Abstract
Traumatic brain injury (TBI) is a common cause of morbidity and mortality worldwide. Early identification of potential TBIs by emergency medical services (EMS) clinicians is critical to improving outcomes. Numerous tools have been developed to support prehospital decision-making, but their diagnostic accuracy and real-world applicability remain unclear. We conducted a systematic review of English-language studies published between January 1, 2015, and December 31, 2024, using MEDLINE, Embase, ClinicalTrials.gov, and Grey literature sources. Eligible studies evaluated tools or protocols used by EMS clinicians caring for suspected or confirmed patients with TBI to help diagnose, prognosticate, or triage these patients. Study selection, data extraction, and risk of bias assessment were performed independently by at least two reviewers. Due to heterogeneity, findings were synthesized narratively. From 961 initial records, 32 studies met inclusion criteria. Studies varied in design, population, and EMS setting. There were 37 unique prehospital TBI assessment tools or strategies. The Glasgow Coma Scale was the most frequently studied tool, though its performance was limited in older adults and patients with mild symptoms. Emerging machine learning-based triage algorithms and point-of-care devices showed promise but lacked large-scale validation. Sensitivity and specificity varied widely, and overtriage was common. Undertriage was more common in geriatric and anticoagulated populations. Current prehospital TBI assessment practices are highly variable, and commonly used tools may fail to detect clinically significant injuries. While novel technologies and decision-support tools show potential, robust prospective studies are needed to confirm their effectiveness in diverse, real-world EMS environments, accounting for population-specific differences.
Introduction
Traumatic brain injury (TBI) is a significant global health concern, affecting millions of individuals each year and contributing to a substantial burden of death and disability. In the United States alone, an estimated 2.8 million people sustain a TBI annually, leading to over 69,000 TBI-related deaths each year and countless cases of long-term cognitive, emotional, and physical impairment. 1 The initial injury, which may be caused by blunt or penetrating trauma, falls, motor vehicle collisions, or assaults, is often compounded by secondary injuries resulting from delayed or inadequate treatment. In elderly patients with a possible TBI, for every minute of delay in obtaining a head computed tomography (CT) to assist with diagnosis, there is an associated 2% increase in in-hospital mortality, a reduced likelihood of anticoagulation reversal within 4 h, and increased length of stay in the emergency department (ED). 2 Accordingly, early recognition and appropriate triage of TBI are essential to improving patient outcomes, particularly in time-sensitive cases such as severe intracranial hemorrhage or rising intracranial pressure.
Prehospital care plays a pivotal role in the trajectory of patients with suspected TBI. Emergency medical services (EMS) clinicians are frequently the first health care professionals to assess these patients, make initial management decisions, and determine the most appropriate destination facility. These early decisions, including the need for airway management and cervical spine precautions, the transport mode, and hospital selection, can influence morbidity, mortality, and long-term recovery. 3 In addition to the critical importance of the prehospital phase, assessing and triaging TBI in the field remains challenging. Clinical signs of brain injury may be subtle, transient, or obscured by intoxication, polytrauma, or language barriers. Furthermore, many tools used to guide in-hospital TBI care, such as neuroimaging, advanced monitoring, or consultation with neurosurgeons and neurointensivists, are unavailable in prehospital environments. 4
To aid in field assessment of TBI, a range of strategies, tools, and scoring systems have been developed for use by the EMS clinician. These include established instruments such as the Glasgow Coma Scale (GCS), Revised Trauma Score, and Field Triage Decision Scheme, as well as more recent innovations such as point-of-care biomarkers, smartphone-based decision aids, and device-based neurological monitoring. 5 Some are designed to identify TBI broadly, while others aim to predict the need for neurosurgical intervention, intensive care unit (ICU) admission, or mortality. However, the diagnostic accuracy of these tools varies greatly. Their generalizability and applicability across a variety of patients and subgroups also remain uncertain. Many have been evaluated only in single-center or retrospective studies, while others have not been validated in diverse populations or EMS systems.
