Abstract
Background:
The association between Helicobacter pylori (H. pylori) infection and metabolic disturbances remains inconsistent across existing studies. Given potential sex-specific differences in metabolic regulation, this study specifically examined the relationship between H. pylori infection and hypertension, glucose metabolism, and lipid profiles in women.
Methods:
We analyzed data from the 1999 to 2000 National Health and Nutrition Examination Survey. Participants were excluded if they were male, pregnant, had missing data, or had indeterminate H. pylori IgG results. Women were categorized as seronegative and seropositive based on antibody levels. Group characteristics were compared, and multivariable logistic regression was used to identify determinants of seropositivity.
Results:
Among 539 women, H. pylori seroprevalence was 41.37%. Independent predictors of seropositivity included elevated hemoglobin (HbA1c) (odds ratio [OR] = 1.75, 95% confidence interval [CI]: 1.11–2.76), older age (OR = 1.02 per year, 95% CI: 1.01–1.04), and lower educational attainment (OR = 0.60, 95% CI: 0.51–0.71). A significant dose-response relationship was observed between HbA1c and H. pylori infection risk (p for trend = 0.005). Women in the highest HbA1c quartile (Q4) had 2.14-fold higher odds of infection (95% CI: 1.25–3.67) compared to those in the lowest quartile (Q1). Restricted cubic spline analysis revealed a nonlinear association between HbA1c and H. pylori infection risk (p for nonlinearity <0.001).
Conclusion:
Elevated HbA1c levels, rather than other metabolic indicators or blood pressure, are significantly and dose-dependently associated with H. pylori infection risk in U.S. women.
Introduction
The World Health Organization (WHO) has classified Helicobacter pylori (H. pylori) infection as a Class I carcinogen. Data from 62 countries show that infection prevalence is highest in Africa (70.1%) and lowest in Oceania (24.4%), with national rates ranging from 18.9% (Switzerland) to 87.7% (Nigeria). 1 The global prevalence has declined from 58.2% (1980–1990) to 43.1% (2011–2022), with the greatest reduction observed in the WHO African region. 2
It is well established that H. pylori infection is associated with a range of gastrointestinal diseases, including chronic gastritis, gastric cancer, and mucosa-associated lymphoid tissue lymphoma. 3 Current evidence also suggests that H. pylori is closely linked to multiple extra-digestive conditions, such as neurological, dermatologic, hematological, ocular, cardiovascular, metabolic, allergic, and hepatobiliary diseases.4–6 However, the potential relationship between H. pylori infection and blood pressure regulation or metabolic disorders remains controversial in the current literature.
A meta-analysis of 22 studies (n = 206,911) suggested a potential association between H. pylori infection and an increased risk of metabolic syndrome and insulin resistance. 7 Nevertheless, this proposed link remains controversial, as subsequent cohort studies failed to identify a significant relationship between H. pylori and diabetes. 8
A key limitation in existing research is the lack of sex-specific analysis, despite evidence that hormonal profiles, body composition, and socioeconomic factors that may differentially impact metabolic outcomes. To address this gap, this study utilizes 1999–2000 NHANES data to focus exclusively on women, aiming to inform targeted strategies for H. pylori management and metabolic syndrome prevention in this population.
Materials and Methods
Data extraction
This cross-sectional study utilized data from the 1999 to 2000 NHANES cycle, the most recent publicly available dataset including H. pylori antibody serology. NHANES employs a nationally representative sample of the non-institutionalized U.S. civilian population. The survey protocol received approval from the Institutional Review Board for Human Subjects of the Centers for Disease Control and Prevention in the United States, and all participants provided written informed consent. Data are accessible on the NHANES website (https://www.cdc.gov/nchs/nhanes/index.htm).
From enrollment of 9,965 individuals in the 1999–2000 cycle, we excluded pregnant participants, men, and those with missing H. pylori serology results or covariate data . The final analytic sample consisted of 539 nonpregnant women, representing a well-defined cohort for sex-specific analysis (Fig. 1).
