Abstract
Introduction
Cleft lip is among the most common congenital anomalies affecting the head and neck, with an incidence of 0.1% in the general population. 1 Delivery of effective care requires a multidisciplinary approach to meet the needs of this complex patient population. From a treatment standpoint, emphasis has always been placed on the surgical approach for defect and structural correction. While various surgical techniques for cleft lip repair have been analyzed to provide tangible guidelines and recommendations for physicians, the surgical approach is only one aspect of the treatment plan2,3 Intraoperative and post-operatively adjuvant therapy for primary cleft lip repair has the potential to optimize surgical recovery, mitigate adverse scar formation, and improve the psychosocial well-being of patients.
A variety of protocols exist to discuss management after surgical repair including feeding protocols, pain management, and prophylactic antibiotics.2,4-6 However, guidance regarding adjuvant therapies for primary surgical repair is limited. Independent studies have examined the use of botulinum toxin type A, revision fat grafting, silicone gel and sheeting, intense pulsed light, and carbon dioxide (CO2) lasers to ameliorate contracture and hypertrophic scar formation.7-12 Other studies have attempted to use fat grafting, fillers, or autologous tissue transfers to achieve the desired contour and improve volume deficiencies.13,14 To date, a comprehensive review of such adjuvant treatment options to inform the cleft provider has yet to be published. Therefore, the aim of this scoping review is to examine different adjuvant techniques and therapies that modulate scar healing to improve the esthetic outcome for primary cleft lip repair outcomes in the perioperative period.
Methods
This scoping review was conducted in accordance with the JBI SUMARI methodology for scoping reviews. 15 All reviews and meta-analyses were excluded. Study quality was assessed using the Jadad et al and Methodological Index for Nonrandomized Studies instruments.16,17 All included studies were Level of Evidence I-IV. 18
Search Strategy
The search strategy, including all identified keywords and index terms, was adapted for each database. PROSPERO, PubMed/MEDLINE, SCOPUS, Cochrane Central Register of Controlled Trials, Web of Science, and ClinicalTrial.gov were searched. Only studies published in English were included. There was no restriction on the publication date range. Experimental, quasi-experimental, and analytical observational studies including prospective and retrospective cohort studies, case-control studies, and analytical cross-sectional studies were included.
Key terms included a combination of Medical Subject Headings (MeSH) terminology including cleft lip or harelip, cleft lip therapy, injections, Botox, botulinum toxin, platelet rich plasma, silicone, hydrocolloid bandage, scar massage, nasal stents, nasoalveolar molding or NAM therapy, presurgical orthopedics or Latham device or Dynacleft, hydrogels, perioperative care, newborn or infant or child or pediatric, and English language. 19 For additional details including the full search strategy, please refer to the Supplemental Materials.
Patients with syndromic and non-syndromic cleft lip, including unilateral and bilateral, who underwent primary cleft lip repair were included. Patients undergoing secondary cleft lip repair or revision were excluded. Interventions had to be performed either intraoperatively or immediately postoperatively. There were no minimum or maximum follow-up duration and no standardized measurement of the outcomes. Studies that examined operative techniques, revision techniques, flap designs, antibiotics, nursing care, feeding techniques, hospital care, nasoalveolar molding, and other forms of presurgical orthopedics were excluded.
Study Selection
Upon search completion, all identified citations were collated and uploaded into JBI SUMARI. 15 Two independent reviewers (M.L., J.P. || medical students) screened studies by title and abstract for assessment against the inclusion criteria for the review. After screening for relevant abstracts, the full text of selected citations was assessed in detail against the inclusion criteria by 2 independent reviewers. Reasons for exclusion of papers at the full-text stage were recorded. Any disagreements that arose between the reviewers at each stage of the selection process were resolved with an additional reviewer (M.J.P. || craniofacial surgeon). The results of the search and the study inclusion process were reported in full in the final review and presented in a Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) flow diagram (Figure 1). 20 Data was extracted by 2 independent reviewers using a standardized data extraction tool. 15 Extracted data included patient demographics, study methodology, study location, context, interventions, and cleft lip surgical outcomes.
