Abstract
Blast-induced traumatic brain injury (bTBI) is a significant health concern for military personnel, causing potential long-term neurological consequences. Given the unique and heterogeneous genetic makeup of the human population, the acute responses and evolving sequelae from blast exposure are difficult to predict. To address these challenges, murine models are invaluable in the study of bTBI, as they allow researchers to carefully control exposure parameters while examining physiological changes across genetic, cellular, and whole-organism levels over time. In this study, we investigated the role of genetic diversity in bTBI by examining six common mouse strains—A/J, 129S1/SvImJ, NOD/ShiLtJ, NZO/HILtJ, C57BL/6J, and CAST/EiJ—and their possible differential response to blast overpressure. We assessed immediate neurological impairment, respiratory symptoms, lethality thresholds, and gross pathological and histological changes following blast overpressure exposure across these strains. We observed significant strain-dependent differences across all the measured outcomes. In particular, C57BL/6J mice exhibited the longest normalized righting times and highest incidence of subdural hematomas. Notably, the strains used as models of type I/II diabetes (NZO/HILtJ and NOD/ShiLtJ) showed the highest resilience to blast-induced lethality. In comparison, the CAST/EiJ strain was the most susceptible to immediate apnea and had the lowest lethality threshold. The NZO/HILtJ mice showed the highest incidence of pulmonary bleeding. Our findings highlight the substantial influence of the genetic background on the bTBI outcomes in mice, even with highly controlled physical exposure conditions. This comprehensive characterization of strain-dependent responses to bTBI provides a foundation for investigating the genetic influences on blast injury outcomes and developing more targeted preventative and/or therapeutic strategies for bTBI.
Introduction
Traumatic brain injury (TBI), particularly blast-induced TBI (bTBI), has emerged as a significant health concern for military personnel. The prevalence of TBI among U.S. service members deployed to recent conflict zones has been estimated at 15–23%, with blast exposure being a primary cause. 1 These injuries can lead to long-term neurological deficits, cognitive impairment, and psychological disorders, significantly impacting the affected individuals’ operational readiness and quality of life. TBI is a heterogeneous condition with many different presentations and outcomes in patients, suggesting that factors beyond the primary injury mechanism(s) or severity affect recovery. 2 For instance, the genetic diversity in the human population may play a significant role in individual responses to TBI, including variations in susceptibility, severity of symptoms, and recovery outcomes. Clinical evidence supports the role of genetic factors in blast-related outcomes. Taylor et al. demonstrate that blast exposure interacts with the 5HTTLPR genetic variant to predict posttraumatic stress symptoms independently of the TBI diagnosis. 84 These clinical findings highlight the critical need for preclinical models that incorporate genetic diversity to better recapitulate the heterogeneity observed in human blast injury outcomes. 3
Preclinical models of bTBI are useful due to their homogeneity, control of experimental variables, and reproducibility, and there is no doubt that there is an emergent need for high-quality preclinical models.4–7 However, the integration of genetic diversity enables the more robust identification of genetic risk factors, gene–environment interactions, and pharmacogenomic effects, which will potentially improve the translational value of preclinical studies. The majority of research uses C57BL/6 mice, which show behavioral and histopathological changes in response to blast exposure. While the importance of genetic predisposition to disease and injury has been stated, most work focuses on how alterations in a specific gene can affect the response to blast. For example, Tran et al. used APOE3 and APOE4 transgenic mouse models to investigate the role of the APOE genotype in blast-induced TBI outcomes, demonstrating gene-specific differences in neuroinflammation and behavior.7,8 Additionally, Alzheimer’s disease-related transgenic mice were used to evaluate the amyloid burden and tau-influenced dysregulation of learning pathways following bTBI.9–11 However, these targeted transgenic studies do not directly mimic broad, genomic-level diversity.
In 2004, eight genetic strains, known as founder strains, were used to develop an inbred mouse population known as the Collaborative Cross (CC). 12 The eight founder strains—A/J, 129S1/SvlmJ, NOD/LtJ, NZO/HILtJ, C57BL/6J, CAst/EiJ, PWK/PhJ, WSB/EiJ—encompass 90% of the genetic diversity of the commonly used laboratory mice and three wild-derived strains. 13 While each of the chosen strains does not represent a well-known human condition, the genome of each strain is fully sequenced, with 45 million segregated single-nucleotide polymorphisms (SNPs).14,15 As a result of intercrossing the CC, two additional multiparental populations (MPPs) were derived—the F1 hybrids of the CC (CC-RIX) and the Diversity Outbred (DO) populations.16,17 These populations provide an even broader spectrum of genetic variation that closely resembles human genetic diversity.14,18
Both the CC and DO murine populations represent the genetic diversity of a whole population and thus provide a method to link phenotypes to potential causal genetic factors. For example, crossing the founder strains led to the identification of gene loci associated with specific neurobehavioral phenotypes. 19 Likewise, collaborative crossing in disease models uncovered additional gene loci that either relate to disease risk, poor outcome, or other key attributes of disease progression.15,20–23 However, none of these collaborative crossing designs have been used in preclinical TBI models, nor correlated with the responses and outcomes to injury. Rather, past work is limited to studying some specific pairings of strains—one study compared the widely used strains C57BL/6J and BALB/cByJ mice with the Jackson Diversity Outbred (J:DO) murine population in a bTBI study, 9 while a second study utilized NOD/LtJ mice to examine adenosine monophosphate (AMP)-activated protein kinase (AMPK) and lymphoid cells following TBI. 24 Murray et al. utilize all the eight founder strains and examined hippocampal gene expression changes following blast-induced TBI. The findings support the influence of the genetic background on transcriptional regulation in response to bTBI. 25 Thus, while past work has addressed targeted genetic questions, including initial transcriptomic profiling across the founder strains, the field lacks a comprehensive foundation for comparing the acute response to blast exposure based on genetic diversity and phenotype, which could be a critical first step toward identifying potential risk factors in blast exposure.
