Abstract
Objective:
The aim of this study was to determine whether patients with syndromic craniosynostosis (SCS) are at increased risk for abnormal speech and language development (ASLD) relative to patients with non-syndromic craniosynostosis (NSCS). Study Design: A retrospective cohort study was completed using the Kids’ Inpatient Database (KID). All patients with craniosynostosis (CS) were included were included. The primary predictor variable was study grouping (SCS vs NSCS). The primary outcome variable was ASLD. Multivariate logistic regression were performed to identify risk factors for ASLD.
Results:
The final study sample included a total of 10 089 patients with craniosynostosis (CS) (37.7% female, 51.6% White, mean age 1.78 years). Patients with SCS were at increased risk for ASLD relative to patients with NSCS (OR 2.1, P < .001). After controlling for all other variables, patients with SCS were no longer at increased risk for ASLD relative to patients with NSCS (OR 1.2, P = .442).
Conclusions:
Relative to NSCS, SCS per se is not a risk factor for ASLD. The significantly greater prevalence of asthma, intracranial hypertension, and compression of brain, all of which are risk factors for ASLD, among patients with SCS explains the greater prevalence of ASLD in SCS relative to NSCS.
Introduction
Craniosynostosis (CS) is a congenital anomaly that is characterized by the premature fusion of the calvarial sutures. CS presents in 1 of 2 forms: non-syndromic CS (NSCS) and syndromic CS (SCS). 1 NSCS manifests as the prematurely fused sutures in isolation of any extracranial anomlaies. 2 As the name implies, SCS is associated with a syndrome, such as Crouzon and Apert syndrome, and often involves multiple sutures. 3 SCS is less common than NSCS. While NSCS occurs in 0.6 for every 1000 births, 4 Crouzon syndrome and Apert syndrome occur in 0.01655 and 0.00156 for every 1000 births, respectively.
Neurocognitive impairments occur among patients with CS with the extent of the impairment being more severe among patients with SCS.7,8 Neurological sequelae are believed to stem from the rapidly growing brain being constricted by the cranium. 9 Exploring the intracranial volume (ICV)-to-cortical surface area (CSA) relationship among patients with SCS and controls, Wilson et al determined that patients with fibroblast growth factor receptors (FGFR)-mediated SCS (ie, Crouzon, Apert, Saethre-Chotzen syndrome) had significantly greater ICV than controls (P < .001). Nevertheless, when CSA was scaled to ICV, patients with FGFR-mediated CS had a significantly reduced exponent (0.68, 95% CI 0.61-0.76) compared to controls (0.77, 95% CI 0.61-0.93), indicating reduced CSA development among the FGFR-mediated group. This trend was especially the case for the parietal and occipital lobes. Coincidentally, when examining the educational data of FGFR-mediated group, virtually every patient required modified education. This led Wilson et al to propose a potential link between cortical development and neurodevelopmental outcomes among these patients. 10
Abnormalities of speech and language have been reported among patients with SCS11,12 and NSCS.13 -17 These abnormalities include language acquisition delay/disorder, speech production delay/disorder, hypernasality, and velopharyngeal insufficiency.14,18 Moreover, hearing loss has been noted in both SCS and NSCS, which substantially impairs language acquisition and development.12,19 -21 Hearing loss is markedly more common among patients with SCS, being as high as 92%, 80%, and 74% in Pfeiffer, Apert, and Crouzon syndrome respectively. 19 Several studies have shown improvements of language and speech after craniofacial surgical correction.18,22
To our best knowledge, no study has yet to compare patients with SCS and NSCS in terms of abnormal speech and language development (ASLD). It is not known whether patients with SCS are at increased risk for ASLD relative to NSCS or vice versa. The purpose of this study was to answer the following question, “are patients with SCS more likely to have ASLD than patients with NSCS?” Specifically, we sought to (1) accumulate data on all patients diagnosed with CS from a large pediatric database, (2) compare the patients according to the type of CS (SCS vs NSCS), (3) examine the prevalence of ASLD across all study variables, and (4) determine independent risk factors for ASLD. We hypothesized that patients with SCS would be at increased risk for ASLD compared to patients with NSCS.
Materials and Methods
Study Design and Sample Selection
A retrospective cohort study design was implemented using the Kids’ Inpatient Database (KID) for the data years 2016 and 2019. Developed for the Healthcare Cost and Utilization Project (HCUP), the KID is the largest publicly available all-payer pediatric (<21 years-of-age) inpatient database in the United States and reports on approximately 3 million discharges annually. 23 The KID database is released on a triennial basis. The 2016 KID dataset includes data for the years 2016, 2017, and 2018. The 2019 KID dataset includes data for the years 2019, 2020, 2021. From 2016 onward, the KID database was coded using the ICD-10 coding system, which distinguishes craniofacial anomalies unlike the ICD-9 coding system.