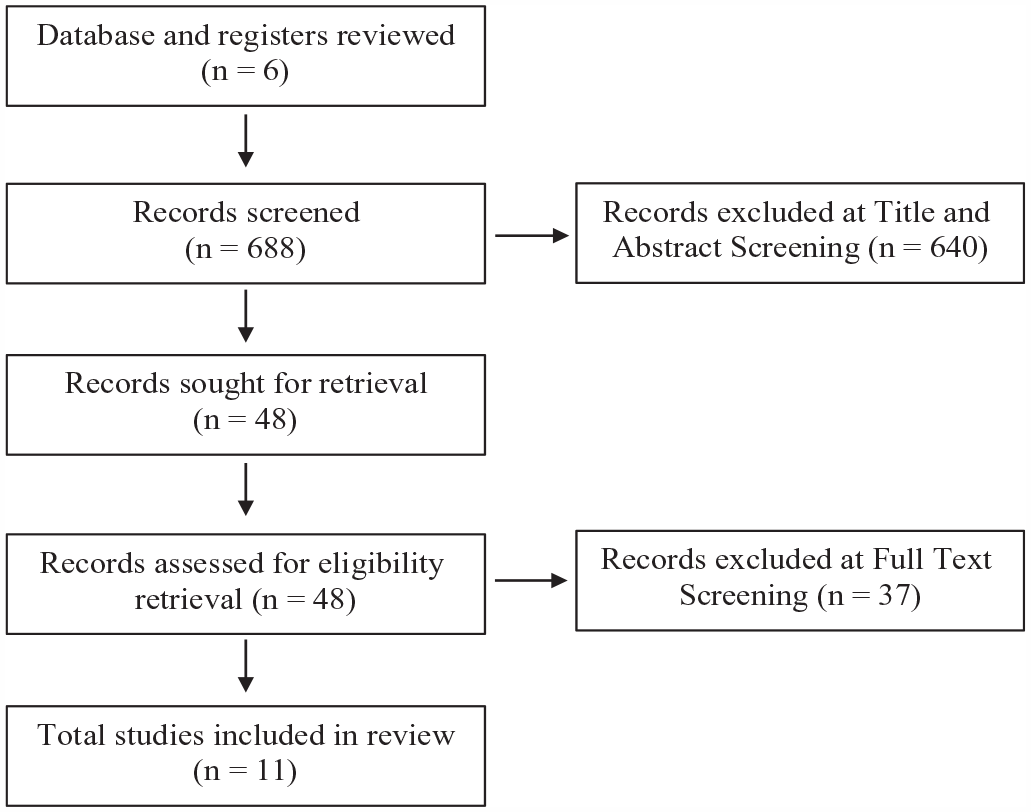
Results from literature search in the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) flow diagram.
Results
Study Inclusion
The review resulted in 688 studies, with 48 studies including relevant key terms in the title and abstract screening. After the full-text screening, 11 studies were included in the final scoping review (Figure 1). Reasons for exclusion included ineligible participant characteristics and ineligible intervention. Included studies were then grouped into 4 groups by associated intervention methods: botulinum toxin studies, fat grafting, nasal stents, and dressings and topical treatments.
Using the Jadad quality index evaluation of the included papers, there was perfect agreement among the 2 raters (weighted κ = 1, P-value < .0001). Using the MINORS assessment of methodological quality on the included papers, there was a strong level of agreement among the 2 raters (κ = 0.817, P-value < .0001).
Characteristics of Included Studies
Characteristics of included studies are tabulated in the Table 1. Four randomized controlled trials, one quasi-experimental multi-center study, 2 case series, and 4 case control studies met inclusion criteria. The included studies took place in the United States of America (3), Taiwan (3), Belgium, Germany, Latvia, Lithuania, Estonia, Mexico, India, and Nigeria. Adjuvant therapies evaluated included botulinum toxin (3), dressings and topicals (3), nasal stent (2), fat grafting (2), care package, silicone gel. There were no studies found that used platelet rich plasma or scar massage alone. The studies were grouped by associated intervention methods: Botulinum Toxin, Fat Grafting, Nasal Stents, and Dressing and Topical Treatments.
Study Characteristics and Data From Included Studies.
