Abstract
Background:
Pediatric cranio-orbital tumors have several etiologies and given the broad spectrum of pathology, it is difficult to predict the extent of resection. Reconstruction in children and adolescents is particularly challenging due to potential craniofacial growth and the relatively limited availability of donor sites. In this review, we analyze 10 pediatric and adolescent cases from a single institution, discuss the existing literature, and propose a reconstruction algorithm for managing these complex defects.
Methods:
A retrospective chart review was performed to identify pediatric and adolescent patients with cranio-orbital tumors who underwent tumor resection and reconstruction at our institution between January 2012 and July 2022.
Results:
A total of 10 patients underwent oncoplastic reconstruction of defects of either the cranium alone or combination of cranium and orbit, with a mean age of 12.4 years (range: 2-20). The defects involved the parietal (n = 3, 30%), parietooccipital (n = 1; 10%), temporoparietal (n = 1; 10%), occipital (n = 1, 10%), and fronto-orbital (n = 4; 40%) regions. Cranioplasty was performed with split-thickness bone grafts, exchange cranioplasty, or alloplastic materials. A pericranial flap was used to isolate intracranial and extracranial contents in 2 cases. Free flaps were utilized for additional soft tissue coverage in 2 cases. Complications included free flap venous thrombosis, CSF leak, hardware exposure, sagittal sinus injury, superior sagittal sinus thrombosis, vertical diplopia, and hypertropia.
Conclusion:
The goals of oncoplastic reconstruction for cranio-orbital defects in pediatric and adolescent patients align with those in adults. However, reconstructive surgeons must consider age-specific differences, such as growth potential and limited donor sites. Effective reconstruction can be achieved through meticulous planning, clear communication, and a multidisciplinary approach.
Introduction
Cranio-orbital tumors in children and adolescents encompass a diverse range of pathologies and often result in complex ablative defects upon resection.1-7 Various etiologies and their subsequent reconstruction have been described, including fibrous dysplasia,8-10 rhabdomyosarcoma,11-14 osteoma, 6 lipoma, 15 neuroectodermal tumors,3,16 Ewing sarcoma, 17 anaplastic ependymoma, 18 and non-Hodgkin diffuse large B-cell lymphoma. 19
Advancements in surgical techniques, technologies, and instruments have enabled more radical tumor resections.2,18,20 Consequently, oncoplastic reconstruction has become notably more challenging, particularly in the pediatric population in which donor site selection is somewhat limited and there is potential for growth disturbance. 21 Adopting a multidisciplinary approach is essential for the optimal management of these tumors.12,17,18,21,22 This series proposes an algorithm for oncoplastic reconstruction of cranio-orbital defects in the pediatric and adolescent population, drawing upon the senior author’s experience and existing literature.
Methods
After obtaining Institutional Review Board (IRB) approval, a retrospective chart review was conducted using our institution’s electronic medical records to identify patients under 21 years of age with cranial and/or orbital tumors who underwent tumor resection and reconstruction by our multidisciplinary team between January 01, 2012, and July 01, 2022. Patients were excluded if they had extra-cranio-orbital malignancy. The recorded data included demographics, tumor diagnosis, use of chemotherapy and/or radiation, defect characteristics, reconstructive methods, and complications. Only complications causing significant morbidity or requiring operative intervention were included in the analysis, while minor complications were excluded.
Results
Between January 2012 and July 2022, a total of 10 patients underwent oncoplastic reconstruction of cranio-orbital ablative defects. Their ages ranged from 2 to 20 years, with an average age of 12.4 years. The average duration of multidisciplinary follow-up was 15.5 months. Additional patient characteristics, treatment modalities, and complications are outlined in Table 1.
Clinical Characteristics, Treatment Modalities, and Reconstructive Outcomes of Patients Undergoing Oncoplastic Reconstruction of Cranio-Orbital Defects.
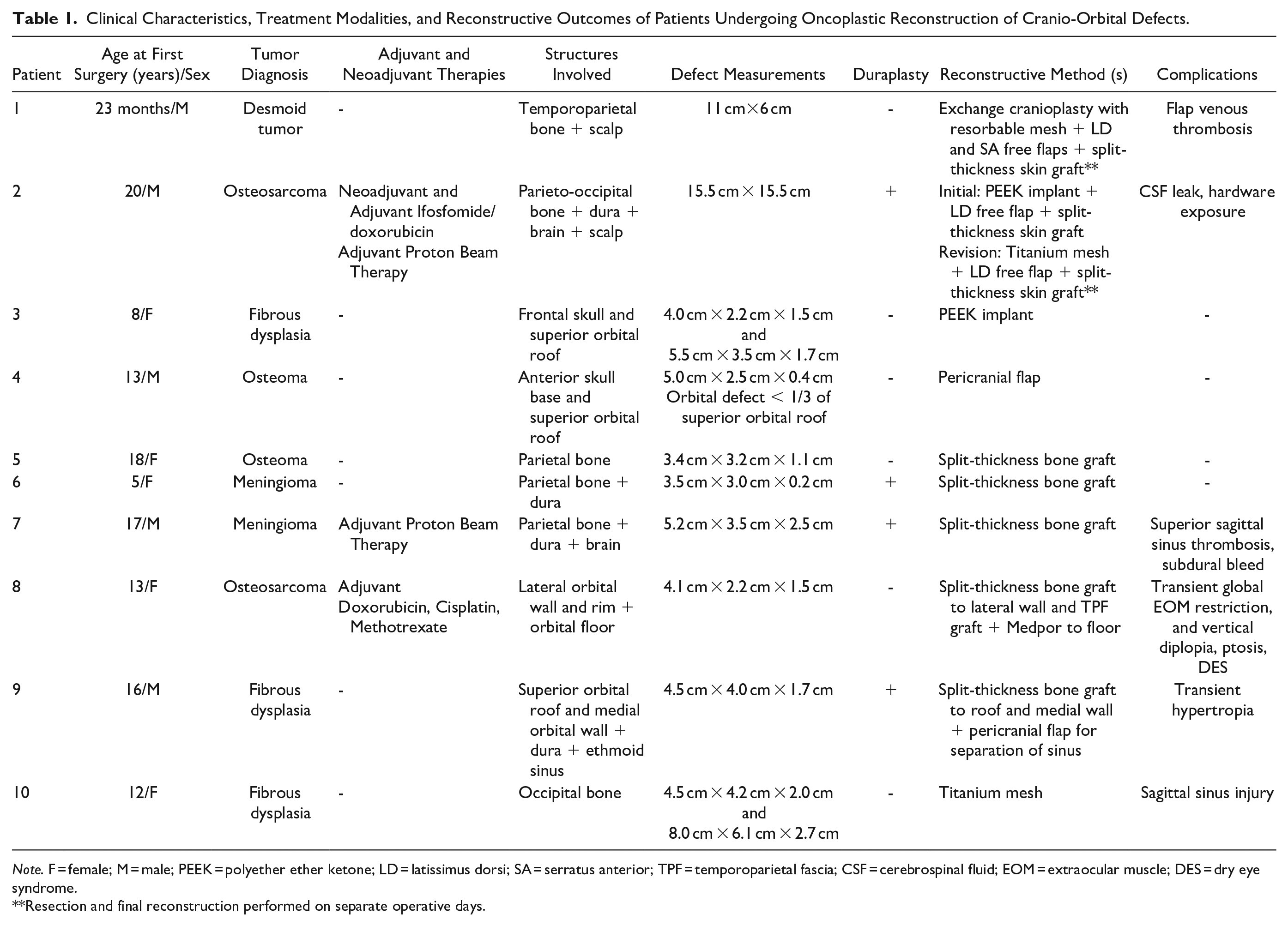
Note. F = female; M = male; PEEK = polyether ether ketone; LD = latissimus dorsi; SA = serratus anterior; TPF = temporoparietal fascia; CSF = cerebrospinal fluid; EOM = extraocular muscle; DES = dry eye syndrome.