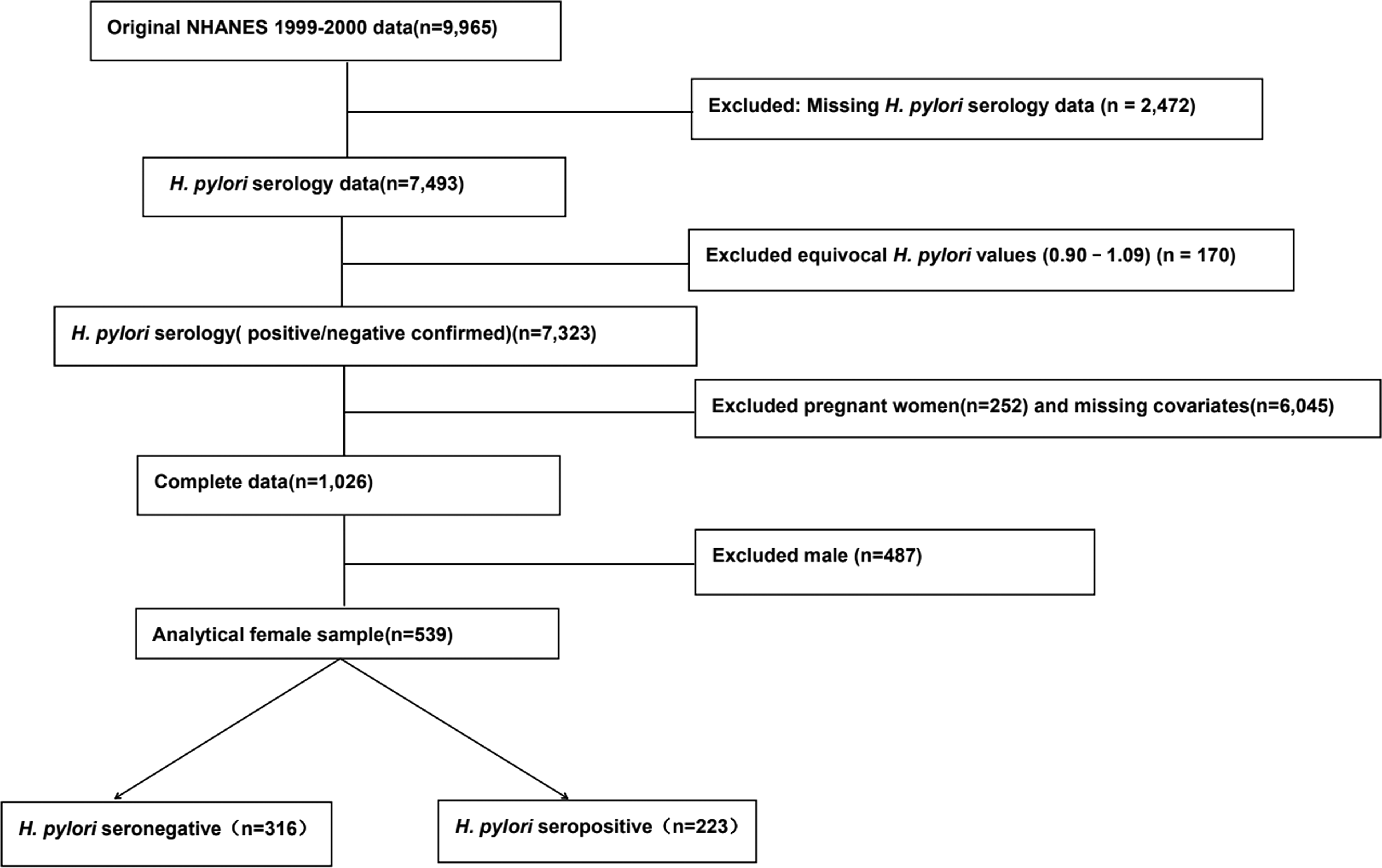
The selection flow chart of study populations.
Methods
Samples were stored at −20°C prior to analysis at designated laboratories. Triglycerides (TG), total cholesterol (TC), and high-density lipoprotein (HDL-C) were measured at the Lipoprotein Analytical Laboratory of Johns Hopkins University. Low-density lipoprotein (LDL-C) was calculated using the Friedewald formula: LDL-C = TC - HDL-C – TG/5. C-reactive protein (CRP) was measured at the University of Washington, while serum fasting glucose (GLU), insulin, and glycated hemoglobin (HbA1c) were measured at the University of Missouri-Columbia. Insulin resistance was assessed using the homeostatic model assessment for insulin resistance (HOMA-IR), calculated as: HOMA-IR = Insulin ×GLU/22.5.