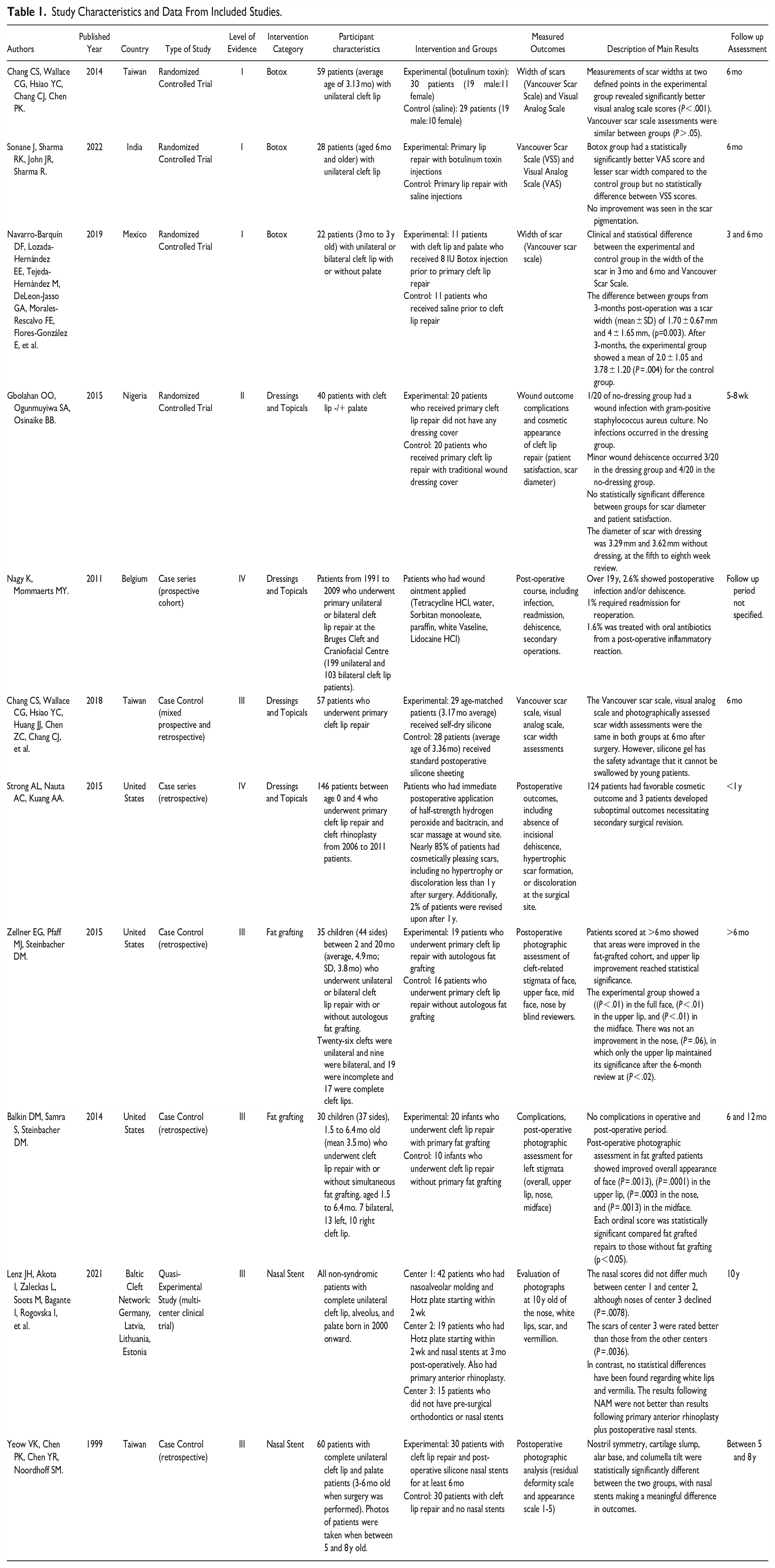
Measured primary and secondary outcomes from adjuvant therapies varied by study, including the Vancouver Scar Scale (VSS), Visual Analog Scale (VAS), complications and patient safety, patient compliance, and general cosmetic appearance or photographic assessment for cleft stigmata.
