Abstract
Background:
Allergic rhinitis (AR), colloquially known as hay fever, is a sinonasal inflammatory disease. Growing evidence suggests that female sex hormones play a significant role in immune-driven conditions. Systemic hormonal contraceptives (SHCs), widely used in reproductive health care, and menopause affect hormonal levels in women, presenting a framework to examine the role of sex hormones in AR.
Objective:
To assess the associations of SHC use and menopause with AR using a diverse, large, nationally representative database.
Methods:
This cross-sectional study utilized data from three cycles of the National Health and Nutrition Examination Survey (NHANES). Exposure variables were current SHC use (yes vs. no), duration of SHC use (years), and menopausal status (yes vs. no). The outcome variable was AR, defined as self-reported hay fever episode in the past 12 months (yes vs. no). Covariates considered included age, race/ethnicity, family Poverty Income Ratio, language, health insurance status, general health, alcohol use, smoking, and recreational drug use. A forward selection approach determined the final model.
Results:
Extended SHC use was associated with increased odds of AR. Current SHC use within one year, however, was not associated with AR. Postmenopausal women did not have a significantly lower risk of AR than premenopausal women.
Conclusion:
SHC use may have a dose-dependent influence on AR, which highlights a potential relationship between female sex hormones and sinonasal inflammation. Prospective and mechanistic studies are needed to further evaluate this association.
Keywords
Introduction
Allergic rhinitis (AR), or hay fever, is a sinonasal inflammatory disease that poses a significant health and economic burden on individuals and health care systems globally. In the United States, approximately 15%–30% of the population is affected.1,2 Direct health care costs of AR in the United States are within the range of $3–4 billion yearly, with an additional annual average of $593 per patient incurred in indirect costs from reduced productivity.3,4
AR is characterized by sinonasal inflammation, mediated by increased local and systemic IgE production following a cascade of events in response to allergen exposure in sensitized persons. 5 Growing evidence suggests that female sex hormones, estrogen and progesterone, are immune modulators, 6 and thus potentially play a role in biological sex-based differences in inflammatory conditions.7,8 In animal studies, female sex hormones and their analogues have been shown to influence immune-mediated conditions, including airway inflammatory conditions, via several mechanisms involving both the innate and adaptive immune systems. 9 Further, recent epidemiological studies show females may be disproportionately affected by immune-mediated diseases such as asthma and atopic dermatitis, which share pathophysiologic similarities with AR. 10
In AR, studies are limited and mostly suggest an increased prevalence of AR in women.11–13 A meta-analysis found a shift in the prevalence of AR from a male predominance before puberty to a female predominance during puberty, coinciding with the period of transition to higher systemic levels of female sex hormones. 14 Additionally, studies have found that prolonged exposure to endogenous estrogen in women with longer reproductive periods is associated with increased incidence of AR, 15 and late-onset menarche has been linked to reduced risk of AR. 16 The elevated estrogen levels during the periovulatory phase have also been associated with increased nasal congestion and heightened reactivity of the nasal mucosa to histamine.17,18 All these findings suggest a role for sex hormones in AR.
While exogenous estrogen sources such as systemic hormonal contraceptives (SHCs) have been found to increase the incidence of asthma,19–21 and other immune-driven inflammatory conditions, 22 further highlighting the potential effects of sex hormones on inflammation, this has not been well-studied in AR. Menopause, which is accompanied by decreased levels of endogenous sex hormones, also presents another opportunity to investigate the hormonal influence on inflammatory conditions. Results from studies investigating the effect of menopause on airway inflammation remain inconsistent, as some studies found an associated reduction in asthma prevalence in postmenopausal women,23,24 while others have found menopause to be linked to exacerbation of asthma symptoms, typically in nonatopic cases. 25
The conventional ideology is that any rhinitis related to a hormonal cause, such as rhinitis of pregnancy or hormone-induced rhinitis, is inherently nonallergic and thus nonimmune, non-IgE-mediated.2,26 This may represent an incomplete understanding of the effects of female sex hormones on the sinonasal mucosa, considering the growing literature on the immunomodulatory effects of female sex hormones and their well-documented role in asthma and other immune-mediated conditions. Given the inflammatory-mediated pathophysiology of AR, it is reasonable to investigate if similar hormonal influences may play a role in AR, a separate clinical entity from the aforementioned, traditionally known hormone-related rhinitis. The objective of this cross-sectional study is to assess the associations of SHC use and menopause, respectively, with AR using data from the National Health and Nutrition Examination Survey (NHANES), a diverse, large, nationally representative database.
