Abstract
Objectives:
Rural-dwelling adults with advanced cancer and pain face obstacles to achieving cancer pain management. We explored patients’ experiences managing cancer pain while living in rural Oklahoma to improve access and overcome barriers to current treatments.
Methods:
We recruited patients from Oklahoma’s only National-Cancer Institute (NCI)-designated cancer center. We included those aged ≥21 years with an incurable solid or hematological malignancy residing in rural Oklahoma (Rural–Urban Commuting Area code ≥4). Interview topics included pain self-management practices, health care utilization, and descriptions of participants’ daily lives. We transcribed interviews verbatim and used an inductive, critical realist approach to reflexive thematic analysis.
Results:
Among 11 participants (mean [SD] = 62 [11]y/o), most were male (73%) and identified as White (64%), with 27% identifying as American Indian. Most (73%) had an annual income ≤$40,000. Participants’ cancer pain management experiences were shaped by the rural context through structural (distance, costs), interpersonal, and intrapersonal barriers. Participants compared greater familiarity within rural care settings with disconnection within the urban cancer center. In the context of the opioid epidemic, barriers to opioids contributed to hopelessness for some participants because within their cancer pain management plans, opioids were often the sole treatment option. Most exhibited stoicism and self-reliance while also utilizing community support, eventually connecting with urban cancer services and experiencing pain relief.
Conclusions:
Structural, cultural, and intrapersonal obstacles coexisted with hopelessness and disconnection from oncology/pain care clinicians. These experiences influenced how rural-dwelling adults with advanced cancer pain engaged with health care, self-managed, and coped with pain. Our findings highlight the importance of multimodal pain care grounded within the local cultural context.
Introduction
Rural residence is associated with worse cancer severity, pain, and other cancer-related symptoms; mental health outcomes;1,2 and consistently higher mortality (+9.6%) relative to suburban and urban residence. 3 Living in a rural area has also been associated with an increased risk for developing and living with chronic pain.4–8 Limited access to specialty cancer and pain care may contribute to these disparities; 9 However, the drivers of these outcomes are poorly understood. In response, the National Cancer Institute (NCI) called for research on the factors contributing to rural–urban cancer care disparities in 2018. 10
People with advanced cancer who live in rural areas experience cancer and cancer-related symptoms, including pain, in a uniquely challenging environment. For many, financial toxicity, the negative financial impact of cancer, including direct costs of care and loss of work,11–13 and long commutes to cancer treatment complicate access to care as symptoms worsen and financial resources narrow. 14 To date, qualitative studies of people with advanced cancer and chronic pain living in rural areas have also described the overlapping impacts of high symptom burden, limited resources, burdensome transportation, communication and cultural barriers, and racism on patients’ experiences.15–18 In addition, some evidence suggests that the impact of rurality on self-rated health may be more substantial in specific regions of the USA, such as in the southern USA.7,19
In Oklahoma, a southern U.S. state, 35.4% of the population resides in rural areas, 20 compared with 20% of the national population. 21 Oklahomans living in rural areas are more likely than their nonrural counterparts to be older, 22 live in poverty, 23 and not have a high school diploma. 23 These experiences occur within the context of broader social challenges in Oklahoma, such as high rates of disability, poverty, and low median household income relative to national rates. 24 In addition, Oklahoma has the largest population of sole heritage American Indians in the USA. 25 Many areas of the state are categorized as Medically Underserved Areas or Health Provider Shortage Areas, 26 which may compound the impact of rurality on health outcomes, including cancer and pain.
On an individual level, evidence suggests that the rural context may shape one’s experiences of living with chronic pain and cancer,15–18 yet a dearth of research has explored the interplay between known social and structural drivers (i.e., socioeconomic and demographic factors, health care access) and their psychological and behavioral consequences that likely produce rural cancer-related pain disparities. Qualitative approaches can clarify how rurality may influence individuals’ experiences of cancer pain management and by what mechanisms. We conducted a qualitative study with patients receiving care at the only NCI-designated cancer center in the state of Oklahoma to understand the unique perspectives of patients living with advanced cancer and pain in rural areas. By enhancing our understanding of the interplay of advanced cancer and chronic pain, findings from this work will help to inform modifications and intervention targets that may improve access and overcome barriers to current treatments.
Methods
Recruitment and study procedures
From February to August 2024, we recruited patients from the Stephenson Cancer Center (SCC) to participate in this qualitative study. Study procedures were approved by the University of Oklahoma Health Sciences Institutional Review Board (IRB#13-725). We screened palliative care, solid tumor, and hematology clinic schedules at SCC. Patients were eligible if they were ≥21 years old, diagnosed with a metastatic cancer being treated with palliative intent (comfort care to improve symptoms, noncurative), lived in a zip code catchment area assigned a Rural–Urban Commuting Area code score ≥4, 27 and spoke English. We purposively sampled patients with an opioid prescription or pain ≥4/10 over the past two weeks (Numeric Rating Scale). Clinicians could also refer their patients to the study. Eligible patients were approached via mail, phone, and in-person during a clinic visit and provided informed consent to participate.
