Abstract
Mild traumatic brain injury (mTBI) is common among adolescents because of their participation in contact sports, and mTBI is more likely to lead to depression-related behaviors in girls than boys. Various neuropeptides, often within the limbic system, have been implicated in the regulation of depression-related behaviors. To identify potential neuropeptide involvement in behavioral effects of mTBI, this study used adolescent-age male and female rats to compare the effects of single and repetitive mTBI on depression-like behavior and limbic neuropeptide expression. Female but not male rats displayed increased immobility in the forced swim test compared with sham-injured rats at 5 weeks (chronic), but not 2 weeks (acute), following closed-head injury, and this effect was estrous cycle-dependent following single but not repetitive injury. In the nucleus accumbens (NAc), a major limbic nucleus, mRNA expression of corticotropin-releasing factor and dynorphin was decreased after single injury only in female rats, particularly during the estrus phase, while expression of enkephalin (ENK) was decreased after repetitive injury. In the paraventricular nucleus of the thalamus (PVT), another limbic nucleus, mRNA expression of pituitary adenylate cyclase-activating polypeptide (PACAP) was increased in repetitively injured female but not male rats at 2 and 5 weeks but was unchanged in single-injured female rats compared with sham-injured rats. These data suggest that depression-like behavior emerges in the chronic phase following adolescent mTBI only in females. Although not statistically analyzed in females receiving repetitive mTBIs, this depression-like behavior only appeared in the estrus phase following a single mTBI. Moreover, reduced ENK in the NAc and elevated PACAP in the PVT may contribute to depression-like behavior in females following repetitive mTBI.
Introduction
One to two million traumatic brain injuries (TBIs) diagnosed in the emergency department annually are classified as mild.1,2 Mild TBI (mTBI) can result in problems with memory, concentration, anxiety, and depression, which can persist in a subset of patients. 3 Athletes who play contact sports are particularly vulnerable to sustaining repeated mTBI, exacerbating the associated injury deficits.4–6 Female athletes typically exhibit different symptoms and take longer to recover, with the menstrual cycle stage contributing to this phenomenon.5,7–9 The vulnerability of female athletes is further magnified in adolescents, as this population may take longer to recover than other age groups and may not exhibit symptoms until years after injury.10–12 People with a history of mTBI are more than three times as likely to experience depression compared with those without mTBI, which begins 6 or more months following injury, with a higher prevalence among adolescents.13,14
Few studies have investigated depression-like behavior after mTBI in both male and female adolescent rodents.15–18 At 2 weeks after a single or repetitive lateral mTBI, adolescent female rats displayed differences in time spent immobile in the forced swim test (FST), 17 while 4 days after repetitive injury, adolescent mice showed no change in behavior in the FST. 18 In contrast, we have demonstrated that 6 weeks following either a single or repeated mTBI during adolescence, injured female but not male Sprague-Dawley rats display greater immobility in the FST, an effect that is dependent on estrous phase only after the single injury.15,16 The specific mechanistic basis for these injury-induced sex differences remains to be understood.15–18
Neuropeptides expressed in the limbic system can regulate both cognitive and emotional behaviors,19–21 and several have been evaluated in the acute period following TBI.22–26 Within the nucleus accumbens (NAc), dynorphin (DYN) and enkephalin (ENK) are major neuropeptides found in medium spiny neurons 27 while corticotropin-releasing factor (CRF) is found more sparsely, 28 and these neuropeptides regulate negative affective behaviors, often in response to stress.29–34 Immunoreactivity for DYN is increased in the brain immediately following repetitive blast mTBI in adult mice, and pretreatment with a DYN receptor antagonist reduces anxiety-like behaviors. 23 However, there are sex-related differences in sensitivity to a DYN receptor agonist in the NAc in ethanol drinking behavior. 35 Immunoreactivity for ENK is increased in the pituitary of adult male rats following a severe TBI. 24 Expression of CRF and receptor mRNA is increased in multiple limbic regions following TBI in rodents, 26 along with an increase in anxiety-like behavior, 22 and a CRF1 receptor antagonist can reduce anxiety-like behaviors. 36 In addition, female rodents have shown differences compared with males and across estrous phases in behavior and brain circuit activity to central infusion of CRF.37,38 Within the paraventricular nucleus of the thalamus (PVT), which sends dense projections to the NAc, 39 pituitary adenylate cyclase-activating polypeptide (PACAP) is highly expressed, especially in female rodents,40,41 and has been implicated in depression-like behaviors in rodents.42,43 In addition, PACAP expression is increased in the perifocal cortical region and dentate gyrus within a few to 72 h following moderate TBI. 25 Thus, it is apparent that TBI can cause acute upregulation of these stress-related neuropeptides,23–26 and literature outside the TBI field has indicated that these neuropeptides within the NAc and PVT regulate many negative affective behaviors.29–34,44 Chronic upregulation of these neuropeptides following TBI remains largely understudied but may underlie depression-like behavior changes.
