Abstract
Biomarkers are needed to characterize mechanistic endophenotypes after neurotrauma and inform prognosis and therapy. Vascular endothelial growth factor-A (VEGF-A) is associated with inflammation and vascular permeability, which are implicated in secondary brain injury. We describe changes in plasma VEGF-A levels at injury (D1) and 2 weeks (W2) postinjury, and their associations with traumatic brain injury (TBI) severity and 6-month (M6) Glasgow Outcome Scale-Extended (GOSE) scores in a subset of Transforming Research and Clinical Knowledge in TBI (TRACK-TBI) participants with TBI (
Introduction
Traumatic brain injury (TBI) is a heterogeneous disease that involves variable injury to neuronal and glial1–4 cell populations as well as cerebral micro-5–7 and macro-8,9 vasculature, which is compounded by dysregulation of downstream inflammatory10,11 and coagulation cascades. 12 Key signaling molecules in these molecular pathways can be readily detected in the bloodstream and may serve as diagnostic and/or prognostic biomarkers in TBI.3,13,14 A better understanding of these potential mediators of secondary injury is important for the development of targeted therapeutic approaches.
One such molecule is vascular endothelial growth factor (VEGF-A), a 35–45 kD proangiogenic glycoprotein and member of the VEGF family that includes VEGF-A through VEGF-E. VEGF-A is uniquely responsive to both mechanical endothelial injury as well as ischemia, two major mechanisms of injury in TBI, and it has been widely studied as a therapeutic target.15–18 When endothelial cells are injured, myriad factors are released to promote repair, including VEGF-A. VEGF-A has four isoforms produced via alternative splicing, all of which bind to receptor tyrosine kinases, VEGFR1, and VEGFR2. These receptors are expressed on endothelial cells, monocytes, and macrophages; VEGF-A binding promotes angiogenesis and vascular permeability19,20 as well as prevents apoptosis.21–23 Therefore, VEGF-A may have both adaptive and/or maladaptive effects after acute brain injury. Repair predominantly occurs via VEGFR-2 activation, resulting in endothelial cell proliferation24,25 and migration to foci of trauma, as well as activation of prosurvival endothelial signaling cascades. 21 Its effect on promoting vascular permeability, however, may contribute to the pathogenesis of inflammation and cerebral edema in acute brain injury.20,26,27 Mechanisms by which this is thought to occur are via inflammation-mediated dilation of arterioles, capillaries, and venules, as well as upregulation of intravascular adhesion molecules, 28 thus contributing to blood–brain barrier (BBB) disruption.
Since traumatic vascular injury commonly accompanies TBI, it is not surprising that VEGF-A levels are elevated after trauma. Broadly, vascular trauma results in the acute release of VEGF in plasma.
29
Prior clinical studies in smaller neurotrauma cohorts (20–40 neurotrauma cases with 40–70 participants in total), including those with GCS predominantly 13–15
30
as well as GCS 3–8,31,32 have demonstrated that following TBI, plasma levels of VEGF-A rise within the first several days postinjury and peak around day 14, followed by a decline by day 21. Although in a cohort with a majority GCS 13–15, there was no significant difference in VEGF-A level between survivors and nonsurvivors, VEGF-A levels at 14 days were significantly higher in subjects that deteriorated clinically compared with those who improved.
30
Our study extends these findings by analyzing a larger sample (
Methods
Subjects and study design
This study included a subset of the TRACK-TBI cohort, a prospective, multicenter observational study that enrolled patients with TBI presenting to one of 18 participating U.S. level I trauma centers between February 26, 2014, and August 8, 2018.33,34 Over its enrollment period, TRACK-TBI enrolled 2,997 trauma participants. 35 Eligible TBI participants included adults age ≥17 years who presented within 24 hours of head trauma requiring computed tomography (CT) imaging per clinical guidelines 36 including altered mental status (i.e., unconsciousness, peritraumatic amnesia, or other signs of altered consciousness). Consent was obtained from the participant or legally authorized representative. The study was approved by the institutional review board of each TRACK-TBI site.
Orthopedic trauma controls (OTCs) were identified from patients presenting to the same level I trauma centers with trauma isolated to their limbs, pelvis, and/or ribs with an associated Abbreviated Injury Score <4; they were ineligible if they sustained alteration or loss of consciousness, post-traumatic amnesia, or other clinical evidence of head strike.