Data collection
The interview guide was developed by the first author and senior author together and reflected aspects reviewed in the literature around cancer and noncancer chronic pain management in rural populations,15–18,28,29 as well as experiences participants shared during a similar project refining a mobile health app to improve cancer pain management.30,31 To avoid shaping participants’ lived experiences with the authors’ standpoints, we did not structure data collection with a framework or theoretical model and allowed participants’ responses to guide the flow of the interview. Interviews were conducted by the first author, a masters-level research assistant trained by the senior author, a pain psychologist specializing in cancer pain. Both had previous experience interviewing the study population.28,30,31 Interviews were semi-structured and focused on participants’ daily routines and local environment, health care utilization, and pain and cancer experience (Table 1). Interviews were audio recorded, transcribed verbatim, and accompanied by written field notes. We conducted interviews until saturation was reached and new interviews were contributing little new information relevant to developing themes. Six authors (S.M.D., D.R.A., J.Z., A.B., R.N., and J.M.) lived and worked in urban Oklahoma during the study period. They had varying levels of affiliation with rural communities prior to the study. Participants were encouraged to describe their environment as though speaking to someone who was unfamiliar with their town.
Interview Topics

Prior to completing an interview, participants completed a survey that quantified their cancer pain experiences compared with other clinical samples. The survey assessed sociodemographic and clinical characteristics, overall health care utilization, and pain medication and cannabis use, as other research found patients used cannabis at high rates to manage cancer pain, 32 especially in Oklahoma. State Area Deprivation Index (ADI) scores were obtained using nine-digit zip codes. 33 Scores are deciles, with a higher score indicating worse socioeconomic deprivation. Pain intensity and interference were assessed with the Brief Pain Inventory on Likert scales ranging from 0 to 10. 34 Persistent, negative thoughts about pain were measured using the 13-item (0–4 Likert scale) Pain Catastrophizing Scale (range: 0–52).35,36 Depression and anxiety were measured with the Patient-Reported Outcomes Measurement Information System with calculated t-scores normalized to other clinical samples, ranging from 0 to 100.37–39 Table 2 provides descriptive statistics for sociodemographic and clinical characteristics.
Participant Sociodemographic and Clinical Characteristics (N = 11)

Data analysis
Data were analyzed using reflexive thematic analysis, which allows for the application of diverse theoretical perspectives, engagement with subjectivity, and iterative analysis.42,43 We took a critical realist approach to acknowledge how rurality as a social construct was likely to unfold as participants selected and described aspects of their identities and experiences during interviews conducted about and, in some cases, within an urban health care institution.44,45 Methods and results are reported according to COREQ. 46
We followed a team-based, primarily inductive approach to coding the interview transcripts. Using NVivo, S.M.D. and D.R.A. developed a codebook by open coding the data, applying codes that represented frequent, intense, or notable concepts, emotions, and events. S.M.D. and J.K., a medical student who received training from S.M.D. and D.R.A. on qualitative coding, used the focused codebook to independently code each interview, then compared coding, discussed, and reconciled discrepancies. S.M.D. presented code memos, coding trees, and nascent categories to the broader study team. In response to the structure of the data, we returned to the literature published on cancer pain, rural health, and social scientific models. S.M.D. and J.K. revisited the coding tree and code memos and then identified themes.
Findings
Of the 28 patients we approached, 7 were not interested, 6 were lost-to-follow up, and 15 consented (53.6% consent rate). Three patients withdrew due to poor health, and one withdrew because of lack of interest. In total, we conducted 11 interviews; 8 were conducted in-person at SCC, 2 over the phone, and 1 over Zoom. Interviews ranged from 30.57 to 93.05 min in length (median = 57.75 min) depending on patient preference, ability, and availability. Five participants were accompanied by their caregivers; two caregivers contributed context during interviews.
Most participants were male (73%), 64% were White, 27% were American Indian/Alaskan Native, and 9% were Black. Half (54%) lived below the state poverty line, and 72% reported a household income ≤$40,000 per year. All participants were diagnosed with a metastatic cancer, and 91% of participants were initially diagnosed at least one year prior to the interview. Most participants were diagnosed with lung (27%), breast (18%), and head and neck (18%) cancers (Table 2, ). Participants lived an average of 112.23 miles from the cancer center (SD = 57.11 miles; median = 107 miles; range = 47.5–196 mi; (Fig. 1). While primarily exploratory, the frequency and severity of negative thoughts and emotional reactions to pain were high compared with similar cancer populations 28 (Pain Catastrophizing Scale sample mean [sd] = 25.73 [16.44]). Most (72.7%, n = 8/11) participants reported experiencing ruminative and negative thoughts about pain to a clinically significant degree (≥18 on the Pain Catastrophizing Scale). Depression (t-score: 52.11) and anxiety (t-score: 52.95) symptom severity scores were within normal range compared with other clinical populations.