The objective of the present study was to further characterize the effect of single and repetitive mTBI sustained in adolescence on sex differences in depression-like behavior in adulthood. Some evidence suggests that mTBI plays a role in depression-like behavior in female rats, but these changes may be estrous-cycle dependent following a single mTBI.15–17 Importantly, this study also sought to investigate the associated expression of neuropeptides within the NAc and PVT, which may mediate affective behaviors differently in females and across the estrous cycle.35,37,38,44
Methods
Subjects
Fifty-nine male and 107 female adolescent Long-Evans rats (35 days old, Charles River Laboratories, Wilmington, MA) were used in this study and housed in a 12-h light/dark cycle with ad libitum access to food and water. Animal numbers are listed in Table 1. All female rats were monitored for phase of estrous cycle using vaginal cytology during the week prior to behavioral testing, 2 to 3 h before behavioral testing, and on the day of sacrifice as described.15,45 Female rats, irrespective of injury status, displayed irregular estrous cycles at the 2-week timepoint, presumably because they were still in late adolescence, but returned to the normal 4 to 5-day cycle by the 5-week timepoint during adulthood. All procedures were performed in accordance with the rules and regulations of the Institutional Animal Care and Use Committee of Drexel University College of Medicine and followed the NIH Guide for the Care and Use of Animals.
Number of Rats Used in Each Experimental Group

*Some rats evaluated in FST were randomly removed from qRT-PCR analysis to fit within the space provided in the well-plate for statistical comparison. In some instances, the estrous phase of the animal during FST was different from that when euthanized for qRT-PCR.
**One group of female rats subjected to single surgery/injury and FST were added in order to only evaluate PACAP expression within the PVT during estrus and were not used in analysis of FST data or neuropeptide expression in the NAc.
FST, forced swim test; qRT-PCR, quantitative real-time polymerase chain reaction; NAc, nucleus accumbens; PACAP, pituitary adenylate cyclase-activating polypeptide; PVT, paraventricular nucleus of the thalamus; NA, not applicable.
Brain injury
Brain injuries were induced using an electronically driven controlled cortical impact device (Custom Design and Fabrication, Richmond, VA) as described,15,16 with identical injury parameters used for each animal. Animals were anesthetized with isoflurane (5% induction, 3% maintenance), a midline incision was made to expose the skull, and then animals were removed from anesthesia for ∼45 sec for the head to be leveled on a foam pad under the impact device. Impacts were induced at the midline of the skull aligned with bregma using a convex metal indenter tip (5 mm diameter), which was fired to 2 mm below the zero point of the surface of the skull (5.5 m/sec velocity, 100 ms dwell time). Sham-injured animals were anesthetized but did not receive an impact. Immediately following impact, the rat was monitored for the time to regain their first breath (apnea) and time to regain normal posture when placed in the supine position (righting reflex). Rats were then reanesthetized, evaluated for skull fractures and hematomas, and then sutured shut. Starting at 6 weeks of age, single-injured rats received only one sham or brain injury, whereas repetitive-injured rats received three sham or brain injuries, each one spaced 3 days apart, with behavior and tissue collection timepoints counted from the final injury day.