Helicobacter pylori antibody
H. pylori IgG was measured at Wampole Laboratories using enzyme-linked immunosorbent assays (ELISA) for qualitative detection. Antibody levels were classified as negative (<0.90) or positive (≥1.10). To ensure analytical clarity, equivocal values between 0.91 and 1.09 were excluded from the study.
Measurement
Anthropometric measurements were performed using standardized procedures. Height and weight were measured using a stadiometer and digital scale, respectively, to calculate body mass index (BMI) as weight (kg)/height2(m2). Waist circumference was measured at the highest point of the iliac crest during minimal respiration. Blood pressure (systolic and diastolic) was recorded by certified technicians in the Mobile Examination Center after a 5-minute rest period, with the final value derived from three consecutive readings.
Assessment of covariates
Potential confounders, including demographic characteristics, lifestyle factors, and health conditions, were evaluated.9–11 Race/ethnicity was categorized as non-Hispanic White, non-Hispanic Black, Mexican American, or Other. Education was grouped into three levels: <11th grade, high school/GED, or college and above. Alcohol consumption was defined as ≥12 alcohol drinks per year (one drink = 12 oz beer, 4 oz wine, or 1 oz liquor). Health conditions, specifically hypertension and diabetes, were documented through participant self-reports.
Statistical analysis
SPSS 26.0 was used for statistical analysis. Categorical variables were described as counts and percentages. The chi-square test was used for comparisons between groups. Continuous variables were expressed as mean ± standard deviation for normally distributed data or as median(P25, P75) for non-normally distributed data. Student’s t test or the Mann–Whitney U test was applied as appropriate. Logistic regression was used to evaluate associations between H. pylori seropositivity and relevant variables. To examine dose-response relationships between H. pylori infection and HbA1c, restricted cubic spline analysis was performed. All tests were two-sided with a significance level of α = 0.05.
Results
Clinical characteristics of study participants
The final analysis included 539 women, with an H. pylori IgG seropositivity rate of 41.37%. Compared with the H. pylori seronegative group, the seropositive group had higher proportions of middle-aged and older people, lower education levels (≤11th grade), alcohol consumption, and diabetes history. Race distribution also differed significantly between the two groups (all p < 0.001). In addition, GLU, insulin, HbA1c, HOMA-IR, TG, CRP, systolic BP, and waist circumference were significantly higher in the H. pylori seropositive group. No significant differences were observed between the two groups regarding household size, TC, LDL-C, HDL-C, diastolic BP, body weight, or BMI (Table 1).
Baseline Characteristics of the Study Subjects [n(%),M(P25-P75)]

Bold value indicates a two-sided p < 0.05 was considered statistically significant.
<12 drinks per year
Logistic regression analysis
The results of the logistic regression analysis are shown in Table 2. HbA1c (odds ratio [OR] = 1.750, 95% confidence interval [CI]: 1.112–2.755), age (OR = 1.020, 95% CI: 1.005–1.036), and education level (OR = 0.601, 95% CI: 0.509–0.711) were independently associated with H. pylori infection.
A Logistic Regression Analysis

<0.001
Association between H. pylori seropositivity and HbA1c levels
After adjustment for potential confounders, the multivariate logistic regression model demonstrated a significant association between HbA1c and H. pylori infection. Compared with the lowest quartile (Q1), the odds of infection increased across successive quartiles of HbA1c, with the highest quartile (Q4) showing the greatest increase (OR = 2.144, 95% CI: 1.252–3.672, p = 0.005) (Table 3, Model 2).
