Abstract
Highly repetitive exposures to low-level blasts (LLBs) during military training (e.g., breaching and heavy weapons exercises) can cause neurological abnormalities, but the pathological mechanisms are poorly understood. We evaluated the effect of repetitive LLB exposures on cellular responses at specific neuroanatomical and cellular strata and on plasma cytokine levels in a mouse model. Animals were exposed to either 5 ± 0.2 sequential LLB exposures with roughly 3 min intervals or sham exposures consisting of anesthesia only. Overpressure properties were 4.49 ± 0.35 pounds per square inch (psi), 1.04 ± 0.13 msec, and 0.0035 ± 0.004 psi × sec impulse occurring during an average total anesthesia of 14.1 ± 0.02 min. Peak pressure was restricted to ∼20 cumulative psi to ensure animal welfare. Following recovery in their home cage, cardiac blood and whole brains were collected at 4 and 24 h postexposures. We found increased tissue levels of the mitochondrial antioxidant SOD2 in protein extracts of the brainstem. LLB did not increase tau phosphorylation (at Ser396), but the acute drop detected in total tau tissue levels resulted in altered ptau:tau ratios and an increase in brain tissue levels of myelin alarmin protein interleukin-33 (IL-33), indicating myelin injury even in the absence of apparent axonal injury. Mass cytometry images indicated astrogliopathy at the medullary surfaces near the foramen magnum and apical inferior cerebellar lobes consistent with interface astroglial scarring detected at 24 h after LLB. In both the medulla and inferior cerebellum, LLB increased the frequency of association of cluster of differentiation-68+ macrophages with IL-33+ myelin and cerebellar Purkinje cells in regions of astroglial scarring. At 4 h, LLB exposures significantly increased the plasma levels of IL-6, Keratinocyte-derived chemokine/Growth-regulated oncogene alpha (KC/GRO-α), and IL-10 (areas under the receiver operating characteristic curve of 0.8, 0.83, and 0.86, respectively) but not that of Neurofilament light chain (NF-L). Our results indicate that repetitive blast exposures near the 4 psi safety threshold can induce diverse neuropathological changes and systemic inflammatory response consistent with the known acute effects of mild traumatic brain injury despite the classification of these exposures as functionally subconcussive.
Keywords
Background
Reports indicate that repetitive exposures to low-level blast (LLB) overpressures from explosive blasts during military weapons training, often referred to as occupational blast exposure (OBE), can result in both acute and chronic neurological impairments. These effects have been documented in military personnel involved in the frequent use of heavy artillery, 1 mortars, 2 and potentially even smaller explosive devices.3,4 The repeated exposure to rapid pressure fluctuations during blast waves has been associated with a spectrum of cognitive and behavioral dysfunctions, as well as persistent neuropathological changes. 5
Reports suggest that the immediate and long-term consequences of an OBE extend beyond the observed transient subconcussive symptoms. Observed symptoms range from common complaints such as headaches, dizziness, and difficulty concentrating to more severe psychiatric manifestations, including depression, anxiety disorders, increased irritability, and even suicidal ideation.1,6 More concerning is the suggestion that repeated low-level exposures can induce and/or contribute to progressive neurodegeneration, with potential links to tauopathy and chronic traumatic encephalopathy-like pathologies.7–12
Despite military safety guidelines suggesting that overpressures of ≤4 pounds per square inch (psi) are within safe exposure limits, 13 emerging evidence challenges this threshold. 14 Several studies now indicate that these so-called “subconcussive” blast exposures, even at levels below conventional safety standards, may lead to cumulative neurobiological changes over time.4,15–20 This is particularly critical for service members who, despite lacking visible injuries or documented concussions, experience persistent cognitive and emotional difficulties postservice. 21 Additionally, Veterans with service-connected blast trauma disabilities have been reported to have a heightened risk of developing delayed-onset cognitive and mental health disorders, irrespective of direct combat-related injuries.22,23
Given these concerns, a history of frequent occupational exposure to LLBs has been proposed as a contributing factor to cognitive and behavioral impairments observed in noncombat deployed personnel, particularly those involved in training environments where exposure is routine.24–27 This issue has gained increasing attention from the U.S. government and military health agencies, prompting efforts to strengthen protective measures and reassess exposure guidelines to mitigate long-term risks.28–30 Understanding the precise relationship between blast exposure parameters (intensity, frequency, and duration) and the resulting neuropathological outcomes is critical for improving preventative strategies and refining medical assessments for at-risk personnel.
Animal models that simulate blast overpressure exposure in controlled settings provide important tools to better understand the pathobiological effects of LLB.31,32 These models provide crucial insights into how repetitive blast waves affect the brain at molecular, cellular, and behavioral levels.16–18,33 However, many existing models fail to fully replicate real-world military exposure scenarios, as they often do not accurately simulate the frequency, intensity, or duration of exposure experienced during weapons training.9,34–37 This limitation reduces their translational relevance and complicates efforts to derive meaningful conclusions applicable to human populations.
To address these gaps, we recently developed a shock tube-based mouse model designed to mimic highly repetitive, LLB exposures4,15 based on Special Operations Forces (SOF) breaching training. 38 Using this model, we analyzed neuropathological changes and plasma biomarkers in the acute postexposure phase to assess injury responses. Subsequently, we investigated the correlation between neuropathological changes and peripheral biomarker levels, allowing for a more comprehensive understanding of how repeated LLB affects brain function.
Our approach introduces new methodologies to expand the study of acute and chronic neurobiological responses to LLB exposure. By identifying key cellular and molecular injury markers, our work aims to facilitate the development of clinical assessment tools and intervention strategies for service members chronically exposed to occupational LLB. Importantly, our findings highlight acute elevations in common plasma cytokines following blast exposure, suggesting their potential utility as biomarkers for blast-related neurotrauma. These insights could aid in the identification of individuals at risk for long-term neurological deficits and mental health changes, supporting early intervention and risk mitigation strategies for those routinely exposed to LLB in military and law enforcement settings.
Methods
LLB exposure in mice
In this study, 3-month-old, male C57BL/6J mice were used in accordance with procedures approved by the Veterans Affairs Puget Sound Health Care System Institutional Animal Care and Use Committee. Mice were group-housed, four to five to a cage, in an Association for Assessment and Accreditation of Laboratory Animal Care-accredited specific antigen-free facility and maintained on a 12-h light cycle with
Sample collection
Mice were euthanized with sodium pentobarbital (210 mg/kg, Intraperitoneal [i.p.]) at study end-points. Whole blood was then collected via cardiac puncture into a heparinized syringe with a 25-gauge needle and followed by transcardial perfusion with ice-cold phosphate-buffered saline (PBS). The collected blood was transferred into a plasma separator tube (Beckton Dickinson, San Jose, CA; Cat# 365985) and centrifuged. The resulting plasma fraction was then aliquoted, flash-frozen in liquid nitrogen, and stored at −70°C until use. Brains of PBS-perfused LLB and sham mice were removed and bisected at midline along the entire frontal cortical to caudal medulla axis. The left side was stored in 10% neutral buffered formalin until standard paraffin embedding and cutting. The right side of each brain was further dissected into cortical hemisphere (including all subcortical structures), cerebellar, and whole brainstem sections. Each tissue piece was immediately flash-frozen in liquid nitrogen and stored at −70°C until protein extraction. Briefly, tissues were homogenized on ice by hand using an Eppendorf tube-fitting pestle (Eppendorf, Hauppauge, NY) in ice-cold standard Radioimmunoprecipitation assay (RIPA) lysis buffer supplemented with Protease and Phosphatase Inhibitor Cocktail (Sigma, St. Louis, MO; Cat# PPC1010). Lysates were then centrifuged at 12,000
Protein analyses
Plasma cytokines were measured on a Meso Sector S 600 instrument using a 19-plex electrochemiluminescent assay validated for mouse (Mesoscale Discovery, LLC, Rockville, MD; Cat# K15255D). Plasma NF-L levels were measured by colorimetric enzyme-linked immunosorbent assay (Novus Biologicals, Centennial, CO; Cat# NBP2-80299).