Resection and final reconstruction performed on separate operative days.
Diagnoses and Neoadjuvant/Adjuvant Therapies
Tumor diagnoses varied and included osteoma (n = 2; 20%), osteosarcoma (n = 2; 20%), fibrous dysplasia (n = 3; 30%), meningioma (n = 2; 20%), and desmoid tumor (n = 1; 10%). Computed tomography (CT) scans in Figures 1 and 2 depict tumor involvement prior to resection. Among the patients diagnosed with osteosarcoma, one received both neoadjuvant and adjuvant chemotherapy with adjuvant proton therapy, while the other received adjuvant chemotherapy only. Additionally, one of the patients with meningioma received adjuvant proton therapy for atypical features. Adjuvant chemotherapy was initiated 2.6- and 2-weeks following surgery, respectively. Adjuvant proton therapy was initiated 5.4- and 6.4-weeks following surgery, respectively.
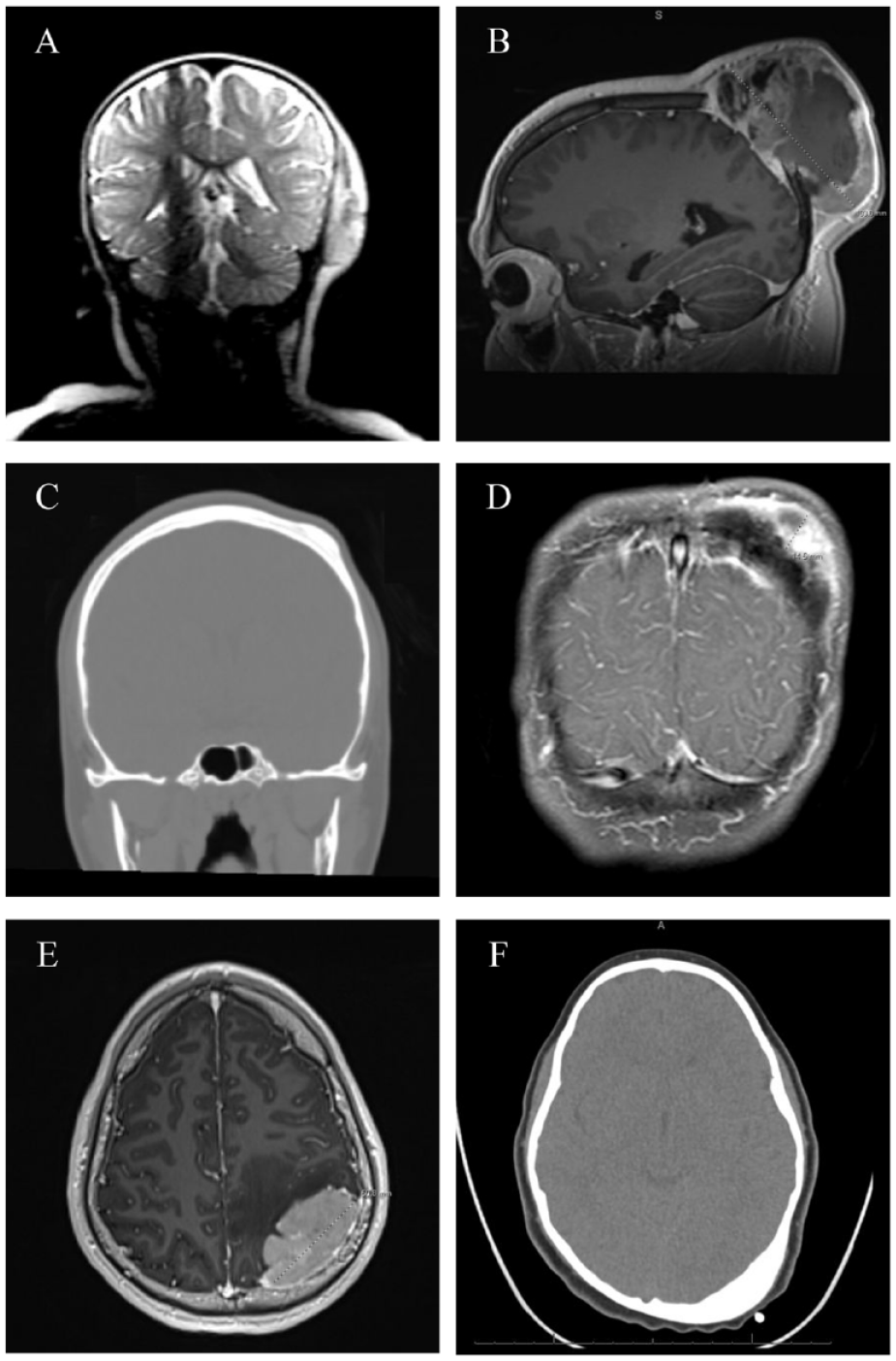
CT scans of patient 1 (A), patient 2 (B), patient 5 (C), patient 6 (D), patient 7 (E) and patient 10 (F), demonstrating defects isolated to the cranium.
