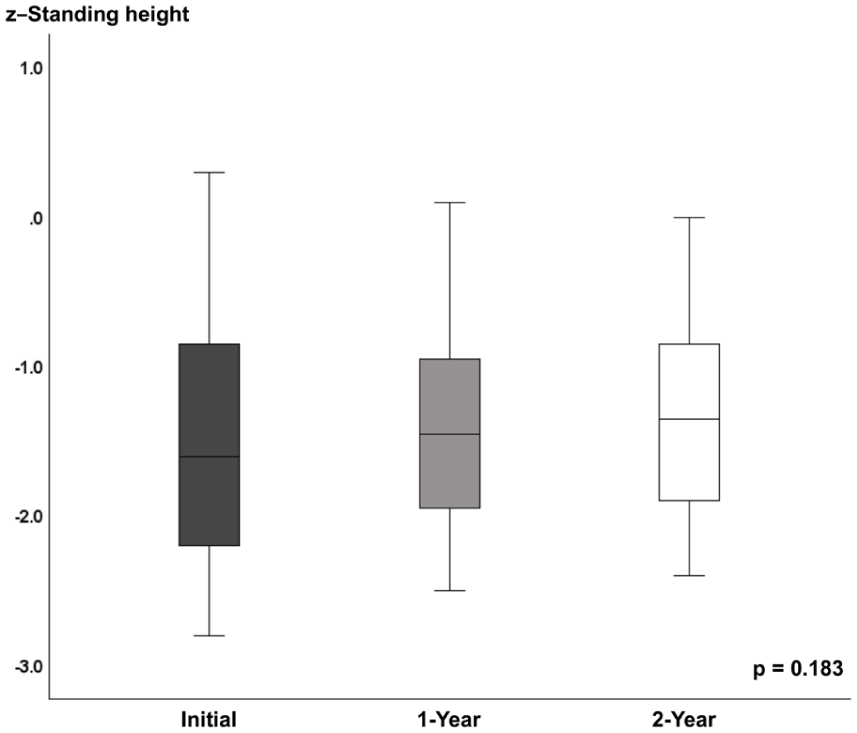
Changes in standing height z-scores at baseline, 12 months, and 24 months.
Discussion
This study evaluated the 2-year effects of burosumab on lower limb skeletal outcomes in pediatric patients with XLH. Significant improvements were observed in rickets severity and lower limb alignment parameters, particularly in MAD and mLDFA. While MPTA and LDTA showed overall significant changes, pairwise comparisons did not consistently reach statistical significance. No significant change was observed in standing height.
The RSS is a validated and widely used tool for assessing radiographic severity in rickets. In our study, burosumab therapy resulted in a significant and sustained reduction in RSS over a 2-year follow-up period. Similar improvements have been reported by Wei et al., Imel et al., and Carpenter et al. supporting the reproducibility of this outcome across diverse pediatric populations.6,20,21 Burosumab is known to restore bone mineralization by suppressing excess FGF23, 22 and it is presumed that similar changes may occur at the growth plate, contributing to the observed improvement in RSS. These findings reinforce the therapeutic role of burosumab in improving skeletal manifestations of rickets in children with XLH.
In terms of the longitudinal effect of burosumab on lower limb angular deformities, we found statistically significant improvements in alignment, particularly in MAD and mLDFA. Although MPTA and LDTA showed significant overall effects over time, pairwise comparisons did not consistently meet the corrected significance threshold. These findings partially align with those of Sawamura et al., who reported improvements or stabilization in lower limb alignment, including mLDFA and mLDTA, after 12 months of burosumab therapy in five children with XLH. 7 Notably, in their study, mMPTA did not show statistically significant improvement, which contrasts with our finding of an overall significant effect in |z|-MPTA. This discrepancy may be attributed to differences in sample size, follow-up duration, and statistical methodology. In particular, the application of z-score standardization in our study may have provided a more age-adjusted and sensitive measure of alignment changes over time.
While MPTA and LDTA exhibited significant overall changes across time points, consistent statistical significance was not observed in all pairwise comparisons. Nonetheless, the distribution of values—including outliers—tended to converge toward the normative range over the study period, suggesting a general trend toward radiographic improvement. This observation aligns with the findings of Frumberg et al., who demonstrated that prolonged burosumab therapy (up to 160 weeks) led to progressive correction of mechanical femorotibial angles and an increasing proportion of limbs achieving age-specific normal alignment. 8 As restoration of growth plate function and subsequent remodeling are likely prerequisites for the correction of alignment, a longer treatment duration may be necessary. Their results support the concept that extended therapy, and by longer follow-up, may be necessary to achieve meaningful correction without surgical intervention.
Nevertheless, surgical correction should not be entirely ruled out in managing skeletal deformities in patients with XLH during the burosumab era. As reported by Mindler et al., guided growth or derotational osteotomy was performed in five of seven patients during burosumab therapy due to the need for timely correction or functional impairment. 5 Similarly, 5 of the 25 initially enrolled in our study underwent surgery during the treatment period and were excluded from the analysis. Surgical intervention was mainly indicated for persistent torsional deformities; however, in some patients with pronounced frontal malalignment at baseline, burosumab therapy alone was unlikely to achieve sufficient correction within the treatment period, necessitating guided growth. These findings suggest that torsional deformities may be less responsive to medical therapy, often requiring mechanical intervention for satisfactory alignment. Notably, the rate of correction following guided growth under burosumab therapy was comparable to, or even faster than, that observed in healthy children. 5 In addition, osteotomy sites achieved union within 6–8 weeks, suggesting a potentially beneficial effect of burosumab on bone healing. 5 Similarly, in our experience, guided growth proceeded uneventfully during burosumab therapy, and no cases of delayed union were observed following osteotomy.