Brain extracts were analyzed using Western blots and cytokine arrays. Proteins (20 μg per lane) from individual animal supernatants were separated on Criterion TGX Stain-Free 4–20% SDS-PAGE gels (Bio-Rad) and transferred to Immun-Blot Polyvinylidene difluoride (PVDF) membranes using the Bio-Rad Trans-Blot Turbo system. Membranes were blocked with 10% BLOTTO in PBS for 1 h and probed with antibodies against total tau (tau5), phospho-tau S396, Superoxide dismutase (SOD2)/MnSOD, and pyruvate kinase. Membranes were stripped with 25 mM glycine-Hydrochloric acid (HCl) (pH 2) and 1% SDS, rinsed, reequilibrated in Tris-buffered saline with Tween-20 (TBS-T), and reblocked before reprobing. Densitometry was performed using Photoshop, with bands standardized to pyruvate kinase or total tau (for phospho-tau S396) and normalized to sham control means.
The RnD Proteome Profiler Mouse XL Cytokine Array (RnD Systems, Minneapolis, MN; Cat# ARY028) was used for the discovery analysis of mouse brain cytokine responses to LLB. Array blots were imaged in pairs, each containing a single unique sham and LLB animal and using lysates from the same brain tissue region. LLB values were normalized against the yoked sham control values. All blots were imaged on a Bio-Rad Chemidoc MP (Bio-Rad, Hercules, CA; Cat# 12003154) using enhanced chemiluminescence (Thermo Fisher Scientific, Bothell, WA, Cat# 45-010-090).
Imaging mass cytometry and analyses
Formalin-fixed, paraffin-embedded brains were sagittally sectioned at 10 μm, mounted on SuperFrost slides, and analyzed via imaging mass cytometry (IMC). Prelabeled metal-conjugated antibodies were obtained from Standard Biotools, while custom-labeled antibodies were prepared using the MaxPar X8 kit per manufacturer’s protocols. Tissue staining followed standard protocols with a reduced DNA intercalator concentration (1:4000 iridium 191/193) for 30 min. Slides were washed, air-dried overnight, and stored at room temperature.
For analysis, 500 μm 2 (or larger) brain regions were ablated using the Hyperion UV laser (1 μm 2 resolution), and the resulting aerosolized material was quantified using the Helios mass cytometer. Imaging datasets were imported into CellProfiler as .tif files, with single-cell segmentation performed using a watershed gradient algorithm based on DNA intercalator and IMC Cell Segmentation Kit markers.
Purkinje cells were identified in inferior cerebellar lobe images by gating Mouse double minute 2 homolog (MDM2)-intensity < 5 in cells with > 90 μm 2 soma area. Other cell types (astrocytes, neurons, oligodendrocytes, and vasculature) were manually annotated based on marker expression. Segmented images were processed in histoCAT, using Barnes–Hut t-distributed stochastic neighbor embedding (t-SNE), PhenoGraph, and neighborhood analyses (proximity = 6). Images were saved in an MCD viewer, then despeckled in Photoshop using a Gaussian blur filter.
Statistical analyses