Methods
This cross-sectional study utilized publicly available data from NHANES, a continuous survey conducted since 1999 to monitor health and nutritional trends in a representative sample of the noninstitutionalized U.S. population. Data used in this analysis were drawn from NHANES cycles 2005–2006, 2007–2008, and 2009–2010 that included relevant information on hormonal factors, including SHC use and menopause, and AR/hay fever. Participants selected for NHANES undergo a structured interview and physical examination and provide biological samples for laboratory testing. NHANES employs a multistage sampling process, oversampling specific subgroups (African Americans, Hispanics, and older adults) to ensure generalizability to the U.S. population. Details on the selection process of NHANES are available in NHANES documentation. 27
Inclusion and exclusion criteria
For the SHC analysis, only biologically female participants between the ages of 12 and 40 years were included to focus on the reproductive-age population and better isolate SHC use as the exposure of interest. Individuals with a history of oophorectomy, those who were pregnant, postmenopausal, or had missing data on any of the study variables were excluded from this analysis.
For the menopause analysis, only biologically female participants between the ages of 40 and 60 years were included to better isolate the sample to the perimenopausal age period. Individuals who were pregnant, had a history of oophorectomy, were using hormonal medications, or had missing data on the variables of interest were excluded.
Exposure, outcome, and covariate variables
The primary exposure variables were current SHC use (yes vs. no), duration of SHC use (years), and menopausal status (yes vs. no). The primary outcome variable was AR, defined as self-reported episode of hay fever in the past 12 months (yes vs. no). These variables were defined using NHANES survey items. Detailed definitions for the primary exposure and outcome variables are in Supplementary Table S1.
Potential confounders, including age, race/ethnicity, education level, family Poverty Income Ratio (PIR; less than 1 means household income is below the federal poverty level), language of interview, health insurance coverage status, general health condition, alcohol consumption, smoking status in the home, drug use history (marijuana, cocaine, heroin, or methamphetamine), and age of the home, were considered.
Statistical analysis
Descriptive statistics of median and interquartile range (IQR) were calculated for continuous variables, while percentages were reported for categorical variables. A forward selection approach was adopted to determine the final model, with age, race/ethnicity, and family PIR forced into all models. All analyses accounted for NHANES sampling weights, and a complete case analysis was used to deal with missing data. Statistical analyses were conducted in R (version 4.2.3). 28
Results
Baseline characteristics and bivariate analyses
Baseline characteristics for participants in the SHC analyses are shown in Table 1. Of the 4006 participants included, 352 (9%) had an episode of AR in the past 12 months, and 3654 had not. For the full cohort, the median age was 25 years (IQR: 18–33), and participants were mostly White (62%), had a high school education level or above (85%), and had health insurance (79%). Compared to participants without AR, participants with AR were generally older (p < 0.001), predominantly White (p < 0.001), had completed higher levels of education (p = 0.007), had higher family PIR (p < 0.001), spoke English (p = 0.031), had health insurance (p = 0.033), and drank alcohol (p < 0.001). Current SHC use was reported by 19% of total participants and was higher in participants with AR (22%) compared with those without (19%), but this was not significant (p = 0.2). The median duration of SHC use in years was significantly higher in participants with AR compared with those without AR (4 vs. 3 years, p < 0.001).
For the Oral Contraceptive Analyses, Baseline Descriptive Statistics Overall and Stratified by the Outcome Variable (Allergic Rhinitis in the Past 12 Months, Yes vs. No) and Bivariate Analyses

Weighted median/interquartile range for continuous variables and unweighted n/column percentages for categorical variables are listed.
Weighted values were used to calculate p values.
*p < 0.05 was considered significant.