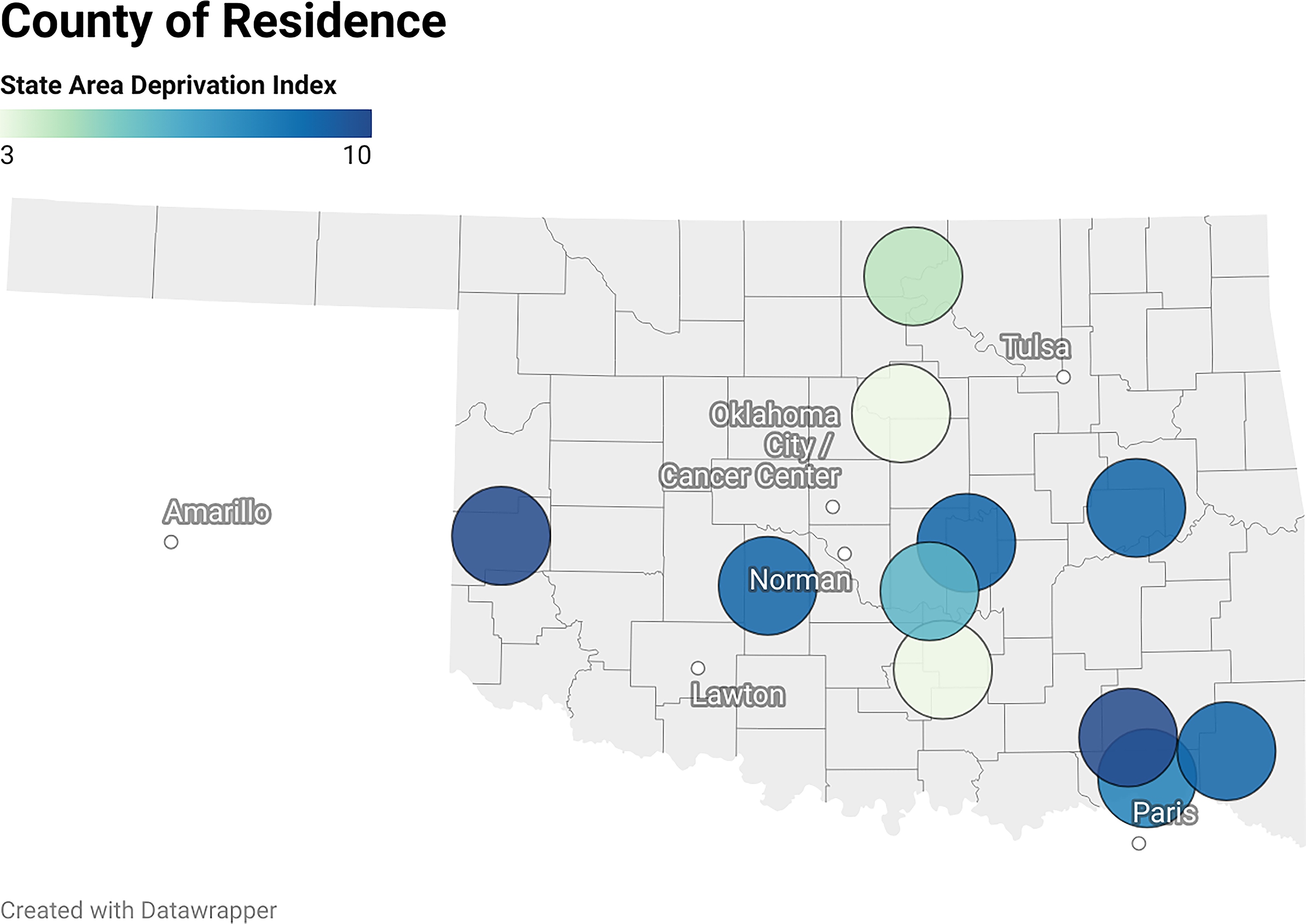
(map): Map of Rural Participants’ County of Residence and State Area Deprivation Index Codes in Oklahoma, United States.
Participants described how their cancer-related pain management experiences were shaped by the rural context. First, in participants’ narratives about their difficulties establishing care at the urban cancer center, they expressed a desire for close and familiar relationships with urban clinicians. They described difficulty seeking care for a life-limiting, symptomatic illness in resource-poor but socially cohesive rural areas. While rural cancer care was inadequate, they described authentic, mutual relationships with rural health care providers. These rural health care experiences shaped expectations in urban cancer care centers, which were experienced as better resourced but distant and impersonal, producing an emotional disconnect between participants and their clinicians. Second, some participants experienced hopelessness while managing pain, given how difficult it was to establish care and have comfortable clinical relationships. This challenge was compounded for some by opioid stigma related to the rural opioid epidemic. Many participants responded to this pain management context with stoicism and self-reliance informed by a rural cultural emphasis on independence and acceptance of mutual community support. See Fig. 2 for a diagram of themes.