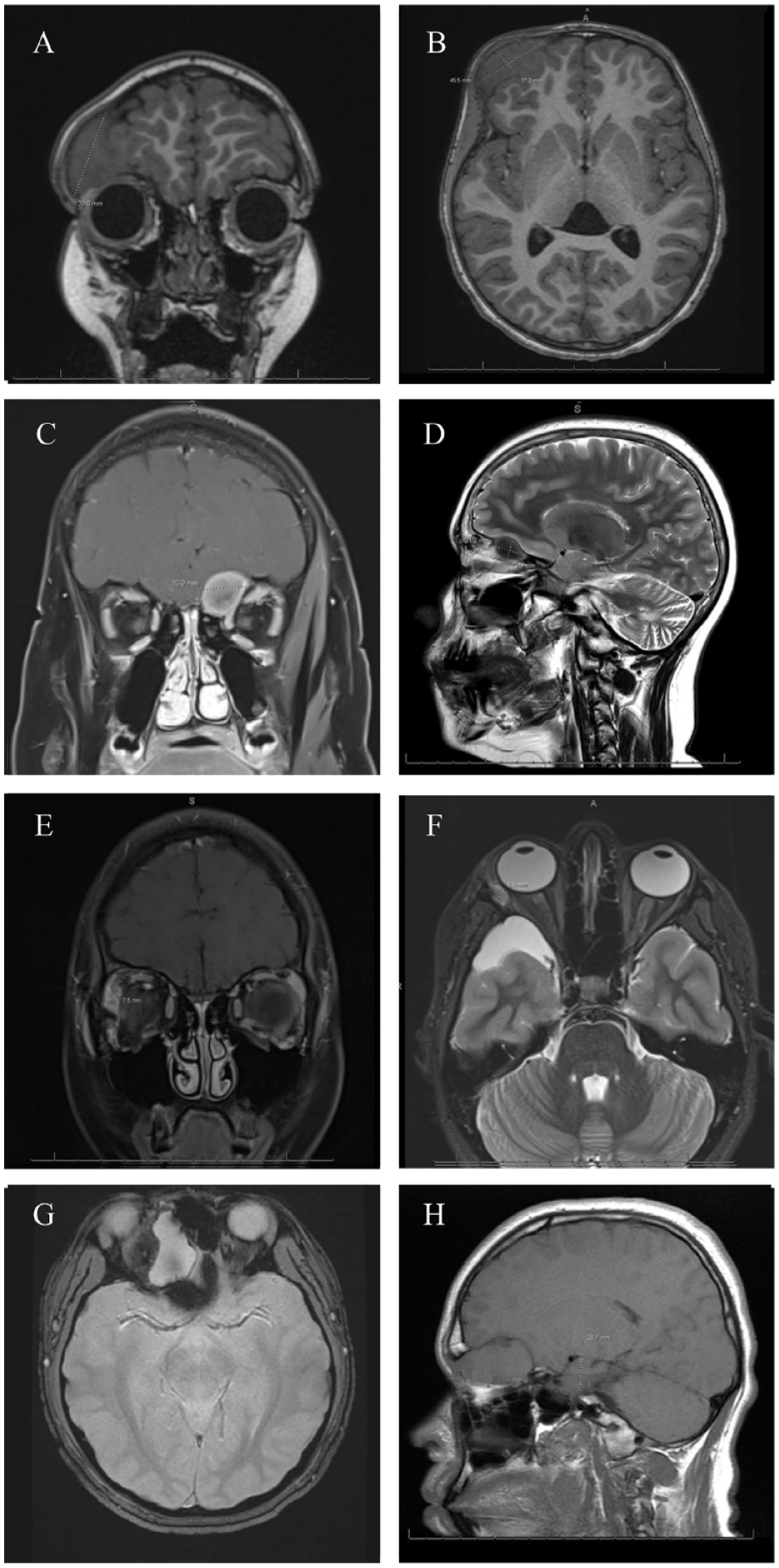
CT scans of patient 3 (A and B) patient 4 (C and D), patient 8 (E and F) and patient 9 (G and H) demonstrating defects involving both the cranium and orbit.
Reconstruction
Defects included those isolated to the cranium (n = 6; 60%), and those involving both the cranium and orbit (n = 4; 40%).
Among the 6 defects confined to the cranium, cranioplasty was performed using split-thickness bone grafts in 3 patients (5, 17, and 18 years old) to reconstruct parietal bone. A split-thickness bone graft was also planned for reconstruction of a 12-year-old with an occipital bone defect; however, this procedure was emergently aborted due to an intraoperative sagittal sinus injury and required the use of readily available titanium mesh. Exchange cranioplasty was performed on a 2-year-old patient with a temporoparietal bony defect. A customized Polyether ether ketone (PEEK) implant was used for a large 15 x15 cm parieto-occipital bony defect in a 20-year-old. The case was complicated by a persistent cerebrospinal fluid (CSF) leak and peri-implant fluid collection, requiring revision reconstruction. The exposed PEEK implant was ultimately replaced with a titanium plate, providing a light, strong, relatively biocompatible, and inert construct.
Among the 4 defects involving the cranio-orbital region, one case resulted in a defect involving the anterior cranial fossa and encompassing less than 1/3 of the superior orbital roof. In this case, reconstruction was achieved using a pericranial flap without the need for bony reconstruction. The second case involved the frontal skull and a larger portion of superior orbital roof, which was reconstructed using a customized PEEK implant to provide precise contour and strength. In the third case, the defect involved both the lateral orbital wall/rim and the orbital floor, which were reconstructed using a split-thickness bone graft and Medpor (Porex Surgical, Inc, Newnan, GA), respectively. Finally, the fourth cranio-orbital case resulted in a defect involving the superior orbital roof and medial orbital wall extending to the ethmoid sinus. A pericranial flap was used to separate the anatomical spaces, and a split-thickness bone graft was utilized to reconstruct the orbital roof and medial wall.
For all 4 tumors involving the dura, intraoperative dural repair was performed by the neurosurgery team with dura substitute and 4-0 nylon suture. For 2 cases requiring extensive soft tissue coverage; a latissimus dorsi (LD) free flap was used for a parieto-occipital soft tissue defect, and a chimeric LD flap with serratus anterior free flap was utilized for a temporoparietal soft tissue defect. Both cases utilized the superficial temporal vessels as recipient vessels, and muscle flaps were covered with a split-thickness skin graft.