HCs were recruited through public outreach at participating TRACK-TBI institutions or by an existing relationship with an enrolled TRACK-TBI participant. HCs were required to provide informed consent and could not have a history of TBI or polytrauma within the 12 months prior to enrollment.
Exclusion criteria for all participants included being pregnant, in police custody, having non-survivable trauma, non-English- and non-Spanish-speaking, and history of pre-existing debilitating neurological or mental disorders.
The samples for the biomarker sub-study were selected based on completeness of 6-month Glasgow Outcome Scale-Extended (GOSE) data, blood sample availability, and selection of equivalent number of GCS 3–12 cases as GCS 13–15 due to cost considerations. Eligible subjects were randomly selected, yielding 200 TBI GCS 13–15 and 194 TBI GCS 3–12 cases. In addition, 100 orthopedic controls and 69 HCs with available blood samples were included. For the current analysis, we further restricted the sample to participants with complete VEGF-A measurements at both day 1 (D1) and week 2 (W2), resulting in final sample sizes of 148 TBI GCS 3–12, 169 TBI GCS 13–15, 82 OC, and 69 HC.
Clinical data collection
Participant demographic data, injury features, and imaging were collected through medical record review and per patient reporting in accordance with the National Institute of Neurological Disorders and Stroke (NINDS) TBI Common Data Elements (TBI-CDE) guidelines.37,38
In-person or telephone outcome assessments for participant-reported disability attributable to the TBI were made by GOSE at 6 months (M6) post-injury. Outcomes were dichotomized as unfavorable (GOSE 1–4) versus favorable (GOSE 5–8) outcome, at least moderate functional disability (GOSE 1–6 vs. GOSE 7–8), and incomplete (GOSE 1–7) versus complete (GOSE = 8) recovery at M6.
Sample collection and biomarker analysis
Blood samples were collected within 24 hours (D1) of injury, and at 2 weeks (W2) and M6 post-injury. Samples were de-identified, dated, and time-stamped, and centrifuged to derive plasma and serum aliquots, which were stored at −80°C for future analysis per TBI-CDE Biospecimens and Biomarkers Working Group consensus recommendations. 37 Specimens were shipped under temperature-controlled conditions to the TRACK-TBI Biospecimens Repository at the University of Pittsburgh Medical Center (Pittsburgh, PA) for storage.
Meso scale discovery technology
Meso Scale Discovery (MSD) MULTI-ARRAY® technology uses sensitive electrochemiluminescence (ECL) detection, which is based on the use of SULFO-TAG™ labels that emit light when oxidized at an electrode. MSD’s assays are carried out directly on the surface of single-use electrodes using the electrode surface as both a solid phase support for arrays of binding reagents and as a source of electrical energy for inducing ECL. MSD® assays display wide dynamic ranges of three to five orders of magnitude, use significantly less sample material per measurement than most other detection systems, use simple protocols, and can support large-scale testing due to the high-throughput capability of MSD instruments. MSD’s ultrasensitive S-PLEX® assay format achieves detection limits in immunoassay formats that can compete with nucleic acid-based detection techniques such as PCR.39–41 MSD assays are a Research Use Only platform.
Samples were analyzed at Meso Scale Diagnostics, LLC. (Rockville, MD, USA) as described previously42,43 and according to the product inserts. Ultrasensitive MSD S-PLEX Neurology Panel 1 (Cat. K15639S) assays were performed manually and read on a MESO SECTOR® S 600MM reader, while the MSD V-PLEX® Angiogenesis Panel 1 (Cat. K15190D) assays were performed on the MSD MESO Parsec™ R 5000MM automated system. Plasma samples were assayed in duplicate at a 1:2 dilution for the Neurology Panel 1 and a 1:4 dilution for the Angiogenesis Panel 1. An 8-point calibration curve and three quality control (QC) samples spanning the assay range were included in duplicate on each plate. The standardized mean of the QC samples all fell within the prespecified acceptance limit of 80–120% of the assigned concentrations. The samples were codified and randomized for blinded testing across 37 total plates per panel. The Lower Limit of Quantification and Upper Limit of Quantification were 0.738 and 7860 pg/mL for VEGF-A, 0.304 and 2840 pg/mL for GFAP, and 2.765 and 10,540 pg/mL for NfL.

