Abstract
Ketamine is a dissociative anesthetic often used for airway management in trauma. While perceived to preserve hemodynamic stability, concerns exist regarding its effects on intracranial pressure and cardiac output in critically ill patients. There is a lack of studies evaluating outcomes after ketamine administration in the setting of traumatic brain injury (TBI). This study aimed to investigate the effects of ketamine on outcomes and physiological responses in a large cohort of TBI patients. We hypothesized that ketamine administration would not be associated with differences in survival, vital signs, or disposition outcomes compared with other induction medications when administered following TBI.
This was a retrospective, observational study utilizing data from the Linking Investigations in Trauma and Emergency Services registry (2017–2021). Subjects were divided into two groups: those who received only ketamine (n = 429) and those who received other induction medications (etomidate and/or propofol; n = 993). We compared 24-h mortality, initial in-hospital vital signs (systolic blood pressure [SBP], respiratory rate, heart rate, and Glasgow Coma Scale [GCS]), and hospital discharge disposition; a propensity score analysis adjusted for potential confounders including race, injury type, pre-hospital GCS, initial pre-hospital SBP and site location.
Ketamine-exposed subjects were younger and presented with a worse clinical profile, including lower pre-hospital GCS (5 vs. 6, p < 0.01) and lower SBP (126.5 vs. 144.0 mmHg, p < 0.01) compared with the ketamine-unexposed group. Unadjusted analysis showed a significantly higher 24-h mortality rate in the ketamine-exposed group (3.5% vs. 1.4%, p = 0.02), as well as lower initial in-hospital vital signs. After propensity score adjustment, the odds of 24-h mortality remained significantly higher for the ketamine-exposed group (OR 2.358, p = 0.042). Hospital discharge disposition was not different between groups in any analysis.
In this retrospective analysis, ketamine administration for pre-hospital airway management in TBI patients was associated with an increased 24-h mortality and lower in-hospital SBP, even after adjusting for baseline differences. However, injury severity and the length of time examined for mortality may explain the significant mortality association for ketamine in this study. Prospective studies are needed to examine the relationship between ketamine administration and mortality following TBI.
Introduction
Ketamine is an anesthetic drug which is administered in trauma situations because of its perceived preservation of respiratory and hemodynamic responses. 1 Compared with other anesthetic medications such as propofol or etomidate, which are gamma-aminobutyric acid receptor agonists, ketamine is an N-methyl-D-aspartate receptor antagonist, and its use is typically associated with higher blood pressure and respiratory rates (RRs). 1 However, ketamine directly suppresses myocardium, which can decrease cardiac output in critically ill patients, 2 and concerns about ketamine’s effects on intracranial physiology, including intracranial pressure and cerebral metabolic rate, remain. 3
Prior studies evaluating medication administration in trauma noted an increasing utilization of ketamine following traumatic brain injury (TBI). 4 However, safety and outcomes data for ketamine administration are lacking. Only one prior study evaluated survival and morbidity associated with ketamine administration following TBI, which observed no difference in survival for ketamine-exposed subjects, but did note a ketamine-associated increased rate of seizure activity and a subgroup with increased cardiac events. 5 Additional evaluation of ketamine’s safety and physiological effects when administered for TBI are needed.
The Linking Investigations in Trauma and Emergency Services (LITES) registry presented an ideal opportunity to further investigate the effects of ketamine on survival, recovery, and physiological responses in TBI in a large cohort of subjects. 6 We identified subjects with airways placed following TBI along with the medications administered, allowing a comparison of ketamine versus other induction drugs that were used. We hypothesized that ketamine administration to TBI subjects for airway placement would not be associated with differences in survival, vital signs, or disposition outcomes compared with other induction medications.
Methods
This study utilized pre-hospital, in-hospital, and Trauma Quality Improvement Program (TQIP) data collected quarterly from eight sites, including Level 1 trauma centers across the United States over the period of 5 years (2017–2021). The data were pooled into a relational database registry for the Task Order 1 (TO1) project of the Department of Defense-funded, LITES clinical trials network.