Complications
A total of 6 complications were reported in 6 separate patients. The first complication was a chimeric LD flap venous thrombus that developed on postoperative day (POD) 6 after temporoparietal reconstruction with exchange cranioplasty and a chimeric LD and serratus anterior flap. On POD 6, the patient was taken to the operating room for exploration of the flap due to a change in clinical exam. A thrombus was noted on the venous side at the site of anastomosis, thought to be a result of patient compression of the flap. The thrombosed segment was excised, and a 2.5 cm reverse saphenous vein graft was harvested from the left lower leg and placed in the area of venous resection. The flap was successfully salvaged, and the patient went on to heal without complications.
The second complication involved a persistent CSF leak following the resection of a parietooccipital sarcoma. This resection resulted in a compound defect involving the cranium, dura, and a large area of soft tissue. During the initial resection, duraplasty was performed using a dura substitute and 4-0 nylon suture to address a 2 to 3 cm defect. Bony and soft tissue reconstruction was then performed in 2 stages. The first stage involved a temporary closure using KLS Martin titanium mesh, xeroform, and wound VAC placement. Final reconstruction occurred 6 days later, after pathology confirmed negative margins, and involved replacement of the temporary mesh with a PEEK implant and a free LD flap with a split-thickness skin graft. Postoperatively, the patient developed a persistent CSF leak despite treatment attempts involving compression, bedrest, and lumbar drain placement. The patient was taken back to the operating room for exploration at which time dural graft dehiscence was identified at the duraplasty site. A watertight dural seal was achieved with a composite of Gelfoam® (Pfizer, New York, NY, USA) and dura substitute, and the exposed PEEK implant was replaced with titanium mesh. The previously elevated LD flap was then readvanced to provide soft tissue coverage. There was no recurrence of CSF leak. Seven months after reconstructive surgery and initiation of radiation therapy, the postoperative course was further complicated by a 2 × 3 cm full thickness wound with titanium plate exposure. At this time, chest CT and positron emission tomography (PET) scan revealed new pulmonary metastatic nodules. Unfortunately, the patient ultimately succumbed to his disease prior to the planned flap revision.
The third complication involved a superior sagittal venous sinus thrombosis following resection of a meningioma involving parietal bone and dura. Dural repair was performed using dura substitute, and cranioplasty with a split-thickness bone graft. The postoperative course was complicated by headache and nausea; imaging obtained on POD 3 revealed extensive superior sagittal sinus thrombosis with extension to the left internal jugular vein. Therapeutic anticoagulation with Lovenox was initiated in the inpatient setting and continued at discharge. The patient returned 1 month later with headache and vomiting; imaging revealed extension of the thrombus and a small subdural bleed, which was thought to be secondary to medication noncompliance and extension of clot. The patient was restarted and maintained on anticoagulation therapy until MRI showed no evidence of residual sinus thrombus.
The fourth complication involved transient global extra ocular muscle (EOM) restriction, vertical diplopia, dry eye syndrome (DES) and ptosis following reconstruction of the lateral orbital wall/rim and orbital floor with a split-thickness bone graft and Medpor, respectively. The EOM restriction and diplopia has since resolved, likely a transient result of immediate postoperative edema. The patient continues to follow with ophthalmology for DES and ptosis.
The fifth complication involved mild hypertropia following reconstruction of the orbital roof and medial wall using a split-thickness bone graft and pericranial flap. Hypertropia was noted only in right gaze, and the resulting diplopia was not bothersome to the patient; thus, further intervention was not required and the hypertropia resolved with time, which may also have been attributable to postoperative edema.
The sixth complication involved an inadvertent injury to the sagittal sinus during resection of an occipitally based tumor. Hemostasis was achieved with Gelfoam® without additional sequelae.
Discussion
Surgical resection remains the definitive treatment for locally invasive tumors involving the cranium and orbit.23,24 In recent decades, advancements in adjuvant and neoadjuvant therapies have significantly improved postoperative outcomes and long-term survival for pediatric patients. However, it is important to note that these therapies do not come without complications. Cranial radiation has been associated with a nearly 50% complication rate, resulting in issues such as craniofacial and soft tissue hypoplasia or asymmetry, as well as visual, auditory, or endocrine dysfunction.2,23,24 Given this high complication rate, surgeons may advocate for upfront, aggressive resection in an attempt to avoid radiation. However, resection of these tumors often results in structural defects involving a combination of soft tissue, dura, cranium, and/or orbit.23,24
Cranio-orbital reconstruction following tumor extirpation aims primarily to maintain a watertight dural seal, isolate and support intracranial contents, maintain orbital support for the globe, and provide soft tissue coverage. Secondary goals of reconstruction include optimization of function and esthetics to augment quality of life.2,23,25 Failure to achieve these goals may lead to complications such as life-threatening infections, functional and visual deficits, or poor esthetic outcomes.8,25 To achieve optimal operative outcomes in pediatric and adolescent patients, surgeons must consider age-specific factors such as thin cranial bone, fragile blood vessels, growth potential, and psychosocial sequelae.2,9,23,26,27 This case series demonstrates the senior author’s experience in reconstructing various cranio-orbital defects and proposes a reconstruction algorithm to circumnavigate these challenges (Figures 3 and 4).