Forced swim test
Sham- and brain-injured animals were tested for depression-like behavior using the FST,46,47 during the first half of the light cycle at 5 weeks after single injury and 2 or 5 weeks after repetitive injury.15,16 Rats swam for 10 continuous minutes in the apparatus (49 cm tall, 29 cm diameter acrylic cylinder filled with 30 cm of 25 ± 2°C water) as described. 44 The time spent swimming (moving in the cylinder using hindlimbs), climbing (upward motion with forelimbs), and immobile (least amount of movement necessary to keep afloat) during the last 5 min of the test was quantified by an experimenter blind to injury status and estrous phase.
Quantitative Real-Time PCR
Sham- and brain-injured animals were sacrificed during the first half of the light cycle. As FST is an acute but not chronic stressor, repetitively injured rats were sacrificed 2 days post-FST to avoid a possible confounding stress interaction with neuropeptide expression. 48 To capture the different estrous phases for statistical analysis, tissue collection occurred ∼1 week after FST for single-injured rats. The NAc and the posterior PVT were dissected out to examine mRNA levels of CRF, DYN, ENK (NAc), and PACAP (PVT), as described.35,41 The NAc slice was made coronally, from Bregma + 2.5 to + 1.5 mm, 49 and the sample was dissected bilaterally as teardrop shapes, starting at the ventral end of each lateral ventricle and going ventrolaterally to the lateral side of the striatum and then medially to dorsally along the ventral pallidum back to the ventral end of the lateral ventricle. The posterior PVT slice was made coronally from Bregma −2.5 to −3.5 mm, 49 and the sample was dissected as an inverted isosceles triangle directly ventral to the dorsal third ventricle. Primer sequences and concentrations are listed in Table 2. Total RNA yield resulted in A260/A280 ratios between 1.90 and 2.20, indicating high purity. Target gene expression was quantified relative to cyclophilin-A using the relative quantification method (ΔΔCT) by an experimenter blind to injury status and estrous phase.
Primer Sequences and Concentrations Used for Quantitative Real-Time Polymerase Chain Reaction Experiments

CRF, corticotropin-releasing factor; PACAP, pituitary adenylate cyclase-activating polypeptide.
Statistics
Statistical tests were performed using Statistica 7 (StatSoft, Tulsa, OK). Body weights, latencies to regain righting reflex, and apnea times during surgeries were compared using a two-way analysis of variance (ANOVA) with sex and injury status as independent variables. For repetitive surgeries, repeated measures ANOVA was used to compare these variables between each surgery day. Chi-square tests were used to analyze fractures and hematomas within the brain-injured rats acutely following injury. Two-way ANOVA was used to assess the interaction of sex and injury status (single and repetitive surgeries) or estrous phase and injury status (single surgery females) on FST behavior (time spent swimming, climbing, and immobile) and qRT-PCR (ΔΔCT values), unless otherwise noted. When unequal variances were detected, a Welch’s test was used. Post hoc analysis using the Newman–Keuls test was performed on significant effects (p ≤ 0.05). As an a priori assumption was made that sex-related differences in PACAP expression could exist in females, 41 Newman–Keuls tests were used to examine injury effects within each sex, even when interaction effects between groups did not reach significance. Behavioral and qRT-PCR data are presented as percentage of sham values for each separate group (i.e., injured females vs. sham females or injured estrus females vs. sham estrus females). All data are presented as mean ± standard error of the mean. Data points in behavior and qRT-PCR analyses were categorized as outliers and excluded if the values were more than two standard deviations away from the mean.