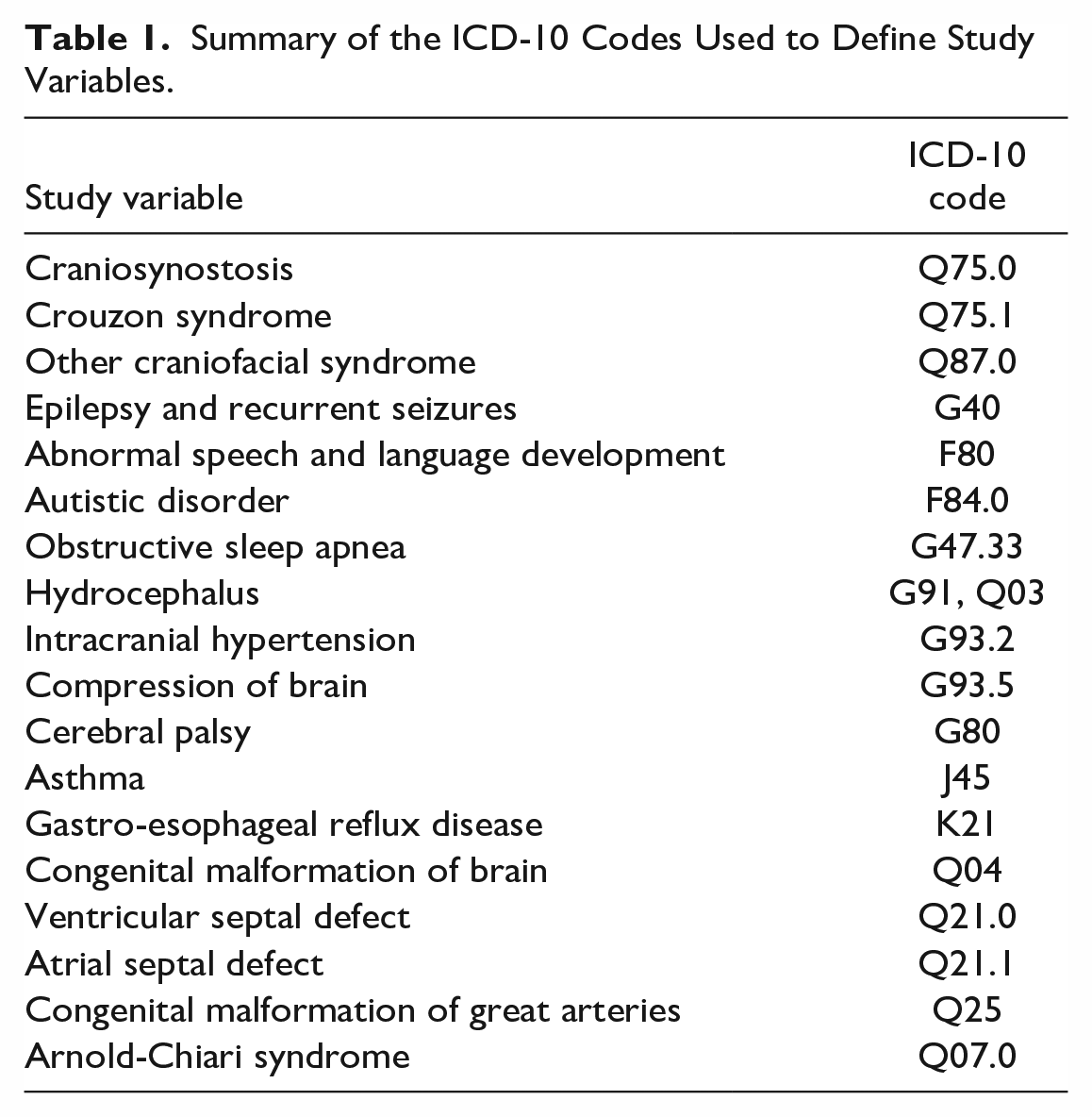
The 2016 and 2019 KID datasets include up to 30 different potential ICD-10 diagnoses for each patient. The final study sample was achieved by including all patients diagnosed with CS via the corresponding ICD-10 code (Q75.0). The SCS study group consisted of patients with CS that were also diagnosed with craniofacial dysostosis (Q75.1) or any other unspecified SCS (Q87.0). The remaining patients with CS comprised the NSCS study group.
Study Variables
The primary predictor variable was study grouping (SCS vs NSCS). The primary outcome variable was diagnosis of ASLD. The secondary predictor variables included patient demographic factors (age, sex, race, median household income, patient location, payer information) and comorbidities. Age was a continuous variable (years). Median household income was determined by ZIP code and was categorized into quartiles (Q1, Q2, Q3, and Q4). Patient location was categorized as large metro area, small metro area, or micropolitan county. Population size distinguished between large (≥1 million) and small (50 000-999 999) metro areas. Payer information denotes primary payer of procedures and services that were received during hospitalization (Medicare/Medicaid, private insurer, or self-pay).