CT imaging evaluation and analysis
TBI participant CT imaging was deidentified and uploaded to a central database at the Laboratory of NeuroImaging (LONI; University of Southern California, Los Angeles, CA). Board-certified neuroradiologists blinded to patient characteristics independently evaluated the scans for trauma-associated common data elements (CDE) in accordance with TBI-CDE Neuroimaging Working Group consensus recommendations. 36 Each analysis was then uploaded to the respective participant’s record in the TRACK-TBI clinical database. Initial cranial CT was considered positive (CT+) if it included the following trauma-associated CDE: epidural hematoma, acute subdural hematoma (aSDH), traumatic subarachnoid hemorrhage (tSAH), contusion (CON), petechial hemorrhage concerning for diffuse axonal injury, or intraventricular hemorrhage (IVH).
Statistical analysis
Demographic and clinical characteristics were summarized for the study cohort. To delineate the association between VEGF-A with injury severity and outcomes, VEGF-A levels at D1 and W2 were compared by TBI diagnosis, severity, CT status, CT lesion type, and 6-month GOSE status using the Wilcoxon Rank Sum test and adjusted for multiple testing using the Benjamini–Hochberg false discovery rate (FDR) procedure as follows: At each timepoint (D1, W2, and M6), VEGF-A levels were first compared between the overall TBI group and control groups (HC and OTC) using Wilcoxon rank-sum tests as