Results
Acute responses to injury
A single impact to the closed skull of 6-week-old Long-Evans rats resulted in increased latency to recover righting reflexes compared with sham animals (F1,87 = 144.22, p = 0.000001, Table 3). Brain-injured animals experienced apnea (1–13 sec) immediately after injury (F1,87 = 209.52, p = 0.000001, Table 3). With a significant interaction between injury status and sex (righting reflex, F1,87 = 4.42, p = 0.04; apnea time, F1,87 = 5.51, p = 0.02), injured males displayed a greater loss of righting reflex and apnea time compared with injured females. For male and female rats, impact was associated with a moderate or mild hematoma (91%) and a small linear fracture on the skull (98%).
Acute Neurological Responses to a Single Impact on the Intact Skull of Adolescent Male and Female Long-Evans Rats

Data are represented as mean values and standard errors of the mean.
p < 0.05 compared with sham-injured animals.
p < 0.05 compared with brain-injured females.
NA, not applicable.
Repetitive impacts resulted in increased latency to recover righting reflexes compared with sham animals (F1,71 = 54.82, p = 0.000001, Table 4). With a significant interaction between injury status and surgery day (F2,142 = 5.43, p = 0.005), the loss of righting reflex was greater for brain-injured animals on the first day of injury compared with injury days 2 and 3; no differences in loss of righting reflex were observed in sham-injured animals across surgery days. Brain-injured animals experienced apnea (1–7 sec) immediately following injuries (F1,71 = 349.62, p = 0.000001, Table 4). There were no differences in apnea times between the three injury days. For male and female rats, impact was associated with a moderate or mild hematoma (98%) and a small linear fracture after the first injury (76%). There were no significant sex differences observed in any acute responses, which suggests similar injury severity in all rats after repetitive mTBI. No animals were removed from the study due to the presence of hematoma or linear fracture after injury.
Acute Neurological Responses to Repetitive Impact on the Intact Skull of Adolescent Male and Female Long-Evans Rats
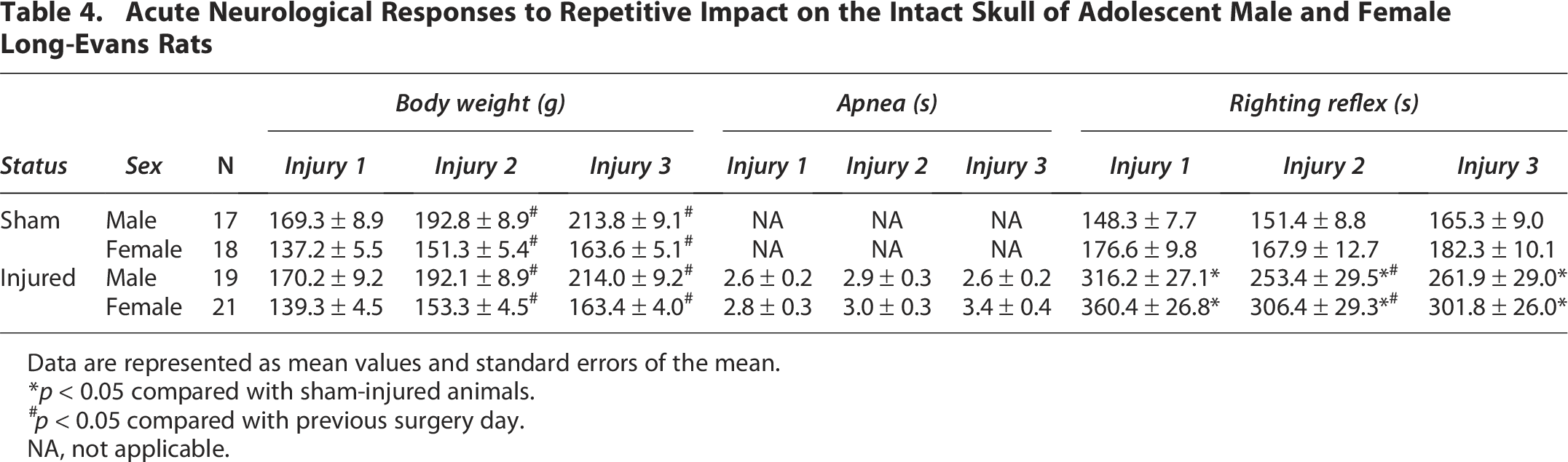
Data are represented as mean values and standard errors of the mean.
p < 0.05 compared with sham-injured animals.
p < 0.05 compared with previous surgery day.