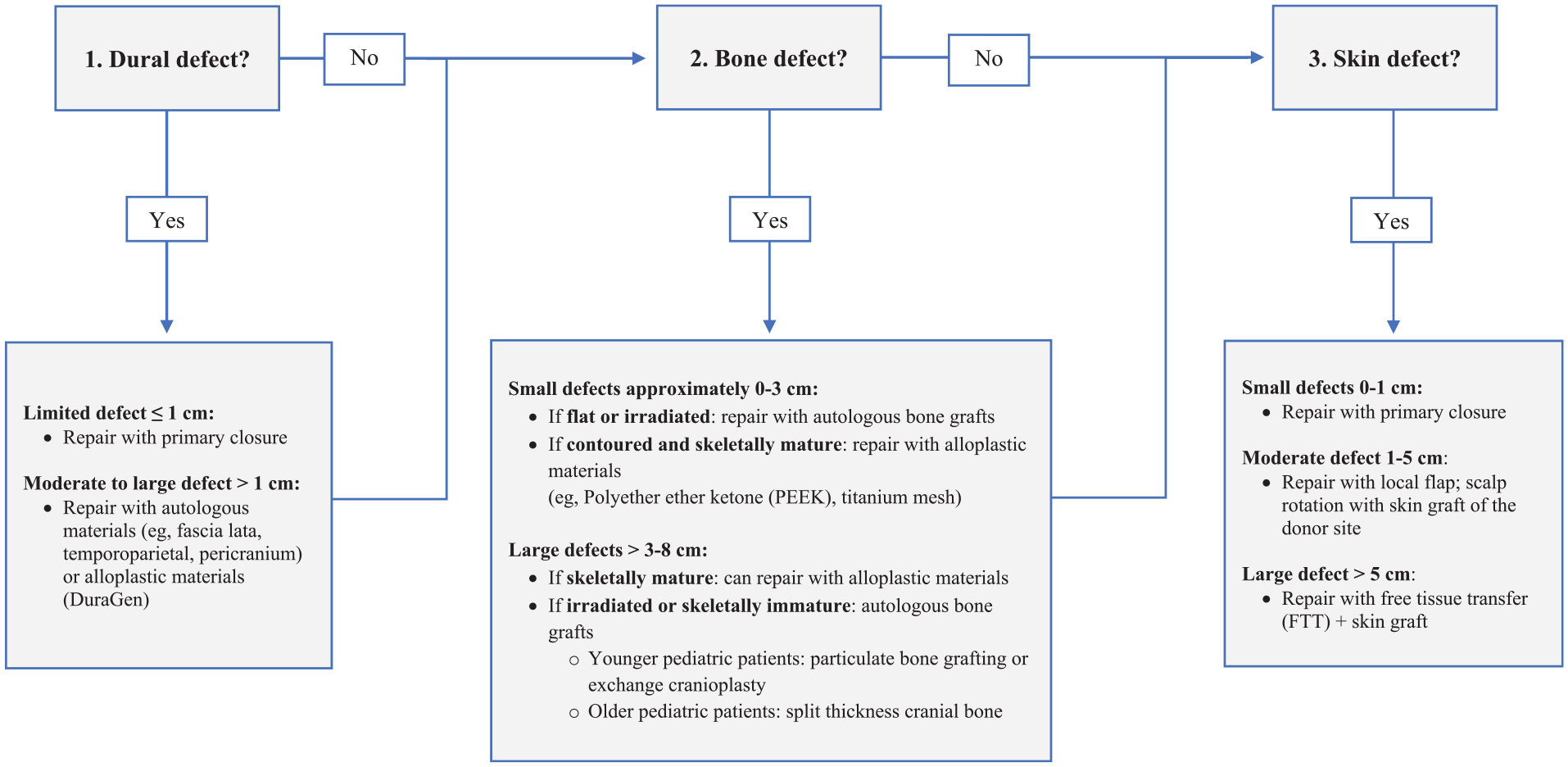
Algorithm for reconstruction of cranial defects.
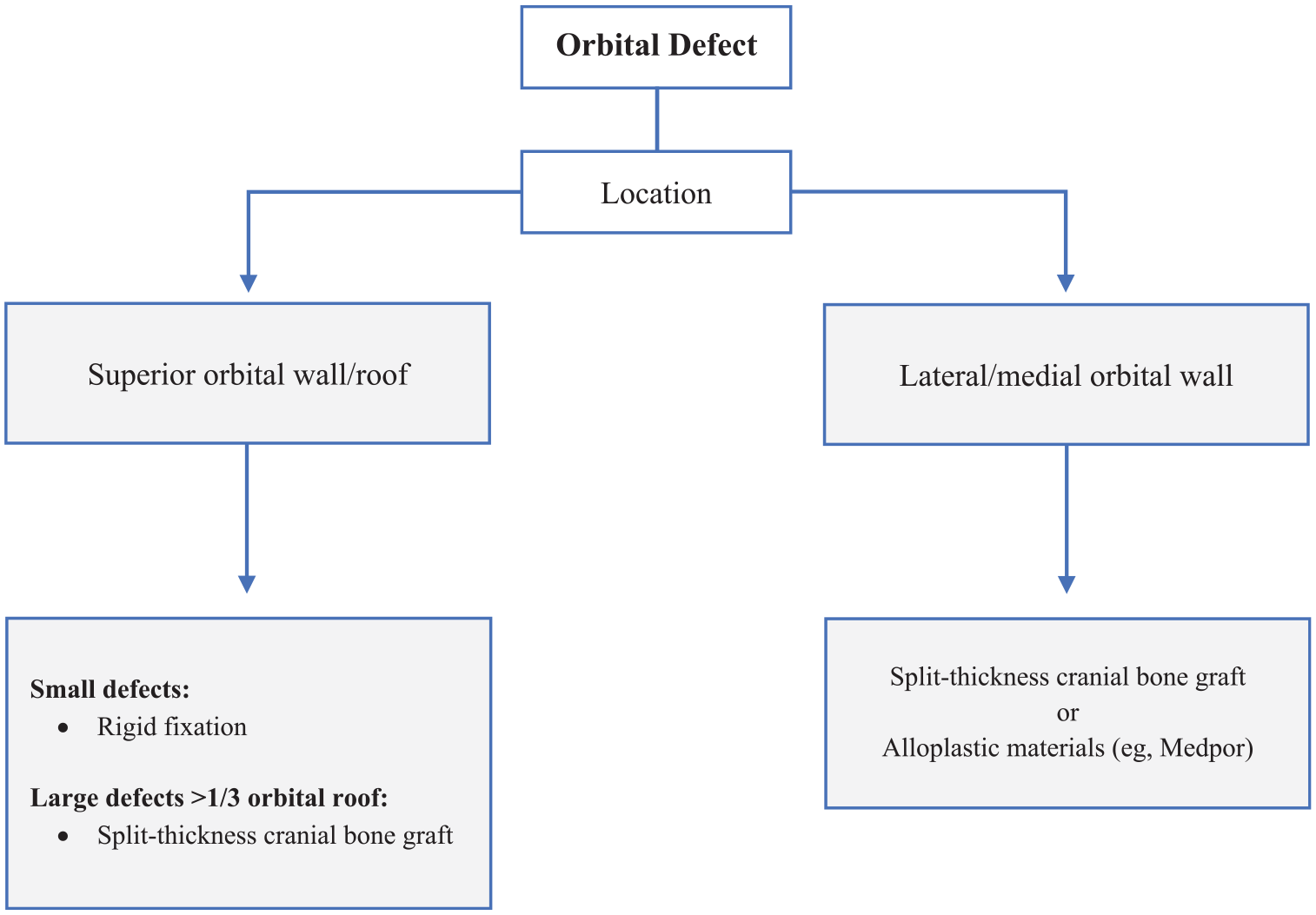
Algorithm for reconstruction of orbital defects.