Comorbidities were derived from the ICD-10 codes as was done with the primary predictor variable (Table 1). Comorbidities included respiratory comorbidities (obstructive sleep apnea [OSA], asthma), cardiovascular comorbidities (ventricular septal defect [VSD], atrial septal defect [ASD], congenital malformation of great arteries), neurological comorbidities (epilepsy and recurrent seizures, hydrocephalus, intracranial hypertension, compression of brain, cerebral palsy [CP], congenital malformation of brain, Arnold-Chiari syndrome) and miscellaneous comorbidities (autistic disorder, gastro-esophageal reflux disease [GERD]).
Summary of the ICD-10 Codes Used to Define Study Variables.
Data Analyses
Descriptive statistics were completed for all variables. Univariate analyses (chi square or fisher exact) were conducted to determine associations between predictor variables and the primary outcome variable. Predictor variables with significant associations with the outcome variable were further analyzed via univariate logistic regression to determine risk factors for ASLD. All confounding variables were controlled for in the multivariate logistic regression model. SPSS version 28 for Mac (IBM Corp., Armonk, NY, USA) was used to perform the statistical analyses. Statistical significance was considered for a P value of less than .05.
Results
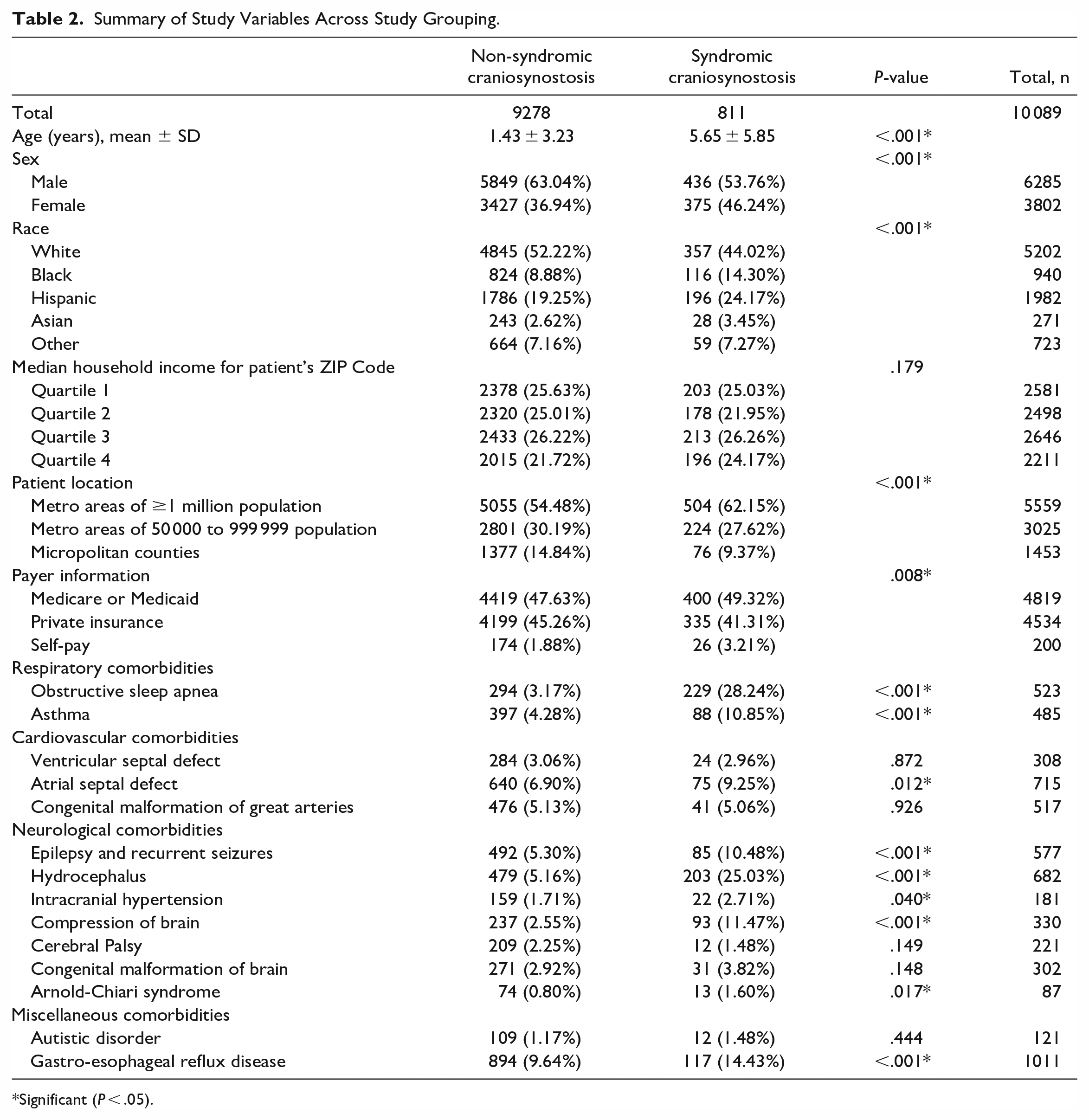
The final study sample included a total of 10 089 patients—9278 patients (92.0%) had NSCS, and the remaining 811 patients (8.0%) had SCS. Of the 811 patients with SCS, 609 had Crouzon syndrome while the remaining 202 had an unspecified form of SCS (ie, Apert syndrome). The mean age of the study sample was 1.78 years (SD, 3.70 years) and 3802 patients (37.7%) were female. Table 2 summarizes the study variables across study grouping (SCS vs NSCS). Age, sex, race, patient location, payer information, OSA, asthma, ASD, epilepsy and recurrent seizures, hydrocephalus, intracranial hypertension, compression of brain, Arnold-Chiari syndrome and GERD were all significant predictors of study grouping.
