Abstract
Background:
The glymphatic system, a relatively recently discovered system for transport of cerebrospinal fluid (CSF) and waste products in the central nervous system, has been implicated in several neurologic disorders. Altered CSF dynamics are seen after decompressive craniectomy, suggesting this procedure may impact glymphatic function. We aimed to systematically review the current literature to understand the effect of the glymphatic system on patients with decompressive craniectomies and Syndrome of the Trephined (SoT).
Methods:
PubMed and Embase were searched for preclinical and clinical studies investigating the glymphatic system and craniectomy, cranioplasty, or SoT in years 2012 to 2023.
Results:
Sixty-three studies were identified for review. Intracranial oscillations, cerebral perfusion, and arterial pulsations have been identified as major drivers of glymphatic influx. Each of these processes are diminished following craniectomy, suggesting craniectomy may impair glymphatic function. Reductions in brain pulsatility and alterations in glymphatic drainage after craniectomy were accompanied by impaired functional outcomes, as seen in SoT. Radiologically, CSF flow alterations are seen after craniectomies, which improve with cranioplasty.
Conclusions:
Glymphatic system dysfunction after craniectomy and in SoT may be a potential therapeutic target for patients with iatrogenically altered cranial vault anatomy.
Introduction
The classic “third circulation” model of cerebrospinal fluid (CSF) flow proposed by Harvey Cushing holds that CSF is produced in the choroid plexus, circulates through the ventricles and subarachnoid space (SAS), and is reabsorbed into the venous system through the arachnoid villi.1,2 However, advancements in research over the past 100 years have challenged the simplicity of this view. 2 Among these findings is the glymphatic system, a recently revisited pathway in which perivascular channels transport CSF out of the brain to aid in clearance of neurological waste products, macromolecules, and immune cells.
Perivascular spaces, space surrounding blood vessels as they penetrate the brain parenchyma, were initially described by Durand-Fardel, Pestalozzi, Virchow, and Robin in the mid-1800s.3,4 The potential role of these spaces in lymphatic drainage was proposed in the late 19th century and, by the early 20th century, Mott, Weed, and others proposed the concept of perivascular fluid flow. 3 Subsequent studies by Cserr et al 5 and the Rennels et al 6 demonstrated that CSF from the SAS could enter the brain parenchyma along perivascular spaces, enter the extracellular space, and flow out via perivascular spaces. Nedergaard et al connected and extended these concepts in a landmark 2012 study. 7 They found that fluorescent tracers in the CSF traveled with penetrating arterioles into the brain parenchyma through an astrocyte-dependent sieve and were cleared via perivenous spaces. Based on its similarity in function to the lymphatic system and dependence on glial cells, this pathway was termed the glymphatic system.
The glymphatic system has garnered much attention over the past decade. Studies have suggested its role in the removal and delivery of important substances to the brain.7-13 As such, glymphatic dysfunction has been implicated in several disorders, including Alzheimer’s disease, traumatic brain injury, and stroke.13-16 With improved medical management, there has been increased utilization of decompressive craniectomies after trauma or stroke to reduce intracranial pressures.17,18 Recently, there is increasing interest in applying the glymphatic system to understanding complications after craniectomies and in Syndrome of the Trephined (SoT). Decompressive craniectomy has been shown to cause alterations in CSF flow dynamics, suggesting this procedure may impair glymphatic circulation.14,19 This systematic review aims to describe the anatomy and physiology of the glymphatic system and examine glymphatic dysfunction after craniectomy and in SoT.