Our study, which included patients with a mean age of 7.5 years, did not demonstrate a significant improvement in standing height z-scores over the 2-year follow-up period, from −1.5 ± 0.8 to −1.3 ± 0.7. By contrast, Imel et al. reported a greater increase in height z-scores in children treated with burosumab, particularly in younger patients with a mean age of 5.8 years. 20 Although the therapeutic benefits of burosumab have generally been thought to be more pronounced in younger patients during periods of active linear growth,3,7,23 both cohorts were within a similar pediatric age range in which the growth plates remain open and responsive. Therefore, the discrepancy in height growth responses is unlikely to be explained solely by age. Instead, it may reflect other factors, such as individual variation or the extent of baseline frontal plane deformities.
Our findings support the hypothesis that skeletal improvements in children treated with burosumab occur in a sequential pattern. Initially, normalization of serum phosphate levels enhances mineralization at the growth plate, as reflected by early improvements in the RSS. This mineralization likely facilitates the correction of physeal abnormalities and promotes more physiological growth dynamics, ultimately enabling the remodeling of angular deformities. In our cohort, alignment improvements were most prominent in the distal femur, as evidenced by significant changes in mLDFA. Additional trends toward improvement were observed in MPTA and LDTA. These angular corrections were accompanied by a clear and consistent improvement in overall limb alignment, as reflected in MAD. While rickets severity appeared to improve early in the treatment course, the correction of frontal plane deformities and subsequent linear growth may require a longer duration. Linear growth may follow limb alignment correction. Consistent with this sequence, no significant increase in standing height was observed during the study period, supporting the idea that linear growth may lag behind skeletal correction. Growth response may depend on factors such as age at treatment initiation and the baseline severity of rickets.
This study has several limitations. First, its retrospective design limited control over data collection, introducing the potential for information bias. In addition, irregular follow-up intervals may have affected the accuracy of longitudinal assessments. Second, this study focused on skeletal and radiographic outcomes; biochemical markers, including FGF23 or serum phosphate, were not assessed as part of this orthopedic-focused retrospective analysis. Third, five patients who underwent surgery during treatment were excluded. Although this may have resulted in a relative underrepresentation of more severe deformities, the exclusion was deemed necessary to more accurately isolate the pharmacological effects of burosumab. Fourth, angular parameters (mLDFA, MPTA, and LDTA) were measured using the mechanical axis rather than separate anatomic axes for each segment. It may distort proximal or distal angles in cases with diaphyseal deformity, but this approach provides a consistent mechanical reference for overall limb alignment. Lastly, rotational alignment was not formally assessed, as pre-treatment CT scans were not routinely performed. This limitation precluded an objective evaluation of rotational deformities. Nevertheless, the need for derotational procedures in some patients suggests that rotational correction may be a potential area for further investigation. Despite these limitations, the study is strengthened by its 2-year follow-up duration and the inclusion of a relatively large cohort. Inter-rater reliability was excellent, supported by independent radiographic measurements conducted by both a pediatric orthopedic surgeon and a senior medical student. Furthermore, the use of age-standardized z-scores for lower limb alignment allowed for a more physiologically appropriate interpretation across a pediatric population with wide age variability. To address these limitations, future studies should prioritize prospective designs with early initiation of treatment and standardized follow-up intervals to improve data quality and enhance the external validity of findings.
In conclusion, burosumab therapy may contribute to improvements in skeletal deformities in pediatric patients with XLH. It led to a rapid reduction in rickets severity and overall improvement in lower limb alignment, with particularly notable changes in the distal femur as reflected by improvements in mLDFA. These alignment corrections were accompanied by corresponding improvements in MAD. However, no meaningful change was observed in linear growth over the study period. Longer-term follow-up beyond 2 years is warranted to draw more definitive conclusions.
Supplemental Material
sj-pdf-1-cho-10.1177_18632521251398403 – Supplemental material for Two-year structural skeletal outcomes of burosumab therapy in pediatric X-linked hypophosphatemia: A radiographic cohort study
Supplemental material, sj-pdf-1-cho-10.1177_18632521251398403 for Two-year structural skeletal outcomes of burosumab therapy in pediatric X-linked hypophosphatemia: A radiographic cohort study by Hayeon Lim, Wonik Lee, Naye Choi, Yun Jeong Lee, Chang Ho Shin, Young Ah Lee, Jung Min Ko and Mi Hyun Song in Journal of Children's Orthopaedics
Footnotes
Acknowledgements
The authors gratefully acknowledge the contributions of Dr. Tae-Joon Cho (Division of Pediatric Orthopedics, Seoul National University Children’s Hospital), Dr. Yo Han Ahn, and Dr. Choong Ho Shin (Department of Pediatrics, Seoul National University Children’s Hospital) for their assistance with patient care and data collection. Their support was valuable to the completion of this study, although they do not meet the criteria for authorship. The authors also thank the Medical Research Collaborating Center (MRCC) for providing statistical consultation and verifying the appropriateness of the analytical methods used in this study.
Author contributions
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical statement
This retrospective study was approved by the Institutional Review Board (IRB No. H-2505-140-1643). Informed consent was waived because of the retrospective nature of the study, and the analysis used anonymous clinical data.
Data availability statement
The data supporting this study are not publicly available due to patient privacy and ethical restrictions. De-identified data may be available from the corresponding author upon reasonable request and with appropriate institutional approvals.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