AR, allergic rhinitis; PIR, Poverty Income Ratio; SHC, systemic hormonal contraceptive.
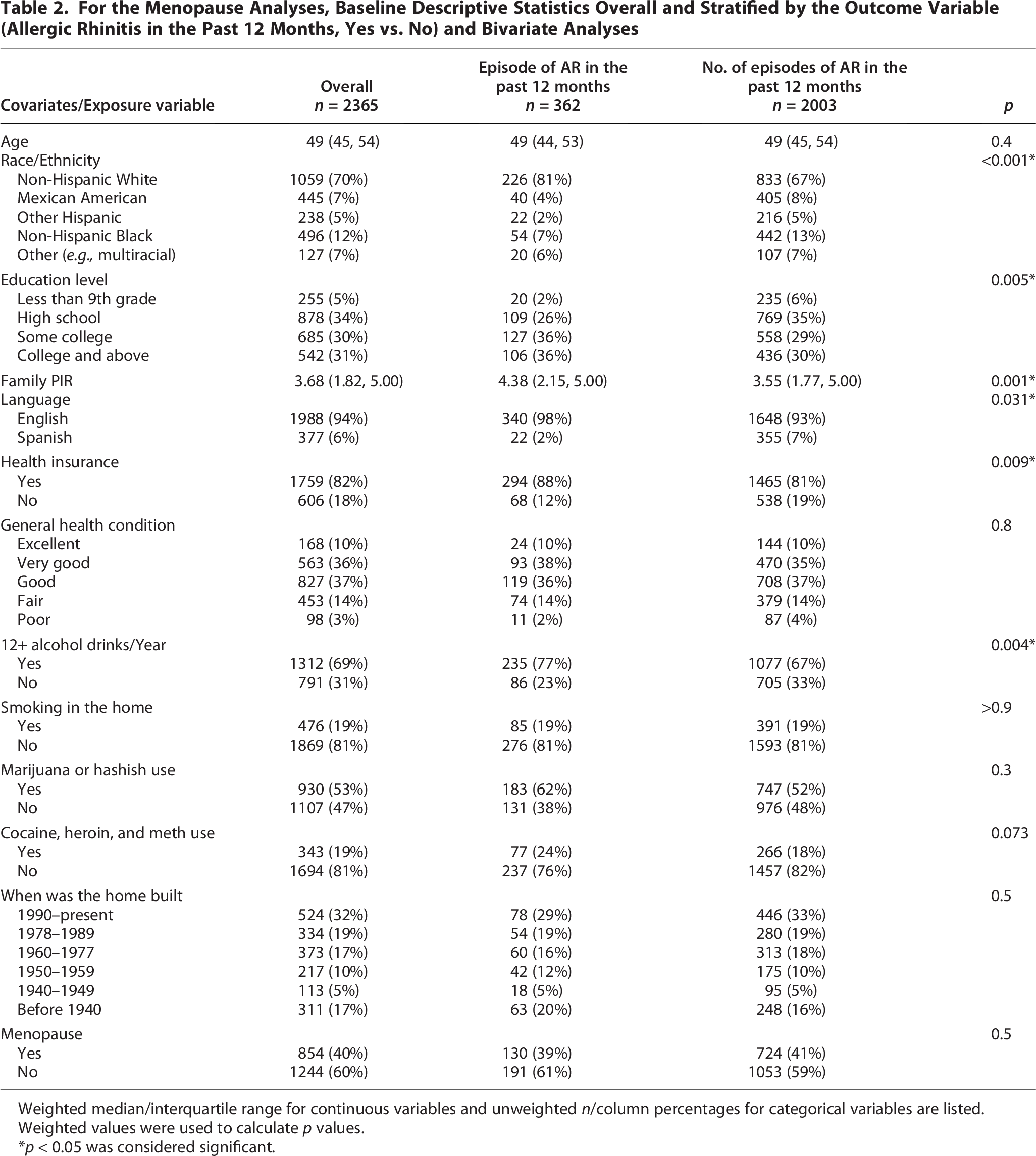
Baseline characteristics for participants in the menopause analyses are shown in Table 2. Of the 2365 participants included, 362 (15%) had an episode of AR in the past 12 months, and 2003 had not. For the full cohort, the median age was 49 years (IQR: 45–54), and participants were predominantly White (70%), had a high school education level or above (95%), and had health insurance (82%). Compared to participants without AR, participants with AR were more likely to be White (p < 0.001), have some college education or above (p = 0.005), have a higher median family PIR (p = 0.001), speak English (p = 0.031), and drink alcohol (p = 0.004). Forty percent of participants had undergone menopause, with no significant difference between those with and without AR (39% vs. 41%, p = 0.5).