(diagram): Diagram of Themes.
Relationships in an under-resourced cancer care environment
Participants shared similar narratives about working to seek and establish specialist care at the urban cancer center. Common elements in the narratives are captured by four subthemes: (1) Perceived need for care following advanced cancer diagnosis, (2) Difficulty obtaining specialist pain and oncology care, (3) Wishing for close relationships with clinicians, and (4) Unacknowledged challenges. At the core of this narrative were participants’ unmet expectations for their relationships with the clinicians to whom they were entrusting their care.
Perceived need for care following advanced cancer diagnosis
All participants understood that they were diagnosed with an incurable cancer. Their understanding of their disease was often informed by memories of friends and family with cancer, some of whom had died painfully (“…You know ‘cause I’ve seen some of my family and friends, their death was miserable. And I don’t want to have a miserable one. I don’t want to lay there and be miserable in front of my family and stuff” [60–69y/o, lung cancer, ADI: 9]). Memories of loved ones, worsening functional impairment, and resulting changes in identity contributed to heightened emotions about participants’ diagnoses, such as shock, sadness, and loss. Others expressed gratitude for and rationalized their survival past the life expectancy that had been communicated to them. For example, one participant explained, “Well, I’ll tell ya the truth. They shocked that I’m still walking and breathing on my own and all that. They figured I’d be in the bed and all that. Nah, man. I got a will to live… Lord’s got a plan for me.” (50–59 y/o, lung cancer, ADI: 3). These emotions informed participants’ decisions about desired palliative care and their perceptions of the care they received for their cancer and pain management. Some felt an urgency to seek symptom management to avoid the hardships loved ones with cancer had previously experienced.
Difficulty obtaining specialist pain and oncology care
Despite this urgency, participants reported few local health care resources in their rural communities. Many found nearby specialist and emergency medical care to be of poor quality and ill-equipped to handle cancer and cancer-related symptoms like pain. One participant described her local hospital as having few oncology clinicians: “Nothing’s just — it’s very basic here, if you need some stitches or something, some people it’s fine for. I mean, I’m a different kind of patient, for your basic, common sickness and stuff, it’s fine, but if you are a complex patient, you need to go somewhere else.” (40–49 y/o, adenocarcinoma with unknown primary cancer, ADI: 3). Given the lack of appropriate local health care resources, participants needed to seek care at the urban cancer center, which required them to navigate a sequence of referrals, significant travel, and associated costs.
Because obtaining care at the urban cancer center was so difficult, some participants endured pain for months before receiving appropriate pain management. For example, one woman reported having to attend appointments at several clinics before receiving appropriate care:
“And then I went to the — you know, living in [previous address in small out-of-state city] I went ‘jack in the box’ [attended many different clinics] all over Oklahoma and Texas. And it wasn’t until — when did I start my treatments? About three and a half months ago. It took that long to get proper care. So, I was in pain for quite a long time before I got proper care at a facility that handles cancer.” (60–69y/o, breast cancer, ADI: 10)
For almost all participants, establishing care at the cancer center and receiving palliative and supportive care was a turning point, marking the transition from uncontrolled to controlled pain.
Wishing for close relationships with clinicians
However, even after establishing care at the cancer center, participants’ health care experiences within the rural context followed them. Participants struggled to build relationships with clinicians at the urban cancer center. For example, one participant had been diagnosed with cancer in 2020 by a rural health care provider after being evaluated for an unrelated concern, but he described that he had not been explicitly told about his diagnosis at that time. Therefore, he was unaware of his 2020 cancer diagnosis until he sought care for a work-related injury multiple years later. He described how this experience affected him and his trust in clinicians:
“You know, that’s when I said, ‘Man, if they’d have told me, I could’ve done something different.’ …You know I’m picky. Ever since this happened to me, I don’t trust nobody — yeah — when it comes to stuff like that, like the doctor. [You] tell me you want me to trust you. I’m like, [I] trusted that man and look what he done to me. Yep.” (50–59y/o, lung cancer, ADI: 3)
He later transferred his care to the urban cancer center, where he believed he would be seen by experts. Even so, it took time to build trust and mutual respect with his clinician.
While some struggled to trust urban clinicians because of prior negative experiences in the rural context, others found that urban clinicians could not provide the warm, familiar relationships they had with rural primary care providers. Participants saw local primary care providers around town, socialized with them, or had grown up with them. One participant spoke to the differences between seeing a local nurse practitioner and receiving care in Oklahoma City, where she had previously lived:
The nurse practitioner here, our sons play together in the clinic, and she’s great with them with physicals and, your routine care and stuff, where, if you get sick, you might sit at [redacted Cancer Center] in Oklahoma City for five hours. That didn’t happen here [local clinic]. (40–49y/o, adenocarcinoma unknown primary, ADI: 3)
This familiarity created a level of accessibility and, potentially, accountability, which some participants felt the cohesion in their towns provided them. For example, one participant knew a physician through her real estate business. She described the physician as subsequently being helpful with her symptom management. The participant connected this to the need to maintain positive local relationships, saying “And in small-town America you don’t stay in business if you don’t try to do things right.” (70–79 y/o, breast cancer, ADI: 4).