In the current study, we used six of the founder strains of the CC (Table 1) to systematically investigate the role of genetic diversity in lethal thresholds, acute neurological impairments, and gross neuropathological and pulmonary damage following blast overpressure exposure. The C57BL/6J strain serves as a widely used control strain in neuroscience research, providing a standard against which other strains can be compared.25,36 The A/J strain, known for its susceptibility to lung diseases,25,36 was included to investigate the potential interplay between pulmonary vulnerability and blast-induced brain injury.39–41 The 129S1/SvlmJ strain offers a well-characterized genetic profile.26,42 The NOD/ShiLtJ and NZO/HILtJ strains were chosen for their utility in studying diabetes,30,34 as clinical studies have shown that diabetes is a risk factor for poor outcomes following TBI. 35 The wild-derived CAST/EiJ strain introduces genetic diversity that may more closely reflect the natural variations seen in human populations. 43 This strain is commonly used to study the long-term effects of viral illnesses, including influenza, West Nile virus, and COVID-19.44–47 Furthermore, as these selected strains collectively represent laboratory mouse genetic diversity and wild-derived genotypes, the findings among these strains can be linked to specific gene loci and haplotypes, rather than generalized human conditions.
Characterization of Strains

Comparison of general, whole body, and central nervous system (CNS) phenotypes, behavioral considerations, and research applications across the six genetic strains.
By embracing genetic diversity in our preclinical model, we aim to provide a more nuanced understanding of blast-induced injuries and their genetic underpinnings. This approach has the potential to inform the development of personalized prevention strategies and treatments for blast-exposed individuals in military settings. Moreover, by correlating our findings with known clinical risk factors, we strive to bridge the gap between animal studies and human applications, ultimately contributing to improved care for those affected by blast-induced TBI.
Methods
Animals
In this study, we enrolled 8–10-week-old (w/o) male mice of each genetic strain (A/J, 129S1/SvlmJ, NOD/ShiLtJ, NZO/HILtJ, C57BL/6J, CAST/EiJ) (The Jackson Laboratory, Bar Harbor, ME, USA). All experimental procedures were approved by the Institutional Animal Care and Use Committees (IACUCs) at both the CMC Veterans Affairs Medical Center and the University of Pennsylvania. We carefully adhered to the guidelines set forth in the Public Health Service (PHS) Policy on Humane Care and Use of Laboratory Animals to ensure animal welfare.
Blast tube configuration and pressure transducer measurements
We adapted the shock tube design from Gullotti et al. 48 and oriented the aluminum tube in a vertical position. 49 To initiate a blast peak overpressure wave, we supplied helium to the load chamber, where it burst through single-use polyethylene terephthalate (PET) membrane(s) of a precalculated thickness (Fig. 1). Pressures measured at the tube exit were used to quantify the blast overpressure exposure, as peak overpressure is linearly correlated with the membrane thickness (Fig. 1).