Logistic regression models were used to assess whether D1 or W2 VEGF-A level was associated with unfavorable outcome (GOSE 1–4 vs. 5–8), at least moderate functional disability (GOSE 1–6 vs. GOSE 7–8), and incomplete recovery (GOSE 1–7 vs. GOSE = 8) at 6-months post-injury. The primary predictor was continuous VEGF-A concentration, which was log-transformed to reduce right skew. VEGF-A levels at D1 and W2 were analyzed in separate models. Covariates in the multivariable logistic regression models consisted of age, sex (female vs. male), psychiatric history (yes vs. no), presence of major extracranial injury (yes vs. no), CT positivity (yes vs. no), and injury severity (GCS 3–12 vs. 13–15). Adjusted odds ratios (aORs) with 95% confidence intervals were reported, where aOR denotes the odds ratio for VEGF-A after adjustment for all listed covariates. To evaluate the incremental predictive value of VEGF-A, models with and without VEGF-A were compared using Nagelkerke’s
Statistical software R (version 4.1.2) was used for the analysis (http://www.r-project.org).
Results
This analysis included a subset of the TRACK-TBI study (Fig. 1): prospectively enrolled participants with TBI (

CONSORT flow diagram of participants.
Demographic and Clinical Characteristics Table
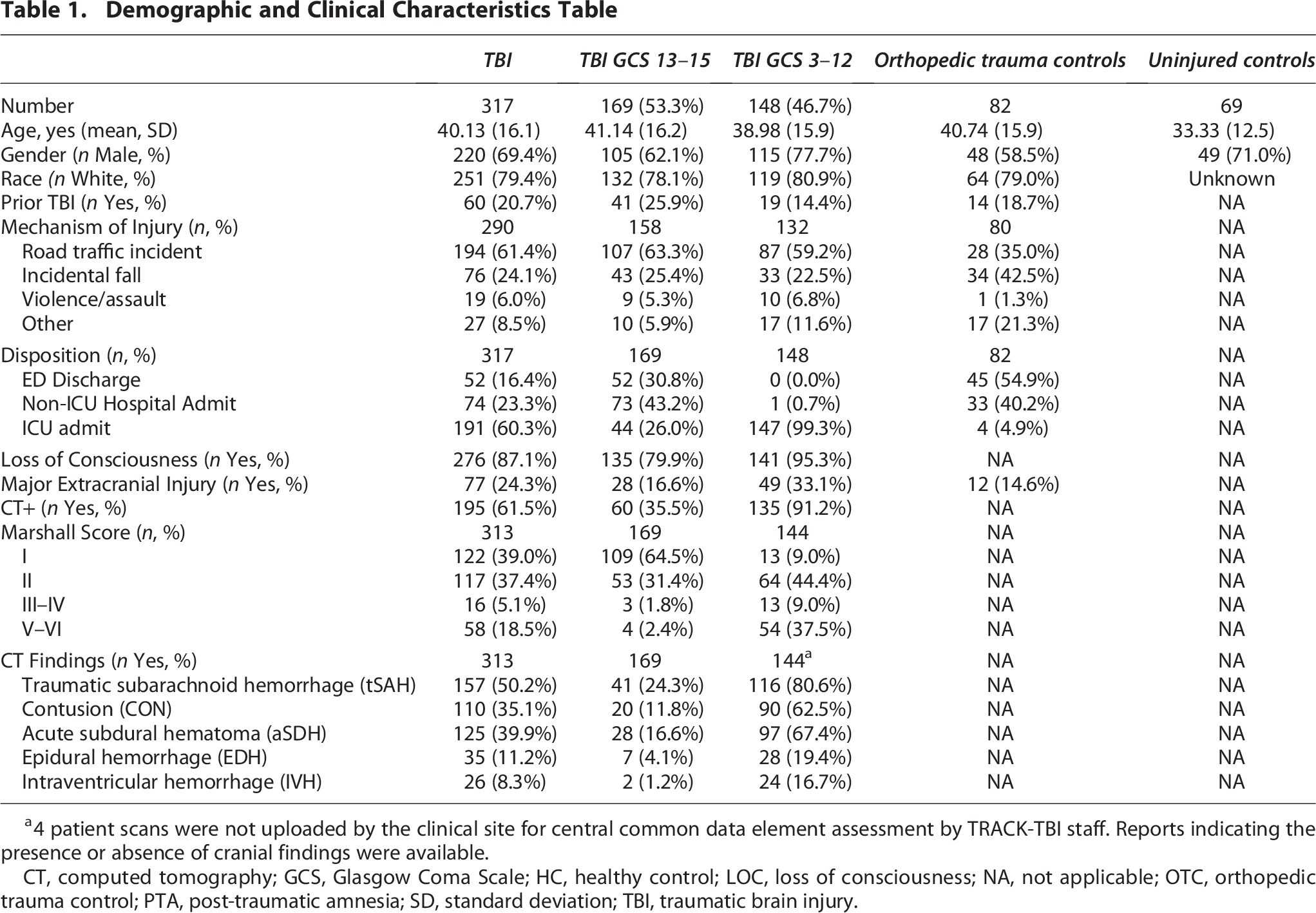
4 patient scans were not uploaded by the clinical site for central common data element assessment by TRACK-TBI staff. Reports indicating the presence or absence of cranial findings were available.
CT, computed tomography; GCS, Glasgow Coma Scale; HC, healthy control; LOC, loss of consciousness; NA, not applicable; OTC, orthopedic trauma control; PTA, post-traumatic amnesia; SD, standard deviation; TBI, traumatic brain injury.