The first consideration in the proposed cranio-orbital reconstruction algorithm involves management of a dural defect. The choice of reconstruction for dural defects primarily depends on defect size and leakage flow rate.28,29 Small, low-flow dural defects, typically less than 1 cm, can be closed primarily. 28 Larger or high-flow defects should be repaired with alloplastic materials such as DuraGen® (Integra Neurosciences, Plainsboro, NJ, USA) or autologous grafts/flaps (eg, pericranium, temporoparietal, or fascia lata) for particularly large or compromised defects (Figure 3).30,31
The next step in the proposed cranio-orbital algorithm involves assessing the extent of the bony cranial defect. The management algorithm for bony reconstruction in pediatric and adolescent craniofacial tumors differs from that in adults. While complete healing via bony ossification may be possible in infants under 1 year old, the potential for complete healing decreases with age.32-36 Though anterior skull base defects may be addressed with soft tissue coverage alone to separate intracranial and sinonasal spaces, cranioplasty is necessary following tumor resection of the remainder of the skull to protect and support the brain. Autologous bone remains the gold standard reconstructive substrate. 37
Autologous bone is preferred due to its innate ability to expand with the growing brain and to provide osteoconduction, osteoinduction, and osteogenesis. Cons of autologous graft include donor site morbidity, poor ability to contour, and high resorption rates—particularly in a pediatric population, with some studies citing up to 50% resorption compared to 6.5% in an adult counterpart. 38 Prevention of bone graft absorption is combated by support from well vascularized tissue and rigid fixation to surrounding bone to allow for osteoblast migration. This may be accomplished with absorbable fixation systems in a pediatric population where growth restriction is of concern. Bone graft for pediatric cranioplasty may be obtained from calvarial split grafting, iliac crest, or rib. Calvarial bone graft is the preferred substrate for cranioplasty in a pediatric population given its lower rates of resorption compared to other sites in the body and ability to replace “like with like.” The diploic space develops around 4 years of age and facilitates splitting of cranial bone through this soft layer of cancellous bone. Splitting of cranial bone before development of the diploic space is difficult and traditionally not recommended—though recent studies have brought this into question. 39 Particulate bone grafting has emerged as a reliable source for cranial reconstruction in this population. 37 This technique involves using small bone segments harvested from the calvaria ectocortex or endocortex, with thicker parietal bone being the preferred source.37,40 Contraindications to particulate bone grafting include defects requiring structural support for contour, such as those in the frontal or occipital cranium, and defects where the dura is compromised. 40 To address these limitations, a method termed “exchange cranioplasty” has been described. In this approach, a full-thickness structural bone graft is taken from an unaffected area of the cranium and contoured to fill the defect. 40 The donor site, which retains intact dura and pericranium, is covered with a particulate bone graft harvested from the endocortex of the full-thickness graft or ectocortex of the intact cranium, hence the name “exchange” cranioplasty. 40 This technique is particularly suitable for temporoparietal defects, utilizing the opposing tempoparietal region as a donor site (Figure 5). The bone grafts should be fixed and covered with well-vascularized soft tissue to minimize resorption and prevent infection, especially in patients receiving adjuvant radiation therapy. Split bone grafts may be secured with plates and screws, while particulate bone can be stabilized with fibrin glue or thrombin-based hemostatic agents. 40 Resorbable mesh or local flaps, such as pericranial or temporoparietal flaps may also be utilized to cover grafts (Figure 6).
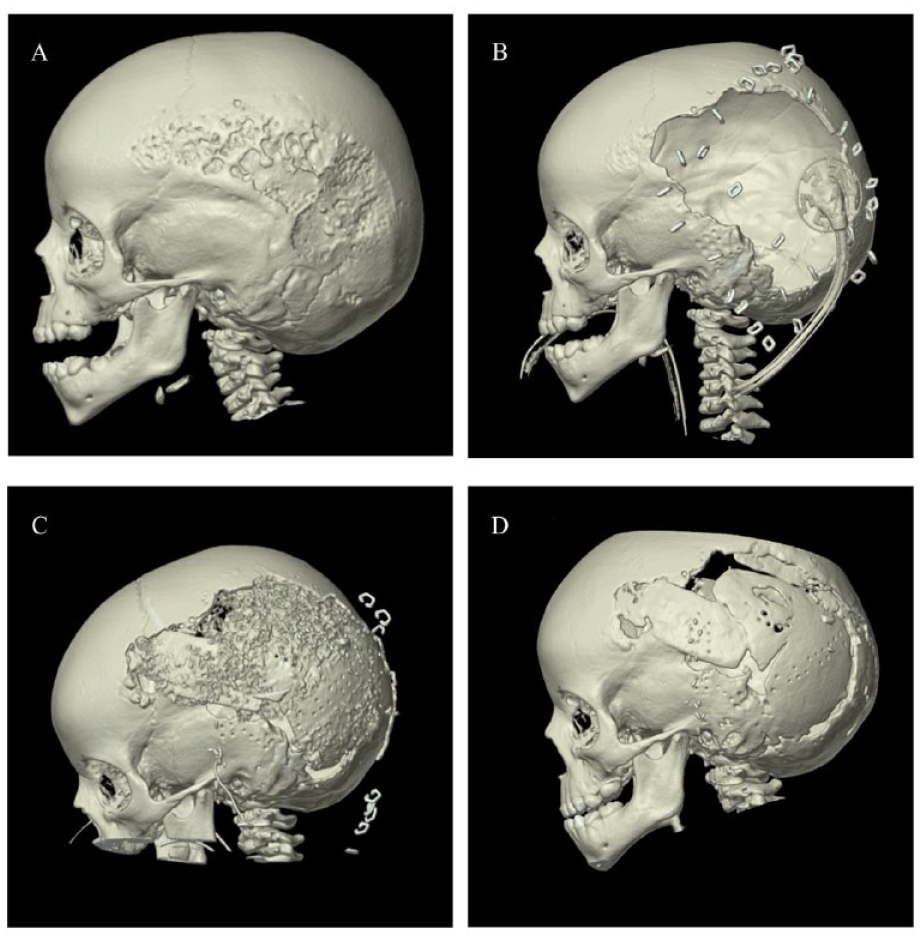
Three-dimensional computed tomography (3D-CT) reconstruction of a 23-month-old male with a large left-sided temporoparietal desmoid tumor before (A) and after (B) tumor resection. Post-operative images (C and D) show the healing of exchange cranioplasty on postoperative day 8 and week 5, respectively.