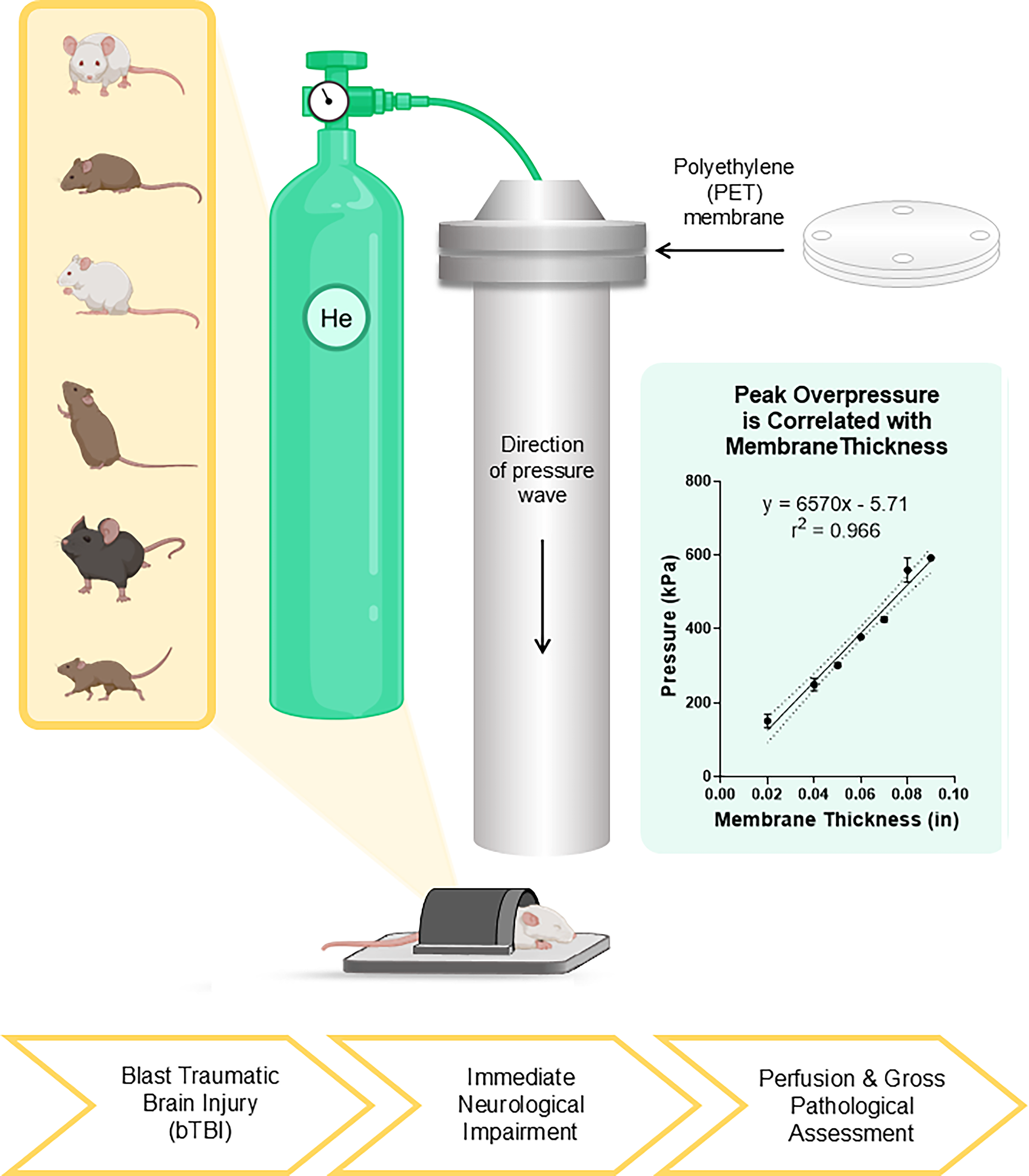
Experimental schematic of bTBI. Blast tube apparatus representative of the peak overpressure wave generated by helium gas bursting through a PET membrane. Following bTBI, immediate neurological impairment assessment (apnea, dyspnea, righting time), gross pathological assessment, perfusion, and further histological analyses. Created in BioRender. Georges, A. (2025) https://BioRender.com/bk34l1p. bTBI, blast-induced traumatic brain injury; PET, polyethylene.
In designing our study, we considered various options for investigating the effects of blast overpressure, such as blast tube orientation (vertical, horizontal, or angled), animal placement (inside or outside the blast tube), and distance from the tube exit.6,48,50 We chose a vertical orientation and fixed animal position at the tube exit to minimize head motion acceleration. 48 We also used a sorbothane-lined aluminum encasing to reduce lung damage and severe organ-wide injury. Our priority was to maintain consistent experimental parameters to preserve the physical characteristics of the loading direction and waveform shape.50,51
We measured the peak overpressure using a pressure transducer (model 8530B-200; Endevco, Irvine, CA, USA) positioned at the exit of the blast tube. To reduce noise in the signal, we used an inline filter conditioning device (linear phase filter, 10 kHz cutoff frequency; Alligator Technologies, Charlottesville, VA, USA). At least two test trials were used to calibrate the blast overpressure level caused by rupturing PET membranes of specified thickness. In each test, pressure transients were recorded using a digital oscilloscope (Tektronix, Beaverton, OR, USA). From each pressure recording at the tube exit, we computed the average and standard deviations for peak overpressure, impulse, and duration of the pressure transient (Table 2, Supplementary Fig. S1).
Pressure, Impulse, and Duration of Peak Overpressure Wave

aNo SD measurements for 0.09 in membrane thickness because only one pressure transducer measurement was obtained.
Polyethylene terephthalate (PET) membrane thickness, peak overpressure measurement, duration, and impulse as calculated from the pressure transducer measurements.
Data are shown as mean ± SD, n = 3.
Additional measurements and graphs shown in Supplementary Figure S1.
Blast injury
We weighed each animal prior to exposure of a mixed inhalation anesthetic of 3.5% isoflurane and 60% FiO2 in an anesthetic induction chamber for 5 min. We confirmed anesthetic depth using a toe pinch. We then transferred the animal to the blast tube apparatus and placed it on the platform in the prone position. After securing the nose within the nose cone and encasing the torso in the sorbothane-lined aluminum encasing, we fired the blast tube, exposing the animal to a shockwave of predetermined pressure wave intensity. The peak overpressure ranged from 224 to 487 kPa. For sham conditions, we exposed the animal to identical anesthetic conditions, placed it in the blast tube apparatus for approximately the same period as pressure-exposed animals, and then placed it in the recovery cage.