Statistical analyses were conducted in Prism 10.0.0 for Mac OS (GraphPad, San Diego, CA) or R version 4.2.3 (R Core Team, Vienna, Austria) using an α of 0.05. Sample sizes were based on previous reports.4,39,40 Standard analysis of variance was used for planned statistical tests involving multiple measures or groups followed by Bonferroni correction for multiple comparisons. We used
Results
LLB induces acute and subtle axonal and white matter injury signatures in the brainstem
We used a high-density cytokine array validated for mouse tissue lysates to discover potential brain injury response markers that may occur 4 h after LLB. One-hundred and eleven cytokines were probed in parallel from tissue lysates of brainstem (i.e., mixed pons and medulla), cerebellum, and whole cortical hemispheres. These results are summarized in Figure 1. Following correction for multiple comparisons using a 1% FDR adjustment, significant elevations in the tissue levels of myelin alarmin protein interleukin-33 (IL-33), the retinoic acid responsive protein Chemerin, IL12p40, leukocyte inhibitory factor (LIF), and serum amyloid P (SAP) were observed in the brainstem (Fig. 1a). No cytokine elevations were detected in cerebellar lysates (Fig. 1b), whereas only minor elevations in cortical levels of lipoprotein homeostatic protein PCSK9 and the pattern recognition receptor Receptor for Advanced Glycation End-products (RAGE) were observed (each,
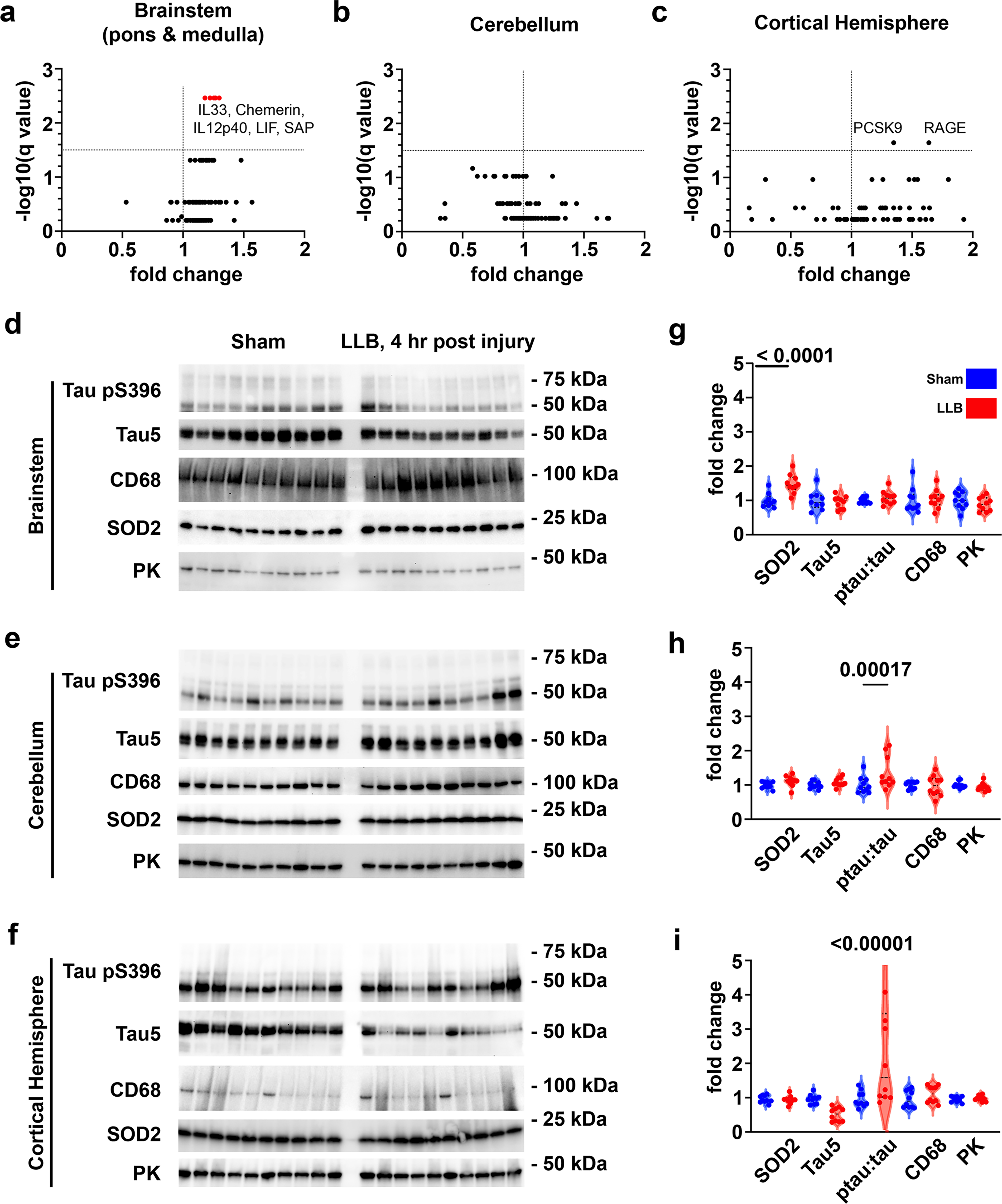
LLB exposure induces subtle axonal and myelin injury within 4 h after repeated LLB exposure. Cytokines in detergent-soluble lysates were measured by microarray from
Elevated brainstem expression of the myelin alarmin IL-33 may indicate that LLB exposure provokes brainstem myelin injury signaling likely related to axon injury. We investigated the potential for LLB to induce signatures of axon injury using Western blotting with tissue homogenates obtained from 10 LLB and 10 sham mice. We found increased tissue levels of the mitochondrial antioxidant protein SOD2/Manganese superoxide dismutas (MnSOD) in brainstem tissues, confirming similar reports by Song and colleagues,
42
but found no similar changes in the cerebellum or cortical hemispheres (brainstem:
LLB causes acutely increased levels of plasma cytokines
To determine whether LLB overpressures near the historical U.S. Department of Defense/War (DoD/W) safety limit of 4 psi can cause systemic inflammatory responses that may inform signatures of probable brain trauma, we measured plasma proinflammatory cytokines and the murine righting reflex (used as a measure of neurobehavioral response to brain trauma) in mice after LLB and sham exposures. LLB mice were subjected to an average of five sequential LLB exposures with a mean peak pressure of 4.49 ± 0.35 psi, a positive phase duration of 1.04 ± 0.13 msec and an impulse of 0.0035 ± 0.004 psi × sec impulse (Fig. 2a). LLB exposure significantly reduced the murine righting reflex as compared with sham control mice (
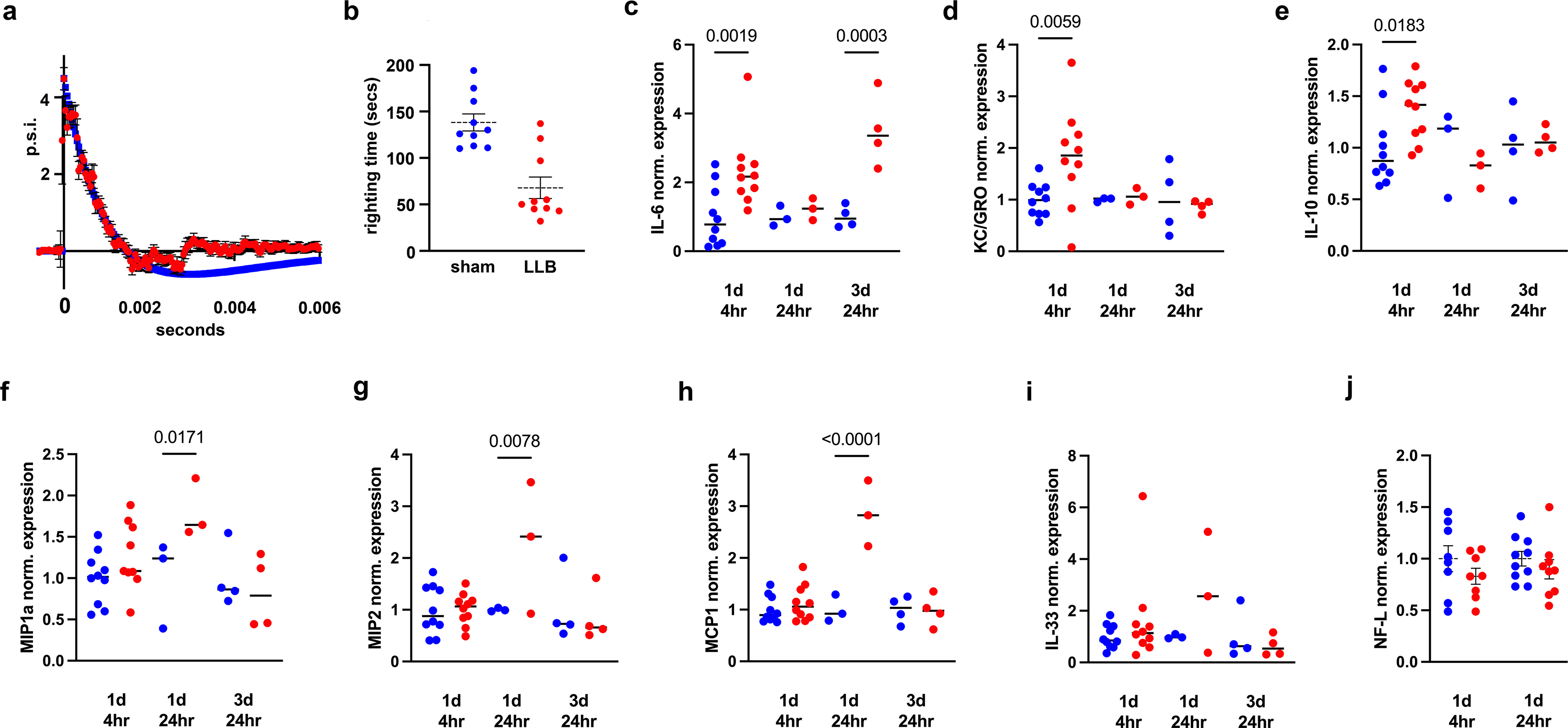
Repeated LLB exposure is detectable in mouse plasma as inflammatory cytokine elevations.