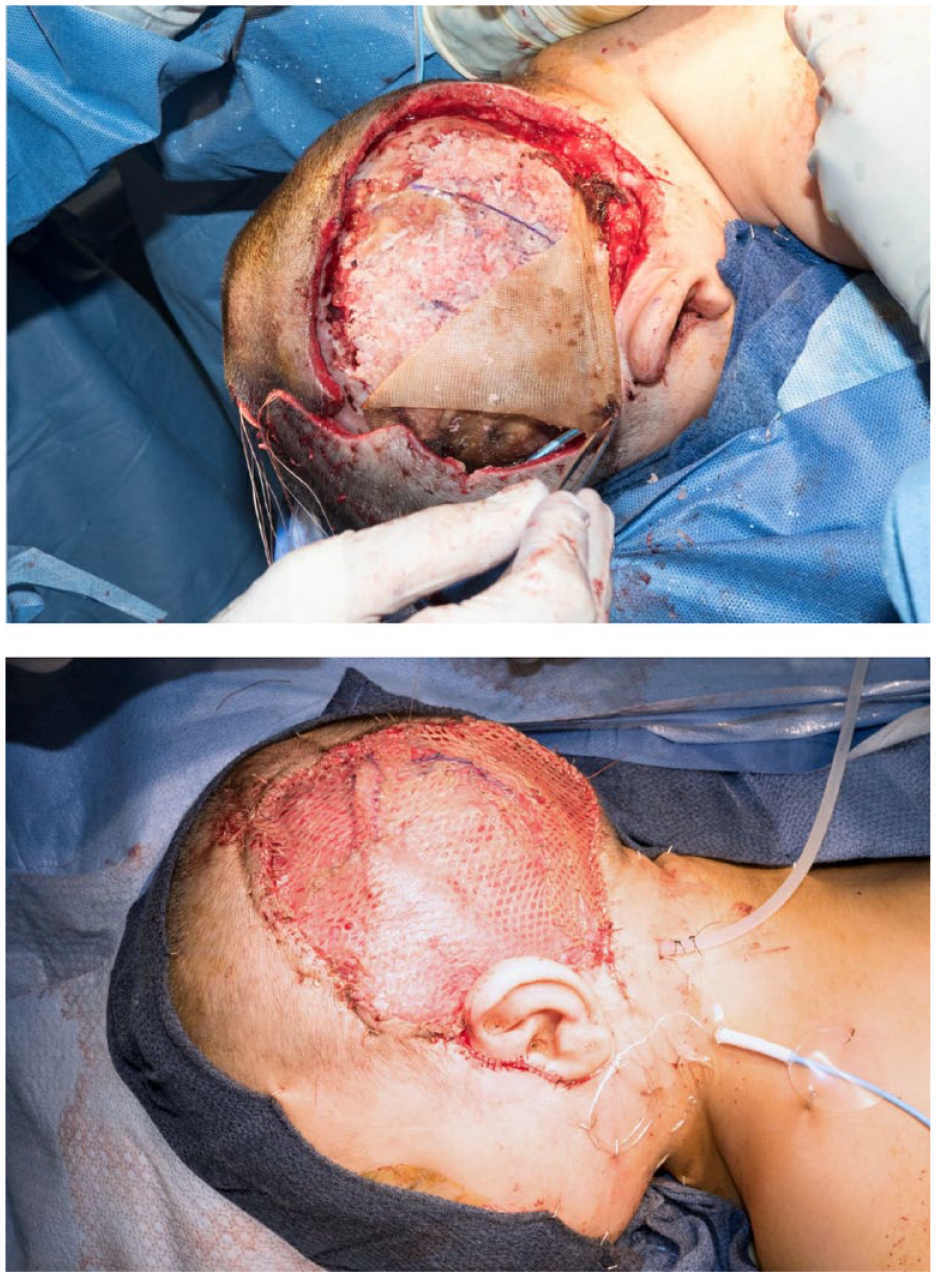
Intraoperative photographs of a 23-month-old male with a large left-sided temporoparietal desmoid tumor. The top photograph shows resorbable mesh overlying the exchange cranioplasty, constructed with harvested bone from the right temporoparietal area. The bottom photograph shows the completed reconstruction, including a chimeric latissimus dorsi and serratus anterior free flap covered by a meshed split-thickness skin graft from the left thigh.
Alloplastic materials are not favored in pediatric calvarial reconstruction due to their lack of growth potential, association with higher infection rates, and risk for extrusion. 37 Strict avoidance of alloplastic materials is recommended in an irradiated field. However, alloplastic materials may be considered in circumstances of large calvarial defects and those crossing multiple sutures lines if the overlying skin is intact, or the skin incision is distant from the cranial defect. Further consideration should be given to the age of the child. The neurocranium is approximately 25% of its adult size by birth, 50% by 6 months and 65% by 1 year. 41 Skeletal cranial maturity is reached around 5 years of age, and surgeons should take this into account when considering alloplastic materials. Though there is concern for growth restriction below the age of 5, Ma et al demonstrates successful outcomes and lack of growth restriction on short term follow up with alloplastic titanium mesh cranioplasty in those patients where cranial maturity had not yet been met. 42 The series demonstrated titanium mesh cranioplasty to be an acceptable alternative to autologous bone in both a younger and older pediatric population, assuming adequate soft tissue coverage. 42
The most commonly used alloplastic materials for pediatric cranioplasty are titanium mesh and polyether ether ketone (PEEK). Titanium is a readily available substrate that is relatively inexpensive, infection resistant, and ideal for contouring given its thin and malleable properties. 43 Titanium can be designed as a custom implant or contoured intra-operatively on the day of surgery. Cons include radiopacity and its proneness to deformational forces. Furthermore, it may be susceptible to extrusion as demonstrated in a series on pediatric titanium mesh cranioplasty. 42 PEEK implants are radiolucent and more resistant to deformational forces; however, they are expensive, more prone to infection, and do not integrate with surrounding bone. 43 Unlike titanium mesh, PEEK cannot be contoured on day of surgery. Regardless of material, alloplastic grafts must be surrounded by well vascularized tissue to prevent extrusion and infection.