Immediate neurological impairment
Immediately following injury, we carefully removed each animal from the blast apparatus and placed it in the original cage in the supine position. We closely observed each animal until the time of death or time of the end-point at 30 min post-injury. We measured immediate apnea as the time from the animal was placed in the supine position to the time the animal resumed breathing. To measure the righting reflex, or the righting time, we recorded the time the animal took to turn from the supine to prone position. If the animal resumed breathing post-injury but became apneic within the 30-min observation period, we recorded this finding as secondary apnea. We also recorded the presence and duration of dyspnea, or irregular breathing. To compare the righting times across the strains, we normalized the righting time of each injured animal with the average righting time for the sham animals within the respective strain. We compared the resulting measure, normalized righting time, across the strains.
Lethal-dose 50
To determine the thresholds for lethality, we exposed six animals of each strain to a blast peak overpressure beginning at 224 kPa. If >50% of animals survived in a given group, we incrementally increased the subsequent cohorts until lethality was observed within 30 min post-injury. We monitored the animals for 30 min post-injury and defined the parameters for lethality post-injury as death occurring within this window. We scored survival as a binary outcome (0 = survived 30 min post-injury; 1 = died within 30 min post-injury). Lethal-dose 50 (LD50) values were calculated using logistic regression based on the relationship between the peak overpressure and lethality rate for each strain.
Perfusion
At the time of death or 30 min post-bTBI, we gave each animal a lethal dose of pentobarbital sodium injection, USP, 250 mg/kg (Sagent Pharmaceuticals, Schaumburg, IL, USA) intraperitoneally (IP). When the animal was unresponsive to a toe pinch, we transcardially perfused it with 10 mL of ice-cold phosphate buffered saline and 25 mL of 10% neutral buffered formalin solution (Sigma-Aldrich, St. Louis, MO, USA) using a peristaltic pump (Avantor, Radnor, PA, USA) at a rate of 6 mL/min.
Gross pathology
Following perfusion, we dissected and examined the relevant organs for gross pathological changes. We grossly photographed the brain and lungs from each animal and scored pathologically on a scale using three categorical rankings. For the brain tissue rating scale, we defined: 0—brain tissue appeared healthy, with no visible gross pathology, bleeding, or damage; 1—bleeding at the brainstem, spinal cord, and/or cerebellum; and 2—subdural hematoma. For the lung tissue rating scale, we rated: 0—lung tissue appeared healthy, with no visible gross pathology, bleeding, or damage; 1—some signs of erythema or bleeding on the lungs, visible, healthy lung tissue; and 2—no visible healthy lung tissue, significant erythema, and bleeding into the chest cavity. We post-fixed all organs in 10% formalin for 24 h and recorded the weight of each brain.
Histology
We sectioned 8-μm-thick coronal sections between bregma 0 and 1.5 mm for each animal using a rotary microtome (Thermo Shandon Finesse 325 Rotary Microtome, Thermo Fisher, Waltham, MA, USA). We used hematoxylin and eosin staining to assess ventriculomegaly and abnormal neuropathology. To measure the ventricle size for each specimen, we captured a 4× image on a microscope system (Aperio ScanScope CS2, Leica Biosystems, Deer Park, IL, USA). After saving the images, we encircled each animal’s left and right ventricles and measured their respective areas using ImageJ software. 52
Statistical analysis
We performed all data analyses and created all graphs using Prism (GraphPad Software, Boston, MA, USA). We normalized the righting time outcomes at a consistent peak overpressure level (224 kPa) with respect to the group sham mean values. We analyzed the data presented in bar graphs nonparametrically using the Kruskal–Wallis test, followed by the post hoc test with Dunn’s multiple comparisons as a result of a significant Shapiro–Wilk test for normality and a Brown–Forsythe test for unequal variance. We fit simple logistic regressions to determine the thresholds for the delayed righting time, immediate apnea, and lethality. From each logistic regression curve, we plotted and analyzed 95% confidence intervals (CIs) of the 50% risk level using an ordinary one-way ANOVA with uncorrected Fisher’s least significant difference. All the error bars and ±values reported denote standard deviation. We determined significance by p ≤ 0.05 and denoted significance as follows: ns p > 0.05, *p ≤ 0.05, **p ≤ 0.01, ***p ≤ 0.001, ****p ≤ 0.0001.
Results
Body weight and brain weight varied across genetic strains
The six genetic strains had unique physical appearances. Additionally, body weight differed significantly across strains (Kruskal–Wallis test, p < 0.0001). Over the same age ranges (8–10 weeks old), the NZO/HILtJ strain had significantly higher body weights than all the other strains (44.3 ± 4.4 g). CAST/EiJs had lower body weights (15.6 ± 1.2 g) (Fig. 2A,B). Across all strains, the brain weights varied slightly, but not significantly (Kruskal–Wallis test, p < 0.0001) (Fig. 2C,D). We next looked at the ratio of the brain weight to body weight across the strains (Kruskal–Wallis test, p < 0.0001). The NZO/HILtJ strain had the lowest brain/body weight ratio (0.011 ± 0.00096 g/g), while the CAST/EiJ strain had the highest brain/body weight ratio (0.025 ± 0.0021 g/g) (Fig. 2E,F).

Six genetic strains, body mass, and brain mass. The six genetically diverse mouse strains and their profile images from the Jackson Laboratory.