Multivariable Logistic Regression Analyses of HbA1c and H. pylori Infection
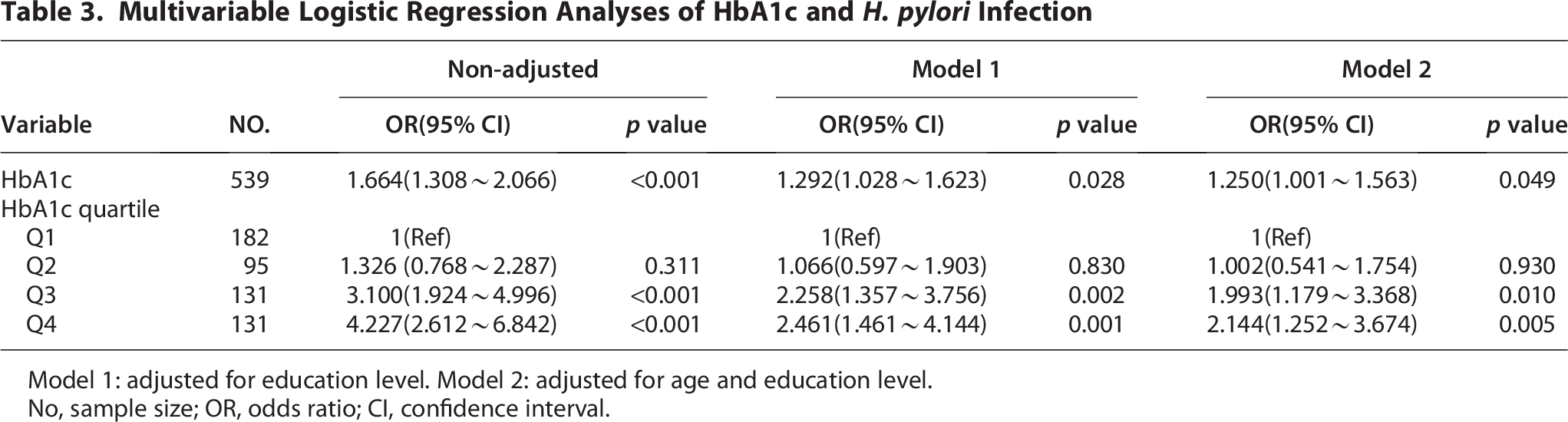
Model 1: adjusted for education level. Model 2: adjusted for age and education level.
No, sample size; OR, odds ratio; CI, confidence interval.
Dose-response relationship
The adjusted smooth curve (Fig. 2) demonstrated a nonlinear correlation between HbA1c and H. pylori infection risk (p for nonlinearity <0.001), with risk increasing steadily alongside HbA1c levels. The horizontal dashed line indicates an odds ratio of 1.0 (reference point). The vertical dashed lines indicate the threshold value of HbA1c at 5.30%.

RCS plots for H. pylori infection outcome by HbA1c.
Discussion
In the United States, the prevalence of H. pylori infection ranges from 30.0% to 41.1%, 1 which is comparable to our finding (41.37%). Lower prevalence has been reported among younger individuals, in high-income countries, and in countries with high levels of universal health coverage. 2
Despite extensive research, the link between H. pylori and metabolic syndrome remains inconsistent, likely due to population diversity, hormonal influences, and methodological variations. Women represent a distinct group, as their metabolic responses to infection may be modified by age-related hormonal changes, pregnancy-related metabolic shifts, sex-specific body composition, and immune regulation differences. These factors underscore the necessity of sex-stratified analysis. Accordingly, this study focused on non-pregnant women from the 1999 to 2000 NHANES to provide more objective and accurate estimates.
Elevated HbA1c was identified as a risk factor for H. pylori infection in women, independent of other glycemic, lipid metabolic indicators, or blood pressure measures. As a stable product of nonenzymatic glycation in red blood cells, HbA1c reflects average blood glucose levels over the preceding 2–3 months. Sustained HbA1c elevation may promote H. pylori susceptibility through three interrelated mechanisms: (1) hyperglycemia-induced microvascular dysfunction, which impairs gastric mucosal perfusion and repair; (2) autonomic neuropathy, which alters gastric motility and acid secretion to favor bacterial colonization; and (3) suppression of both innate and adaptive immunity—including neutrophil chemotaxis, macrophage function, and secretory IgA production—thereby reducing host clearance of the infection.
However, the association between H. pylori infection and glycemic control remains controversial in the current literature. A large cross-sectional study (n = 37,263) linked the infection only to dyslipidemia rather than GLU or HbA1c. 12 Several meta-analyses similarly reported no significant glycemic association in diabetic populations. 13 Our findings align with Chen et al., 14 in their cohort of 9,266 healthy individuals, H. pylori infection risk increased significantly when HbA1c levels exceeded 5.7%. Differences in HbA1c inflection points across studies may be explained by methodological variations in both HbA1c measurement and H. pylori detection. Chen et al. Used 13C/14C urea breath testing, which has high specificity but may miss low-grade infections. Our study used ELISA-based serology, which reflects cumulative exposure (higher sensitivity but potential cross-reactivity) through antibody detection . Serological methods generally report higher H pylori prevalence than nonserological methods (53.2% vs. 41.1%) 2 . Discrepancies in HbA1c measurements may also arise from varying analytical principles, operational complexity, accuracy, and specificity among laboratories.