The next consideration in the proposed cranio-orbital algorithm involves assessing the involvement of the orbital cavity (Figure 4). If a defect exists such that there is contact between cranium and sinonasal contents, isolation must first be achieved. This may be obtained with local flaps such as pericranial flaps or temporoparietal fascial flaps. Bony reconstruction of the orbital cavity depends on the size of the defect and its anatomical location. Failure of bony orbital reconstruction may lead to ocular dystopia (ie, orbit asymmetry) with subsequent visual disturbances, brain herniation into the orbit, or cosmetic deformities. Bony reconstruction is warranted if the defect results in a change in the position of the eyeball, a clear loss of structural support, or cosmetic deformity. 44 Bony reconstruction is indicated for orbital roof defects greater than one third of the roof to prevent pulsatile exophthalmos.6,17,45,46 Additionally, the supraorbital bar and frontal bone should be reconstructed to achieve an optimal esthetic outcome. 2 Defects in the lateral/medial walls and floor are repaired on a case-by-case basis, guided by the defect size and involvement of the periorbital soft tissue structures. 30
Reconstruction of the orbital roof can be accomplished through rigid fixation in small defects, 2 or autologous bone grafting as previously described.37,40 Grafts should be fixed and covered with vascularized tissue, similar to cranial defects. Alloplastic materials in this region should be avoided, given the vital overlying structures and potential risk of erosion. Reconstruction of the medial/lateral walls and floor can be achieved by utilizing autologous bone or alloplastic materials, such as titanium mesh or Medpor. 30
The final consideration in the proposed cranio-orbital reconstruction algorithm involves assessing the soft tissue (Figure 3). Limited soft tissue defects may be closed primarily, while medium-sized defects should be reconstructed utilizing adjacent tissue transfer.2,47 Significant soft tissue defects may necessitate free tissue transfer.2,48 Free tissue transfer should be considered for large defects that encompass more than one anatomical region or are within an irradiated field. In our algorithm for pediatric cranio-orbital reconstruction, small defects are defined as approximately 0 to 1 cm, moderate defects as approximately 1 to 5 cm, and large defects as greater than 5 cm in length. Common free muscle flaps include the latissimus dorsi, which may be extended as a chimeric flap, or the rectus abdominus flap—both of which provide excellent pedicle length. Fasciocutaneous flap options may include the anterolateral thigh (ALT) flap and radial forearm flap (RFF).25,49 Muscle flaps may offer superior contour compared to fasciocutaneous flaps but require coverage with split-thickness skin grafts both externally and for the nasal sinus.
Pediatric cranioplasty alone is associated with relatively high overall complication rates, ranging from 12% to 50%. 50 These complication are heavily dependent on the underlying pathology, anatomical location, and materials used for reconstruction. 50 Complications include events both intraoperatively (such as bleeding, damage to venous sinuses, dural tears, and injury to the dura/brain during graft harvest) and postoperatively (including CSF leak, infection or exposure of graft/implant, graft or flap failure, contour defects, and alopecia). Fortunately, severe complications, such as meningitis, air embolism, and death are rare. 50 Defects that encompass the anterior skull base and extend to include the sinuses and/or orbital cavity inherently pose an even greater risk for complications. In our case series, we report complications including CSF leak, sagittal sinus injury, superior sagittal sinus thrombosis, venous thrombosis of a chimeric latissimus flap, and vertical diplopia.
CSF leak is reported in approximately 4% of cranial pediatric surgeries, with the highest risk for leak associated with craniectomy, larger dural defects, and high flow regions. 51 CSF leak may be controlled successfully in the postoperative period with conservative measures alone or lumbar drain placement. If CSF leak is persistent, such as in our reported case, operative exploration and repair is mandated. An understanding of risk factors prompting a more complex repair will help to mitigate complication and prevent future occurrences.
Sagittal sinus injury is overall rare and may occur during tumor resection or graft harvest. 50 Postoperative venous sinus thrombosis (POVST) is even more rare, with few instances reported in the literature. In a review of 14 cases of POVST, management was found to be variable. The authors recommend anticoagulation only if the clot is symptomatic, involves the superior sagittal sinus, or propagates. In our case of superior sagittal sinus POVST, the patient went on to resolve the thrombosis with anticoagulation management. 52 On reflection, the incidence of this complication may have occurred from an abnormal pressure that was exerted from reconstruction and will be a consideration moving forward.
The perception of microsurgery in the pediatric population has evolved over the decades. Initially, concerns about vessel size, donor site morbidity, and growth potential of the flap led to apprehension about successful outcomes. However, we now understand that microsurgery in pediatric cases is both safe and effective. 53 A study of 108 pediatric microsurgery cases across various anatomic locations showed a 93% success rate, which was comparable to the 95% success rate in the adult population by the same surgeon. 54 More specifically, a study reviewing 49 pediatric head and neck free flaps demonstrated a 92% flap survival. 55 Besides comparable outcomes, there are some factors that are even beneficial in a pediatric vessel compared to an adult counterpart. For example, the relative size of the flap pedicle to its corresponding soft tissue is larger compared to that in the adult patient. Additionally, vasospasm and atherosclerosis are infrequent in this younger population. In our case, we encountered a complication of venous thrombosis on POD 6. It is likely that the venous clot in this reported case resulted from compression of the pedicle at 6 days post-surgery, without evidence of technical error. However, this instance of venous thrombus should not be overlooked, as poor compliance is specific to a pediatric population and should be considered preoperatively.
Both ocular complications in this series were transient. The first of which was extra-ocular muscle restriction and vertical diplopia following a lateral orbital wall/rim and floor reconstruction with split calvarial graft and Medpor, respectively. This was likely attributed to swelling in the perioperative period. Diplopia is not an uncommon complication following orbital reconstruction and is reported to be as high as 5% to 18% in those undergoing orbital floor reconstruction. 56 The etiology of diplopia is likely due to the restriction of the inferior rectus muscle.56,57 While many cases resolve independently, there are some instances which may require eventual surgical intervention with strabismus surgery, despite imaging which reveals satisfactory implant position. The second ocular complication included transient hypertropia following reconstruction of the orbital roof and lateral wall with split thickness bone graft and pericranial flap, which can likely also be attributed to postoperative edema.
Conclusion
General principles of cranio-orbital reconstruction include achieving a watertight dural seal, isolating and supporting intracranial contents, maintaining orbital support for the globe, and providing soft tissue coverage. In the pediatric and adolescent patient population, surgeons must additionally account for age-specific differences. In this series, the authors report on 10 unique pediatric cranio-orbital reconstructions, along with their respective outcomes. Furthermore, the authors propose an algorithm for reconstruction.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by the Baylor College of Medicine Institutional Review Board (H-48931).