53
Righting reflex is strain-dependent and significantly different between sham and bTBI conditions
To assess immediate neurological impairment, we measured the righting reflex, or the righting time, of each mouse following exposure to a peak overpressure wave. The righting reflex has been used in prior work as an indicator of immediate neurological impairment. 54 We observed differences in the righting reflex in animals exposed to sham conditions across the strains. For this reason, we compared the normalized righting times across the strains exposed to the same blast pressure pulse (224 kPa peak, 22.4 kPa*msec impulse, 0.651-msec duration). The righting times, normalized to the mean sham righting time of each respective strain, varied significantly across the strains (Kruskal–Wallis test, p < 0.0001). When ranked by the duration of the normalized righting times, the post hoc analysis showed that the C57BL/6J strain (7.73 ± 4.88) had significantly greater normalized righting times than most other strains (NZO/HILtJ, 2.52 ± 1.77, p = 0.0099; NOD/ShiLtJ, 3.44 ± 2.07, p = 0.0052; 129S1/SvlmJ, 2.92 ± 1.70, p = 0.0015; CAst/EiJ, 1.64 ± 1.04, p = 0.0003) (Fig. 3A,B).

Immediate neurological impairment following bTBI.
We next considered how the immediate neurological impairment varied with the blast overpressure level. Logistic regression analysis on animals exposed to a range of overpressure conditions showed that the genetic strain was a significant factor in the range of the righting times observed (ANOVA, p < 0.0001). Post hoc comparison across strains revealed two distinct, nonoverlapping groups in normalized righting times. Group 1 consisted of NZO/HILtJ (298 ± 179), NOD/ShiLtJ (279 ± 152), and C57BL/6J (267 ± 158), which required higher peak overpressures to induce prolonged righting times. Group 2 consisted of A/J (161 ± 186), 129S1/SvlmJ (159 ± 176), and CAst/EiJ (82.3 ± 185), which had lower thresholds for prolonged righting times. Both groups were significantly different from sham exposure levels (Fig. 3C,D).
Strain-dependent differences in respiratory symptoms occurred over a range of blast overpressure exposures
To explore the relationship between blast exposure and pulmonary damage, we considered the acute (<30 min) response to blast overpressure. For all overpressure levels (224–487 kPa), most strains showed normal respirations and respiratory rates in the majority of cases (>50%). The exceptions being the A/J and CAST/EiJ strains, showing respiratory impairments post-injury in 51.4% and 52.2% of cases, respectively. The A/J strain had the highest percentage of cases with dyspnea (17.1%), and the NOD/ShiLtJ had the lowest (3.5%). The CAST/EiJs had the most frequent instances of immediate apnea (47.8%) but no instances of delayed apnea. The percentage of cases with delayed apnea post-blast was minimal across the strains, but the most prevalent was in the 129S1/SvlmJ (7.02%) and NZO/HILtJ (4.08%) strains (Fig. 4A).

Respiratory symptoms following bTBI.
We considered if the overpressure threshold for immediate respiratory apnea following bTBI was different across the strains. After separating the response into either the presence (=1) or absence (=0) of apnea, we plotted the 95% CIs of each strain and determined that there was a significant relationship between the genetic background and apnea threshold (one-way ANOVA, p = 0.0018). The NOD/ShiLtJ (468 ± 210 kPa) and NZO/HILtJ (444 ± 498 kPa) strains have the highest thresholds of the peak overpressure level for immediate apnea, which were not different from one another (ns, p = 0.492) (Fig. 4B). Post hoc testing showed that the NOD/ShiLtJ and NZO/HILtJ strains also had significantly higher thresholds than the A/J strain (275 ± 132, p = 0.0262, p = 0.0065, respectively). The CAST/EiJ (207 ± 339 kPa) strain was significantly more susceptible to immediate apnea at lower blast levels than most of the other strains (NOD/ShiLtJ, p = 0.0009; NZO/HILtJ, p = 0.0002; 129S1/SvlmJ [395 ± 213], p = 0.0084; C57BL/6J [369 ± 376], p = 0.0109) (Fig. 4C,D).
Lethality thresholds were also strain-dependent
To determine whether the genotype was a contributing factor to the lethality of a particular strain, we established LD50 thresholds for each of the six strains of mice. Broadly, a genetic strain was a significant factor in the blast overpressure needed to achieve 50% likelihood of lethality. The LD50 values, ranked from most susceptible to least susceptible strain, were: CAST/EiJ (200 ± 245 kPa), A/J (310 ± 155 kPa), 129S1/SvlmJ (390 ± 154 kPa), C57BL/6J (429 ± 155 kPa), NZO/HILtJ (474 ± 347), and NOD/ShiLtJ (517 ± 272 kPa) (Fig. 5A). Post hoc comparisons of the 50% CIs of LD50 thresholds show that the NOD/ShiLtJ and NZO/HILtJ strains showed the highest resilience to injury, and these thresholds were not different from each other (ns, p = 0.8349). Moreover, the threshold for CAST/EiJs was significantly less than all the other strains (NOD/ShiLtJ, NZO/HILtJ, C57BL/6J, 129S1/SvImJ, p < 0.0001; A/J, p = 0.0054). The NOD/ShiLtJ thresholds were significantly higher than most of the other strains (C57BL/6J, p = 0.0297; 129S1/SvImJ, p = 0.066; A/J, p = 0.0003; CAST/EiJ, p < 0.0001). C57BL/6Js do not have significantly different lethality thresholds than the NZO/HILtJs (p = 0.0581), 129S1/SvImJs (p = 0.5043), or A/Js (p = 0.0693) (Fig. 5B,C).