NA, not applicable.
Depression-like behavior in the FST
When tested in the FST at 5 weeks following a single mTBI, there was a main effect of sex for time spent immobile (F1,70 = 44.27, p = 0.0001) and swimming (F1,67 = 56.60, p = 0.0001), where females spent more time immobile (Fig. 1A) and less time swimming (Fig. 1B) than males. There were no differences in time spent climbing (Fig. 1C). Evaluating females in different estrous phases revealed a significant effect of injury status (F1,44 = 7.01, p = 0.01) and an injury status and estrous phase interaction (F2,44 = 8.06, p = 0.001, Fig. 1D). During estrus, brain-injured females spent more time immobile than their sham-injured counterparts, and sham-injured rats in met/diestrus and proestrus spent more time immobile than sham-females in estrus. For time spent swimming, injury status and estrous phase interaction neared but did not reach significance (F2,44 = 3.17, p = 0.052, Fig. 1E). There was a main effect of estrous phase on time spent climbing (F2,43 = 6.12, p = 0.005), where females in estrus spent more time climbing than in other phases (Fig. 1F).

FST behavior 5 weeks following a single surgery in male and female Long Evans rats
Two weeks following repetitive mTBI, there was a main effect of sex for time spent immobile (F1,27 = 4.62, p = 0.04) and swimming (F1,28 = 9.02, p = 0.006), where females spent more time immobile (Fig. 2A) and less time swimming (Fig. 2B) than males, with no significant difference in time spent climbing (Fig. 2C). Five weeks following repetitive mTBI, there was a main effect of injury status (F1,28 = 5.57, p = 0.03) and an interaction of injury status and sex on immobility time (F1,28 = 5.38, p = 0.03), where brain-injured females spent more time immobile than sham females and injured males (Fig. 2D). There was a main effect of injury on swimming time (F1,28 = 14.55, p = 0.0007) and a main effect of sex on climbing time (F1,27 = 4.67, p = 0.04), where injured rats spent less time swimming than shams (Fig. 2E) and females spent less time climbing than males (Fig. 2F).