The LITES TO1 dataset was used to identify TBI subjects with an abbreviated injury scale score > 2, and with pre-hospital intubation and medication administration data available. Subjects were stratified into two groups: those who only received ketamine for intubation (ketamine-exposed), and those who received other induction medications (propofol and/or etomidate; ketamine-unexposed), as well as sedative medications including midazolam and lorazepam. Demographic variables included: age, sex, race (White, Black, other), ethnicity (Hispanic vs. non-Hispanic), as well as initial vital sign measurements collected in the pre-hospital setting: systolic blood pressure (SBP), RR, heart rate (HR), and Glasgow Coma Scale (GCS). Outcome variables available for analysis include: 24-h mortality, initial in-hospital vital signs (SBP, RR, HR, and GCS), and hospital discharge disposition (home without health services, left against medical advice, home with health services, short-term general hospital or inpatient rehabilitation, long-term general hospital or inpatient rehabilitation, deceased, or other disposition including jail or mental health facility).
Descriptive statistics for baseline and pre-hospital clinical characteristics were summarized by group (ketamine-exposed vs. ketamine-unexposed) as frequencies and percentages for categorical data or as the mean ± standard deviation or median and interquartile range for normally or non-normally distributed continuous data, as appropriate. Examination of the normal distribution assumption for continuous data was determined using histograms. Chi-square or Fisher’s exact test, as appropriate, was used to compare the frequency distribution of the categorical variables between the two groups. Two-sample t-test or Wilcoxon rank sum test was used to determine the differences in baseline and clinical characteristics (e.g., initial pre-hospital vital sign) between the two groups for normally or non-normally distributed continuous data, respectively. Hospital discharge disposition was defined as a dichotomous variable categorized as favorable versus unfavorable for analysis purposes, with favorable involving: home without health services, left against medical advice, home with health services, and short-term general hospital or inpatient rehabilitation; unfavorable disposition involved: long-term general hospital or inpatient rehabilitation, deceased, and other (jail, mental health).
An unadjusted logistic regression model was applied to assess the strength of the association between groups and the dichotomous outcome of interest (e.g., 24-h mortality). Unadjusted ordinal logistic regression was used to assess the association between group and ordinal outcome (i.e., initial in-hospital GCS). Unadjusted linear regression was used to assess the relationship between group and continuous outcome (e.g., initial in-hospital vital signs). For adjusted analysis, a propensity score analysis was used to reduce the potential impact of confounding and selection bias effects. The propensity score is the probability of group assignment conditional on observed baseline characteristics. We estimated the propensity scores using the generalized boosted models approach. We used the Toolkit for Weighting and Analysis of Nonequivalent Groups (twang) software package and SAS macros to estimate and evaluate the propensity scores.7,8 All baseline and pre-hospital clinical characteristics were evaluated to be included in the propensity score model. Based on statistical analysis, characteristics that revealed imbalance between exposure groups and were associated with the outcome of interest were included in the propensity adjustment; in addition, characteristics that were associated to the outcome but unrelated to exposure were also included in the propensity score adjustment model.9,10 Race, injury type, pre-hospital GCS, initial pre-hospital SBP, and site were included in the propensity score adjustment; although there was a difference in age between groups, it was not associated with imbalance in outcomes and was therefore not included in the propensity score adjustment model. A propensity score weighted logistic, ordinal logistic, or linear regression model was used to estimate the association between group and outcome after controlling for potential confounders by including propensity score as weight (inverse probability of treatment weighting). Results were summarized as odds ratio (OR) or regression coefficient and the corresponding 95% confidence interval. All statistical tests were two-sided, and the significance level was set to p = 0.05. All analyses were conducted using SAS statistical software (version 9.4, SAS Institute, Cary, NC).
Results
An initial query of the LITES TO1 database for TBI identified 23,770 TBI subjects. We excluded 558 subjects with no prehospital airways (intubation), 18,560 subjects who had missing medication documentation, and an additional 2,999 subjects were excluded for not receiving any intubation-related medications. Of the remaining 1,653 intubated subjects, 231 were excluded due to receiving both ketamine and another intubation-related medication, leaving 1,422 TBI subjects, of which 429 were identified to have received ketamine only following TBI, and 993 subjects received other medications for intubation (Fig. 1).

STROBE flow diagram showing subject consideration, exclusion and inclusion from the LITES Task Order 1 dataset. LITES, Linking Investigations in Trauma and Emergency Services.