Methods
A comprehensive search of PubMed and Embase was performed on 5/29/2023, adhering to PRISMA guidelines, with queries of “(glymphatic)” AND ((“syndrome of the trephined”) OR (“sinking skin flap syndrome”)), (glymphatic) AND ((craniotomy) OR (craniotomies) OR (craniectomy) OR (craniectomies)), (glymphatic) AND ((cranioplasty) OR (cranioplasties) OR (cranioplastics)), and “glymphatic” [Title]. This yielded 446 articles from 2012 to 2023. The abstracts were reviewed by one reviewer. Articles were excluded if there was no full-text article available in the English language. Review papers and book chapters were also excluded. Articles were included if they were peer-reviewed articles of interventional studies, observational studies, case reports, case series, or animal studies investigating the anatomy and/or physiology of the glymphatic system or exploring the relationship between the glymphatic system and craniectomy, cranioplasty, or SoT. Studies exclusively investigating the relationship between the glymphatic system and other disorders (eg, Alzheimer’s disease, traumatic brain injury) were excluded. These criteria yielded 63 studies for review, of which 8 explored the relationship between the glymphatic system and craniectomy, cranioplasty, or SoT.
Anatomy and Physiology
The traditional understanding of CSF flow and physiology states that a majority, approximately 80%, of CSF is produced in the cerebral ventricles by the choroid plexus, with the remainder produced by other structures such as the brain parenchyma.20,21 CSF flows from the ventricles to the SAS and, based on early animals models, was primarily thought to exit the intracranial SAS through arachnoid villi into the large dural venous sinuses.1,22,23 Yet even in those early models, evidence of CSF tracking within the perivascular spaces questioned the amount of attention paid to arachnoid granulations and laid the groundwork for a separate CSF lymphatic pathway.22,23 From the SAS, CSF can enter the glymphatic system to interface with the brain parenchyma or can egress from the intracranial and intraspinal SAS directly into the peripheral circulation via separate meningeal lymphatics, perineuronal pathways, parasagittal spaces, arachnoid granulations, or adventitia of large cerebral vessels (Figure 1). 20
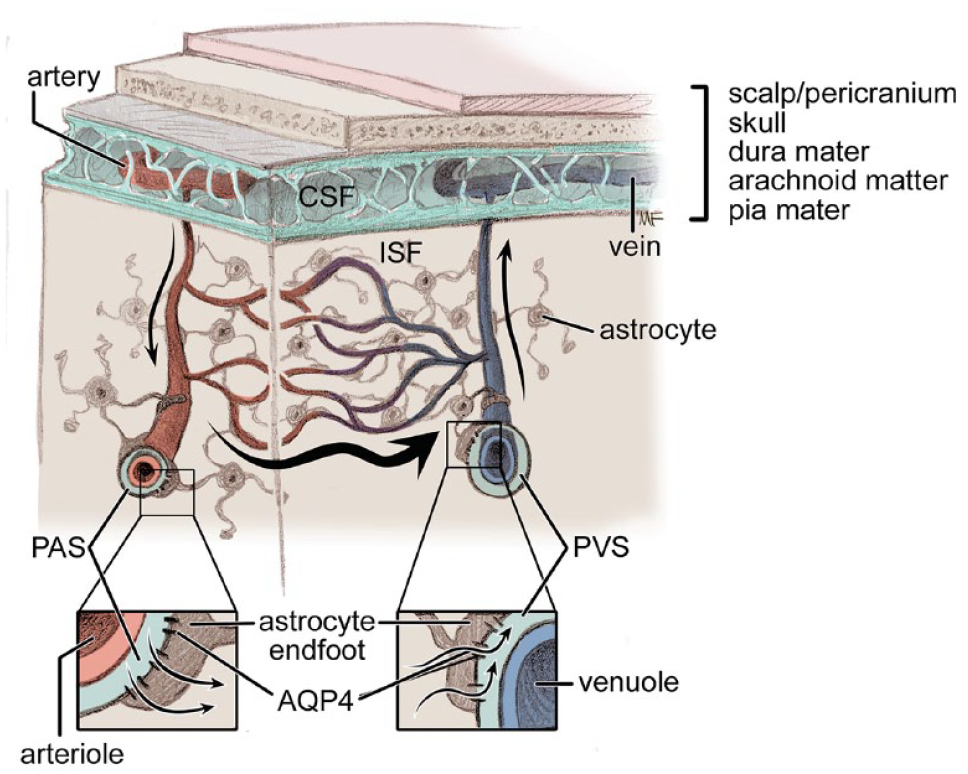
Fluid movement through the glymphatic system. CSF moves from the subarachnoid space into the perivascular spaces around arterioles. CSF then moves into the brain parenchyma with the help of AQP4 on astrocyte end-feet. From the parenchyma, CSF and interstitial fluid move toward the perivenous spaces, carrying metabolic waste, ultimately exiting along major draining veins and can exit via the meningeal lymphatic system.