VEGF-A level increases following TBI
D1 VEGF-A levels (median [25th-75th percentiles]) were higher in TBI participants (
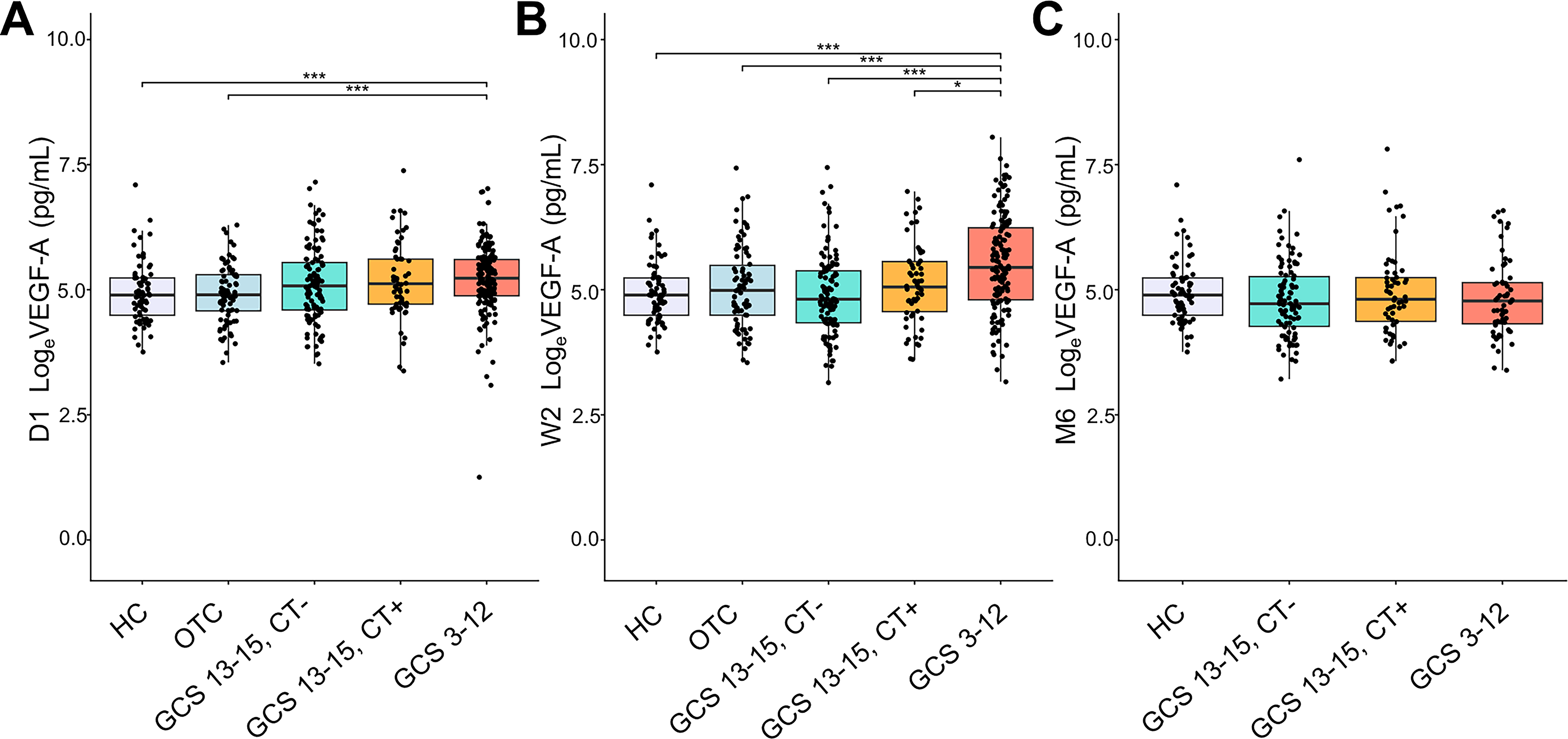
Comparison of D1, W2, and M6 levels between groups. VEGF-A levels (pg/mL) at D1 for HC compared with
At W2, VEGF-A returned to levels comparable to those of HC (
Levels of VEGF-A are poorly correlated with NfL and GFAP
Assessment of correlation (Spearman’s correlation coefficient) of VEGF-A with NfL and GFAP revealed weak correlation at both D1 (rho = 0.11 with NfL and GFAP among GCS 13–15; rho = −0.01 with NfL and 0.05 with GFAP among GCS 3–12) and W2 (rho = 0.18 with NfL and 0.13 with GFAP among GCS 13–15; rho = 0.23 with NfL and 0.25 with GFAP among GCS 3–12; Supplementary Fig. S2).
Higher levels of VEGF-A are associated with trauma-related abnormalities on cranial CT scans
Table 1 describes the cranial CT findings among the TBI participants. The most common acute intracranial abnormality overall was tSAH (157 of 313 [50.16%]), followed by aSDH (125 [39.94%]), CON (110 [35.14%]), and IVH (26 [8.31%]; Supplementary Fig. S3). Isolated tSAH was found in 24 of 195 positive CT scans (12.3%). In participants with TBI, VEGF-A levels (median ([25th–75th percentiles]) were higher for CT+ cases as compared with CT− cases at D1 (CT+: 183.4 [128.9–277.4] pg/mL; CT−: 160.2 [95.8–242.5] pg/mL,
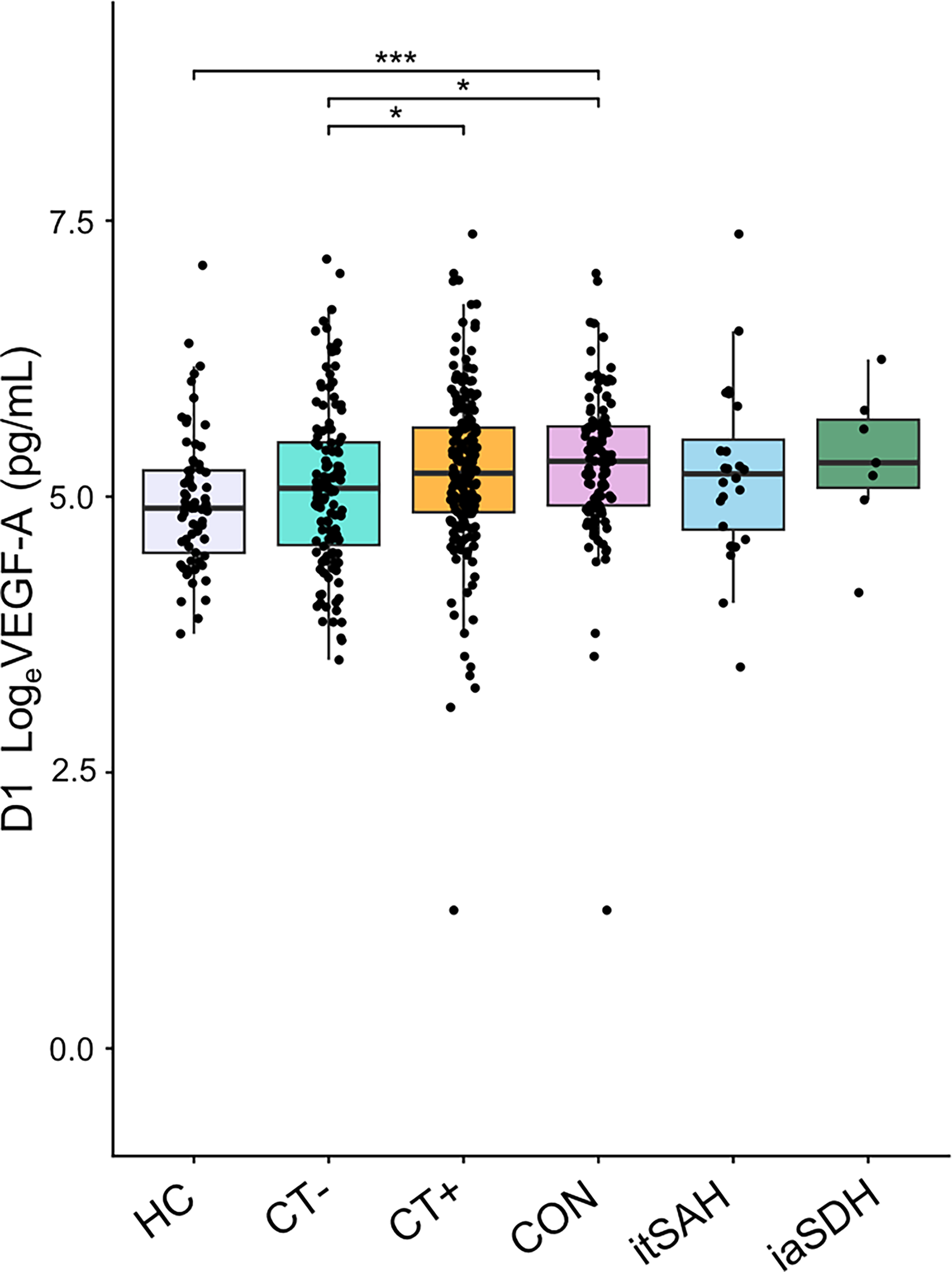