In contrast, though participants credited clinicians at the cancer center with providing life-changing care, they wished that they had closer, more authentic relationships with their oncologists, in particular:
Yes, ‘cause I would like to have a little bit more of a personal relationship with her [oncologist], I guess. And that’s something, you know, that when you deal with rural health care vs. big city. You know, you get to know your care providers here more-so than you would when you see your doctor once every three months here. And where here in [rural town of residence] you probably see them at the movie theater, they go out to the Chamber of Commerce, they would, you know, your paths would cross. You see them at a city commission meeting. Or in your church and your church isn’t so big that you wouldn’t get to know them, that type of thing. (70–79 y/o, breast cancer, ADI: 4)
These prior, positive experiences created expectations for the care participants received at the cancer center, all of which related to a desire for mutual connection.
Unacknowledged challenges
By the time participants established care at the cancer center, they had experienced health care inaccessibility, poor quality local specialist and emergency care, taken on financial burdens, and endured long and uncomfortable travel. Despite the impact of life-changing care received at the cancer center, participants’ path through the cancer care system was mostly described as difficult. One participant described how hard it had been to access specialist care, a challenge that felt more acute given his dissatisfaction with the care he received: “There’s just one doctor in Oklahoma that does it [a procedure] that the insurance bills. They take a lot of gas to go over here, [inaudible] been coming up here for over two months and they still haven’t helped me one time,” (50–59 y/o, laryngeal cancer, ADI: 10). Like this participant, many had experienced a string of clinical encounters that they perceived as incompetent, stigmatizing, and even unjust while striving to receive specialist care.
Others reported frustration stemming from poor communication, especially given distance from the cancer center:
“Yeah, I drove two hours. To talk with some other goofball that, ‘Oh, I’m Dr. [redacted], an associate or whatever.’ And he had no idea who I was and, you know, he wasn’t up to my deal. And they had to find him somewhere to talk to me. So, he wasn’t even expecting me to take [redacted]’s place because [redacted] wasn’t there. Because nobody told me he wasn’t gonna be there. Yeah, so it cost me, you know, I spent a day, basically, for nothing.” (60–69 y/o, lung cancer, ADI: 9)
At times, participants perceived that cancer center staff, clinicians, and pharmacies did not understand how difficult it had been to access care at the cancer center to begin with. This perception exacerbated the inconveniences caused by delays, rescheduled appointments, referrals, and miscommunications. A scheduling error did not always feel like a small mistake when participants had driven hours, spent limited funds, and waited months to attend an appointment; it felt like a lack of understanding and respect.
Opioid-epidemic-related stigma and fractured cancer pain care systems
Even after establishing care with the rural cancer center, participants struggled to self-manage their pain. This experience is captured by two subthemes and the connection between them: (1) the opioid epidemic and subsequent opioid-related stigma and (2) stoicism, self-reliance, and mutuality reinforced by rural culture while self-managing cancer pain. Participants’ awareness of public opioid stigma and their personal experiences with stigmatizing clinical interactions around opioids exacerbated the challenges they experienced within difficult-to-navigate health care environments. In response, participants were stoic about pain, relied on themselves to manage their pain, self-advocated, and accepted community support. For some participants, these strategies were an advantage of living in a rural area. Others were forced to rely on themselves or community members because there were no options for professional support.
The opioid epidemic and stigma
The opioid epidemic and related opioid stigma impacted participants’ comfort taking opioid medications and, in some cases, damaged participants’ relationships with clinicians. Participants described the impact of public opioid stigma, other people’s opioid misuse, and stigmatizing clinical encounters.
Impact of public opioid stigma. Some, particularly participants living in southeast Oklahoma where opioid misuse and related mortalities were more prominent, were aware of local opioid and methamphetamine use, which informed and influenced participants’ experiences with opioids and opioid stigma. One participant and his daughter stated that the historical and cultural context in their rural town influenced perceptions and fears about opioid use for cancer pain:
Daughter: “And where we’re from, [redacted] County has been forever a prescription drug abuse area. So, in our area it’s—it’s just notorious for people to be in that mindset that if you take pain medicine—you’re a—you know,—addict… They used to have this saying. They called it [redacted] County Cocktail.—it was some hydrocodone and Xanax. It was being so highly prescribed to people in our area for the longest time.”
Participant: “You know, especially this—pain pills. You know, it is—that’s one of my biggest ones [fears] right there. Getting strung out on pain meds.” (60–69 y/o, lung cancer, ADI: 9)
As the participant voiced frustrations with people who engage in substance use behaviors, his language (i.e., “strung out”) suggests that he may have internalized public stigma about opioid addiction and generalized these stigmatizing attitudes to his own use of opioids for cancer pain.