Baseline and injury characteristics are summarized in Table 1. Ketamine-exposed subjects were younger than unexposed subjects (45.9 ± 19.8 years vs. 50.4 ± 20.6 years, p < 0.01). Sex and racial/ethnic distribution, initial field RR, initial field SpO2, and injury type were similar between groups. Comparison of initial pre-hospital vital signs measurements demonstrated that ketamine-exposed subjects had a lower GCS (5 [3–10] vs. 6 [3–12]), lower initial field SBP (126.5 ± 36.1 mmHg vs. 144.0 ± 37.8 mmHg), and higher initial field HR (102.5 ± 29.9 beats/min vs. 96.3 ± 28.0 beats/min) compared with ketamine-unexposed subjects. Site location demonstrated a significant difference for ketamine administration rates (p < 0.01) (Table 1).
Demographic and Pre-Hospital Clinical Characteristics by Ketamine Group

GCS, Glasgow Coma Scale; HR, heart rate; RR, respiratory rate; SBP, systolic blood pressure; SD, standard deviation; SpO2, oxygen saturation.
Descriptive statistics for primary and secondary outcomes are summarized in Table 2. For 24-h mortality, ketamine-exposed subjects had a statistically significantly higher mortality rate than ketamine-unexposed subjects (3.5% vs. 1.4%, p = 0.02). In addition, the ketamine-exposed subjects had lower initial in-hospital SBP (119.6 ± 41.1 mm Hg vs. 134.5 ± 30.5 mmHg, p < 0.01), lower initial in-hospital RR (16.3 ± 8.3 breaths/min vs. 18.1 ± 7.8 breaths/min, p < 0.01), lower initial in-hospital SpO2 (96.9 ± 9.8% vs. 97.6 ± 6.9%, p < 0.01) and lower initial in-hospital GCS (3 [3–3] vs 3 [3–7], p < 0.01) compared with ketamine-unexposed subjects. There was no difference between ketamine-exposed and ketamine-unexposed subjects in regard to initial in-hospital HR and hospital discharge disposition.
Descriptive Statistics for Primary and Secondary Outcomes by Ketamine Group
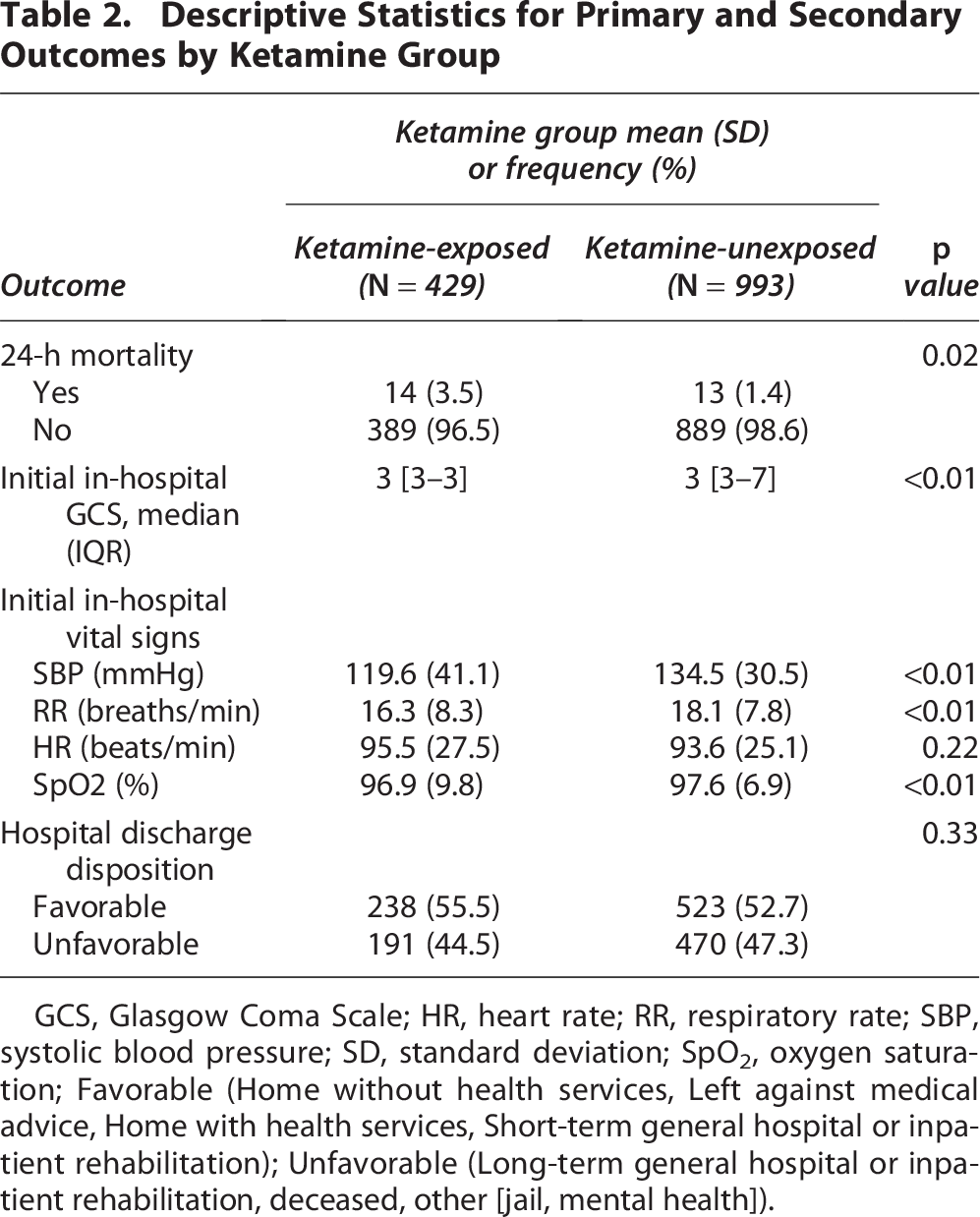