Glymphatic Influx
Following the Nedergaard group’s initial study, 7 several rodent and human studies have tracked the course of fluorescent tracers injected into the CSF. In these studies, CSF tracers traveled alongside large cerebral arteries running toward the cortical surface of the brain within the SAS.7,9,20,24-28 Iliff et al. found rapid movement of intrathecally injected tracer along arteries and arterioles, but not along veins or venules in a mouse model.7,29 They found the periarterial space then continues to narrow after entering the parenchyma until it is continuous with the basement membrane of capillaries. Evidence of paravascular flow of fluid is not limited to animal models. 30 Utilizing intrathecal injection of gadobutrol tracer in humans, the tracer was present early in the brain tissue, most prominently in brain tissue around major cerebral arteries. 24 After 24 hours, there was a more global distribution of the tracer in the parenchyma, indicating the tracer moved from the large paravascular spaces to smaller perivascular spaces and ultimately the interstitial space. The mechanism of entry into the paravascular space is not well understood but may be aided by brain pulsatility. Microspheres injected into the CSF were seen to travel alongside branches of the middle cerebral artery in concordance with the cardiac cycle, suggesting a role of arterial pulsatility in cortical CSF flow. 31
From the SAS, CSF travels into the brain parenchyma in perivascular spaces known as Virchow-Robin spaces.7,32-35 Flow in these spaces is fast and interestingly does not seem to depend on molecular size.36-39 Once fluid reaches the end of the perivascular space, the mechanism of entry into the interstitium is not well understood, but probably relies on astrocytes.34,37-41 Iliff et al. proposed that CSF then enters the brain interstitium via narrow clefts in the astrocyte end-feet as part of the neurovascular complex, which would act like a sieve for molecules. In this case molecules would enter the interstitium in a size- and structure-dependent manner. 7 This is not universally accepted as the major mechanism of CSF influx into the interstitial space, but an in-depth discussion of the basic science pathways are outside the scope of this review.42,43
Astrocytes appear to play a key role in the glymphatic system. Astrocytic end-feet provide near complete coverage of the capillaries in the brain and are an essential part of the blood-brain barrier.35,42 These end-feet express high levels of water-specific aquaporin-4 (AQP4) channels on their plasma membranes and are implicated in fluid transport between the extracellular and perivascular spaces.7,35,42 Fluid can also move between the perivascular space and the interstitial space in a paracellular fashion, around the astrocytic end-feet, which is indirectly facilitated by AQP4. Deletion of AQP4 in mice resulted in virtually no movement of fluorescent tracers from the perivascular space into the interstitial space and an overall decrease in clearance of interstitial solutes, with preservation of periarterial CSF flow. 7 Similar impairment of the glymphatic system was seen in mice with intact AQP4 but with loss of polarization, highlighting the importance of its perivascular location. 26 Subsequent work by the Nedergaard group and several other groups have supported these findings.8,10-12,26,44-47 The role of AQP4 was disputed by one study, 48 but a recent meta-analysis of these preclinical studies supports the role of AQP4 in normal CSF influx and interstitial fluid clearance. 26
Glymphatic Efflux
After fluid is exchanged between the CSF and interstitial fluid (ISF), ISF exits the brain via perivenous spaces. Ex vivo evidence demonstrated CSF tracers rapidly exited the brain along large midline and lateral veins. 7 Additional studies have similarly shown tracers exiting the parenchyma in perivenous spaces, highlighting the role of these pathways in ISF efflux.7,26,28,34,49 Fluid within the perivenous space can then exit the cranial compartment via separate meningeal lymphatic vessels lining the venous sinuses, along cranial and spinal nerve sheaths, or across the cribriform plate.26,49,50 Lymphatic drainage may play a more significant role than it has historically been given credit for. The meningeal lymphatic system is formed by lymphatic channels within the dura mater and exit along arteries, veins, and nerves primarily at the skull base. The meningeal lymphatic system therefore contributes to efflux of waste from the glymphatic system to the lymph nodes. 51 Deep and cervical lymph nodes have been identified as major drainage sites for tracers injected intrathecally in humans and cisternally in rodents.49,52 In rabbits, 50% of a radioiodinated tracer injected into the brain parenchyma drained along perivascular spaces and cleared to the deep cervical lymphatics. 53 Additionally, 14% to 47% of radiolabeled albumin injected into brain parenchyma or CSF passed through the lymphatic system in other animal models. 54 Efflux through the glymphatic system is a growing area of research and may contest the superiority of arachnoid granulations.