Previous reviews have addressed aspects of prehospital TBI care, including transport decisions, airway management, and triage protocols.5–8 However, a comprehensive synthesis of the tools used specifically for field-based assessment and triage of TBI, encompassing both diagnostic and prognostic utility, is lacking. This narrative review seeks to identify and evaluate available tools, instruments, and strategies used in the prehospital setting to assess or triage patients with suspected TBI. Our objectives are to (1) catalog the full range of field-based TBI assessment and triage tools; (2) assess the diagnostic or prognostic performance of these tools based on available evidence; and (3) identify gaps in the literature related to specific populations, settings, or implementation factors. This review aims to support a more evidence-based approach to prehospital neurotrauma care.
Methods
Study design
This systematic review was designed to evaluate how EMS clinicians assess and triage patients with suspected TBI. The planned approach was registered with the Open Science Framework (OSF) registry for systematic reviews (“Prehospital Assessment and Triage of Traumatic Brain Injury”). Our PICO framework (P: Problem, I: Intervention, C: Comparison, O: Outcome) focused on (P) patients with suspected TBI in a prehospital environment, (I) the prehospital tools or triage protocols used to identify or evaluate patients with TBI, (C) standard assessment or no assessment tools, and (O) outcomes related to diagnostic accuracy, triage decisions, and clinical consequences of suspected TBI. Clinical consequences of interest included the need for neurosurgical intervention, presence of intracranial hemorrhage on CT imaging, in-hospital mortality, ICU admission, and trauma center activation status, as well as overtriage (the proportion of patients with minor injuries initially identified by EMS as having a moderate or severe TBI) and undertriage (the proportion of patients with serious injuries that are missed by initial assessment) rates when reported. 9
We included studies that evaluated prehospital assessment strategies, tools, or triage protocols used by EMS clinicians for the identification and management of suspected or confirmed TBI. Only full-text articles published in English between January 1, 2015, and December 31, 2024, were considered. Included studies were required to report outcomes related to the prehospital diagnosis of TBI, triage destination decisions, or the prognosis of patients with TBI. Additional exclusions included conference abstracts without full data, editorials, commentaries, protocols, narrative reviews, and studies involving nonhuman subjects.
Search strategy
To retrieve the studies meeting the inclusion criteria, C.E. performed comprehensive searches across MEDLINE, Embase, and clinicaltrials.gov. The search covered all databases during the period of January 1, 2015, to December 31, 2024. The keywords included prehospital emergency medicine, TBI, assessment, triage, and their related terms (see OSF for detailed search strategy: https://osf.io/4zpfg). In addition to these databases, Grey literature was reviewed through Grey literature sources such as medRxiv, Research Square, and OSF Preprints. Additional sources were hand searched, including the National Association of EMS Physicians and Prehospital Emergency Care, the International Journal of Paramedicine, the Journal of Emergency Medical Services, Google, and Google Scholar for government reports, white papers published by EMS agencies or academic medical centers, and national or international guideline repositories such as the Guidelines International Network. The search strategy is provided in Supplemental Data. Each abstract was double-screened for inclusion and exclusion criteria by a team of seven authors (C.E., C.F., I.E., M.L-.V., J.B., N.T., and L.D.). Full-text articles were then assessed in duplicate by a team of four authors (C.E., P.I., A.B., and L.D.) to ensure that the inclusion and exclusion criteria were satisfied. For any cases where there was disagreement regarding inclusion/exclusion, a third author (C.E., L.D., or P.I.) also assessed the article for a final decision.
Data extraction and assessment
Study design, population type, sample size, EMS clinician type, prehospital assessment tool used, triage or prognostic outcome, and reference standard were extracted independently by C.E. and verified by L.D. Quantitative data, including sensitivity, specificity, positive predictive value (PPV), negative predictive value (NPV), and area under the curve (AUC), were recorded when available.
Risk of bias
Risk of bias for included studies was evaluated using the QUADAS-2 tool, which is specifically designed to assess the quality of diagnostic accuracy studies. The tool includes four domains: patient selection, index test, reference standard, and flow and timing. Each domain was independently assessed for risk of bias, contributing to an overall risk assessment. For nondiagnostic studies, a simplified assessment framework was applied, focusing on participant selection, outcome ascertainment, and reporting transparency. Risk of bias judgments were incorporated into the narrative synthesis to contextualize findings, but no studies were excluded based on quality alone.
Data synthesis
Search, data extraction, and presentation of results were all performed according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement. The summary of the findings reported includes sensitivity, specificity, PPV and NPV, and AUC for each included study in relation to the prehospital identification, triage, and/or prognostication of TBI.