Thresholds for lethality.
Gross brain and lung lesions after blast overpressure differed with genetic background
Given the difference in the lethality thresholds, we next examined if there were distinct patterns of primary blast and lung injuries across genetic strains. Using a semi-quantitative scoring method for the primary brain pathology appearing after blast exposure, we observed a considerable difference in the type and frequency across genetic strains. Subdural hematoma is one primary type of brain bleeding that appears after blast loading,55,56 and C57BL/6Js had the highest incidence of subdural brain hematoma (71.42%). In contrast, the NZO/HILtJs had no subdural bleeding (Fig. 5A). In both the A/Js and CAST/EiJs, more than half of the animals had some type of primary cerebral pathology in fatal cases. The NOD/ShiLtJ strain had the fewest cases of macroscopic cerebral bleeding (80% with no gross cerebral pathology) (Fig. 6A).
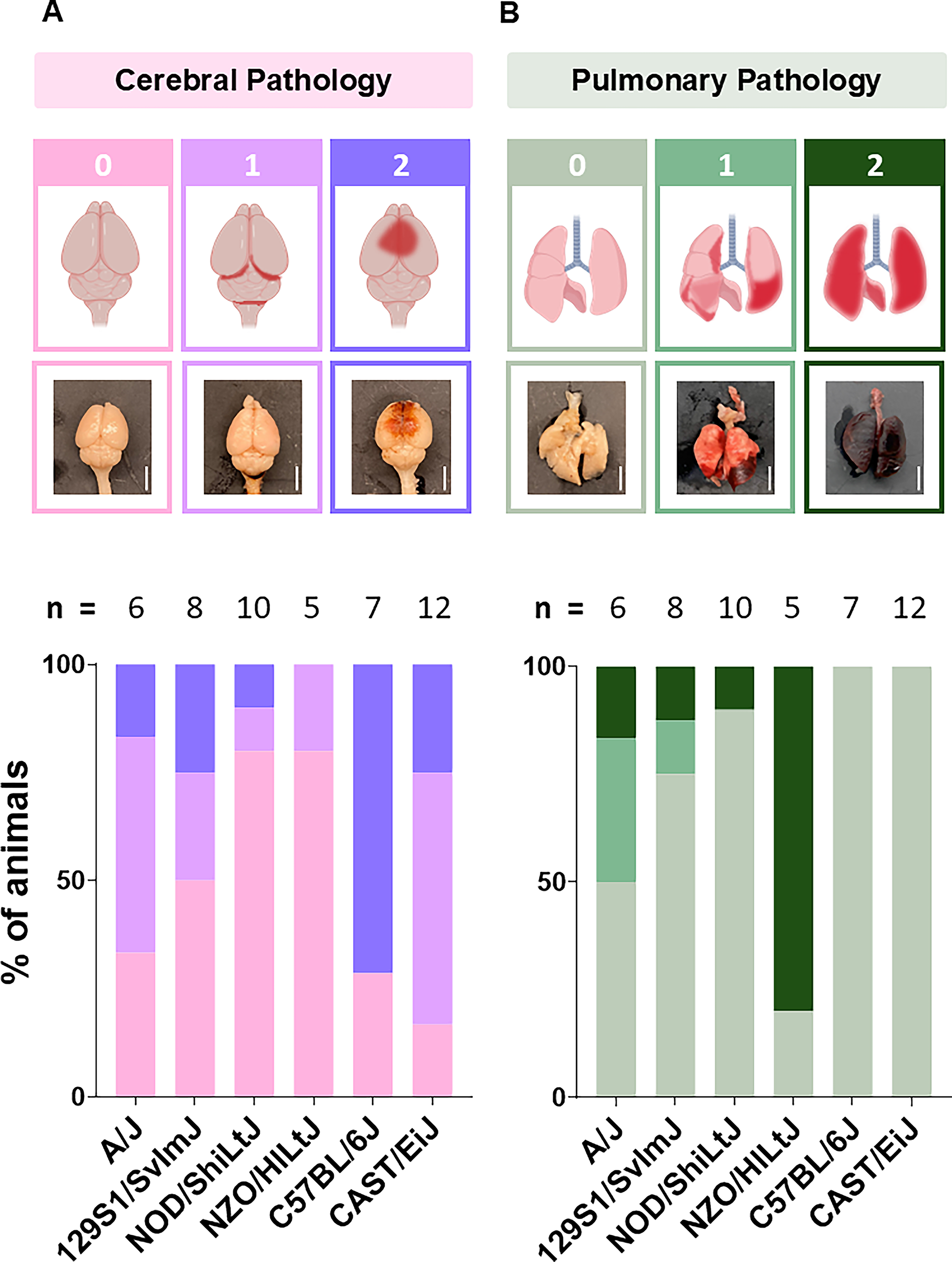
Gross cerebral and pulmonary pathology.