Summary of Study Variables Across Study Grouping.
Significant (P < .05).
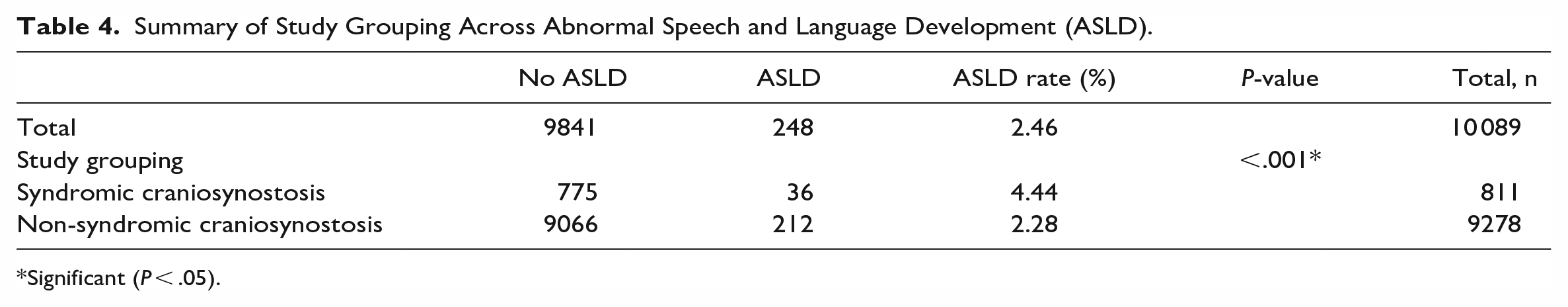
A total of 248 patients (2.5%) exhibited ASLD in our study. Table 3 summarizes the study variables across ASLD. Age, sex, payer information, OSA, asthma, epilepsy and recurrent seizures, intracranial hypertension, compression of brain, CP, Arnold-Chiari syndrome, autistic disorder, and GERD were all significant predictors of ASLD. Patients with SCS had a significantly higher rate of ASLD than patients with NSCS (4.4%vs 2.3%, P < .001) (Table 4).
Summary of Study Variables Across Abnormal Speech and Language Development (ASLD).
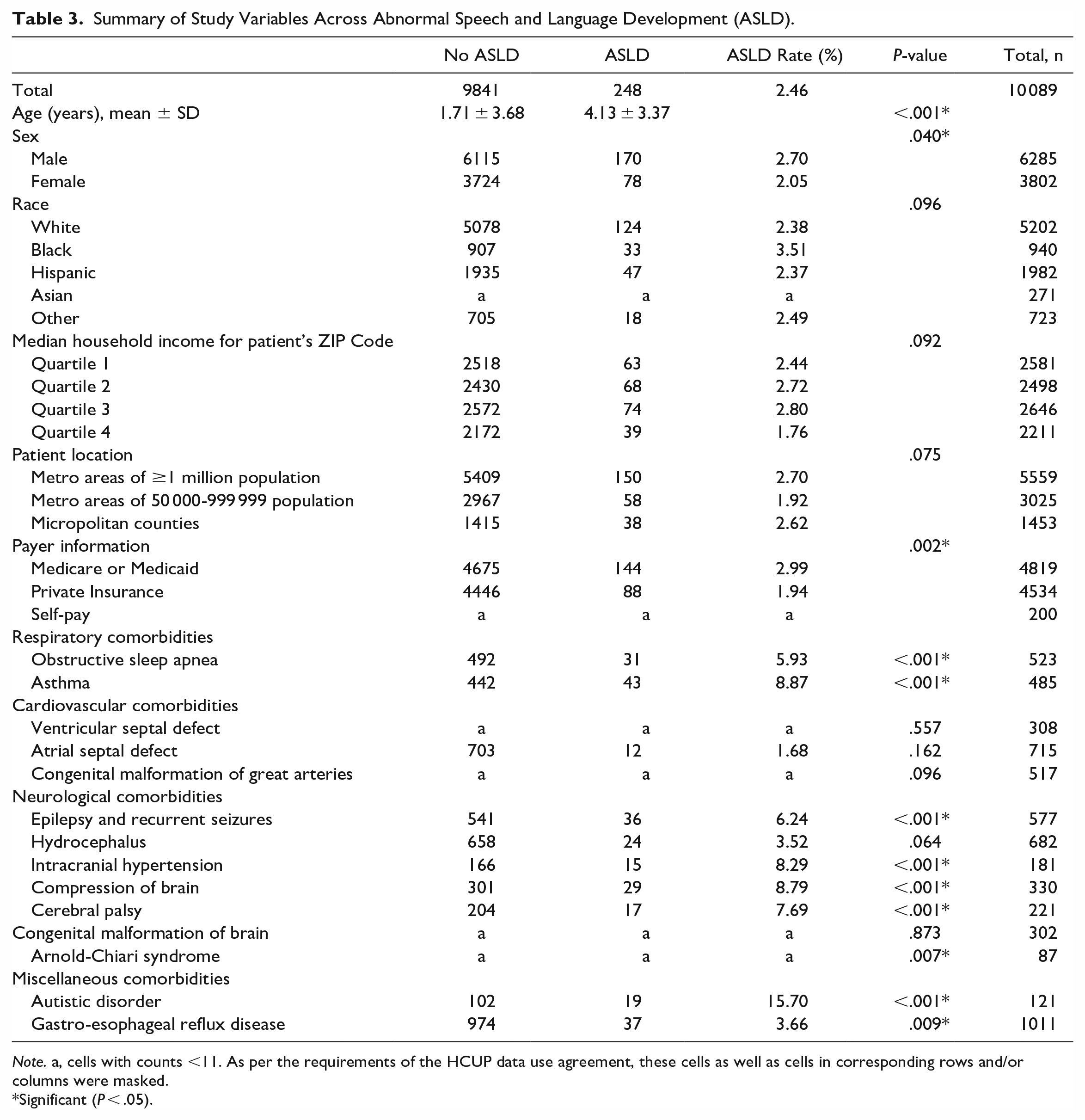
Note. a, cells with counts <11. As per the requirements of the HCUP data use agreement, these cells as well as cells in corresponding rows and/or columns were masked.
Significant (P < .05).
Summary of Study Grouping Across Abnormal Speech and Language Development (ASLD).
Significant (P < .05).
Univariate logistic regression was conducted to determine risk factors for ASLD. Patients with SCS were at increased risk for ASLD relative to patients with NSCS (OR 2.1, P < .001) (Table 5). After controlling for all other variables, multivariate logistic regression was conducted. Patients with SCS were no longer at increased risk for ASLD relative to patients with NSCS (OR 1.2, P = .442). Every additional year in age increased the risk for ASLD (OR 1.1, P < .001). Regarding respiratory comorbidities, asthma was an independent risk factor for ASLD (OR 2.6, P < .001). Concerning neurological comorbidities, intracranial hypertension (OR 2.6, P = .001) and compression of brain (OR 3.1, P < .001) were independent risk factors for ASLD. Finally, autistic disorder was the strongest independent risk factor for ASLD (OR 3.8, P < .001) (Table 6).
Univariate Logistic Regression for Risk of Abnormal Speech and Language Development (ASLD).