Results
Study selection
The initial database and registry search yielded 961 records: 762 from Embase, 175 from MEDLINE, and 24 from ClinicalTrials.gov. Following deduplication (101 were removed via Covidence and three manually removed), 857 unique records remained for screening. After title and abstract review, 698 records were excluded, and 159 full-text reports were sought for retrieval. All were successfully obtained. An additional five studies were identified through backward citation tracking or Grey literature searches, yielding 164 full-text articles assessed for eligibility. Of these, 132 were excluded due to wrong focus, setting, design, publication type, or patient population. A total of 32 studies met all eligibility criteria and were included in the final synthesis. A PRISMA flow diagram summarizing this process is provided in Figure 1.
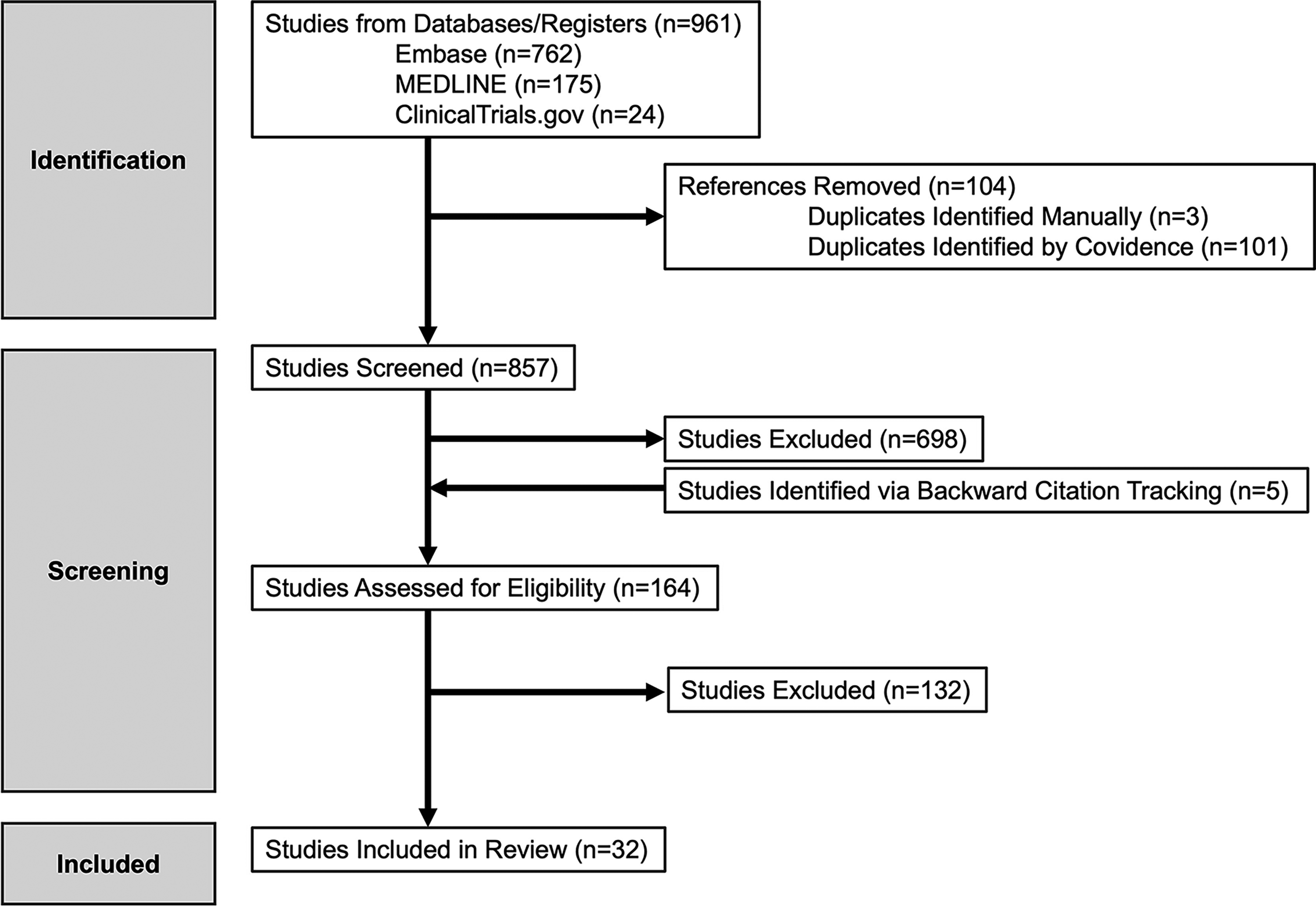
Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) diagram of prehospital assessment and triage of traumatic brain injury.