Similar to the primary brain pathology observations, the type and incidence of pulmonary changes varied considerably across the genetic strains. Macroscopic pulmonary bleeding was observed in 80% of the NZO/HILtJs following a lethal bTBI. In contrast, both the C57BL/6J and CAST/EiJ strains had no observable pulmonary damage. The results were mixed with the A/J and 129S1/SvlmJ stains, with ≥50% of fatal cases with no observable pulmonary damage and some cases in each group with mild and severe pulmonary damage (Fig. 6B).
Brain histology and cerebral bleeding varied across genetic strains following bTBI at respective LD50 levels
Across the strains and within individual strains, we observed significantly different patterns of primary cerebrovascular lesions (Fig. 7A). Subarachnoid hemorrhage (SAH) was the most common across all animals blasted at their respective LD50 levels, regardless of the genetic strain. Intraparenchymal hemorrhage (IPH) was the least common, observed only in a subset of animals; in the C57BL/6J strain, IPH was in the cerebrum and cerebellum; in the NZO/HILtJ strain, IPH was only in the cerebrum; in the A/J strain, we only observed IPH in the cerebellum. Intraventricular hemorrhage (IVH) and intracranial hemorrhage (ICH) had mixed occurrences. Notably, the A/J strain had the highest frequency of microscopic hemorrhage, as all animals had ICH and SAH in both the cerebrum and cerebellum, and all animals had IVH in the cerebrum (Fig. 7B,C). A preliminary examination of the ventricular size at LD50 levels showed that the strain (p = 0.0039), rather than blast exposure, was a more relevant factor to explain the changes in the ventricular size (Supplementary Fig. S2).

Histological cerebral pathology.
Discussion
Our study characterized the responses of six distinct murine genetic strains to bTBI, revealing significant strain-specific differences in the righting times, respiratory symptoms, lethality thresholds, and gross pathological changes. Broadly, these findings highlight the substantial role of the genetic background in the acute response and outcome to blast exposure. Among the most striking results were the unexpected resilience of the NOD/ShiLtJ (T1DM) and NZO/HlLtJ (T2DM) strains to blast injury, implying that brain metabolism homeostasis and immune system state may play a particularly important role in the acute effects of blast TBI.
We observed substantial strain-dependent differences in multiple outcome measures, suggesting the involvement of biophysical, genetic, or other factors in the response to blast injury. The C57BL/6J strain, serving as a baseline, had significantly longer righting times and the highest susceptibility to subdural hematomas. The A/J strain was the most susceptible to dyspnea and had a lower apnea threshold, while the 129S1/SvlmJ strain showed similar responses to C57BL/6J. The CAST/EiJ strain was the most susceptible to lethality at lower blast levels, possibly related to their smaller body size.
One potential explanation for these differences across the strains could be the physical characteristics, such as skull shape, brain size, and vasculature differences across the strains. For acceleration-induced injuries, there is a clear consensus that differences across the brain size and shape, and even their network structure, should be accounted for when considering injury risk.57–60 Although we observed changes in the brain size across the strains, the proposed scaling relationships for blast loading57,61 do not fully explain the differences in lethality that we observed across the strains. For example, the largest brain sizes did not show the lowest LD50 threshold, and the smallest sizes did not show the highest threshold. In addition, it is possible that other physical features may also affect the lethality thresholds or immediate neurological impairment. For example, computational models suggest that skull properties play a crucial role in the transmission of blast waves to the brain.51,52,60,62 Similarly, the skull shape exposed to the incident blast wave may also affect the reflection and transmission of the shock wave. The NZO/HlLtJ strain has a characteristically flat skull relative to other strains. 63 This flatter skull geometry may influence blast-wave reflection, potentially explaining the increased resilience to blast exposure observed in this strain. Both the physical properties and geometric shapes of the skull are not well-defined across these strains and would require a more precise characterization before determining the relative role that each plays in transmitting a blast wave into the brain parenchyma.
While we cannot rule out physical factors, genetic variations among the strains may also contribute to the observed differences in outcome. For example, the A/J strain’s susceptibility to lung diseases 25 could relate to their vulnerability to respiratory symptoms post-bTBI. Alternatively, past work shows that the Muc5b gene, associated with lung inflammation and fibrosis, is highly expressed in NZO/HlLtJ and minimally expressed in CAST/EiJ, 64 aligning with our gross pathology findings, which showed that primary lung damage was more apparent in the NZO/HlLtJ strain than other strains.