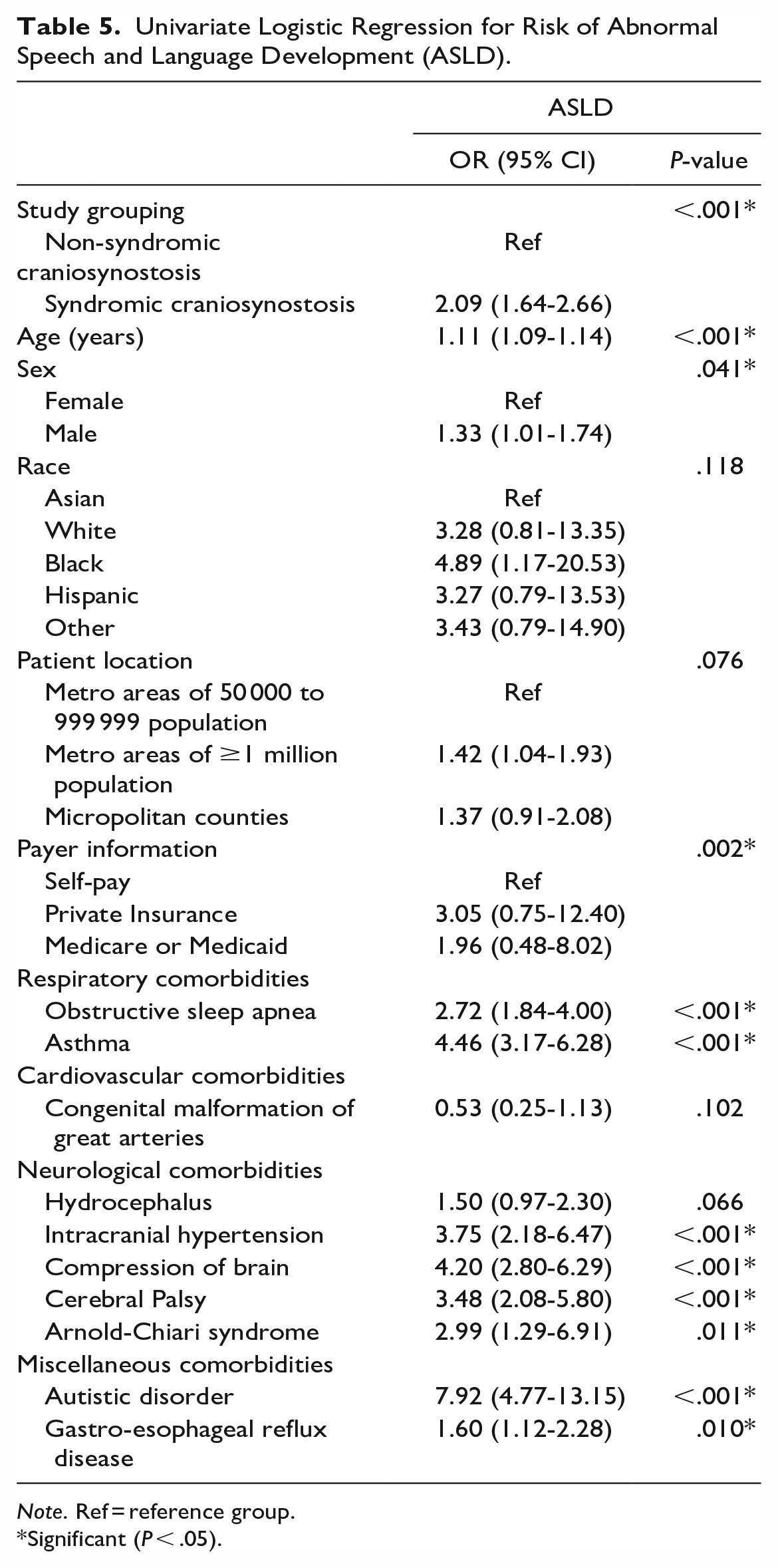
Note. Ref = reference group.
Significant (P < .05).
Multivariate Logistic Regression for Risk of Abnormal Speech and Language Development (ASLD).
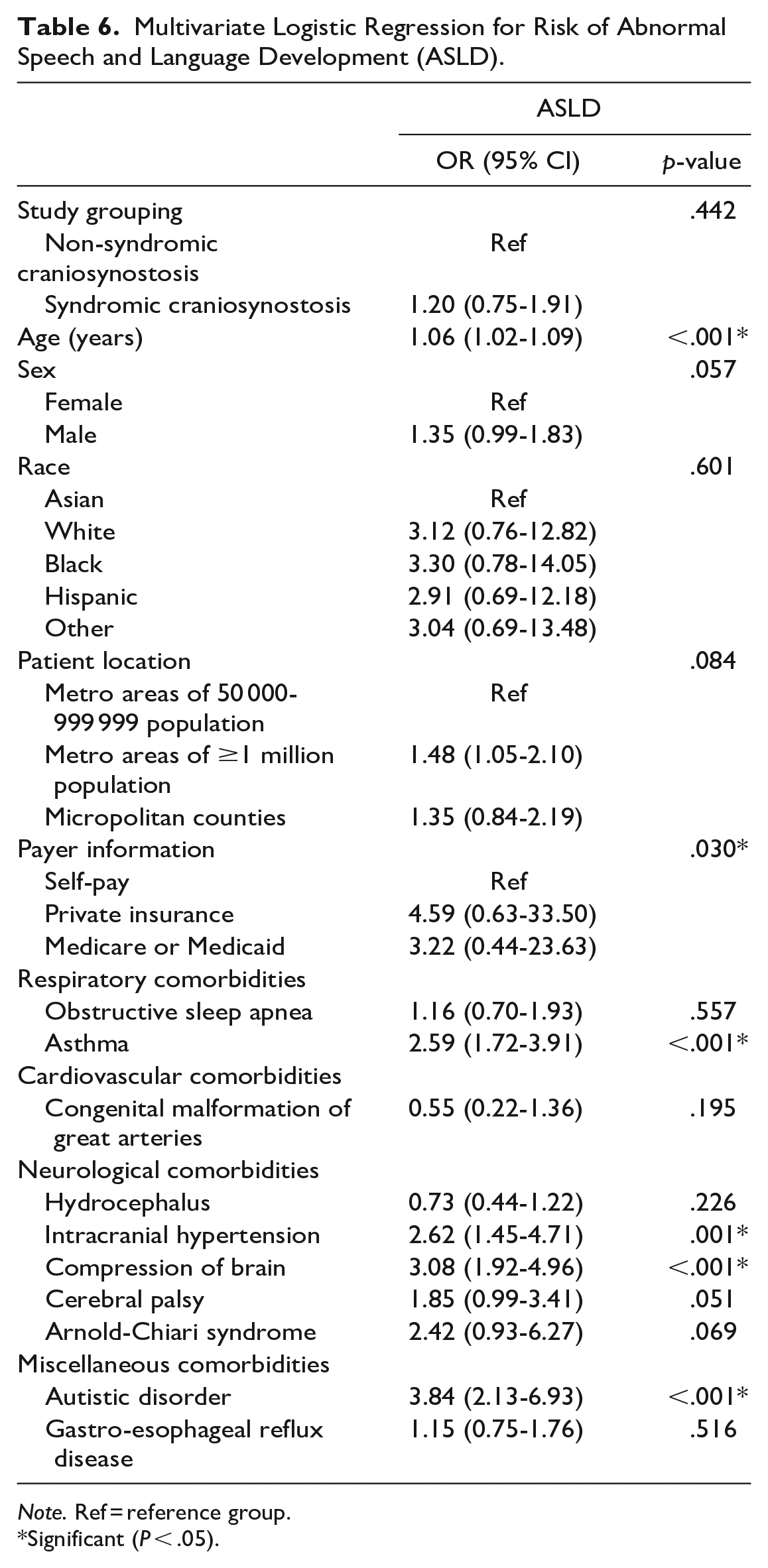
Note. Ref = reference group.
Significant (P < .05).
Discussion
The purpose of our study was to determine whether patients with SCS are more likely to have ASLD than patients with NSCS. We hypothesized that patients with SCS would be at increased risk for ASLD compared to patients with NSCS. We ultimately failed to reject the null hypothesis since, after controlling for all significant covariates, patients with SCS were not at an increased risk for ASLD relative to patients NSCS. Nevertheless, age was a risk factor for ASLD. Asthma was a respiratory risk factor for ASLD. Intracranial hypertension and compression of brain were each neurological risk factors for ASLD. Lastly, autistic disorder was the strongest risk factor for ASLD.
Intracranial hypertension and compression of brain were significantly more common among patients with SCS than NSCS. Further, each comorbidity was a risk factor for ASLD in our study. Intracranial hypertension (ICH) has been shown to occur more often among patients with SCS than patients with NSCS. 24 Regarding SCS, it is most common among patients with Apert syndrome and Crouzon syndrome, with reported prevalences of 71% and 61%, respectively.25,26 ICH is thought to arise from venous hypertension due to stenosis of the jugular-sigmoid complex and transverse sinuses. 27 Patients with CS have a significantly smaller mean jugular foramen diameter in the greatest anterior-posterior direction relative to controls. 28
The link between ICH and ASLD among patients with CS has a neurological basis. Wilson et al sought to identify the impact of ICH on cortical thickness among patients with SCS. The authors determined that papilledema was significantly associated with cortical thinning. Fundoscopic presence of papilledema has been shown to be a reliable indicator of ICH among children with CS. 29 Papilledema manifests only after a prolonged period of ICH. By the time papilledema is apparent, cortical neurons have already been stressed to an extent sufficient to result in irregular cortical remodeling and, on a large scale, cortical thinning. 10 On the same topic, Thiele-Nygaard et al conducted a systematic review of the literature on the association between ICH and negative cognitive outcomes in patients with sagittal CS. The authors determined that certain areas of the brain in charge of language and speech were different among patients with sagittal CS compared to age-matched controls. 30
According to the ICD-10 coding system, compression of the brain is applicable to Chiari type I and herniation of the brain. Similar to ICH, Chiari malformation (CM) is much more common among patients with SCS than patients with NSCS. According to Matsuhashi et al, the prevalence of CM among patients with SCS and NSCS is 31.8% and 10%, respectively. 31 CM is thought to develop among patients with SCS due to ICH and an exceedingly small posterior cranial fossa that cannot keep up with the hindbrain’s growth.32,33