Study characteristics
The 32 included studies varied in design, population, and geographic location. The majority were retrospective cohort studies (n = 16), followed by prospective cohort studies (n = 13), randomized controlled trials (n = 2), and cross-sectional studies (n = 1) (Table 1).10–41 Sample sizes ranged from 13 to 3,221,995 participants.17,24 Most studies focused on adult civilian populations, though several included pediatric cohorts, elderly patients, or military personnel. Settings included ground EMS, air medical services, and combined systems across North America, Europe, and Asia.
Complete Study List with Overall Description and Bias Assessment
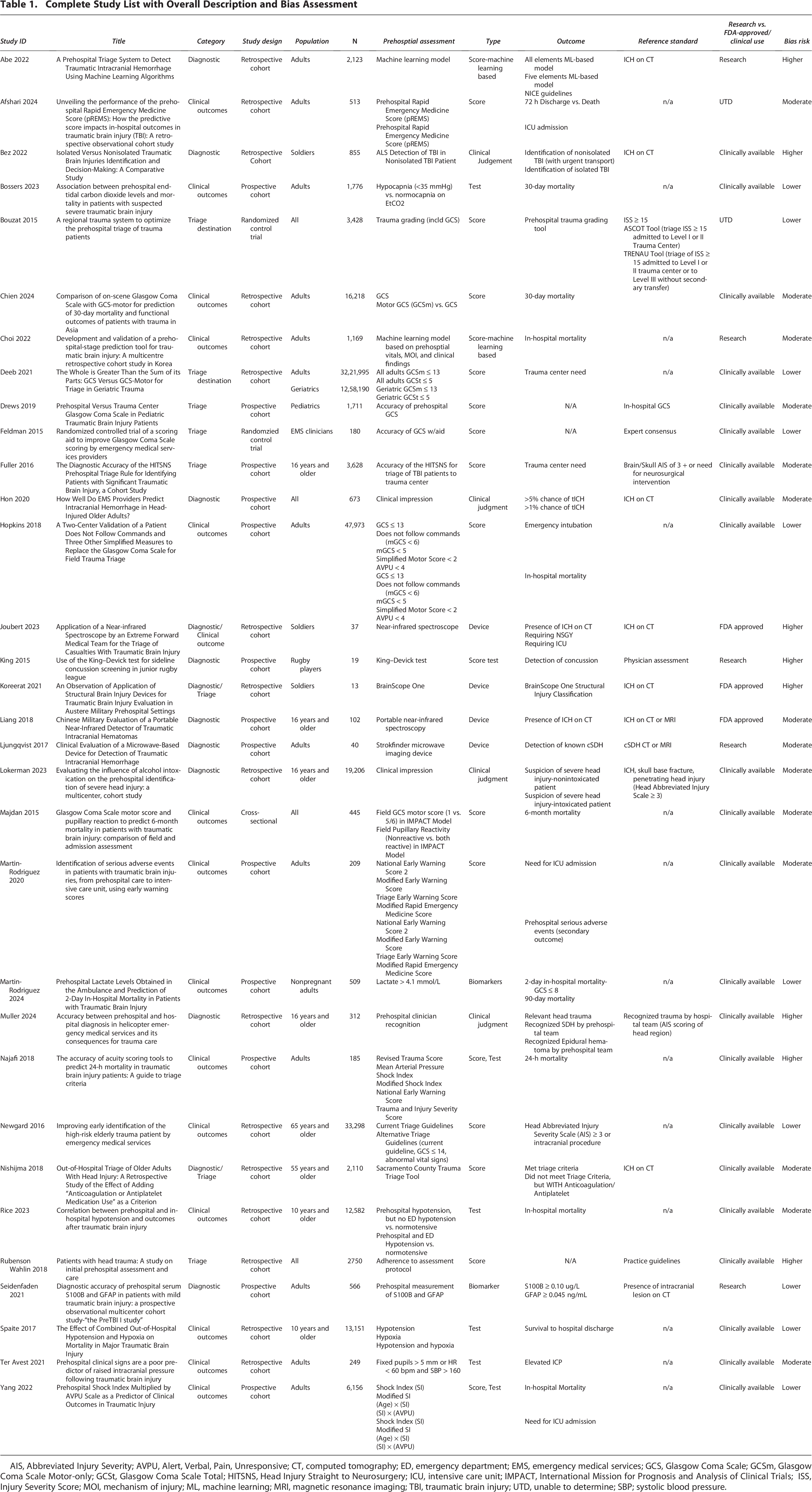
AIS, Abbreviated Injury Severity; AVPU, Alert, Verbal, Pain, Unresponsive; CT, computed tomography; ED, emergency department; EMS, emergency medical services; GCS, Glasgow Coma Scale; GCSm, Glasgow Coma Scale Motor-only; GCSt, Glasgow Coma Scale Total; HITSNS, Head Injury Straight to Neurosurgery; ICU, intensive care unit; IMPACT, International Mission for Prognosis and Analysis of Clinical Trials; ISS, Injury Severity Score; MOI, mechanism of injury; ML, machine learning; MRI, magnetic resonance imaging; TBI, traumatic brain injury; UTD, unable to determine; SBP; systolic blood pressure.
Characterization of prehospital assessments
Across the included studies, 37 distinct prehospital assessment tools or strategies were identified. These tools were grouped into the following characteristics to facilitate comparison of their utility for diagnostic accuracy, prehospital triage, and clinical outcomes: biomarkers, decision-making scores, tests, and devices (Table 1). The most studied assessment tool or score was the GCS. It appeared either as a stand-alone measure, as part of broader triage criteria, or as part of the clinical gestalt or scoring systems used by providers in 15 studies.11,14,15,17–21,27–29,32,33,36,41 Other frequently evaluated methods included mechanism-of-injury-based algorithms, physical examination findings (e.g., pupil reactivity, motor response), and structured field triage guidelines such as those from the Centers for Disease Control and Prevention (CDC) or the Brain Trauma Foundation. 