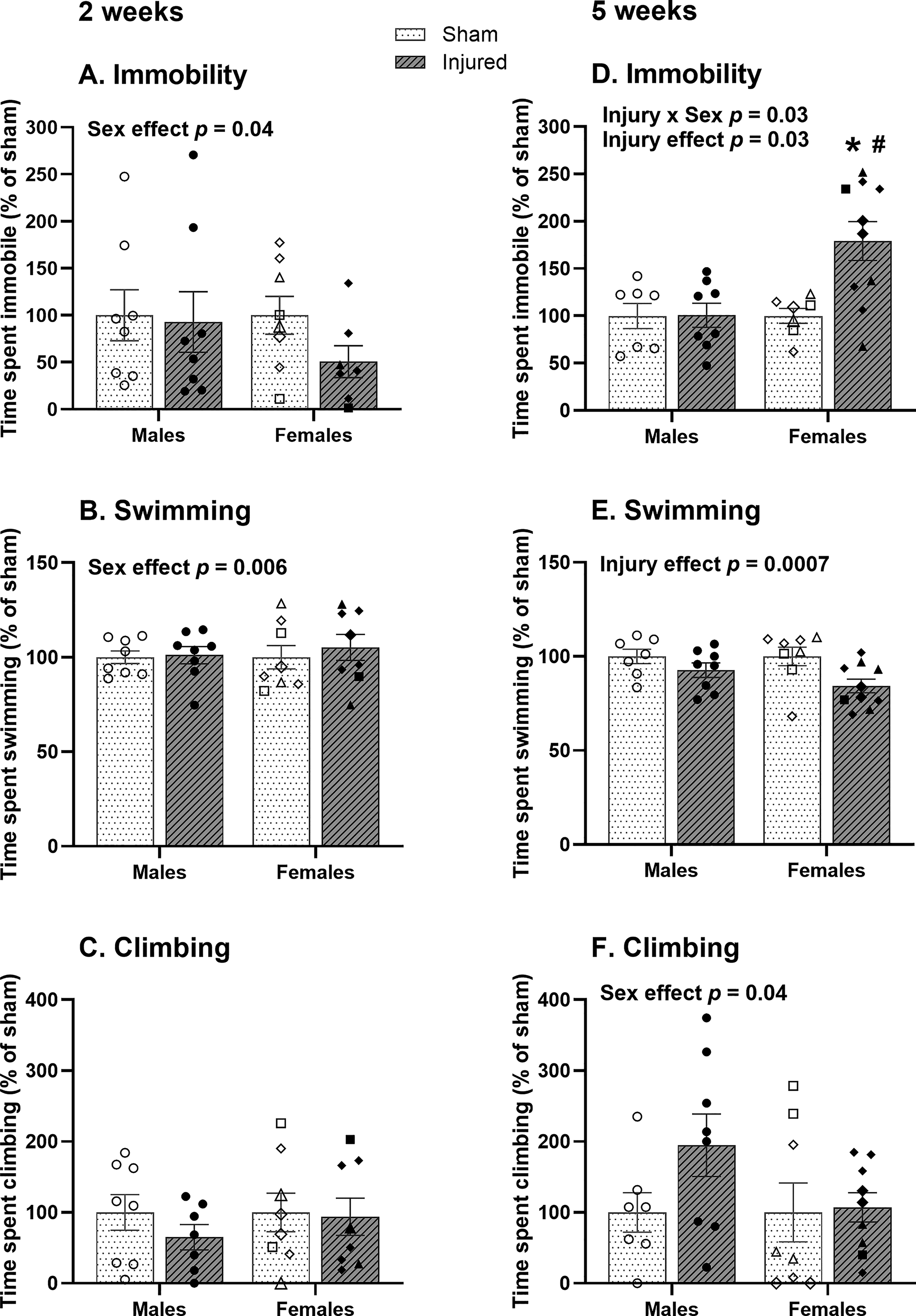
FST behavior in male and female Long-Evans rats at 2 weeks
Neuropeptide mRNA in the NAc
Five weeks following a single mTBI, there was a main effect of sex on CRF expression (F1,53 = 4.84, p = 0.03), where females showed a higher expression than males (Fig. 3A). There were no differences in DYN expression due to sex or injury status (Fig. 3B). There was a main effect of sex on ENK expression (F1,10.385 = 10.50, p = 0.008, Fig. 3C), where females showed less expression compared with males. Due to largely uneven group sizes, the interpretation of Figure 3A–C should be taken with caution. When comparing females by estrous phase, there was a main effect of injury status (F1,40 = 4.03, p = 0.05) and estrous phase (F2,40 = 5.91, p = 0.006) on CRF expression. Injured females showed reduced CRF expression compared with sham females, and estrus and met/diestrus phases had reduced expression compared with proestrus (Fig. 3D). There was a main effect of injury status on DYN mRNA expression (F1,40 = 4.15, p = 0.05), where injured females showed reduced expression compared with shams (Fig. 3E). There were no differences in ENK mRNA expression (Fig. 3F).

Neuropeptide mRNA expression in the nucleus accumbens (NAc) 5 weeks after a single surgery in male and female Long-Evans rats
Five weeks after repetitive mTBI, there were no significant differences in CRF expression in the NAc (Fig. 4A). A main effect of sex on DYN (F1,26 = 10.91, p = 0.003) revealed that females had reduced expression compared with males (Fig. 4B). A main effect of injury status on ENK (F1,26 = 4.30, p = 0.05) showed that injured rats had reduced expression compared with shams (Fig. 4C).