Factors Affecting Flow Through Glymphatic System
Arterial pulsations are a potential driver of CSF flow and thus flow through the glymphatic system (Figure 2). Arterial pulsations were shown to drive fluid flow along perivascular spaces in as early as the 1970s. 6 Since then, CSF flow in the perivascular space has been visualized to change in synchrony with the cardiac cycle, with displacement of the arterial wall during the cardiac pulse wave matching changes in CSF movement.36,55,56 Factors which reduced vessel pulsatility, such as ligation, hypercapnia, or vasoconstriction, reduced CSF influx into the interstitial space and reduced clearance of interstitial solutes indicating glymphatic dysfunction. 57 Similarly, increasing arterial pulsatility with dobutamine improved CSF influx. 45 The mechanism through which arterial pulsation enhances glymphatic flow is not well understood. It has been postulated that arterial pulsation mechanically propels CSF through the perivascular space and through AQP4 channels. 55 However, a fluid flow model mimicking arterial pulsations failed to enhance CSF transport into the parenchyma, indicating that arterial pulsations may improve perivascular flow, but bottlenecks at the level of the astrocytes. 38 Inspiration is another important driver of CSF flow. 58 The negative intrathoracic pressure during inspiration promotes venous outflow and may improve glymphatic drainage by increasing the perivenous space. 36 Imaging studies demonstrated respirations induced fluid flow changes in the perivenous areas in the brain in humans. 55 The cardiac and pulmonary systems therefore may be potent drivers of periarterial and perivenous flow of the glymphatic system, respectively.
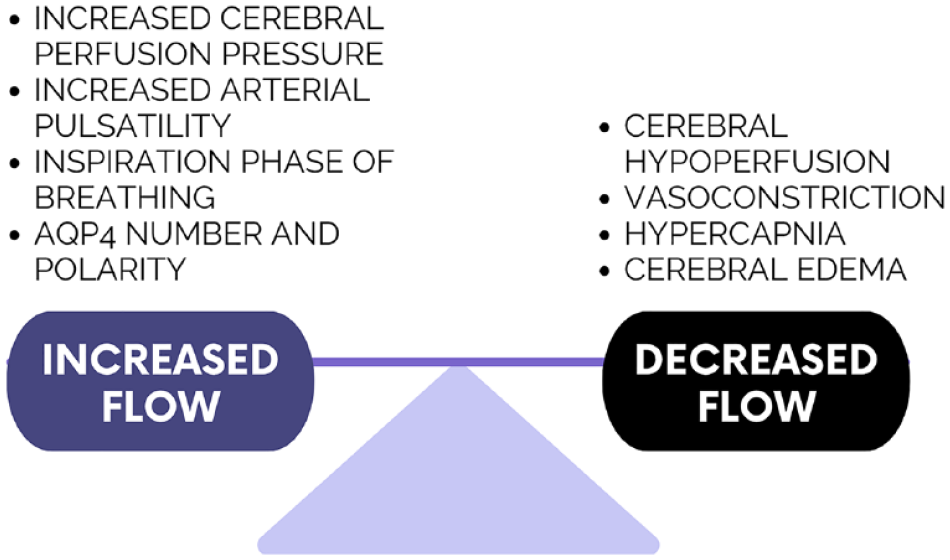
Diagram illustrating factors that increase and decrease flow of fluid through the glymphatic system.