9 Many of these other scoring methods for diagnosis or prediction of complications or mortality use items that are clinically available, such as early warning scores that are used to identify patients at risk for decompensation, serum lactate levels, end-tidal carbon dioxide levels, and shock index.13,29,30,32,40 While lactate levels may not be routinely assessed in the prehospital setting, point-of-care testing could yield this as an option for future use.
Emerging or novel tools included machine learning-based predictive models, point-of-care electroencephalography devices such as BrainScope One, Infrascanner Model 2000, and microwave technology-based devices. Of these devices, both the Infrascanner Model 2000 and BrainScope One are Food and Drug Administration (FDA) approved. Other tools listed in Table 1 are being evaluated, but their components are clinically available. In a few cases, it was not possible to determine if they were clinically available or for research purposes only (listed as “UTD”—unable to determine). However, the degree to which they are being used clinically in prehospital assessments for TBI is unknown. One study incorporated telemedicine-assisted triage.
Several studies evaluated provider gestalt or judgment, especially in air medical settings where prehospital clinicians need to recognize neurological deficit without specialized equipment.12,20,27,31,20,27,31 In these contexts, clinical judgment commonly incorporates routine neurological and physiological assessment elements, most notably GCS, along with other features available at the point of care, such as mechanism of injury and vital signs.12,20,27,31
A summary of assessment tools and the studies evaluating each is presented in Table 1. The absence of reporting and tremendous variation in EMS protocols, unfortunately, hindered the ability to identify what advanced tools have transitioned from experimental use to routine clinical use, despite being FDA approved and/or clinically available.