For the Menopause Analyses, Baseline Descriptive Statistics Overall and Stratified by the Outcome Variable (Allergic Rhinitis in the Past 12 Months, Yes vs. No) and Bivariate Analyses
Weighted median/interquartile range for continuous variables and unweighted n/column percentages for categorical variables are listed.
Weighted values were used to calculate p values.
*p < 0.05 was considered significant.
Adjusted associations of current SHC use and AR
Table 3 shows the adjusted associations between current SHC use and AR. Females who reported current SHC use had a 1.13 times higher odds of reporting AR in the past year, although this was not significant (odds ratio [OR]: 1.13, 95% confidence interval [CI]: 0.76–1.69, p = 0.54). Age and alcohol consumption were significant predictors, with each additional year of age increasing the odds of AR (OR: 1.08, 95% CI: 1.05–1.10, p < 0.001) and those consuming alcohol showing a 1.85-fold increased odds of AR (OR: 1.85, 95% CI: 1.34–2.56, p < 0.001). Individuals of Mexican American race/ethnicity showed a significantly lower risk of having AR (OR: 0.36, 95% CI: 0.20–0.65, p < 0.01).
Adjusted Association Between Current Systemic Hormonal Contraceptive Use and Episode of Allergic Rhinitis in the Past 12 Months with Included Covariates in the Final Model

*p < 0.05 was considered significant.
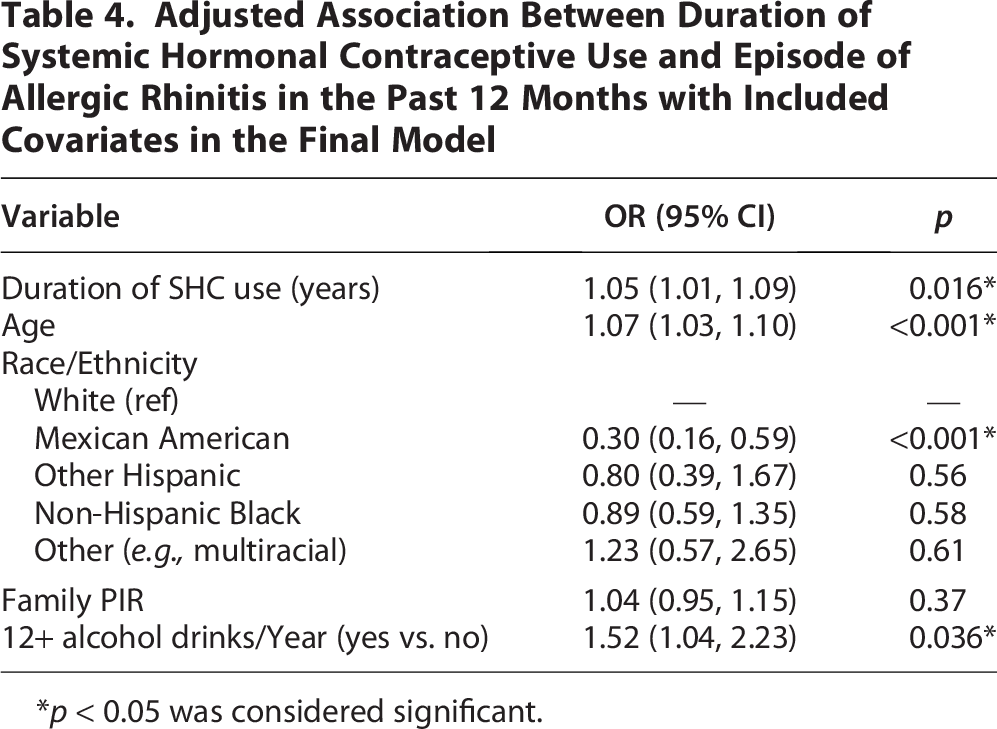
Adjusted associations of duration of SHC use and AR
As shown in Table 4, longer SHC use was associated with increased AR risk after adjusting for covariates. Each additional year of SHC use was associated with a 1.05 times higher risk of experiencing AR (OR: 1.05, 95% CI: 1.01–1.09, p = 0.016). Age (OR: 1.07, 95% CI: 1.03–1.10, p < 0.001). Alcohol consumption was also a significant predictor in this model (OR: 1.52, 95% CI: 1.04–2.23, p = 0.036). Additionally, Mexican Americans were significantly less likely to report AR (OR: 0.30, 95% CI: 0.16–0.59, p < 0.001).