GCS, Glasgow Coma Scale; HR, heart rate; RR, respiratory rate; SBP, systolic blood pressure; SD, standard deviation; SpO2, oxygen saturation; Favorable (Home without health services, Left against medical advice, Home with health services, Short-term general hospital or inpatient rehabilitation); Unfavorable (Long-term general hospital or inpatient rehabilitation, deceased, other [jail, mental health]).
Table 3 summarizes the association between ketamine exposure and outcomes after propensity score adjustment for race, injury type, pre-hospital GCS, initial pre-hospital SBP, and site. The adjusted estimated odds of 24-mortality were 2.358 (95% CI [1.033, 5.382], p = 0.042) times higher for the ketamine-exposed group when compared with ketamine-unexposed group. For initial in-hospital vital signs, there was a significantly lower mean SBP (−13.76 mmHg, 95% CI [−18.68, −8.84], p < 0.001) for the ketamine-exposed compared with unexposed group after propensity score adjustment. Initial in-hospital GCS, initial in-hospital RR, HR, and SpO2, and hospital discharge disposition were all similar between groups following propensity score adjustment.
Unadjusted and Adjusted Associations of Ketamine-Exposed Versus Ketamine-Unexposed with Primary and Secondary Outcomes

aKetamine-unexposed group is the reference category.
bOrdinal logistic regression, GCS = 15 is the reference category.
cPropensity score adjusted analyses included race, injury type, pre-hospital GCS, initial pre-hospital SBP and site.
CI, confidence interval; GCS, Glasgow Coma Scale; HR, heart rate; RR, respiratory rate; SBP, systolic blood pressure; SpO2, oxygen saturation.
Discussion
From analysis of the LITES TO1 registry, we identified 1,422 TBI subjects with known induction medications and outcome variables recorded, and 30% (429/1,422) of these subjects received ketamine as a sole medication for induction. Population data for ketamine administration following TBI isn’t known, but a prior North American study identified roughly 16% of TBI subjects received ketamine, 5 and therefore, this retrospective analysis of the LITES cohort data demonstrates an increased incidence of ketamine administration following TBI.