Several surgical interventions can correct CM among patients with CS. According to Park et al, posterior cranial vault distraction osteogenesis (PVDO) can effectively alleviate CM type I in patients with CS. After PVDO was completed, the intracranial volume and posterior skull base increased significantly. Concurrently, cerebellar tonsillar descent significantly decreased, with some patients achieving complete resolution of herniation. 34 Foramen magnum decompression (FMD) is another surgical means that can alleviate CM among patients with CS. Among 5 children with CS, Addo et al determined that FMD alleviated central sleep apnea, a proxy of CM, by at least 60%. 35
Asthma was a risk factor for ASLD in our study. Further, asthma occurred more frequently among patients with SCS than patients with NSCS. According to the literature, we found no publications on Asthma in the context of CS. Asthma is often comorbid with OSA and a bidirectional relationship exists between the 2 disorders. 36 This is important since OSA has established neurocognitive implications, which are mediated by intermittent hypoxia and neuroinflammation.37,38 Shi et al sought to compare the levels of Neurofilament light (NfL), a marker of neuroaxonal damage, among patients with and without OSA. The authors determined that patients with OSA had significantly higher levels of NfL relative to patients without OSA (P < .001). The level of NfL was also significantly correlated with via various parameters of OSA, including apnea–hypopnea index (AHI), obstructive apnea index (OAI), obstructive apnea–hypopnea index (OAHI), average oxygen saturation (SaO2), and respiratory arousal index (RAI) as well as Verbal Intelligence Quotient. 39 In another study, Lewin et al sought to explore the daytime cognitive sequelae of OSA in children. The authors found a negative correlation between OSA severity and verbal ability. 40
OSA is significantly more common among patients with SCS than patients with NSCS. This is because patients with SCS, particularly Apert syndrome and Crouzon syndrome, are likely to suffer from midfacial hypoplasia that translates to a narrow retropalatal region. 41 A study exploring the course of OSA among 97 children with SCS determined that the prevalence of OSA was 68%. 42 In another study, patients with SCS were approximately 5 times more likely to have OSA relative to patients with NSCS (P< .001). 43
Within this discussion, it is important to mention that the ICD-10 code for ASLD also includes phonological disorder, which concerns the appropriate production of sounds. Apart from the neuropsychiatric mechanisms, such as ICH and CM, it is important to mention that the midfacial hypoplasia-induced malocclusion (angle class III) among patients can hinder their speech. What is more, surgical correction of midfacial hypoplasia among patients with SCS can acutely worsen speech proxied by symptoms of velopharyngeal insufficiency (VPI), which is characterized by failure of the soft palate as part of the velopharyngeal sphincter to separate the oral and nasal cavities during speech. 44 Zimmerman et al explored the effect of midfacial distraction osteogenesis (MDO) on speech among patients with SCS. The authors determined that, relative to the pre-operative phase, patients had significantly (P = .01) worse VPI 27.3 months post-operatively (ie, distractor removal). However, 90 months post-operatively, VPI was no longer significantly different than that pre-operatively (P = .31). 18