Highly repetitive LLB near 4 psi induces a significant increase in plasma IL-6 levels
Since plasma cytokine levels correlated with measures of blast exposure, we sought to investigate the utility of plasma cytokines to distinguish sham from LLB mice at 4 and 24 h after exposure—a timeframe relevant for routine acute clinical care testing and decisions. Cytokines elevated at 4 h after LLB exposure had areas under the receiver operating characteristic curve (ROCC Area under the curve [AUC]) of 0.86 (IL-10,
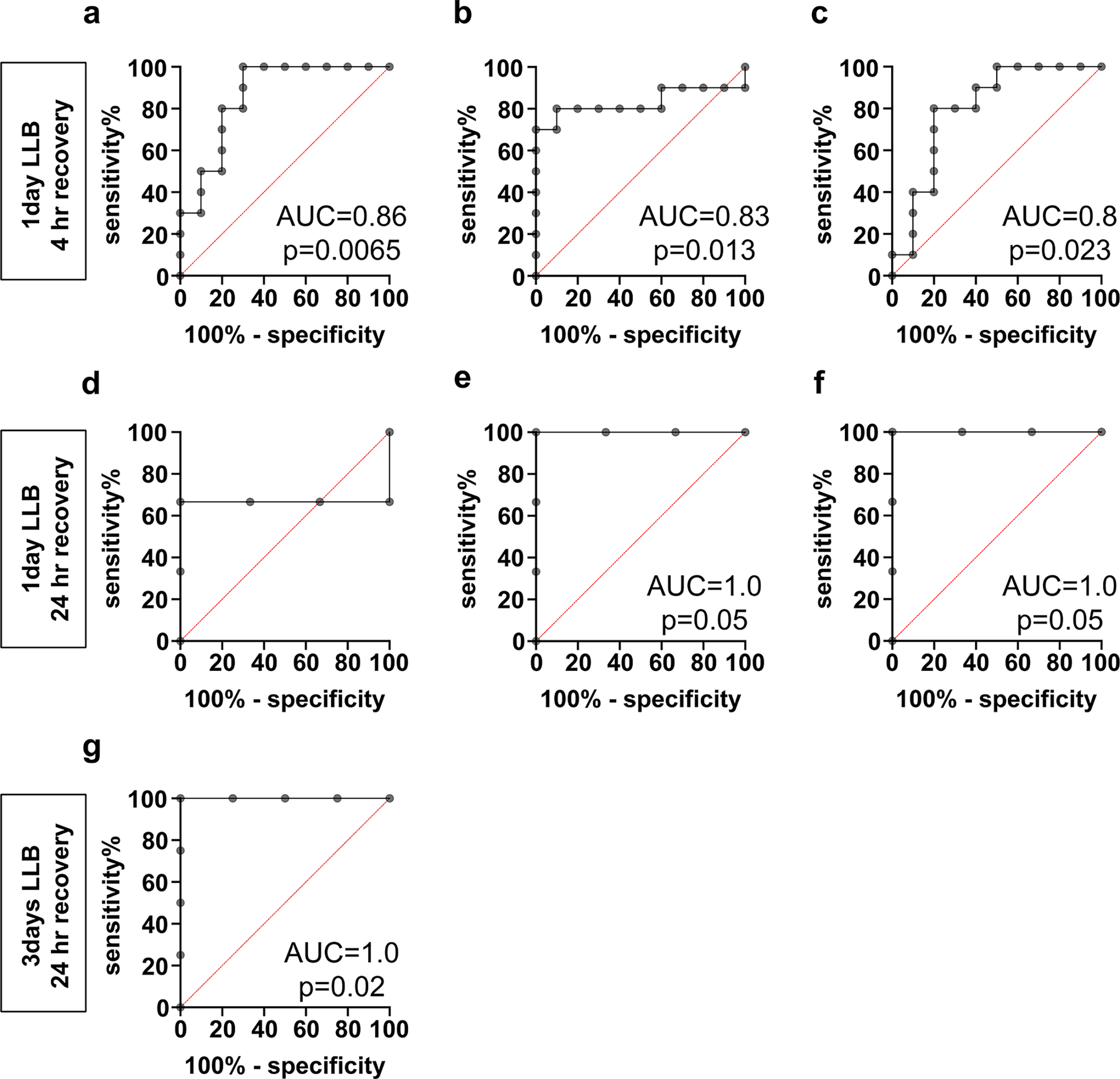
Inflammatory plasma cytokines have high diagnostic performance for LLB in mice.
Elevated levels of plasma cytokines correlate with LLB exposure measures and cytokine changes in brain tissue
Candidate peripheral cytokines may offer an effective diagnostic triage tool for persons with blast-related symptoms following significant LLB exposure. To investigate the potential for informative relationships between plasma cytokine levels, measures of blast exposure, and brain injury responses, we performed a comprehensive nonparametric Spearman’s correlation analysis controlled for multiple comparisons using a rigorous 1% FDR adjustment among the collected variables in these mice (Fig. 4). Plasma IL-6 levels at 4 h after LLB significantly correlated with the number of total LLB exposures (
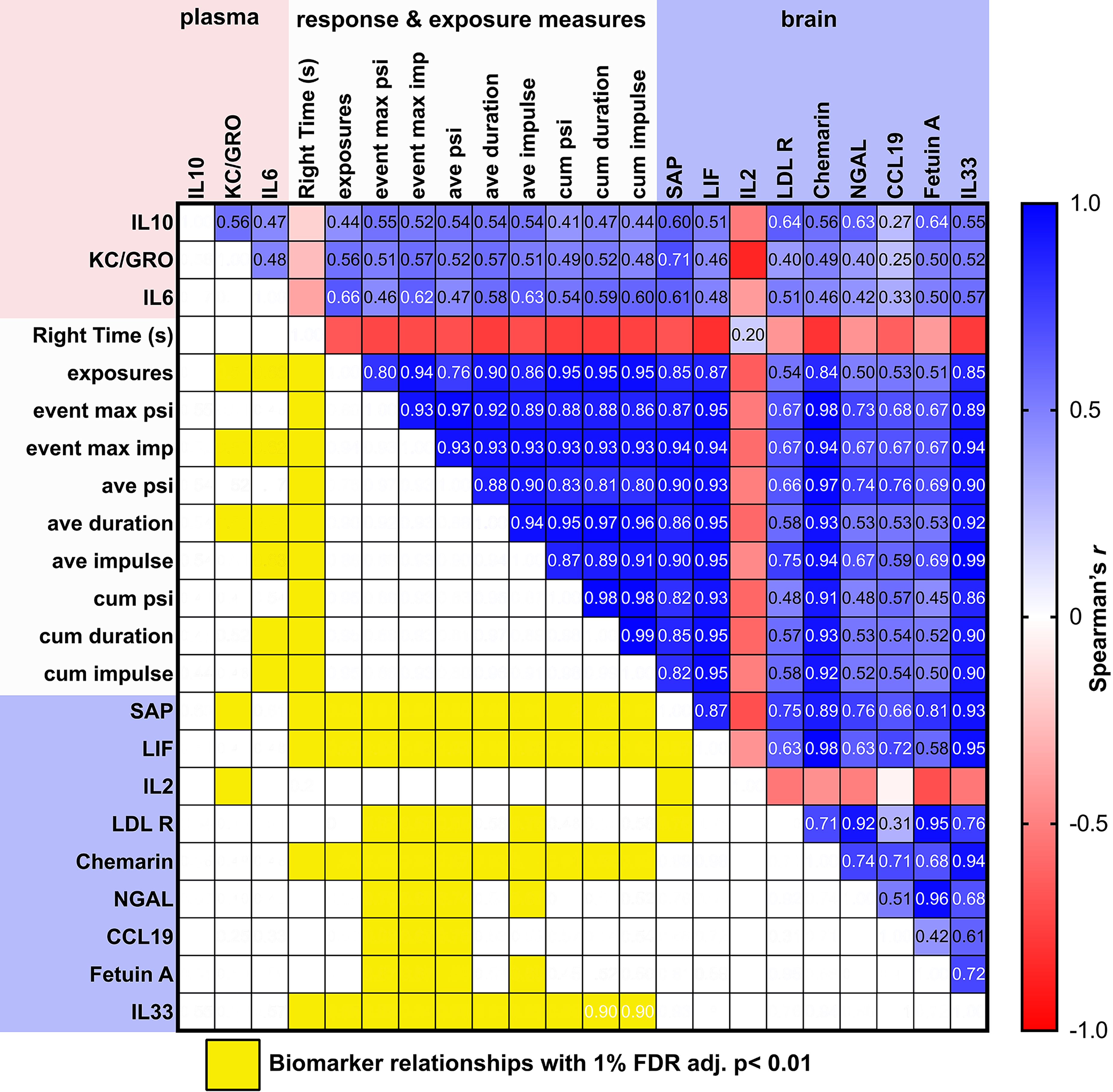
Correlogram of blast exposure measures versus cytokine expression 4 h after repeated 4.5 psi LLB. Correlation plot of plasma cytokines against study measurements. Cells denote results of Spearman’s
Brainstem myelin and cerebellar Purkinje cell injury recruit macrophages after repeated LLB exposure