Several other factors may also affect glymphatic flow. Cerebral hypoperfusion was found to reduce glymphatic flow through depolarization of AQP4 channels, whereas drugs that improved perfusion, such as digoxin and nimodipine, improved glymphatic flow.13,59 Sleep,15,60 anesthetics, 61 posture, 55 stress, 62 and age52,61,62 appear to affect glymphatic function as well, but differences in sex/gender have shown mixed results.60,61
Dysfunction of Glymphatic System in Craniectomy and Syndrome of the Trephined
Decompressive craniectomy (DC) is a common surgical procedure utilized to control intracranial pressure (ICP) and prevent brain herniation.14,19,63 While effective at attenuating ICP acutely, chronically it carries a risk of conditions related to abnormal CSF circulation, such as hygromas, hydrocephalus, and SoT. 64 SoT is a constellation of symptoms, including motor and cognitive deficits, which develop after craniectomy, thought to be due to brain compression when exposed to atmospheric pressure. 65 There are a number of hypotheses about the cause of brain injury in SoT. Some think the atmospheric pressure causes mass effect and mechanical compression of the brain, whereas others believe that the atmospheric pressure causes irritation and gliosis of the underlying cortex leading to neurologic deficits.19,66
In a closed cranial vault, vascular pulsations lead to cyclical changes in cerebral blood flow (CBF) and subsequent pulsatile movements of the brain and brainstem which act like a piston or a pump. These pulsatile movements have been shown to enhance CSF flow and mixing. 64 Additionally, the pulsation of the soft brain against the fixed skull is thought to compress the ventricles, further promoting flow of CSF. DC creates a compliant cranial defect, thereby decreasing CBF-induced cranial pulsations and ventricular squeezing. 64 The CSF stagnation, reduced arterial pulsatility, and diminished CBF seen after DC can be restored with cranioplasty.14,64,67 Clinically, this improvement in intracranial homeostasis is seen after cranioplasty as hygromas, brain shift, and hydrocephalus improve with restoration of the rigid, completed cranial vault.64,68,69 In the neurosurgical literature, there has been recent interest in early and ultra-early cranioplasty to reduce the risk of developing hydrocephalus after a DC. 69 In addition to the DC itself, one must consider the reason for craniectomy as a driver of glymphatic dysfunction. Many patients undergoing DC suffer loss of brain tissue either as a direct result of the initial insult or subsequent injury from elevated ICPs. This tissue is no longer perfused, causing further decrease in local arterial pulsatility and CBF.
Animal Studies
Glymphatic dysfunction after DC is of increasing interest in the literature. Borha et al. 63 examined the role of hemicraniectomy alone on fluid and solute dynamics in mice. After performing a unilateral hemicraniectomy versus a sham operation, they found a global decrease in cerebral distribution of tracer injected into the CSF at 2 and 15 days compared to the sham mice. 63 Mice that underwent hemicraniectomy followed by cranioplasty at 24 hours demonstrated no change in tracer distribution at 2 or 15 days compared with sham procedure. Based on these findings, the authors postulated early cranioplasty may restore glymphatic function by allowing for physiological ICP, enhanced cerebral perfusion, and increased arterial pulsation. 63 This was an exciting finding as this study did not induce injury to the brain itself, through a stroke or traumatic brain injury model, so the effect of hemicraniectomy alone was examined. Conversely, as the mice did not have elevated ICPs, this study does not mimic the ICP problems seen in patients undergoing DC. In this model, care was paid to leave the dura intact, which meant that the effect seen on the glymphatic system was due to bone-only decompression. In DCs, large incisions are made often into the dura to relieve this tension on the brain and relieve intracranial pressure. They are not often closed acutely, which may cause an even greater effect on the glymphatic system.