Adjusted Association Between Duration of Systemic Hormonal Contraceptive Use and Episode of Allergic Rhinitis in the Past 12 Months with Included Covariates in the Final Model
*p < 0.05 was considered significant.
Adjusted associations of menopause and AR
As shown in Table 5, menopause was not significantly associated with AR after adjusting for covariates (OR: 1.13, 95% CI: 0.72–1.75, p = 0.600). Individuals with higher PIR were more likely to report AR (OR: 1.10, 95% CI: 1.02–1.18, p = 0.012). Those who completed the interview in Spanish were less likely to report AR (OR: 0.48, 95% CI: 0.26–0.90, p = 0.027). Finally, unlike with the previous two models, there was no significant difference in AR in Mexican Americans; however, the p value approached statistical significance (OR: 0.55, 95% CI: 0.31–0.99, p = 0.052). Additionally, individuals of non-Hispanic Black race/ethnicity were significantly less likely to report AR (OR: 0.63, 95% CI: 0.33–0.86, p = 0.013).
Adjusted Association Between Menopause and Episodes of Allergic Rhinitis in the Past 12 Months with Included Covariates in the Final Model

*p < 0.05 was considered significant.
Discussion
Our analysis found that a longer duration of SHC use was associated with increased odds of AR. This finding, which is unique from a large sample size representative of the U.S. population, aligns with results from a cross-sectional study in China that found that hormonal therapy use in perimenopausal women was linked to a duration-dependent increase in the risk of AR. 29 This study, however, was in perimenopausal women aged 45–55 years, in contrast to our analysis, which focused on reproductive women aged 12–40 years.
The exact duration of hormonal exposure needed to exert significant influence on airway inflammation, however, remains unclear. While our study found no significant association between current SHC use and AR, a recent U.S. cross-sectional study found that SHC use was associated with AR prevalence. 30 Additionally, a small clinical trial involving 23 patients found that women taking oral contraceptives had an increased risk of rhinitis symptoms within the 12 days of the trial. 31 This suggests there may be a more nuanced and complex interplay between estrogen and other biological factors influencing airway inflammation that warrants further investigation.
In our analysis of perimenopausal women, the odds of AR in postmenopausal women were not significantly reduced when compared with premenopausal women. A similar cross-sectional study we conducted using another U.S.-based population database, the All of Us Research Program, found that menopause independently reduced the likelihood of AR in women. 32 Notably, this dataset is designed to provide a more diverse sample of the U.S. population and thus likely represents a different population than NHANES and has a significantly larger sample of women in the 40–60-year-old age group. Additionally, the primary outcome variable (AR) was defined using the Systemized Nomenclature of Medicine and International Classification of Diseases codes (ICD-9 and ICD-10) from participants’ medical records and not self-reported as in NHANES. Generalizing to rhinitis broadly, another study of a large sample of perimenopausal women in France reported increased risk of rhinitis in postmenopausal women. 33 In a recent scoping review of studies examining the influence of estrogen on sinonasal symptoms, one Korean national survey found no association between postmenopausal duration and rhinitis but did find that older age at menopause was associated with having rhinitis; four studies reported reduced sinonasal function in menopause, and one study found no difference in sinonasal function of premenopausal and postmenopausal women. 8 These conflicting findings represent the broader inconsistencies in the limited literature and underscore the need for further investigation.