We developed an unbiased computational approach based on quantitative imaging mass cytometry (IMC) data to identify alterations in tissue characteristics and their association with macrophages after exposure to four LLB exposures, considering that the response and recovery of tissue pathology following concussive high-level blasts are primarily influenced by microglia and macrophages. In pursuit of this objective, a collection of 25 IMC antibodies with pure isotopic metal labels was designed to investigate the nuanced pathology of murine TBI. This panel encompasses a diverse range of markers covering prominent neural cell types, structural components, pathological characteristics, and crucial signaling pathways (Supplementary Data S1). Following the application of these antibodies to tissue slides, we ablated 500 μm
2
regions at a 1 μm
2
subcellular resolution across the medulla, pons, cerebellum, and cortical hemisphere using a Hyperion scanner equipped with a UV laser. The ablated material was then quantified using a Helios time-of-flight mass cytometer. High-dimensional images for each region, analogous to a series of highly multiplexed fluorescent micrographs, were then reconstructed from the rasterized and timestamped metal abundances (Fig. 5). By utilizing CellProfiler for segmenting the image into spatially resolved single cells and performing analyses in histoCAT and R, we successfully detected 220,859 cells from 101 images representing 10 brain regions from 10 mice each (
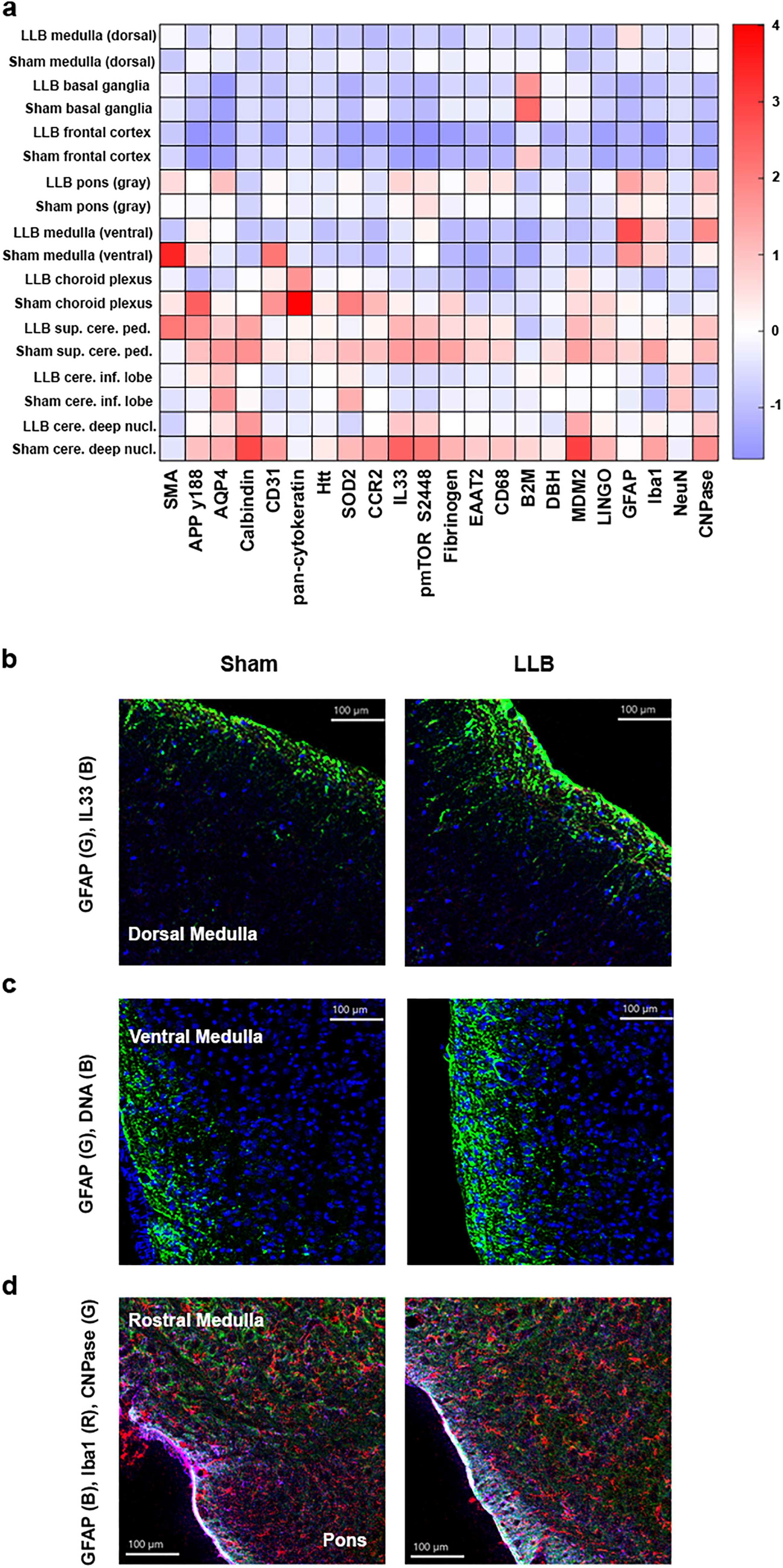
LLB increases hindbrain gliosis within 24 h of exposure.
Following nonlinear dimensionality reduction in single cells with Barnes–Hut t-SNE, we applied an unbiased PhenoGraph-based clustering method to identify 27 distinguishable and informative phenotypes, including microglial macrophages (cluster 14: CCR2-, CD68+, and Iba1+ cells), neurons, blood vessels, glial cells, and peripheral immune cells (Fig. 6).