Neuropeptide mRNA expression in the nucleus accumbens 5 weeks after repetitive surgery in male and female Long-Evans rats.
PACAP mRNA in the PVT
Five weeks after a single mTBI, brain-injured females during estrus did not display a difference in PACAP mRNA expression compared with shams, as shown using a t-test (Fig. 5A). Two weeks after repetitive mTBI, there was a main effect of sex (F1,27 = 9.55, p = 0.005) and an interaction of injury status and sex on PACAP expression (F1,27 = 6.88, p = 0.01) that revealed injured females had greater expression of PACAP compared with sham females and injured males (Fig. 5B). Five weeks after repetitive injury, there was a main effect of injury status (F1,26 = 6.22, p = 0.02), where injured rats showed increased PACAP expression compared with shams (Fig. 5C). There was no significant injury and sex interaction, but post hoc comparisons, based on an a priori assumption, revealed that higher expression in injured rats was driven by a significant difference in sham/injured females.

Pituitary adenylate cyclase-activating polypeptide (PACAP) mRNA expression in the paraventricular nucleus of the thalamus 5 weeks after a single surgery in female rats during estrus phase or 2 weeks and 5 weeks after repetitive surgery in male and female Long-Evans rats.
Discussion
In this study, we found that female Long-Evans rats showed increased depression-like behavior in the FST after single and repetitive mTBI. In single-injured animals, this increase appeared during the estrus phase only and, as previously reported, 16 was independent of estrous phase in the repetitive-injured animals. Following a single injury, CRF and DYN mRNA expression in the NAc was reduced in injured females, with the decrease in CRF most pronounced during the estrus phase. There were no significant changes in CRF or DYN expression following repetitive injury across all phases of the estrous cycle. Within the PVT, PACAP expression was only increased in females (across all phases of the estrous cycle) following repetitive mTBI. These differential changes underscore the importance of sex, estrous cycle, and injury severity in governing changes in behavior and associated molecular responses.
The present data in Long-Evans rats recapitulate our previous work in Sprague–Dawley rats,15,16 suggesting that strain differences may not be prevalent following mTBI. It has been reported that female rats in the estrus phase display increased immobility even after treatment with antidepressants.50,51 Similarly, women may experience worse depression symptoms and higher rates of suicide when estrogen levels are lowest. 52 We observed that repetitive mTBI increased immobility time at the 5-week timepoint postinjury, similar to our observations in Sprague–Dawley rats. 16 For the first time, we showed that repetitive mTBI did not induce behavior changes in the FST at the 2-week time point. As this was similar to our observations in Sprague–Dawley rats, 15 we decided not to repeat this 2-week timepoint following single injury. Previous studies in rats injured at 30 days of age have reported significant differences earlier than 5 weeks postinjury,17,53 which may be explained as a function of age-at-injury (early vs. late adolescence). 54 In this regard, repetitive mTBI in 48-day-old mice (late adolescence) did not result in depression-like behavior. 18
Five weeks after a single mTBI, injured females showed reduced mRNA expression of CRF and DYN in the NAc. Estrous phase did not significantly affect DYN expression, but a decrease in CRF expression was the most striking among injured females in estrus. These observations suggest that CRF may interact with both female ovarian hormones and injury, while DYN expression may only change in females as a function of injury, independent of estrous cycling. Previous work has identified increased grooming behavior in females treated with CRF during proestrus, suggesting an interaction between CRF and higher levels of ovarian hormones. 37 This may explain why single-injured females during their estrus phase display increased immobility and decreased CRF: ovarian hormones interact with CRF to produce active behaviors in female rats, and injury may cause a change in CRF synthesis that dysregulates the natural balance. Interestingly, neither DYN nor CRF expression was altered following repetitive TBI. However, ENK mRNA in the NAc and PACAP mRNA in the PVT only changed following repetitive injury. Other studies have observed a downregulation of ENK in the NAc that accompanied increased anhedonia or susceptibility to stress in rats and mice.33,34 PACAP overexpression in the PVT of female rats reduced time spent swimming in the FST. 44 Although we observed PACAP upregulation in the PVT after both 2 and 5 weeks, this suggests that it may be causal of the observed FST behavior 5 weeks after repetitive injury, as the behavioral effects of PACAP may be age-dependent. 55 These opposing limbic neuropeptide mRNA changes after single or repetitive mTBI highlight possible mechanistic differences in mTBI-induced regulation of behavioral changes specific to females. Previously, we did not observe pathology in the NAc following repetitive mTBI, 16 but other studies have observed alterations in the release of dopamine and upregulation of inflammatory genes following a single mild-severe TBI.56,57 The PVT remains largely understudied in the TBI field but has been found to have increased neuronal activation following a moderate lateral TBI. 58 The implications of the PVT-NAc pathway in depression-like behavior, 59 along with our neuropeptide findings in these regions, suggest that mTBI may induce abnormal pathway activity in females that contributes to FST behaviors.