Comparison of VEGF-A level by CT+/CT-status and trauma CDE. D1 VEGF-levels (pg/mL) in TBI participants with and without intracranial findings on CT as well as further comparisons between the presence/absence of trauma CDE. *
W2 VEGF-A level is associated with M6 outcome
In TBI participants, D1 VEGF-A levels (median [25th–75th percentiles]) were not significantly different between those with unfavorable (GOSE 1–4:

VEGF-A and 6-month GOSE outcome after TBI:
Logistic regression model adjusting for age, sex (Female vs. Male), psychiatric history (Yes vs. No), major extracranial injury (Yes vs. No), CT (+ vs. −), GCS (3–12 vs. 13–15) also showed a significant association of W2 VEGF-A levels with unfavorable outcome (GOSE 1–4) at M6 (aOR = 1.44 per log unit increase, 95% CI: 1.02–2.05,

Unfavorable, GOSE 1–4 vs Favorable Outcome, GOSE 5–8, at Least Moderate Functional Disability, GOSE 1–6, and Incomplete Recovery, GOSE 1–7 versus Complete Recovery, GOSE = 8.
GCS, Glasgow coma scale; GOSE, Glasgow outcome scale extended; MEI, major extracranial injury; OR, odds ratio; 95% CI, 95% confidence interval.
Discussion
Plasma VEGF-A levels are elevated within 24 hours of TBI as compared with both HCs and participants with orthopedic injuries. Moreover, D1 VEGF-A rises to significantly higher levels in those with intracranial lesions on CT, particularly in participants with contusions. Of note, early VEGF-A elevation for the GCS 13–15 group appears dependent upon CT status, with D1 levels for the CT + participants mirroring that of the GCS 3–12 group. VEGF-A levels remain elevated up to 14-days post-injury only in those with more severe injuries.