Autistic disorder (AD) was a risk factor for ASLD in our study. This result was not surprising since AD is characterized by impaired social interaction and communication. It is believed that 30% of Autistic children fail to acquire spoken language, even after access to intervention. 45 That said, we believe that AD is purely coincidental among patients with CS. The prevalence of AD is 1.20% of our study sample of patients with CS, which is similar to the prevalence of AD in the general population (one in 100). 46 Further, to the best of our knowledge, no association between CS and AD has been published in the literature as of yet.
This study is not without limitations. First, the results of this study are only as reliable as the ICD-10 codes and how accurately they were coded. It is possible that certain comorbidities might not have been coded during hospital admission since they were either not clinically apparent or not a substantial problem. For instance, ASLD includes phonological disorder, expressive language disorder, mixed receptive-expressive language disorder, speech and language development delay due to hearing loss, childhood onset fluency disorder, social pragmatic communication disorder, and other unspecified disorders of speech and language. It is quite plausible that some of these specific comorbidities were not clinically urgent issues next to the cause of hospitalization and, thus, were not coded for certain patients. Second, the KID contains data for only one hospital admission per patient. The comorbidities present reflect only what was diagnosed during the patient’s hospital admission. Hence, longitudinal analysis over a period of time was not possible. Third, the ICD-10 codes only specify one type of SCS (Crouzon syndrome). Being able to distinguish other types of SCS would have enabled a sub-group comparison as it relates to ASLD. In spite of these limitations, the following database enabled a large accumulation of patients with SCS. Further, the following database is nationally representative rather than being limited to a certain geographical region within the US.
Conclusion
Without controlling for other significant variables, patients with SCS were at increased risk for ASLD relative to patients with NSCS. However, after controlling for significant variables, patients with SCS were not at increased risk for ASLD. We believe this is due to the significantly greater prevalence of asthma, intracranial hypertension, and compression of brain among patients with SCS. Autistic disorder, albeit a risk factor for ASLD, was evenly distributed among patients with SCS and NSCS.
Footnotes
Acknowledgements
None
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Consistent with our medical center’s policies, research using anonymized and publicly available does not require institutional review board (IRB) approval.