Others’ opioid misuse. Several participants suggested that people experiencing substance use disorders “made it difficult” for people with a “more legitimate” reason, such as advanced cancer, to obtain opioids; “So, yeah, I guess it is understandable [that clinicians are cautious about prescribing opioids] because there are so many people that don’t have control [over their opioid use] or something… Don’t need them and sell them or, you know, uh, so, it’s just the way we are, I guess. But those of us that need pain medicine…” (40–49 y/o, prostate cancer, ADI: 8). Notably, this participant endorsed taking prescribed opioids for cancer pain and using nonprescribed opioids and methamphetamines for uncontrolled pain. Multiple participants perceived that their clinicians were unable to prescribe opioids because of widespread misuse: “So, southeast Oklahoma, I guess, there’s epidemics, you know. I guess it’s everywhere, but it’s the point to where the doctors there can’t really prescribe pain medications.” (40–49 y/o, prostate cancer, ADI: 8) For others, opioid fears were driven by internalized stigmatizing beliefs about people who experience addiction and witnessing substance misuse within their rural communities. For one participant, this fear was grounded in the deaths of multiple loved ones:
“But see, my son was on meth and he was going through punching holes in the walls and stuff. … And then it got him killed. He spend 17 days over there at [redacted]. He was in a coma for 10 of them. Oh yeah. That’s why I say nah — now you know why I was scared about taking drugs [opioids].” (50–59 y/o, lung cancer, ADI: 3)
Stigma in clinical encounters. In addition to broad-scale social issues like the opioid epidemic, some participants also noted opioid-related stigma and judgment in clinical and pharmacy encounters. Given how difficult it was to be seen in their own communities by a clinician who was able to prescribe opioids to manage cancer pain (e.g., many rural advanced practice providers are in effect unable to prescribe opioids in Oklahoma due to supervision requirements), many participants perceived oncology clinicians’ reluctance to prescribe opioids to be an expression of distrust. One participant said, “face it, I mean, everybody’s suspicious of it.” (40–49 y/o, prostate cancer, ADI: 8).
In the face of poorly controlled pain, limited access to pain management, and stigma, many expressed feelings of hopelessness and loss of control. For some, opioids emerged as a desirable solution despite the risks. Some participants expected near-total relief from pain with opioid therapy. A few described using opioids to avoid pain and suffering altogether, leading to risky opioid use or substance use when opioid medications were not available or denied. Others managed hopelessness by turning to their faith and faith communities for support and working to let go of what they could not control. These responses occurred within a social context that necessitated independence and intra-community mutuality alongside reliance on unimodal opioid therapy, with very limited pain management education otherwise.
Stoicism, self-reliance, and mutuality while self-managing cancer pain
The limited availability of resources and clinical support led some participants to take matters into their own hands. Participants’ cultural beliefs about stoicism, their self-reliance, and community mutuality shaped how they managed cancer-related pain in the rural context.
Participants’ communities were social, cultural, and practical resources. Although participants lived in different regions of Oklahoma, they described their rural towns similarly: quiet, spacious, and close to natural spaces. Many participants were active in their local “close-knit” communities through church, community events, local government, and public-facing work. Engagement within small communities made many feel they were known within their communities. Participants relied on themselves and their community because they valued the interpersonal connections and because they felt they did not have another choice. Most participants mentioned that there were no local mental health professionals, case managers, or cancer treatment centers that could provide appropriate help or professional guidance. Therefore, when difficulties arose, participants had to manage them on their own or with the support of their community.
Stoicism. Within participants’ communities, cultural norms dictated how participants received and gave help. Many indicated that there was an internalized cultural expectation that they endure symptoms and try to handle problems themselves. Some who were stoic about their pain chose not to report or ask for help with their uncontrolled pain. Stoicism was related to participants’ conception of themselves as self-reliant and healthy people who did not “need” help, to stigma against people who use opioids, and to identification with behavioral norms perceived to be specific to their rural identity, such as initial rejection of external help and independence. For example, one woman said, “That has been really hard for me [accepting help] because I’m so independent, and I don’t want to complain. And, you know, I’m a pioneer woman, and I want to do it. And I don’t want any help.” (60–69 y/o, breast cancer, ADI: 10).
Self-reliance. Self-reliance in pain management sometimes took the form of self-advocacy, which participants had to engage in to receive adequate care. Many participants effectively and persistently self-advocated for quality care, pain management, financial support, and opioid therapy. This participant described scheduling and self-managing their opioid prescriptions to overcome inaccessibility:
“Being in a small town, my pharmacist can’t order drugs on Friday. So we have to look ahead…So you have to figure out ahead when it is you might run out and let them know ahead of time to make sure they have them in stock. And call them up and say, you know, I’m going to be able to get these Tuesday next week. I just want to make sure since you can’t order them till Monday, make sure that you might have some in stock, but yeah, but it’s [done].” (60–69 y/o, colon cancer, ADI: 9)
For some, self-advocacy involved managing opioid stigma within clinical encounters and taking action to remedy perceived medical errors that occurred prior to receiving care at the cancer center.