The effects of DC on glymphatic function were further demonstrated in a study by the Nedergaard group. Following DC, there was a significant decrease in the pulsatility of penetrating arteries, global reduction in CSF influx into the brain parenchyma, and a proliferation of reactive astrocytes in mice. 14 Functionally, this was accompanied by impaired memory and coordination performance. These changes persisted for several weeks before spontaneous recovery was seen: recovery of CSF influx occurred by day 28 and improvement of astrogliosis by day 56. Cranioplasty at day 14 resulted in enhanced recovery of CSF influx on day 28 in addition to improvement of astrogliosis and microgliosis by day 28. Mice that underwent cranioplasty at day 14 showed complete return of motor and cognitive function by day 28, whereas mice that did not undergo cranioplasty showed persistent impairment. 14 Again DC was sufficient to impair glymphatic function, in part due to a decrease in penetrating arterial pulsatility. Moreover, decreased glymphatic function may drive a neuroinflammatory reaction and behavioral deficits like those seen in SoT. Early cranioplasty may enhance the speed of histologic and functional recovery.
In addition to the altered brain pulsatility, damage to direct lymphatic channels between the skull and meninges may contribute to glymphatic dysfunction. Vascular channels from the bone marrow that traverse the inner table of the skull to the dura are seen microscopically in mice and were confirmed in 3 human specimens from DCs. 70 After stroke and a chemically-induced model of brain inflammation, there was recruitment of myeloid cells from the bone marrow of the skull through these vascular channels to the dura. Furthermore, in a transparent rodent model, tracer injected into the CSF spaces was seen to be transported into these meningeal vessels and ultimately the skull bone marrow. 71 After stroke, the identified meningeal vessels were seen to fill with immune cells, indicating a potential lymphatic connection between the bone and underlying structures. The connection appears to be bidirectional as tracers indicate the potential for CSF to egress from the dura to the skull and lymphoid cells can traverse the vessels from the skull to the dura. The exact role of these meningeal vessels, and the impact of disrupting them through a craniotomy or craniectomy, are unknown. As extensive craniotomies and dural stripping are necessary for cranial vault remodeling, the effects of this on the glymphatic system remain unknown. For more information about how the glymphatic system relates to craniosynostosis, we refer the readers to the recent review by Zibitt et al. 72 More research is needed to determine if dural stripping disrupts the glymphatic system and if the meningeal vessels grow back. Additionally, understanding whether these vessels grow back after autologous cranioplasties and whether this loss of lymphoid cell recruitment affects infection rates and healing from alloplastic cranioplasties could be areas of future research.
Human Studies
The effect of DCs on CSF flow has been observed in humans, but the exact role of the glymphatic system is not well understood. Much of the understanding of glymphatic dysfunction in humans after DC is inferred based on observable flow of CSF. In a prospective study of 16 patients who underwent DC for trauma or stroke, Lilja-Cyron et al 19 quantified what clinicians have been assuming for many years: DCs significantly altered brain pulsatility and ICP regulation. 19 The normal postural changes in ICP present when transitioning from supine to standing were absent after a DC. The large compliant cranial defect also reduced the intracranial pulsation amplitude to nearly zero, indicating much less brain pulsatility. As such, large skull defects significantly impaired CSF flow, which could contribute to the hygromas, hydrocephalus, and SoT seen after craniectomies. 19 Such complications occurred in 2 of the 16 patients included in this study: one developed hydrocephalus 2 weeks after discharge, and the other developed a hygroma ipsilateral to the cranial defect 3 weeks after discharge. Both complications resolved with cranioplasty alone. 19 Furthermore, an imaging report of a patient 3 weeks after DC was found to have stagnation of CSF flow in the cerebral aqueduct as seen on cine MRI, accompanied by progressive hydrocephalus. 67 Flow through the aqueduct was restored on an MRI obtained 3 days after cranioplasty. Though this was only one case, when paired with the prior study, it is hard to ignore the evidence in humans that there are significant alterations in CSF flow after DC.