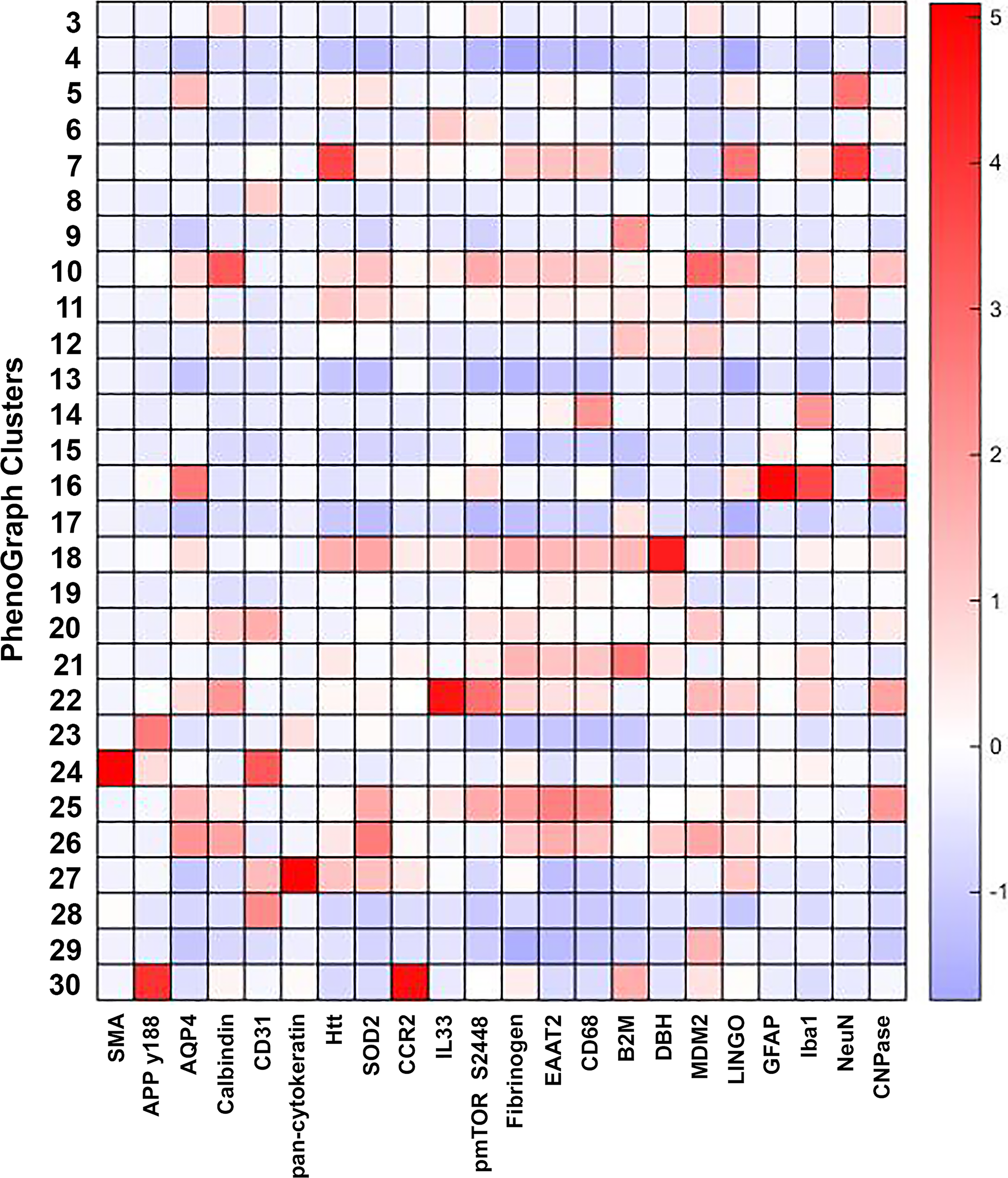
Cell phenotypes identified by PhenoGraph. The heatmap reports scaled marker intensities across cellular phenotypes (i.e., Clusters) present in the data and identified by PhenoGraph analysis. IL-33, interleukin-33.
The analysis of local cellular environments allows for an objective and methodical examination of the reciprocal relationships between cells within a tissue or across multiple tissue samples. We used the neighborhood analysis algorithm of histoCAT, which employs a permutation test to determine interaction frequencies among all cell types in an image with those in a random sampling of cells.
44
By employing this approach, the statistical significance of cell-to-cell interactions is determined; exposing patterns of attraction or avoidance that indicate the overall changes in cellular arrangements provides a data-driven description of the brain’s cellular landscape following LLB exposure. The frequency of intercellular associations, as measured by neighborhood analysis, was significantly increased between spatially resolved macrophages with IL-33+ myelin in the dorsal medulla (one/five sham mice vs. three/five LLB mice), dorsal rostral pons (two/five sham vs. four/five LLB mice), and the inferior cerebellum (zero/five sham vs. four/five LLB mice) (cluster 6, light green boxes, Fig. 7a,b). Macrophage attractions also increased for Purkinje cells in the inferior cerebellum (cluster 26, dark green box, Fig. 7a,c), where trending reductions in CNPase (
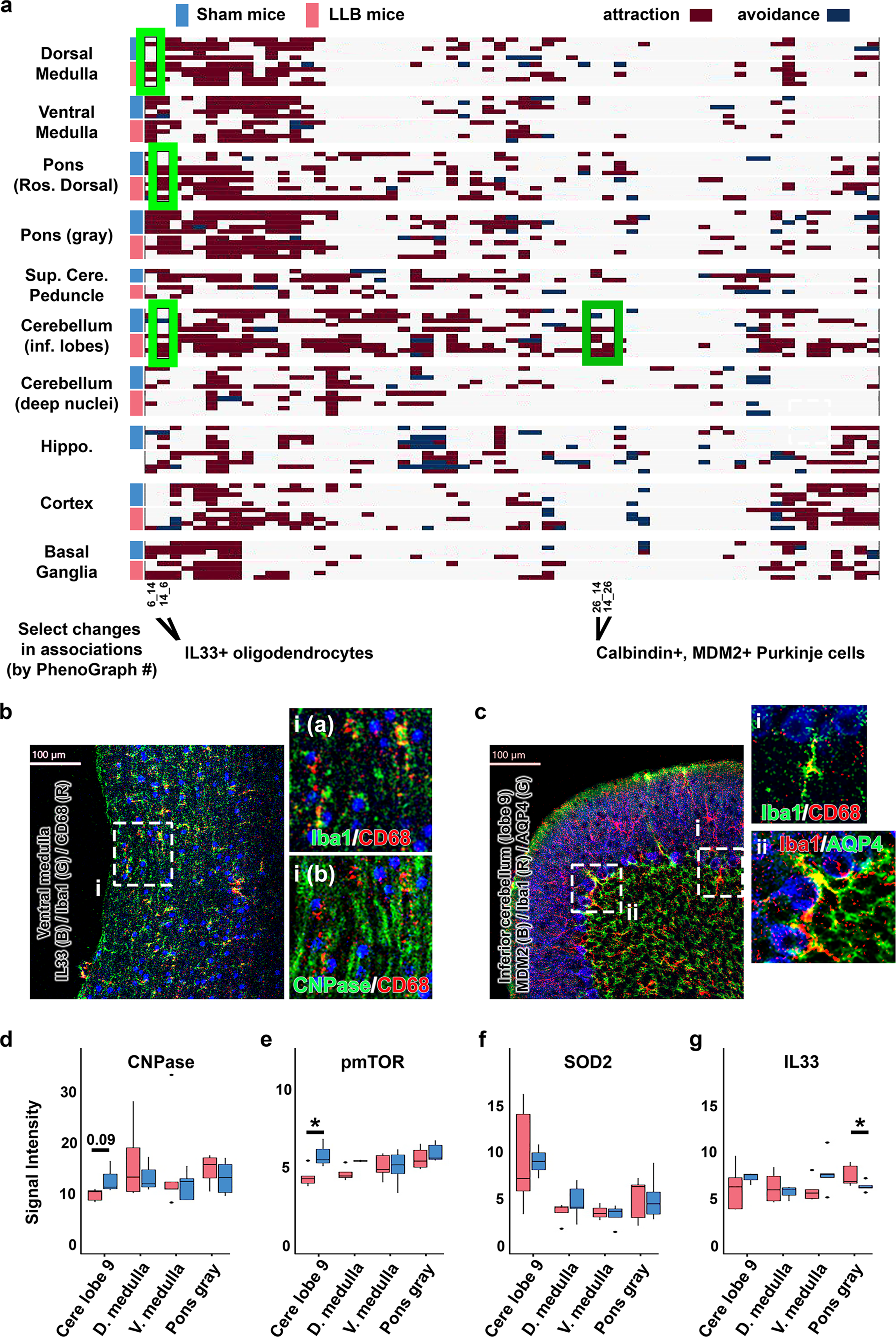
Repeated LLB increases macrophage associations with hindbrain white matter and cerebellar Purkinje cells.