Blood glucose regulation and H. pylori infection may interact in a bidirectional manner. Poor glycemic control increases susceptibility to H. pylori infection, which may in turn exacerbate glycemic dysregulation. This bidirectional relationship may be mediated through inflammatory cytokine release and H. pylori–induced alterations of the gut microbiota.15–17 Wan et al. 18 conducted a cross-sectional study involving 58,482 Chinese adults. Their results indicated that diabetes prevalence, GLU, and HbA1c levels were significantly higher in the H. pylori-positive group than that in the uninfected group, demonstrating a positive association between the infection and diabetes.
H. pylori infection may elevate HbA1c levels, and whether HbA1c meaningfully improves after eradication in patients with diabetes has long been a subject of investigation. Evidence suggests that the glycemic benefits of eradication are time-dependent: while a 2015 meta-analysis (Dai et al. 19 ) found no significant HbA1c improvement at 3 months post-eradication, a 5-year prospective study (Kim et al. 20 ) in 2022 reported a significant long-term reduction in HbA1c among patients with type 2 diabetes or prediabetes. These divergent findings suggest that H. pylori eradication may influence long-term glycemic control in individuals with type 2 diabetes.
Our study identified age as a risk factor for H. pylori infection (OR = 1.020, 95% CI: 1.005–1.036). This age-related risk may be attributed to several physiological mechanisms, including (1) age-related immune dysfunction—particularly reduced mucosal immunity, and (2) age-related fluctuations in steroid hormones that alter the gastric micro-environment and influence bacterial colonization. H. pylori is a unique bacterium capable of incorporating various steroids into its membrane lipids. Hosoda et al. 21 found that estradiol, androstenedione, and progesterone exert inhibitory effects on H. pylori growth. Among these steroids, with progesterone exhibiting the strongest anti-H. pylori activity. Progesterone and free cholesterol may compete for the same binding regions on the H. pylori cell surface and interfere with each other’s effects. The decline in estrogen and progesterone levels with advancing age is associated with an increased risk of H. pylori infection.
Despite providing valuable insights into the relationship between H. pylori infection and glycemic control, our study has several limitations. First, although a clear association was identified, the underlying biological pathways remain incompletely understood and require validation through experimental models, such as in vitro cell models or animal studies. Second, the lack of information on potential confounders, including physical activity, diet, 22 and detailed anthropometric measures, limits our ability to fully account for their influence on HbA1c. Third, because ELISA-based serology cannot distinguish between virulent and non-virulent H. pylori strains, hindering the evaluate of strain-specific metabolic effects, particularly those associated with CagA+/VacA+ strains.23–25 Finally, as our findings should be interpreted within their historical context, monitoring these trends in contemporary populations remains a vital public health objective. Despite these limitations, our findings highlight the necessity for future research incorporating mechanistic investigations, strain-specific analyses, and more comprehensively adjusted epidemiological approaches.
Conclusion
Elevated HbA1c levels, rather than other metabolic parameters, are significantly and dose-dependently associated with the risk of H. pylori infection in U.S. women.
Authors’ Contributions
Q.G.: Study design (lead), data analysis (lead), article revision (equal), and final approval. Y.G.: Data analysis (equal), article revision (supporting), and final approval. J.G.: Data analysis (supporting), article revision (supporting), and final approval. X.X.: Study design (equal), original draft (lead), article revision (equal), and final approval.
Footnotes
Acknowledgments
The authors acknowledge the NHANES program for providing the access data utilized in this study.
Ethics Statement
NHANES is a publicly accessible database. Studies involving human participants were approved by the National Center for Health Statistics Ethics Review Board. Approval details are available on the CDC website (![]() ). This study involved secondary analysis of anonymized data only. The research using publicly accessible and anonymized databases are exempt from review by local institutional ethics committees.
). This study involved secondary analysis of anonymized data only. The research using publicly accessible and anonymized databases are exempt from review by local institutional ethics committees.
Data Availability Statement
Author Disclosure Statement
No competing financial interests exist.
Funding Information
This study was supported by Scientific and Technological Talents Support Program of Shaanxi Provincial People’s Hospital in China (2021BJ-11).