Botulinum Toxin
Three randomized controlled studies used botulinum toxin as adjuvant therapy and reported outcomes using the VAS, VSS, and scar-width scales.21-23 In all 3 studies, the VAS score and scar width measurements were significantly improved postoperatively after 6 months, and some showing improvement in 3 months. Two of the 3 randomized controlled trials did not show significant improvement in the VSS scale after 6 months.21,22
Fat Grafting
Two perioperative, primary cleft lip repair studies, which were categorized into a prospective study and retrospective analysis respectively, both showed significant improvement in overall patient satisfaction with fat grafting.24,25 Weeks after the operation, independent reviewers were asked to assess the full face, upper lip, nose, and midface on a five-point scale (ranging from “invisible stigmata” to “very visible” stigmata) for cleft-related facial stigmata postoperative photos on a five-point scale. In both studies, results showed significant improvements in the full face, upper lip, midface.24,25 In the retrospective study, there was lasting significant improvement in all regions after 6 months for unilateral cleft lip and 12 months for bilateral cleft lip while the prospective study only showed improvement in the lip region after 6 months.24,25
Nasal Stents
Two studies examined nasal stents, with one study retrospectively comparing the application of postoperative nasal stents alone and the other examining the use of nasal stents in combination with other inventions compared to other presurgical and postoperative methods in a multicenter trial.26,27 With at least 6 months of nasal stent use, 5-to-8 years postoperative photographs showed that nostril symmetry, alar cartilage slump, alar base level, columella tilt, and overall esthetic appearance were significantly improved compared to the group that did not use it. 26 In the multicenter clinical trial, 3 centers had different interventions: center 1 consisted of 42 patients that had presurgical nasoalveolar molding (NAM) and presurgical Hotz plates, center 2 consisted of 19 patients who had a presurgical Hotz plate and postoperative nasal stents, and center 3 consisted of 15 patients without presurgical orthodontics or postoperative nasal stents. 27 Postoperative photographs were evaluated using a visual rating scale of the nose, lip skin, lip scar, and vermillion. Nose evaluations were comparable from NAM (center 1) and nasal stents (center 2). 27 However, lip scars from operations without NAM or nasal stents (center 3) were rated better compared to operations with NAM and nasal stents. 27 All centers experienced no statistical difference regarding lip skin and vermillion. 27
Dressings and Topical Treatments
In a randomized controlled trial in Nigeria, dressings did not have an impact on scar formation as there was no statistically significant difference between the 2 groups on postoperative complications and scar diameter. 28 Topical treatments for primary cleft repair include ointments, silicone sheets, and hydrogen peroxide application. One center described their specific postoperative regimen and examined their incidence of postoperative complications and scar appearance after primary cleft lip repair. This regimen included intravenous amoxicillin for 2 days postoperatively, and oral amoxicillin during feeding, Logan bar with adhesive tape, immediate post-operative local anesthetic solution, saline cleaning, elbow restraints, and custom ointment, including tetracycline, sorbitan monooleate, paraffin, white Vaseline, and lidocaine, which was applied twice daily at the hospital and for 2 weeks after discharge. The results showed that 2.6% of patients had a postoperative infection and/or dehiscence, 1% had a secondary operation, and 1.6% had a post-operative inflammatory reaction that was treated successfully with oral antibiotics over 19 years. 29 In another study comparing standard silicone sheet and self-drying silicone gel on the incision site immediately postoperatively, after 6 months, VSS and VAS scores were not different between the 2 groups. 12 In a retrospective study examining the application of hydrogen peroxide, where primary cleft lip incision sites were treated with half-strength hydrogen peroxide, bacitracin, and a scar massage in all patients, about 85% of patients had cosmetically favorable scars, which includes no hypertrophy or discoloration at the surgical site, at less than 1 year postoperatively. 30
Discussion
As demonstrated in this review, adjuvant surgical techniques can improve cosmesis, aid healing, and prevent fibrosis after primary cleft lip repair. Many of the current adjuvant therapies are derived from adult scar revision and modulation. However, the pediatric population presents unique challenges such that stability of the wound and adherence to treatment cannot be guaranteed. The results of our scoping review have illustrated many of the options available to surgeons for primary cleft lip repair, including botulinum toxin, fat grafting, use of nasal stents, silicone gel, hydrogen peroxide, antibiotic ointment.