Purkinje cells of the inferior cerebellum are vulnerable to repetitive blast.40,45–47 We examined the expression of antioxidant and antiapoptotic proteins in spatially resolved Purkinje cells 24 h after a single day of repetitive LLB, testing the hypothesis that the expression of these proteins would be compensatorily increased following LLB. To our surprise, we observed a significant loss of Huntingtin (HTT) and Leucine-rich repeat and Ig domain-containing NOGO receptor-interacting protein 1 (LINGO1) expression and trending reductions in SOD2, pmTOR S2448, and MDM2 (Fig. 8a). Example IMC images are seen in Figure 8b and c. Pyknotic Purkinje cells were not observed. However, surprisingly, GFAP+ astrocytic hypertrophy along the Purkinje cell interface with the granule cell layer was observed in a pattern consistent with astroglial interface scarring (Fig. 8c), where local CD68+ macrophages were observed exclusively interacting within this region. Taken together, these findings suggest that even briefly repeated LLB exposure may increase risk for hindbrain cellular injury responses, evidenced by macrophage-mediated recruitment to the affected areas.
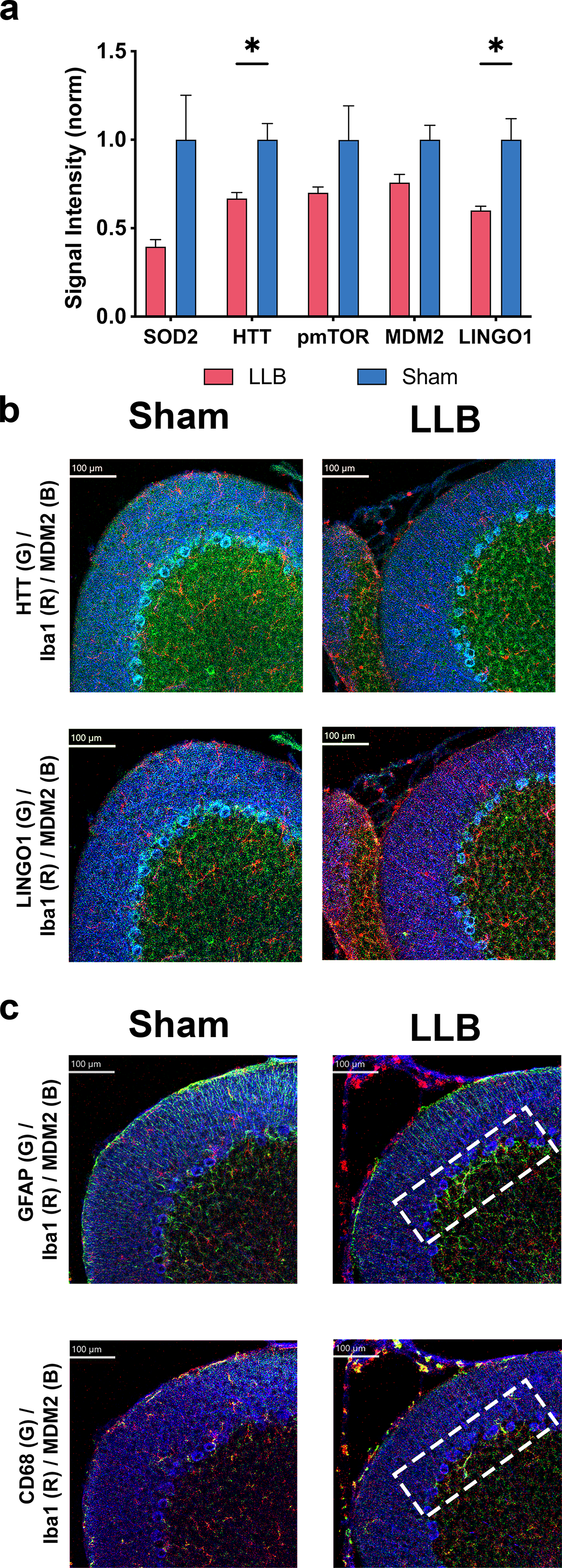
Repeated LLB reduces Purkinje cell expression of antioxidative injury and antiapoptotic proteins.
Discussion
Exposure to military blast overpressures may occur in combat and during training with specific weapons systems.4,38 While large explosive events are well established to cause peripheral and central injuries, the U.S. DoD/W has historically considered blast overpressures of 4 psi or less as relatively safe, intentionally creating guidance reducing exposure to unnecessarily higher overpressures based on the best scientific and epidemiological data available at the time.11,48–50 However, recent reports indicate that the negative effects of exposure to LLBs (≤4 psi) may require significant repetition and/or time to manifest. Our results indicate that in blast-naïve animals as few as four repetitive blast exposures near 4 psi may provoke measurable adverse cellular responses consistent with acute mild TBI and peripheral polytrauma. 51
Pervasive patterns of physical and mental health changes have been noted, particularly among SOF and others who are exposed to repetitive occupational LLB exposures, such as artillerymen and mortarmen. These changes appear to be the result of multiple pathological mechanisms and result in the onset of a variety of mental health disorders and other symptoms, such as anxiety, impulsivity, insomnia, suicidality, depression, and cognitive decline.52–54 Past research has revealed cumulative, repetitive LLB exposure, even when under 4 psi, yields newly elevated Neurobehavioral Symptom Inventory scores and new onset of mental health symptoms, such as decreased concentration, slowed thinking, difficulty sleeping, and irritability, particularly among those with high cumulative LLB exposure.4,21
Though blast-induced neuropsychiatric and behavioral health changes can present acutely, adversely affecting force readiness, they may also develop over time, potentially leading to disability claims and chronic health management in the military and the Veterans Affairs Health Care Systems. Some adverse responses to repetitive LLB exposure, such as neuroinflammation, perivascular astrocyte alterations, and synaptic alterations, are expected to take months to years for the full extent of their cumulative injury to manifest clinically. 55
One such injury is that of neurovascular damage. LLB damages the neurovascular unit, in effect stripping the blood–brain barrier of astrocytic endfeet along injured sections of neurovasculature. 56 Astrocytes provide both lactate shuttling for neuronal energy supplies 57 and proper flow of cerebrospinal fluid through transporters localized at the astroglia endfoot–endothelium interface. 58 Damage to these critical structures can lead to insidious changes in neuronal network efficiency and glymphatic waste clearance that are expected to worsen with repeated LLB exposure in the absence of mitigating factors leading to chronicity of these injuries.59,60 In support of this, several large, well-powered studies have concluded that Veterans who had high occupational exposure to repetitive LLB have an increased risk for delayed cognitive impairments and mental health issues,3,4,61,62 indicating that a single evaluation after leaving military service may not adequately capture the range, development, and prognosis of LLB-associated injuries.