Glymphatic dysfunction following DC has been implicated as a possible cause of hygromas and hydrocephalus. Atkins and Guppy hypothesized that abnormal cranial fluid collections developed after DC for several reasons: (1) the compliant skull and dural defect reduces brain pulsatility, ventricular squeeze, and CSF circulation; (2) reduced pulsatility leads to reduced flow of fluid through perivascular spaces; (3) reduced clearance of extracellular fluid by the glymphatic system; and ultimately (4) redistribution of CSF in the subarachnoid, subdural, and subgaleal spaces. 64 With that in mind, it is not hard to hypothesize why hygromas are seen in the parafalcine region, in the vicinity of large draining veins, and even contralateral to the craniectomy, since the disruption of the glymphatic system is global. 22 If severe enough, CSF may accumulate in the ventricular system causing communicating hydrocephalus 64 or pool in the interstitial fluid leading to intraparenchymal edema. 22
Less is known about glymphatic dysfunction in SoT, but it is a burgeoning area of research. A prospective study of 5 patients with prior DC did not intend to look at SoT, but sought to examine the effect of cranioplasty on CBF, CSF flow, and cognitive outcomes. Cranioplasty resulted in improved CBF globally as well as velocity of CSF flow through the cerebral aqueduct on imaging. 66 Clinically, there were improvements in the patients’ cognitive functional assessment outcomes scores at 1 and 6 months after cranioplasty. Interestingly, these patients underwent cranioplasty 15 to 65 months after their initial craniectomy, indicating that restoring the rigid cranial vault had positive effects on neurologic function even if delayed.
As stated before, there are multiple hypotheses about the cause of persistent neurologic deficit after DC, but glymphatic dysfunction may play a role. According to some, persistent neurologic deficit is due to impaired venous return and scarring of the cortex by atmospheric pressure. 73 This hypothesis would be supported by the reactive astrocytosis seen histologically after DC in a mouse model accompanied by persistent neurologic impairment. 14 Cranioplasty improved the scarring and functional outcomes. Mechanical atmospheric forces may also cause persistent neurologic dysfunction. Increased levels of strain, or deformation, in axonal fiber tracts are seen after DC in humans, with distortions sufficient enough to disrupt neurochemical events.74,75 Part of the ipsilateral axonal strain may be due to the underlying injury and edema necessitating DC, but was clearly worsened after DC on imaging. The large compliant defect of a DC, therefore, may allow for continued neuronal deformation. Others hypothesize neuronal dysfunction may be due to fluid accumulation in the brain parenchyma as a direct result of glymphatic dysfunction. 19 Though brain edema is not seen in all patients with SoT after craniectomy, it is an interesting idea supported by other authors and termed “glymphedema.” 22 More information is needed to understand the role of the glymphatic system in human with SoT and persistent neurologic dysfunction after DC.
Conclusion
The glymphatic system is a recently proposed pathway for brain-wide delivery of molecules and efflux of neurologic waste products. DC has been shown to decrease glymphatic CSF influx in animal studies, leading to a neuroinflammatory response and behavioral dysfunctions similar to those seen in SoT. Early cranioplasty restored glymphatic function and may enhance recovery from these complications. These studies suggest glymphatic dysfunction after DC may play a role in the pathogenesis of the SoT, hydrocephalus, and hygroma. Further studies are needed to explore the effects of DC on glymphatic function in humans and the mechanisms through which glymphatic dysfunction may increase the risk of these complications.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Institutional Review Board approval was not required.
Previous Presentations
None