Efforts to self-advocate were taxing and made some participants feel that they were receiving care in an unjust or dysfunctional system. For example, one participant described pain exceeding his clinicians’ dosage recommendations. He felt forced to use nonprescription opioids, in part because of difficulty maintaining acceptable pain control:
“I mean, — if they make me fill out that I’m trying to do something for the wrong reason, I’m not gonna mess with them
While this may be evidence of opioid misuse, the participant’s experience of stigmatization is worth noting.
Others found that stigmatizing experiences forced them to self-advocate. One patient described being denied opioids and receiving a referral to an interventional pain clinic without appropriate education as to why. As a result, she felt stigmatized and anticipated that her long-suffered pain would go untreated. She recounted,
“They wanted to stick a needle in my shoulder [pain interventional procedure] for what reason I can’t remember. I just dismissed it like ‘No.’ I said, ‘Can you just give me something.’ Before I started doing cancer stuff [before diagnosis], uh, ‘Can — can you just prescribe me something since you are in pain management to just relieve the pain until I can get to the bottom of it?’ They treated me like I was a drug addict. And I said, ‘That is so disrespectful.’ they all treat you — especially if they don’t know you.” (60–69 y/o, breast cancer, ADI: 10)
Notably, she draws a distinction between how she was treated by clinicians “who know her” versus clinicians who did not know her, a difference that contributed to perceived stigma. She divulged that she now shows clinicians pictures from her life to develop trust.
In terms of receiving education from their cancer team, some felt they did not understand what clinicians were trying to convey because of unclear explanations or use of an inauthentic or an overly professionalized tone. Even still, participants exhibited resilience, self-advocated, and worked to “stay cool” when they experienced health care issues.
Mutuality. Despite their self-reliance, participants reported that their community offered support before it was requested and occasionally before it was desired. Community support and mutuality were cultural expectations. Some leveraged mutuality and connections to local community members to obtain emotional, social, informational, and practical support where it was available. One man described the importance of social support to his coping despite the emotional complexity added by his diagnosis with an incurable cancer:
“But I may go to Walmart and walk around. And I see a lot of people there. A lot of time, you’ll see somebody in the Walmart that you haven’t seen in a long time. And they’re like, “Hey, how you doing?” “Oh, hey—” and I’ll have to try to — without getting all emotional. “Well, okay, but I’ve got cancer and I’ m just trying to get by.” You know, and sometimes it’s hard. When I first had it, first was diagnosed, it seemed like it really would affect me emotionally trying to tell people, you know, that I got cancer and fighting it… But I’ve had so much encouragement from people, and even strangers, you know. …And they’re always telling me I’ m gonna make it. So, [you]try to take it, you know, as it is … you always need people in your life or something. (60–69 y/o, lung cancer, ADI: 9)
He stated that community members often shared stories about loved ones with cancer who had survived, which he considered encouraging. All participants used their social networks to meet health-related needs.
Discussion
In this qualitative study, we found that the rural context shaped participants’ cancer pain management through participants’ experience of their relationships with urban clinicians and the complex challenges posed by self-managing pain during the opioid epidemic. Our analysis highlights structural (distance, costs, fragmented services), interpersonal (expectations for relationships formed in close-knit rural care), and intrapersonal barriers (stoicism, self-reliance). Familiarity was an important component of participants’ experiences, informing their perception of medical care and experience of stigma. Despite a desire for familiarity, local specialty cancer pain services were limited to nonexistent. Participants needed to receive care from a specialty urban cancer center despite feelings of disconnection. While receipt of these services resulted in improved pain, accessing specialty cancer pain care required extensive temporal and financial resources, self-advocacy, and community support. For many, this situation contributed to hopelessness.
Participants described opioid stigma within rural and urban settings, which aligns with prior work. Other studies of chronic pain have noted the presence of opioid stigma in rural areas47,48 and more generally during the cancer experience.28,49 Clinicians have reported experiencing pressure to prescribe opioids in a challenging social context and in clinical encounters that include a high degree of uncertainty.50–53 Our data extend the literature by highlighting the importance of trust and familiarity in clinical decision-making about opioid prescribing, which may be reduced when a patient from a rural area is receiving care in an urban health care setting.