Botulinum Toxin
Preventing adverse scar formation in the lip area is challenging due to essential role of the orbicularis oris in feeding and speaking. Since tensile and mechanical forces are key players in the formation and widening of scars, the injection of botulinum toxin in the early phases of healing can help to immobilize the local area until collagen matures. Botulinum toxin blocks the presynaptic release of acetylcholine, temporarily paralyzing the muscle to reduce tensile forces on the repair. 31 Additionally, botulinum toxin directly inhibits the release of inflammatory molecules, such as serotonin, that can mitigate scar hypertrophy. 32 For over 25 years, botulinum toxin has been used safely and successfully in the pediatric population, mainly for cerebral palsy and other spastic muscle conditions. While botulinum toxin has been shown to successfully improve facial scars during revision surgery of hypertrophic and depressed scars, it has not been widely adopted for use in primary cleft lip repair.33,34 In our review, the measured outcome played a large role in the success of the botulinum toxin intervention, with all studies showing scar improvement when measured by scar width and VAS scale, and not all showing improvement with VSS scale, which was also confirmed in a recent systematic review of botulinum toxin treatment for scar prevention in primary cleft lip repair. 35 Given these results, botulinum toxin injection in combination with other scar modulating treatments is a promising adjuvant treatment for primary cleft lip repair. Botulinum toxin injections have also been successfully used for cleft lip revisions in adults immediately after wound closure, resulting in significant improvement of lip scarring. 11 However, a limitation for widespread use is insurance coverage and the cost of each treatment.
Fat Grafting
Fat grafting has been found to improve skin quality and scar appearance while enhancing contour and structure, and incorporating this procedure into primary cleft lip repair is believed to help mitigate potential postoperative complications. 36 Mechanically, fat grafting helps to mitigate physical depressions and balance the contours of the nasolabial complex. Previous research has shown that pediatric and adult patients who had additional lip irregularity after primary cleft lip repair and underwent a revision operation with fat grafting had significantly improved satisfaction, cosmetic outcome, and decreased cleft stigmata.13,37-41 Some studies have even examined how fat grafting can improve functional and speech outcomes for patients with velopharyngeal insufficiency.42,43 The studies in our review have shown that fat grafting does have a lasting impact on both scar appearance and contour. However, fat retention and contour retention over long periods vary, so additional fat grafting procedures may have to be performed. In addition to its structural and functional impact, fat grafting can also have regenerative benefits, as adipose tissue contain adipose-derived stem cells (ADSC) can support wound healing and regeneration.44-51 Therefore, fat grafting techniques have the potential to assist in the correction of asymmetry by soft tissue augmentation and provide a milieu for tissue regeneration, making it an attractive option in primary cleft lip repair. 52 However, higher level of evidence studies are needed to further define its role in primary cleft lip repair.
Nasal Stents
Nasal stents or splints are often used immediately postoperatively after primary cleft lip repair for at least 6 months, as they help to prevent the lower lateral cartilage and alar of the nostrils from collapsing during the wound healing process. A nasal stent or splint is comprised of 2 hollow tubes made from silicone, rubber, or other pliable materials, with a connection between the 2 tubes that will lie along the columnar bridge of the nose. 26 The literature mainly consists of studies examining the effect of nasal stents in combination with preoperative regimens such as presurgical orthopedic devices, NAM, the Latham appliance, and Dyna cleft, so evidence is limited on the effect of nasal stents alone. While nasal splints have been used in other fields such as trauma or burns to keep the nasal airway open, their use in cleft lip care is limited to the last 2 decades. However, the limited studies have been impactful, impacting postoperative care guidelines for all cleft lip and palate patients. Nasal stents or splints are also inexpensive and easily understood by caretakers, likely facilitating compliance.