While the exact mechanisms for this potential influence of sex hormones in AR remain unclear, there is emerging evidence at the cellular level. For instance, studies have demonstrated that estrogen exposure induces significant histopathologic changes in nasal mucosa, including interepithelial edema, fibrotic tissue accumulation, squamous metaplasia, and enhanced vascularization accompanied by endothelial proliferation.34,35 Nasal epithelial cells have been shown to express estrogen receptors, which in turn may modulate disease processes. 36 Moreover, estrogen influences the activities of immune cells involved in the pathogenesis of AR. It promotes eosinophilic adhesion to human mucosal endothelial cells and their recruitment into the airway. 37 Additionally, both estrogen and progesterone upregulate the expression of H1 histamine receptor mRNA in nasal epithelial cells, 38 and have been shown to enhance immediate hypersensitivity reactions through increased mast cell degranulation. 39 Concurrent research at the translational and epidemiological levels is needed to definitively establish if female sex hormones have a role in AR.
While beyond the study’s primary scope, it is noteworthy that Mexican-American participants in SHC and AR analyses and Spanish-language respondents in the menopause and AR analyses were significantly less likely to report AR. This is consistent with previous literature suggesting a lower risk of allergic airway diseases among individuals of Mexican descent who were born outside of the United States, potentially due to differing environmental exposures, 40 genetic factors like Amerindian ancestry, or underreporting due to significant limitations to health care access. 41
This study has a few limitations. First, its cross-sectional design precludes establishing a causal relationship. Thus, the association between longer duration of SHC use and increased odds of AR identified in our study should be interpreted cautiously, particularly because it was observed in the context of otherwise null findings in the association of SHC use and menopause with AR. Also, while our multivariable model was adjusted for age as a covariate, it is important to note that the participants who had increased odds of AR were, on average, 5 years older and, as such, may have had a lengthier opportunity for cumulative exposure to SHCs. Nonetheless, the persistence of this association after adjustment for age suggests a potential duration-dependent effect of SHC use that should be further investigated. Second, the variables examined in this study were self-reported. Most notably, the primary outcome variable, hay fever/AR, was self-reported, as the NHANES database did not have more definitive diagnostic measures of interest, for example, diagnostic codes and objective data. Thus, there is the potential for especially diagnostic misclassifications, as objective data are generally stronger related to this. Self-reported data, however, have been extensively utilized for surveillance of prevalence patterns, particularly in airway conditions, and studies have shown a strong concordance between well-collected self-reported data and clinical outcomes.42,43 The NHANES database is also one of the largest ongoing surveys representative of the U.S. population, and the questions used in NHANES have been standardized and validated over several decades. Third, variable definitions were limited by the data available in NHANES. For example, the definition for menopause did not allow further granularity than separating perimenopausal participants into those who did not have a menstrual period in the last 12 months (postmenopausal) and those who had one (premenopausal). This binary classification may not reflect the full spectrum of perimenopausal transition but does still allow for meaningful comparisons of menopausal effects on health outcomes.
This study represents initial efforts to investigate the relationship of reproductive health-related factors such as SHC use and menopause with AR. There is a need for further investigation using longitudinal study designs to better investigate the relationship of reproductive health-related factors such as SHC use and menopause with AR and move beyond determining association to assessing causality. Understanding this relationship could have far-reaching clinical implications, particularly for tailoring risk assessment strategies, optimizing therapeutic interventions, and developing preventive strategies and health education, based on biological sex and reproductive health-related factors.
Conclusion
Our study found an association between longer SHC use and AR, highlighting the potential influence of female sex hormones on AR. There was, however, no significant association between current SHC use or menopausal status and AR. Further research is needed, particularly using a longitudinal study design, both at the clinical and translational levels to better understand the role of female sex hormones and reproductive health-related factors in AR.
Footnotes
Author Disclosure Statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability
The dataset analyzed during the current study is available in the online NHANES data repository.
Funding Information
Dr. Lee is supported by the National Institutes of Health (NIH) through Grant Award Number K12AR084225. This project was supported by the