Perhaps the most intriguing observation from our dataset is the resilience of the NOD/ShiLtJ and NZO/HlLtJ strains to blast injury. On its own, the NOD/ShiLtJ mouse’s autoimmune predisposition makes it a worthwhile candidate for studying post-TBI autoimmunity given the substantial clinical evidence of autoantibody production after TBI. Clinical studies have documented diverse autoantibody responses following TBI, including antibodies against GFAP,65,66 S100B, 67 and neural antigens such as MAG. 68 The NOD/ShiLtJ mouse’s defects in central tolerance and abnormal T-cell responses mirror mechanisms implicated in human post-TBI autoimmunity, where the blood–brain barrier (BBB) disruption exposes normally sequestered neural antigens to the immune system.67,69 Both clinical data and animal models suggest that repeated TBI exposures lead to more pronounced autoimmune responses, 65 while CD8+ T cells appear to play a key pathogenic role in chronic neurodegeneration after TBI. 70 Indeed, reports of the BBB compromise and subsequent activation of the immune system and inflammatory pathways after blast exposure are now appearing in the literature and may make the NOD/ShiLtJ strain even more relevant for examining the relationship between autoimmunity and blast TBI.71–74 The baseline immune dysregulation in NOD/ShiLtJ mice, which includes impaired antigen-presenting cell function and abnormal natural killer cell responses, could clearly affect the course of recovery after blast exposure.
In comparison, the NZO/HlLtJ (New Zealand Obese) mouse provides a relevant model for studying TBI outcomes in diabetic patients due to its polygenic obesity and the spontaneous development of type 2 diabetes with associated chronic inflammation.65,75 Clinical studies demonstrate that diabetes is an independent risk factor for worse outcomes after TBI, including increased mortality (hazard ratio (HR): 1.89, 95% CI: 1.4–2.6),76,77 and is associated with greater neuroinflammation 73 and impaired functional recovery.76,78 The NZO mouse’s phenotype of early-onset obesity, progressive insulin resistance, and chronic low-grade inflammation77,79 mirrors key pathophysiological mechanisms seen in human diabetic patients who experience worse outcomes after TBI. 75 Moreover, diabetic patients show higher rates of neurodegenerative complications post-TBI, 80 making the NZO mouse’s metabolic phenotype particularly relevant for studying the intersection of diabetes, inflammation, and poor neurological recovery after brain injury.
Several limitations should be considered when interpretating these findings. First, this study exclusively used male mice, which may limit generalizability given the known sex differences in TBI outcomes and neuroinflammatory responses. Additionally, all consisted of age-matched adult mice evaluated at a single acute end-point (30 min post-injury), precluding the assessment of sex-specific, age-dependent, or temporal evolution of blast-induced pathology. These constraints highlight the need for future studies examining female mice, varying ages, and multiple time points to fully characterize the genetic determinants of blast-induced TBI outcomes.
Our data from all evaluated founder strains demonstrate the substantial influence of genetic background on acute bTBI response and outcome in mice. The intriguing resilience of diabetic strains to blast injury, despite their metabolic status, highlights the complex interplay between genetics, immune function, brain metabolism, and blast tolerance. Much like how the collaborative crossing of founder strains identified gene loci responsible for vascular network differences, which could underlie stroke impairment, 81 bone remodeling after hind limb unloading, 82 and DNA damage after ionizing radiation, 83 these observations provide a path to produce a collaborative crossing of founder strains to better pinpoint the possible genetic origins of the different outcomes observed after blast exposure. As some of the genetic strains may be better suited to study the long-term effects of TBI, our results will aid in guiding these studies by characterizing the thresholds and acute responses for a range of loading conditions. Ultimately, a better understanding of these genetic influences could guide the development of personalized therapies and improve the prevention and management of blast-induced neurotrauma. Additionally, future research should aim to link post-injury findings with specific gene loci and explore how genetic factors influence the outcomes across the spectrum of blast injuries, from single high-magnitude exposures to multiple subconcussive exposures.
Transparency, Rigor, and Reproducibility Statement
All animal procedures were conducted according to the protocols approved by Institutional Animal Care and Use Committees (IACUC) at both the University of Pennsylvania and the CMC VAMC. To study the lethality thresholds of each genetic strain, we started at low peak overpressure levels, in which we presumed 100% of the animals would survive. We incrementally increased the peak overpressure that we exposed each animal to in order to minimize the blast pressure needed to complete the logistic regression curves. To confirm the validity of our calculated lethality thresholds, in another cohort, we enrolled each strain (n = 12) at their respective LD10 levels. Approximately 10% of animals of the A/J, NOD/LtJ, NZO/HlLtJ, C57BL/6J, and CAST/EiJ strains died immediately post-injury, confirming our results. The exception was the 129S1/SvImJ strain—30% died at the precalculated LD10 level. We added the additional lethality data to our logistic regression plots, and all strain LD50 levels remained the same, except the 129S1/SvImJ LD50 decreased. This verified our results and increased the accuracy of the 129S1/SvImJ lethality data. An experimenter blinded to the strain and condition of each animal analyzed the brain histology.
Authors’ Contributions
A.P.G.: Conceptualization, methodology, data analysis, figures, writing. K.D.B.: Conceptualization, methodology, writing—review and editing. D.V.A. and A.H.B.: Methodology, data analysis. D.K.C.: Supervision, conceptualization, funding acquisition. D.F.M.: Supervision, conceptualization, funding acquisition, writing.
Footnotes
Acknowledgments
The authors are grateful to Kathryn R. Jacobson, Adam C. Rayfield, and Erin D. Anderson for editorial review and comments.
Author Disclosure Statement
No competing financial interests exist.
Funding Information
This material is based upon work generously supported by the Department of Veterans Affairs, Office of Research and Development through Merit Review I01-BX005017 (PI: D.K.C.).
Supplemental Material
Abbreviations
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