Based on the known biology of VEGF-A, elevations detected in plasma may reflect a secondary vascular injury through injury of endothelial cells resulting in VEGF-A release and its subsequent role in angiogenesis and inflammation.44,45 The poor correlation of VEGF-A with axonal and astrocytic injury markers (NfL and GFAP) suggests that VEGF-A levels reflect a mechanistic endophenotype independent from axonal and glial injury. VEGF-A levels were most elevated in participants with CT scans that demonstrated contusions, but not to the same degree in isolated tSAH or aSDH. Given the role of VEGF-A in vascular permeability, this may reflect the degree of associated edema as well as burden of inflammation with microthrombi, which has been found most dramatically associated with contusion. 46 Further work incorporating magnetic resonance imaging and tissue specimens to better assess this association is needed to fully characterize the potential role of VEGF-A as a biomarker of a distinct endophenotype, but the work presented here suggests that it is an attractive candidate.
VEGF-A levels at W2 were significantly elevated in patients with clinical and neuroimaging evidence of more severe injury as well as poor outcomes, further supporting VEGF-A as a marker of a secondary injury mechanism that persists over time. This supports the potential role of VEGF-A measured over the first two weeks post-injury as a biomarker of traumatic microvascular injury. Given the dual nature of inflammation, embodying both adaptive and maladaptive roles, 47 perhaps the early rise in VEGF-A represents a beneficial, adaptive response while its delayed/prolonged elevation reflects ongoing, smoldering pathological inflammation.
It is important to note that VEGF-A is not elevated to the same degree following orthopedic injuries. Since the brain is a highly vascular organ, it may not be surprising to find that TBI results in relatively greater VEGF-A release compared with orthopedic injury, depending upon its location (i.e., tissue affected) and severity.
Limitations
In addition to the methodological strength of leveraging one of the largest prospective TBI cohorts, some limitations should be recognized. The TRACK-TBI cohort as a whole is not meant to be a reflection of the typical TBI population, but is designed to identify biomarkers and endophenotypes. Therefore, caution in generalizing these findings should be taken. Moreover, due to cost considerations, this study incorporates a subset of the full TRACK-TBI population based on completeness of 6-month GOSE data, blood sample availability, and selection of equivalent number of GCS 3–12 and GCS 13–15 cases. In particular, the loss of participants without M6 outcome data and the selection of equivalent GCS group cases may introduce selection bias. In addition, VEGF-A is one of many potential traumatic vascular biomarkers, and therefore, further work focused on its incorporation into more comprehensive biomarker panels is warranted.
The role of VEGF-A after TBI is likely to be complex and may vary from patient to patient and by time from injury. VEGF-A is a candidate neuroprotective agent, 15 and in animal models including controlled cortical impact in mice 16 or fluid percussion injury in rats 17 intraventricular administration of recombinant VEGF over 1–2 weeks after injury promoted neurogenesis, and in the mouse model 16 reduced lesion size and improved neurological outcomes. Recombinant VEGF-A has also been investigated as a neurorestorative therapy after stroke, and while pre-clinical studies show substantial promise, 18 early-stage clinical trials so far show that while VEGF-A administration is safe, evidence of clinical efficacy is lacking. 48 Alternatively, in the early stages after TBI VEGF-A may promote BBB disruption and increase cerebral edema and inflammation.44,49 A sophisticated understanding of how VEGF levels change over time after TBI and how these relate to imaging and physiological biomarkers will be required for therapies targeting the VEGF-A pathways to be successfully implemented.
Conclusions
In conclusion, our study confirms and extends upon prior work by demonstrating that plasma VEGF-A levels are elevated after TBI. There are suggestions that VEGF-A is particularly elevated in those with parenchymal contusions. VEGF-A levels at 2 weeks are associated with poor outcomes. VEGF-A levels correlate weakly with widely-studied protein biomarkers of neurodegeneration (GFAP, NfL) and potentially show promise as a biomarker of microvascular pathophysiology.