Diagnostic accuracy
Of the 32 included studies, 14 reported at least one diagnostic accuracy measure such as sensitivity, specificity, PPV, NPV, or AUC relating to prehospital diagnosis of a TBI (Table 2). Sensitivity estimates ranged from 6.2% (using glial fibrillary acidic protein [GFAP] as prehospital biomarker) to 100%, with the highest values associated with tools designed to prioritize sensitivity over specificity.14,23,25,26,37 These diagnostic strategies and tools included BrainScope One, portable near-infrared spectroscopy, Strokfinder microwave imaging device, and the King–Devick Test.23,25,26,37 Specificity similarly had large variability, ranging from 15.4% (using S100B as prehospital biomarker) to 99.3% (using GFAP as prehospital biomarker). 37 Most tools or assessments for diagnosis of TBI generally favored a high sensitivity in exchange for accepting a lower specificity, suggesting detection of all potential TBI cases was of greater concern than questions of resource utilization and undertriage. While undertriage is clearly defined (the inappropriate categorization of patients with severe injuries as only having minor injuries on initial assessment), the precise application of this definition can vary, with some using Injury Severity Score (ISS) scores to help define injury severity and others using triage status, that is, triage to trauma center versus nontrauma center.8,42
Prehospital Diagnostic Assessments/Tools

ALS, advanced life support; AUC, area under the curve; CT, computed tomography; ICH, intracranial hemorrhage; ML, machine learning; MRI, magnetic resonance imaging; NICE, National Institute for Health and Care Excellence; NPV, negative predictive value; PPV, positive predictive value; TBI, traumatic brain injury.
Triage outcomes
In addition to diagnostic metrics, two studies reported on downstream triage outcomes (Table 3).14,17 Bouzant et al. evaluated a trauma grading scale that included a prehospital GCS score to predict patients admitted to Level I and Level II trauma centers based on varying definitions of appropriate triage destinations (e.g., ISS, American College of Surgeons Committee on Trauma [ASCOT] tool, and the The Northern French Alps Trauma System [TRENAU] tool). 14 Sensitivity ranged from 83% to 92% with a specificity of 23–41%. 14 Deeb et al. evaluated the ability of the total GCS and motor-only GCS to predict the need for a trauma center among all adults and a geriatric population, reporting low sensitivity (less than 35%) for both the total and motor GCS in elderly patients. 17
Prehospital Assessments/Tools for Destination Triage

GCS, Glasgow Coma Scale; ISS, Injury Severity Score; NPV, negative predictive value; PPV, positive predictive value.
While overtriage was a common concern, particularly in studies involving mechanism-only triage criteria or conservative decision-making protocols, overtriage was clearly preferred over undertriage. Concerns for undertriage were noted in older adults and patients with subtle neurological findings, especially when GCS was in the normal or mildly abnormal range. Few studies provided outcome stratification by age or anticoagulation status, though several identified these as high-risk subgroups with poorer triage performance.
Clinical end-points
A total of 16 studies evaluated a prehospital assessment in relation to patient-level clinical outcomes such as the need for neurosurgical intervention, ICU admission, or mortality (Table 4).11,13,15,16,21,22,28–30,32–35,38–40 While GCS was often included as part of the prehospital assessment used to predict patient-level clinical outcomes, the presence of prehospital hypotension and hypoxia had the best predictive ability with an AUC of 0.94 (95% confidence interval [CI] 0.93–0.95) with regard to survival to hospital discharge. 38 The sensitivity of prehospital assessments to predict mortality (in-hospital to 6 months) ranged from 17.31% (shock index) to 100% (lactate > 4.1 mmol/L with GCS ≤ 8).30,32 Specificity of these assessments also varied greatly, with the greatest specificity among patients found to have a fixed and dilated pupil and either bradycardia or hypertension (93.2%, 95% CI 88.2–96.6%). 39
Prehospital Assessment/Tool for Prognostication

AVPU, alert, verbal, pain, unresponsive; AUC, area under the curve; ED, emergency department; GCS, Glasgow Coma Scale; ICU, intensive care unit; ICP, intracranial pressure; SI, shock index; SBP, systolic blood pressure; NPV, negative predictive value; PPV, positive predictive value.
Prehospital Assessment/Tool for Prognostication

Risk of bias and methodological quality
Risk of bias was assessed for all included studies using the QUADAS-2 tool, which evaluates four domains: patient selection, index test, reference standard, and flow and timing. Patient selection was at high or unclear risk of bias in 12 studies due to convenience sampling or retrospective design without clearly defined inclusion criteria. Index test bias was often low. However, in eight studies, blinding of EMS providers to outcome or use of prespecified thresholds was not reported. Reference standards were judged at high or unclear risk in 10 studies, particularly those using nonuniform CT criteria or delayed confirmation in hospital records. Flow and timing bias were moderate overall; several studies lacked clear documentation of timing between prehospital assessment and definitive diagnosis. Overall, 11 studies were judged as having a low risk of bias across all QUADAS-2 domains, while the remaining 21 had at least one domain rated as high or unclear risk. Applicability concerns were generally low, although four studies included populations not representative of typical EMS settings (e.g., military-only cohorts or specific sporting settings) (Table 1).