In the present study, we investigated early changes in brain cellular and molecular responses and plasma biomarker levels in an LLB mouse model. We found that several factors affected the extent of these acute injury responses. First, within 4 h after a single LLB, IL-6, KC/GRO-α, and IL-10 showed strong diagnostic performance with AUCs of 0.86, 0.83, and 0.8, respectively (Fig. 2). Though these factors returned to baseline within 24 h, repeated daily LLB exposure extended the duration of these elevations (Fig. 2g). These results indicate the repetitive LLB triggers the elevated release of IL-6. Second, the brainstem and, to a lesser degree, the cortex showed increased tissue levels of specific cytokines. Among these, increased expression of the myelin alarmin IL-33 was detected in brainstem homogenates but not elsewhere in the brain. Increased levels of chemerin, IL12p40, LIF, and SAP were also detected in the brainstem following LLB, indicating a complex inflammatory response. These cytokines play crucial roles in immune cell recruitment, inflammation, and tissue repair. Elevated levels not only suggest the selective vulnerability of the brainstem to LLB but also suggest that excessive inflammation may lead to further tissue injury. Third, the brainstem showed evidence of oxidative stress. We observed increased SOD2 expression 4 h following repetitive LLB exposure. Dysregulation of tau, however, was observed more broadly, with significant changes occurring in the cerebellum and cortex. LLB was associated with reduction in total tau levels and not strictly increased levels of tau pS396. Fourth, we observed several strong correlations between plasma cytokine levels and both blast overpressure properties and brainstem cytokine levels at 4 h after repetitive LLB exposure. While plasma IL-6 levels strongly positively correlated with brainstem SAP and IL-33 levels, neither survived statistical correction for our discovery approach in these exploratory analyses. Although this is likely due to our small study sample, plasma levels of KC/GRO-α (C-X-C motif chemokine ligand [CXCL1]) did, however survive these adjustments, correlating with brainstem levels of SAP and IL-2. Plasma KC/GRO-α showed a strong trending correlation with brainstem IL-33 levels 4 h after LLB. It is critical to note that significant elevations in neither plasma IL-33 nor brain IL-6 or KC/GRO-α were observed. Fifth, brainstem levels of IL-33 strongly correlated with single-event and cumulative measures of blast exposure as well as righting time. This observation indicates an underappreciated and elusive causal relationship between even “low levels” of blast exposure and mechanisms of axonal injury and dysmyelination in at least the brainstem. Increased GFAP+ astrogliosis in the pons and medulla suggests that the lateral regions of the brainstem may have increased vulnerability to LLB. An increase in microglia expressing the macrophagic marker CD68 was seen in the pons (Fig. 7a). CD68+ microglia also increased their cellular association with IL-33+ oligodendrocytes in the brainstem and cerebellum and calbindin+/MDM2+ Purkinje cells of the inferior cerebellum, where these cells showed a significant reduction in antioxidative injury and antiapoptotic proteins. (Fig. 8). How these trends might change between the context of acute injury as examined by the present study and chronic injury, such as that experienced by SOF, requires further investigation since acute injury processes in naïve subjects and conditioned subjects are known to differ greatly. 4
Our study adds further support for biomarker development for periacute serial blood draws to inform medical interventions and exposure. 10 Acute peripheral IL-6 levels have been reported to have high diagnostic accuracy for detecting and predicting injuries following head injury. 63 Our results demonstrate that changes in plasma IL-6 levels may have similar utility for positive diagnosis of acute LLB injury at overpressures near 4 psi. This may be important for screening for suspected head injuries that are negative for increased plasma levels of NF-L, similar to the results obtained herein and reported elsewhere. 64 Chronically increased plasma IL-6 and neuroinflammation have been correlated with suicide. 65 Neuroinflammation and increased cytokine activity are present in other mental health conditions and symptoms, including depressed mood, bipolar disorder, and schizophrenia. 66
Elevated plasma IL-6 levels after blast exposure may affect long-term outcomes in high-risk populations. In a study of 50,864 participants from the UK Biobank, including 1391 new dementia diagnoses, higher plasma IL-6 levels were associated with regional brain atrophy, poorer cognitive performance, and increased risk of dementia in aged adults. 67 In a recent study, 68 higher plasma IL-6 levels predicted worse cognitive performance, lower hypothalamic/hippocampal volumes, and increased T2 white matter hyperintensities among both Alzheimer’s disease (AD) and cognitively normal control participants. Interestingly, in the same study, experimental IL-6 neutralization in AD mouse brains rescued memory deficits, peripheral glucose intolerance, and circulating IL-6 levels. While this study suggests that controlling brain IL-6 levels may have positive peripheral effects during late-life neurodegenerative disease, it similarly begs the question of whether controlling peripheral IL-6 levels may aid in the recovery of the brain after polytrauma earlier in life.
This study has several limitations. First, our analysis focuses on only two early postexposure time points (4 and 24 h). Our study does not inform how LLB responses change with conditioning or mitigation strategies and therefore can only be interpreted in the context of injury responses in naïve subjects with little similarity to well-conditioned subjects such as SOF. Nonetheless, while the magnitude and type of injury response likely differ, we find the current results to be informative as to where similar changes may be found in conditioned subjects. Importantly, the current study suggests a pattern of injury similar to Veterans and mice exposed to high-level blast overpressures that occurs in combat. 40 Second, our study does not inform how mitigation or conditioning strategies that are undoubtedly critical for maintaining human health in high-risk occupations alter responses to LLB. The observed increases in inflammatory cytokines and the apparent gliopathy offer objective markers to support the evaluation of candidate prophylactic and treatment efficacies for acute LLB-related injuries. Treatments reducing microglial, astrocytic, and myelin injury responses are expected to be important to prevent the transition from deranged acute injury responses to chronic neuropathology and functional deficit. Last, while our study does establish that LLB near the 4 psi safety threshold produces robust peripheral and central inflammatory polytrauma, it is based on a small sample size. It should also be noted that animals were exposed to whole-body overpressures without chest protection. This raises the possibility that the observed chemo- and cytokine elevations in the systemic blood were due to injury to peripheral organs, especially the lungs.
An important challenge in translating experimental findings to human scenarios is the vastly different and currently not well-understood differences in the temporal aspect of pathobiologies between rodents and humans.32,69,70 Inflammation occurs faster in rodents than in humans, and furthermore, the exact “conversion factor” appears to be different for cellular versus humoral components of inflammation.
Understanding the effects of LLB exposures has direct implications for the military readiness of SOF units. By monitoring potential neuroinflammatory responses and cellular changes caused by LLB, proactive measures can be implemented to prevent long-term neurological impairments, improving force lethality and preserving quality of life after military service.
Mechanistic and interventional studies are advised to use cohorts powered to achieve their specific study measures. This study sheds light on the poorly understood pathology caused by chronic exposure to LLBs. By demonstrating the inflammatory responses and cellular changes that occur after a single day of LLB exposure, our findings extend current knowledge regarding the effects of blast-induced injuries. Such insights may yield significant implications for military readiness and the assessment of U.S. Department of Veterans Affairs (VA) disability claims. Further research is warranted to explore potential therapeutic interventions and preventative strategies to mitigate the long-term impact of LLB exposure on brain health.
Transparency, Rigor, and Reproducibility Statement
This study was not preregistered. The analyses of experimental materials were performed by investigators blinded to relevant characteristics of the subjects. The analyses of IMC data were performed by an unbiased computational pipeline. The information on equipment and analytical reagents not otherwise listed in the Methods section is available upon request. The statistical tests, assumptions, and corrections for multiple comparisons are detailed in the Methods section. A limited number of histological samples from each of the experimental groups are available for future analyses upon request. The authors have agreed to publish the article using the Mary Ann Liebert, Inc. “Open Access” option under an appropriate license.
Authors’ Contributions
J.S.M.: Conceptualization, data curation, formal analysis, investigation, methodology, visualization, writing—original draft, and writing—review and editing. A.C., C.M., R.I., and D.A.: Conceptualization, formal analysis, writing—original draft, and writing—review and editing. M.O., J.R., T.C., and D.M.: Data curation, investigation, and methodology.
Footnotes
Acknowledgments
The authors thank Andrew Shutes-David for editorial assistance; Molly Chinn and Hollie Holmes for administrative support; and Elaine Peskind and Jeff Iliff for their expert scientific consulting.
Author Disclosure Statement
The opinions expressed herein are those of the authors and do not necessarily represent those of the DOD/W, the Department of Veterans Affairs, or any other U.S. government agency. The authors declare no conflicts of interest.
Funding Information
This work was supported by the Biomedical Laboratory Research and Development, US VA Office of Research and Development, under a VA Merit Review award (J.S.M., 5I01BX004896) and a Congressionally Directed Medical Research Programs award (J.S.M., HT94252310755).