Transparency, Rigor, and Reproducibility Statement
The TRACK-TBI study was preregistered at clinicaltrials.gov (clinicaltrials.gov NCT02119182). The analysis plan, while not formally pre-registered, was conducted through a formal collaboration proposal and approved by the TRACK-TBI Executive Committee prior to analysis. This study was developed after the completion of data collection for TRACK-TBI, and therefore, power analyses were not performed, given that the sample size was fixed per the inclusion and exclusion criteria of the current analysis. Participants were selected for the biomarker sub-study based on completeness of M6 GOSE data, blood sample availability, and selection of equivalent number of GCS 3–12 cases as GCS 13–15 within the available participants in the TRACK-TBI database. The CONSORT diagram is presented in Supplementary Figure S1. Participants were blinded to the results of biomarker and clinical assessments throughout the study and following study completion. Data were collected between 2014 and 2018. The key inclusion criteria and outcome measures are standards in the field and were assessed by trained study staff with oversight by clinicians and investigators with extensive experience in the assessment of persons with TBI. Statistical analysis was performed by Sonia Jain, PhD, and Xiaoying Sun, MS, of the TRACK-TBI Biostatistics and Analytics Core (BAC). No replication or external validation studies have been performed at this time to our knowledge. Data for this study are available upon request to TRACK-TBI at https://tracktbi.ucsf.edu/ and may also be accessed via FITBIR (https://fitbir.nih.gov/).
The TRACK-TBI Investigators
Ann-Christine Duhaime, MD (MassGeneral Hospital for Children); Shawn Eagle, PhD (University of Pittsburgh); Brandon Foreman, MD (University of Cincinnati); Etienne Gaudette, PhD (University of Toronto); Shankar Gopinath, MD (Baylor College of Medicine); Ramesh Grandhi, MD MS (University of Utah); Ruchira Jha, MD, MSc (Barrow Neurological Institute); Frederick K. Korley, MD, PhD (University of Michigan); Vijay Krishnamoorthy, MD (Duke University); Christine Mac Donald, PhD (University Washington); Michael McCrea, PhD (Medical College of Wisconsin); Randall Merchant, PhD (Virginia Commonwealth University); Laura B. Ngwenya, MD, PhD (University of Cincinnati); Eric Rosenthal, MD (Massachusetts General Hospital); Andrea Schneider, MD PhD (University of Pennsylvania); David Schnyer, PhD (UT Austin); Sabrina R. Taylor, PhD (University of California, San Francisco); John K. Yue, MD (University of California, San Francisco); Ross Zafonte, DO (Harvard Medical School)
Footnotes
Authors’ Contributions
R.T.: Conceptualization, formal analysis, writing—original draft preparation, and review and editing. C.D.: Resources. N.P.: Resources. T.G.: Resources. G.B.S.: Resources. J.N.W.: Resources. C.L.: Resources. S.J.: Data curation, formal analysis, and visualization. X.S.: Data curation, formal analysis, and visualization. J.T.G.: Conceptualization, funding acquisition, supervision, resources, and writing—review and editing. M.A.M.: Conceptualization, funding acquisition, supervision, resources, and writing—review and editing. D.O.O.: Conceptualization, funding acquisition, supervision, resources, and writing—review and editing. C.S.R.: Conceptualization, funding acquisition, supervision, resources, and writing— review and editing. N.T.: Conceptualization, funding acquisition, supervision, resources, and writing—review and editing. P.M.: Conceptualization, funding acquisition, supervision, resources, and writing—review and editing. K.K.W.: Conceptualization, funding acquisition, supervision, resources, writing—review and editing. A.M.P.: Conceptualization, funding acquisition, supervision, resources, and writing—review and editing. A.L.C.S.: Conceptualization, funding acquisition, supervision, resources, writing—review and editing. D.K.S.: Conceptualization, funding acquisition, supervision, resources, writing—review and editing. G.T.M.: Conceptualization, funding acquisition, supervision, resources, and writing—review and editing. R.D.A.: Conceptualization, funding acquisition, supervision, resources, formal analysis, writing — review and editing and writing — review and editing.
Author Disclosure Statement
R.T. has no conflicts to declare. K.K.W is shareholder of Gryphon Bio, Inc. and Owl Therapeutics, LLC.
Funding Information
The TRACK-TBI study (PI: Manley) was funded by the U.S. National Institute of Neurological Disorders and Stroke (NINDS) grant # U01 NS1365885, One Mind, and Neurotrauma Sciences LLC. The NINDS played an advisory role in the design and conduct of the TRACK-TBI study but did not play a direct role in this secondary analysis project. The article’s contents are solely the responsibility of the authors and do not necessarily represent the official views of the NINDS or other study sponsors. This study was also supported by the NINDS grant # UE5/R25 NS065745 to R.T., and the NINDS grant # U01 NS114140 to RDA.