Synthesis summary
Due to heterogeneity in tools, populations, outcome definitions, and reporting formats, a formal meta-analysis was not performed. Instead, a structured narrative synthesis was conducted. A key finding was high variation in both the type and application of prehospital TBI assessment tools. GCS remains the most widely used tool, but its predictive accuracy varies significantly by age and injury severity. Emerging technologies show promise but require further validation in prospective, real-world settings. Sensitivity tends to be prioritized over specificity in most triage systems, likely representing low tolerance for potential missed cases of TBI, with significant implications for resource use and trauma center activation. Limited data exist on pediatric and geriatric subpopulations, as well as on rural or resource-limited EMS settings.
All extracted data and evidence tables are available in the supplementary materials and OSF repository (https://osf.io/4zpfg).
Discussion
This systematic review synthesizes the current evidence on prehospital assessment and triage of suspected TBI by EMS clinicians. These findings highlight substantial variation in the tools and protocols used across EMS systems, as well as in the diagnostic accuracy and clinical utility of those approaches. While some assessment strategies, most notably the GCS, are nearly ubiquitous, their performance in isolation remains limited and inconsistent. This is of particular concern when assessing vulnerable populations such as older adults. At the same time, newer technologies and decision-support tools show potential but require further validation in real-world prehospital settings. Further consideration is to assess our risk tolerance for missed cases of TBI and the costs associated with overtriage to both the patient and the health care system.
Key findings in context
The GCS continues to serve as the foundational tool in most EMS assessments for suspected TBI. In the analysis of statewide EMS protocols for care of patients with suspected TBI, most protocols included assessment of GCS. 8 However, consistent with prior research, this review found that GCS alone lacks sensitivity for detecting clinically significant TBI, particularly in patients with mild or evolving symptoms. 43 Several studies in this review reported undertriage of patients who had normal or near-normal GCS but later required neurosurgical intervention or were found to have intracranial hemorrhage on imaging.20,39,43 This is particularly concerning in populations where baseline neurological function or response to injury may differ, such as the elderly or those on anticoagulants.
Importantly, the diagnostic and triage performance of GCS differed substantially depending on whether it was used as a stand-alone assessment or incorporated into a multimodal evaluation. Studies evaluating GCS in isolation consistently demonstrated limited sensitivity for clinically significant TBI, particularly among older adults and patients with mild or evolving neurological findings.17,43 In contrast, studies incorporating GCS alongside additional clinical variables—such as physiological derangements, focused neurological findings, patient age, or structured decision rules—demonstrated improved discrimination for severe injury, need for trauma center care, or mortality.29,30,38,40 Multivariable approaches combining GCS with hypotension, hypoxia, early warning scores, or anticoagulation status were associated with higher predictive accuracy than GCS alone, supporting the use of GCS as a contributory rather than stand-alone prehospital triage tool.21,33,34
Emerging technologies such as portable electroencephalogram devices (e.g., BrainScope One) and machine learning-based triage algorithms reported higher diagnostic accuracy, with some studies noting AUC values above 0.85. These tools may support improved identification of TBI in cases where traditional clinical signs are ambiguous or other clinical components confound the assessment using more traditional tools. However, their integration into EMS workflows is limited, and evidence of feasibility, cost-effectiveness, and scalability is sparse. In addition, many studies of novel tools had small sample sizes or were conducted in controlled settings, limiting generalizability.
The heterogeneity in prehospital triage protocols is notable. National field triage guidelines, such as those from the CDC, have demonstrated variable diagnostic performance across EMS systems,44 likely reflecting differences in implementation and in how triage accuracy was defined rather than clear evidence that guidelines were superseded.9,44 This variability may contribute to both overtriage and undertriage, with important implications for trauma system performance and patient outcomes. Several studies reviewed highlighted that overtriage was common, especially when protocols favored high sensitivity, yet this often came at the cost of specificity and efficient resource utilization. Conversely, some studies involving pediatric or elderly patients reported undertriage, reflecting challenges in applying adult-focused criteria to other populations.