One limitation of this study is that we used estrous cycle phases as an independent variable after single injury and not repetitive injury. This was guided by our previous report that behavior following repetitive TBI was not estrous cycle-dependent. 16 In addition, while males after single injury had longer apnea and righting times than females, this was not observed following repetitive injury. As these acute injury effects are commonly used to determine injury severity, 60 this may suggest a difference in severity between single-injured males and females. However, our behavior results here align with our previously published results in Sprague–Dawley rats,15,16 suggesting that the observed differences have negligible effects on long-term outcomes. In addition, histological and cellular pathology beneath the injury site for male and female Sprague–Dawley rats was similar between the sexes following both single and repetitive injury.15,16 This suggests both sexes experience similar injury severities using our model of midline mTBI. Another limitation is that only one behavior test (FST) was employed to assess depression-like behavior. Although immobility behavior is commonly referred to as “depression-like” behavior, recent commentaries suggest that FST may instead evaluate coping behavior.61,62 Other studies report that mTBI induces affective behavioral deficits, such as anxiety-like behavior and anhedonia.17,63–65 We have not observed anxiety-like behaviors following either single or repetitive injuries in adolescent rats.15,16
Conclusion
Our study shows that both single and repetitive mild midline impacts during adolescence result in chronic depression-like behavior that is specific to female Long-Evans rats and that this is dependent on the estrous cycle following a single injury. It also reveals differences in the level of expression of different limbic neuropeptides after single or repetitive impacts in females, similarly, estrous cycle-dependent after single injury. These observations suggest that chronic depression-like behavior and limbic neuropeptide changes following adolescent mTBI may be specific to females and are dependent on estrous cycling after single injury. However, it may be an overstatement to conclude that males do not show depression-like behavior following mTBI without the use of additional behavioral assays. Nonetheless, our findings still offer insight into sex-specific behavioral and molecular changes following mTBI. There is a need to develop more appropriate treatment for TBI-induced depression that takes these sex-specific and injury-type differences into consideration.
Authors’ Contributions
C.R.M.: Conceptualization, formal analysis, investigation, visualization, writing—original draft, and writing—review and editing. L.L.G.: Conceptualization, investigation, and writing—review and editing. J.R.B. and R.R.: Conceptualization, funding acquisition, methodology, project administration, supervision, and writing—review and editing.
Footnotes
Acknowledgments
The authors thank Lilia Sanzalone for her assistance with qRT-PCR analyses and Dana Lengel, Andrew DiMatteo, and Moriah Harton for their assistance with injury surgeries and behavioral assessments.
Author Disclosure Statement
No competing financial interests exist.
Funding Information
These studies were funded in part by a grant from NIH/NINDS R01 NS110898 (R.R.), a grant from NIH/NIAAA R01 AA028218 (J.R.B.), and a grant from the Pennsylvania Department of Health SAP 410-007-9710 (R.R., J.R.B.).