The 24-h mortality rate for TBI subjects receiving ketamine was significantly higher than for subjects who received other medications (3.5% mortality for ketamine-exposed vs. 1.4% for ketamine-unexposed). Ketamine-exposed subjects had a lower initial pre-hospital GCS, lower initial pre-hospital SBP and higher initial pre-hospital HR, but even after controlling for these factors with propensity score adjustment, mortality rate was higher with ketamine exposure (mortality OR 2.358 compared with other medications). This is only the second study which has evaluated mortality in TBI subjects exposed to ketamine, and the first which has identified a mortality association. 5 The previous retrospective study of ketamine exposure and outcomes following TBI also had a worse injury profile in the ketamine-exposed subjects (lower initial GCS and higher likelihood of intubation), demonstrating a trend of ketamine being administered to more unstable trauma subjects. 5
Ketamine-exposed subjects in our current study had an initial pre-hospital GCS of 5, whereas the prior ketamine TBI outcomes study had an initial pre-hospital GCS of 7, and involved subjects randomized into a drug study that might have excluded subjects likely to die within 24 h. Moreover, our average age for ketamine exposure was 46 years old (vs. 50 years old in ketamine-unexposed); the prior study had an average age of 37 years old for ketamine exposure (vs. 42 years old in ketamine-unexposed). The older age and worse GCS of ketamine exposed subjects in the current study may represent an indication bias and explain some of the differences in survival as older age and poor GCS are associated with overall worse outcomes following TBI. 11 Furthermore, the prior study evaluated mortality over a 6-month period; it’s possible that a longer window of mortality evaluation beyond 24 h might result in more mortality in the ketamine-unexposed group. Importantly, although the overall mortality rate was higher with ketamine exposure in our current study, there was no difference in discharge disposition between groups, suggesting that in subjects who survived for longer than 24 h, ketamine exposure did not confer additional morbidity.
Ketamine-exposed subjects arrived at the hospital with a significantly lower SBP and RR compared with ketamine-unexposed subjects. Ketamine is known to preserve or increase blood pressure and RR 12 ; however in our study population, the ketamine-exposed subjects started with a lower initial pre-hospital SBP which likely resulted in the lower initial SBP on hospital presentation. Notably, although ketamine-exposed subjects had lower SBP compared with unexposed subjects, their blood pressure was within the normal range for blood pressure. Furthermore, although ketamine is known to preserve RR better than other intubation medications like propofol, 13 ketamine can still depress overall respiratory drive, which was observed even in the original ketamine studies. 14
Limitations
As this was a retrospective, observational study, no causality can be determined from the administration of ketamine, only statistical associations. Furthermore, the decision to administer ketamine versus another drug was made by pre-hospital medics in trauma situations, which may be impacted by an indication bias to select more hemodynamically unstable subjects for ketamine, as well as the possibility that retrospective records filled out at a later time may be incorrect. Additional unaccounted for confounders may also bias the current results. As data for this study were provided by centers throughout the United States, there may be significant practice or injury variability between regions or sites, which could have influenced ketamine administration, as indeed there was an overall difference in ketamine administration between sites. Lastly, the specific timing and dosage of drugs administered were not available for analysis, and therefore it was not known if medications were administered prior to or after collection of the initial pre-hospital vital signs and GCS. Future prospective studies should evaluate timing and dosages of induction medications relative to vital signs changes if possible.
Conclusion
In this retrospective analysis of subjects who received induction medications for the placement of airways following TBI, ketamine was administered to younger, more hemodynamically unstable subjects, and was associated with increased 24-h mortality compared with ketamine-unexposed subjects. Subject age, injury severity, and the length of time examined for mortality may explain the significant mortality association for ketamine in this study. These findings represent an important cautionary signal for ketamine use in TBI and warrant further examination of ketamine use with prospective studies.
Transparency, Rigor, and Reproducibility Statement
The dataset collected and analyzed during the current study was obtained from the TQIP registry for the TO1 project of the Department of Defense-funded LITES initiative. All study methods are described in detail in the Methods section. The STROBE Flow Diagram in Figure 1 details subject inclusion and exclusion. All statistical analyses were performed using SAS statistical software (version 9.4, SAS Institute, Cary, NC). Investigators responsible for outcomes interpretation were blinded to data collection and statistical analysis.
Authors’ Contributions
A.P.: Conceptualization, writing, analysis. R.M.: Writing. S.R.W.: Conceptualization, writing, analysis. E.V.Z: Statistical analysis. B.A.C.: Conceptualization. J.W.C.: Conceptualization. M.A.S.: Conceptualization. E.E.M.: Conceptualization. N.M.: Conceptualization. J.P.M.: Conceptualization. J.L.S.: Conceptualization, writing. B.L.R-R.: Statistical analysis. F.X.G.: Conceptualization, analysis, writing.
Footnotes
Author Disclosure Statement
The authors have no conflicts of interest to disclose.
Funding Information
This research is supported by Department of Defense Contract No. W81XWH-16-D-0024, (Task Order W81XWH-16-D-0024-0001).