While opioids remain central to cancer pain management, there are few rural clinicians available to prescribe opioids for cancer pain in Oklahoma. 54 Concurrently, comprehensive pain management treatment options (e.g., physical therapy, occupational therapy, and pain psychology) are generally less accessible in rural settings. 55 As such, it is unsurprising that patients with advanced cancer living in rural areas feel hopeless in the face of their pain. Participants felt defeated when confronted with continued pain treatment barriers, including poor communication, lack of pain management education, stigmatization, travel, and financial hardship. Technology-based programs for pain education, palliative care services, pain management interventions, and nonpharmacologic treatment may increase access to comprehensive pain care for patients living in rural areas.56,57
Participants’ cultural perspective informed how they managed difficulties, including uncontrolled pain or inaccessible specialty care. Many were stoic and self-reliant, self-advocated, and leveraged community support, which can both support coping and sometimes delay help-seeking for uncontrolled pain. For them, these behaviors reflected cultural norms that prioritize independence rather than indicating unwillingness to seek care. Others described relying on community support, which was grounded in mutuality, a value that allowed participants to exchange help while maintaining a sense of self-reliance. In this scenario, self-reliance and community support were a “pro” and a “con.” While it is beneficial for individuals to be connected, take an active role in their self-management, and have a high sense of self-efficacy, patients were relying on themselves without evidence-based clinical care for complex medical symptoms such as cancer pain. Moreover, the disconnection that participants experienced from clinicians is emblematic of the circumstances, cultural norms, and emotions that might make self-reliance the most viable behavioral option. This points to the need for accessible pain care that is compatible with cultural norms related to self-reliance and improved pain management education and communication between clinicians and their patients. Opioid stigma and hopelessness were related to a need for self-reliance and community support and also associated with reduced engagement in evidence-based care in favor of a reliance on opioids and endurance of pain.
Limitations
The sample was comprised of participants who were receiving care at an NCI-designated cancer center. It is possible that people with advanced cancer pain who had not established care with the cancer center living in rural Oklahoma would have different experiences. It is also possible that engaging with patients through the cancer center may have biased responses, though participants freely offered nuanced impressions of the cancer center during interviews. Inclusion of field work may have contributed to our ability to understand participants’ experience of their care and ethnographic methods present an opportunity to understand the impact of health care institutions’ procedures on patients’ experience and symptom management.
Future directions
Future studies should triangulate qualitative and quantitative data, as done in mixed-methods research that applies ecological momentary assessment (EMA)-assisted interviewing, to better define how factors interact with one another and where behavioral (e.g., pain-specific mHealth interventions30,31), health care system (e.g., telehealth-integrated palliative care programs56,57), or state-level interventions (e.g., increased access to nonphysician opioid prescribing) can be applied. Such mixed-methods approaches could identify the most important touchpoints and inform the designs of multicomponent, telehealth, and/or mobile health interventions to improve key barriers of rural cancer care, increasing accessibility, communication, engagement, and satisfaction. Further research on the spatial distribution of opioid stigma, factors that cause local intensification in rural areas (e.g., local opioid mortality rates and stigma), and how to target upstream contributors to stigma to improve pain-related outcomes is needed.
Conclusion
In this study, we explored and identified how the rural setting may inform participants’ experience of cancer pain management. Findings suggest that intervention targets could include increased clinician- and system-level recognition and patient-centered pain management education and access, along with patient-centered communication of the barriers to treatment (e.g., distance to cancer center) and treatment expectations (e.g., relationship quality, opioid efficacy, comprehensive pain management) that may reduce perceived stigma and disconnection. These changes may help to improve cancer pain symptom disparities among adults living in rural areas. Additional work applying social scientific theories about culture and health behaviors is needed to provide an integrated picture of how social factors may influence patients’ pain experiences.
Data Sharing Statement
Data may be made available upon request to the corresponding author.
Ethics Approval
Study procedures were approved before initiation by the University of Oklahoma Health Sciences Institutional Review Board (IRB#13-725). Participants provided informed consent to participate.
Authors’ Contributions
S.M.D.: Conceptualization, methodology, formal analysis, investigation, data curation, writing—original draft, writing—review and editing, visualization, and project administration. J.J.K.: Formal analysis, data curation, writing—original draft, and writing—review and editing. J.M.: Methodology, formal analysis, writing—original draft, and writing—review and editing. A.K.: Writing—review and editing. H.W.B.: Writing—review and editing. K.L.K.: Methodology, formal analysis, writing—original draft, and writing—review and editing. J.Z.: Formal analysis, investigation, writing—original draft, and writing—review and editing. A.B.: Methodology and writing—review and editing. R.N.: Writing—review and editing. D.R.A.: Conceptualization, methodology, formal analysis, investigation, writing—original draft, writing—review and editing, supervision, and funding acquisition.
Footnotes
Author Disclosure Statement
The authors have no conflicts to report.
Funding Information
D.R.A. is funded by NCI-K08 CA266937. H.W.B. is funded by NCI K08CA263317. A.K. is funded by K99CA279651. This work was funded by the Oklahoma Tobacco Settlement Endowment Trust (TSET) contract STCST00400_FY25 and the OU Health Stephenson Cancer Center via an NCI Cancer Center Support Grant (P30CA225520).