Implications for practice and policy
These findings have several implications for EMS agencies, trauma systems, and policymakers. The reliance on GCS alone for prehospital triage decisions should be reconsidered, particularly for mild TBI presentations and the elderly. Multimodal assessment strategies, combining GCS with mechanism of injury, neurological findings, patient history, and emerging diagnostic tools, may improve triage accuracy and reduce preventable delays in definitive care. In addition, the integration of structured triage algorithms, decision-support tools, or new diagnostic technologies may standardize care across EMS systems and reduce variability in provider judgment. However, adoption of such tools must be accompanied by targeted training, usability testing, and systems-level evaluation. Furthermore, the potential for newer technologies to augment EMS decision-making is promising but remains contingent on broader validation studies and demonstration of operational feasibility. Finally, this review underscores the potential need for population-specific triage strategies. The physiological and anatomical presentation of TBI varies significantly by age and comorbidities, and current field triage criteria may not adequately account for these differences. Age-adapted triage thresholds, anticoagulation screening, decision aids, and new diagnostic tools for pediatric and geriatric populations warrant further research and implementation attention.
Strengths and limitations
This review was conducted according to a registered protocol using a comprehensive and reproducible search strategy, including dual independent review at all screening stages. The inclusion of a wide range of study designs and assessment tools provides a broad view of current prehospital TBI practices. However, several limitations must be acknowledged. The heterogeneity of study designs, populations, and outcome definitions precluded formal meta-analysis. Many included studies were retrospective, had small sample sizes, or lacked detailed reporting of diagnostic metrics, limiting the strength of inferences. In addition, publication bias may be present, particularly for novel tools with favorable early-phase findings. Finally, this review was limited to studies published in English over the past 10 years, which may have excluded relevant older or non-English research.
Future directions
Future research should prioritize large-scale, prospective validation studies of promising assessment tools across diverse EMS systems. Comparative effectiveness research may be especially valuable in determining how best to integrate novel technologies with standard protocols. Studies focusing on pediatric, geriatric, and anticoagulated populations are particularly needed to improve equity and precision in triage. In addition, the incorporation of EMS perspectives and operational realities, such as time constraints, training burden, and operational costs, will be critical for real-world implementation of any new tool or guideline.
Transparency, Rigor, and Reproducibility Summary
The study was preregistered with the OSF registry for systematic reviews (“Prehospital Assessment and Triage of Traumatic Brain Injury”) (https://osf.io/4zpfg). This includes preregistration of the analytic plan. However, meta-analysis was ultimately deferred due to the significant heterogeneity of the data. A total of 857 unique records were identified for abstract screening by at least two team members. At least two team members also double-screened 164 full-text articles. For any cases where there was disagreement regarding inclusion/exclusion, a third author also assessed the article for a final decision. Data were acquired between May 27, 2025, and August 1, 2025. Study design, population type, sample size, EMS clinician type, prehospital assessment tool used, triage or prognostic outcome, and reference standard were extracted independently by a single author and then independently verified by a second author. All extracted data and evidence tables are publicly available and available through the OSF repository (https://osf.io/4zpfg). This article will be published under a subscription service model for this journal.
Authors’ Contributions
C.E.: Conceptualization, methodology, formal analysis, investigation, data curation, writing—original draft, writing—review and editing, and visualization; P.I.: Investigation, data curation, writing—original draft, and writing—review and editing; C.F.: Investigation, data curation, and writing—review and editing; I.E.: Investigation, data curation, and writing—review and editing; M.L-V.: Investigation, data curation, and writing—review and editing; J.B.: Investigation, data curation, and writing—review and editing; N.T.: Investigation, data curation, and writing—review and editing; A.B.: Investigation, data curation, and writing—review and editing; L.D.: Conceptualization, methodology, formal analysis, investigation, data curation, writing—original draft, and writing—review and editing.
Footnotes
Author Disclosure Statement
No competing financial interests exist.
Funding Information
Grant money for commercial research: L.D. reports grant money to Yale University School of Medicine to conduct research conceived and sponsored by TETMedical, Inc.