Dressings and Topicals
Postoperative wound dressings are utilized as a simple, cost-effective physical barrier to enhance wound healing, reduce pain, protect the wound from foreign material and bacteria, and reduce healing time. 53 The variety in effectiveness and benefits of dressings is centralized on maintaining the correct amount of moisture in the wound bed. Effective mediums and materials such as topical pain medication, silicone gel sheets, and antibiotic ointments can also be beneficial tools in cleft lip post-operative care to impact healing and scar formation. Semi-permeable dressings serve as a barrier for infection while allowing for moisture regulation and evaporation. Hydrogels, which provide a moist environment for wounds to heal, are shown to have high patient satisfaction when utilized in primary cleft lip repair. 53 Silicone products have also been described to be an effective therapeutic strategy to prevent pathologic scarring. A review of randomized controlled trials of silicone products compared to onion extract gel (Contratubex®; Merz Therapeutics; Frankfort, Germany) illustrated that silicone products, including both silicone sheets and gel, were superior in the treatment of hypertrophic scars. 54 However, in more recent systematic reviews studying the efficacy of silicone gel sheeting for prevention of abnormal scarring, there has been limited evidence demonstrating its effectiveness.55,56 Although the exact mechanism of action has not been defined, proposed theories include providing bacterial and barrier protection, modulation of inflammation, and hydration to the wound bed. 57 The benefits of silicone gel include its comparable efficacy profile, inability to be ingested (unlike silicone sheets), and its easy application to irregular surfaces.29,55,58,59 As another proposed topical treatment, hydrogen peroxide is a useful adjunct in postoperative care; however, its efficacy and potential toxicities are heavily debated in other wound use cases. 60 However, the study included in our review found significantly reduced rates of wound dehiscence, hypertrophic scar formation, or discoloration at the surgical site. 30 This benefit, in combination with the low cost and accessibility of hydrogen peroxide, can make it an effective option for wound care management.
Other Post-Operative Therapies
Laser treatments for the treatment of hyperpigmented, atrophic, keloid, or hypertrophic scars have long been utilized with pulsed dye, carbon dioxide, diode, potassium titanyl phosphate, and erbium glass lasers shown to be safe, effective, and useful for the prevention of unfavorable scars postoperatively.61-63 However, laser treatment as a prevention measure has not been well-studied in the cleft lip setting. For patients who had undergone primary cleft lip repair, postoperative laser treatments, CO2 laser, and intense pulsed laser specifically, have been shown to be a successful way of giving scars more pliability and flattening, with an improved appearance compared to patients using scar cream and scar massage.10,64,65
Limitations
From this scoping review, it can be concluded that there are many effective adjuvant treatments that can be implemented for scar modulation in primary cleft lip repair. However, these studies are varied in measured outcomes, typically combined with other treatments, and limited in strong evidence for each of the reviewed interventions. Many of these studies have therapies without a control group, are retrospective, have small sample sizes, or combine multiple treatments, such as scar massage, silicone gel, and ointments, some that are unreported, so it is difficult to definitively conclude a specific treatment’s efficacy alone. Our review only found a few studies per treatment category, with most of the current literature on adjuvant treatments for scar modulation studying scar modulation as a whole, but not primary cleft lip scars. Outside of our review, there is a lack of data summarizing adjuvant treatments cleft providers can use perioperatively for primary cleft lip repair.
Conclusion
Adjuvant therapies to mitigate scar formation can be beneficial tools in a cleft provider’s postoperative care regimen. From this scoping review, using botulinum toxin and immediate fat grafting intraoperatively, utilizing nasal stents and topical treatments such as antibiotic ointment, lidocaine, hydrogen peroxide, and silicone gels immediately post-operatively, and using laser treatment later in the cleft lip patient’s care are worthwhile to consider as a combination of adjuvant treatments to prevent adverse scar formation in patients undergoing primary cleft lip repair. The most robust evidence lies with botulinum toxin use, which is supported by several randomized studies.
Supplemental Material
sj-docx-1-fac-10.1177_27325016231212564 – Supplemental material for A Scoping Review of Adjuvant Perioperative Therapies for Primary Cleft Lip Repair
Supplemental material, sj-docx-1-fac-10.1177_27325016231212564 for A Scoping Review of Adjuvant Perioperative Therapies for Primary Cleft Lip Repair by Melinda Lem, Jason T Pham, Jagmeet Arora, Shivang Trivedi, Omotayo Arowojolu, Ruben Castro, Joseph Mocharnuk, Raj Vyas and Miles J. Pfaff in FACE
Footnotes
Acknowledgements
We would like to thank Linda Suk-Ling Murphy for her assistance in this manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Institutional Review Board approval was not required.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
