Abstract
Background:
Previous research on endometriosis and uterine fibroids has primarily focused on single-disease analyses and specific health outcomes. This investigation examined potential links between both conditions and early mortality.
Methods:
The analysis was based on the NHANES data in a prospective cohort design, with 5402 female participants grouped based on self-reported medical histories. Standardized interview questionnaires were used to gather baseline demographic characteristics and disease-related covariates. We conducted weighted Cox proportional hazards analyses, performing both unadjusted and adjusted regression models. Further stratification was conducted to examine potential variations across subgroups.
Results:
A total of 273 premature deaths were recorded. Endometriosis was significantly associated with premature mortality in the unadjusted model (hazard ratio [HR] = 2.15, 95% confidence interval (CI) 1.35–3.42), with persistent significance after demographic adjustment (HR = 1.68, 95% CI 1.08–2.60) and full adjustment (HR = 1.77, 95% CI 1.15–2.75). Uterine fibroids showed initial significance (unadjusted HR = 1.77, 95% CI 1.18–2.68) but were weakened after demographic adjustment (adjusted HR = 1.07, 95% CI 0.69–1.66). Subgroup analysis results indicated that the risk of premature mortality in patients with endometriosis varied significantly across different populations. The significance was elevated in individuals with less than a high school education (HR = 2.99, 95% CI 1.53–5.84) and nondrinkers (HR = 2.45, 95% CI 1.27–4.73) in the stratified analysis of uterine fibroids. No other significant interactions were observed.
Conclusions:
Our study demonstrated that endometriosis independently contributes to heightened premature death risk. The link between uterine fibroids and premature mortality presents risk patterns specific to subgroups.
Introduction
Endometriosis and uterine fibroids are the most common benign gynecological disorders in women of reproductive age, with clear clinical implications.1–3 Endometriosis, defined by the growth of endometrial-like tissue outside the uterus cavity, impacts roughly 10% of women globally during their childbearing years. 4 Its primary symptoms include painful menstruation, persistent pelvic discomfort, and infertility, frequently paired with long-term mental health challenges. 5 Uterine fibroids are benign tumors derived from uterine smooth muscle cells, with a lifetime prevalence of 40%–89% among premenopausal women. This prevalence varies depending on the diagnostic method, study cohort, and age distribution. 6 Although most of them are asymptomatic, some patients require medical intervention due to symptoms such as menstrual abnormalities and abdominal mass. 6 Beyond imposing direct reproductive health challenges, such as reduced fertility and diminished quality of life, these two conditions share key pathophysiological features, including persistent low-level inflammation and hormone dysregulation.7,8 These underlying factors are known to link to noncommunicable diseases such as cardiovascular disease (CVD), metabolic syndrome, and ovarian malignancy.9–13 This suggests that these diseases may elevate long-term health risks via these pathways and potentially heighten the likelihood of premature death (defined as nonaccidental death before age 70).
There are still shortcomings in the current research on the association between these two diseases and premature death. Previous studies have been mostly limited to analyses of single diseases and specific health outcomes, such as endometriosis and increased risk of ovarian cancer, uterine fibroids, and adverse pregnancy outcomes, but the lack of systematic assessment of the ultimate outcome of “all-cause premature death” makes it difficult to reflect the overall impact of the disease on women’s long-term survival.14,15 Current research on this topic is constrained by several methodological shortcomings, including limited sample sizes (predominantly drawn from single-center clinical cohorts), relatively brief follow-up durations (typically under 10 years), and inadequate adjustment for potential confounders such as lifestyle factors (smoking, alcohol use) and comorbid conditions. More importantly, there is insufficient existing evidence regarding key scientific issues such as whether these two diseases independently increase the risk of premature death, the differences in risk levels between uterine fibroids (high incidence but mostly asymptomatic) and endometriosis (low incidence but significant symptoms), and whether there is a synergistic effect when both conditions are comorbid. Therefore in-depth research based on large-scale populations is urgently needed.3,4,16,17
Using data from the NHANES (1999–2006), we intended to apply a multivariate Cox proportional hazards regression model to evaluate the associations between endometriosis/uterine fibroids and the risk of premature mortality among women in the United States. We adopted key potential confounders to adjust the analysis, including age, ethnicity, socioeconomic status, smoking history, alcohol consumption, and hormone therapy history. Furthermore, we sought to conduct comprehensive subgroup analyses to verify the robustness of the findings. This investigation aims to provide new epidemiological insights into the long-term health consequences of these prevalent gynecological disorders, thereby informing future clinical strategies for early screening and personalized risk management in susceptible populations.
Materials and Methods
Study population and outcome
NHANES, administered by the National Center for Health Statistics (NCHS) under the Centers for Disease Control and Prevention, is an ongoing, multiphase investigation designed to evaluate the health and nutritional profiles of U.S. residents aged 2 months and older. This cross-sectional initiative uses standardized interviews and physical assessments to gather detailed information on demographics, socioeconomic status, dietary habits, physiological metrics, and laboratory analytes.
For this study, we analyzed data from four consecutive NHANES cycles (1999–2006). Mortality outcomes were accessed via data linkage with the NCHS, and all datasets are publicly accessible through the NCHS official website (data release as of December 31, 2019; available at https://www.cdc.gov/nchs/data-linkage/mortality-public.htm). These files link NHANES participants to death records from the National Death Index via a probabilistic record linkage algorithm. The study protocol was approved by the institutional review board (Protocols No. 98–12, 2005–06, 2011–17, and 2018–01), and written informed consent was obtained from all participants or their legal representatives before enrollment. Premature death was defined as death before the age of 70 years.
Determining endometriosis and uterine fibroids
Endometriosis status was ascertained through participants’ response to the “RHQ360” item: “Has a doctor or other health professional ever told you that you had endometriosis?” Participants who reported “yes” were categorized as endometriosis cases, whereas “no” respondents formed the reference group. Similarly, self-reported uterine fibroids were diagnosed using the “RHQ380” questionnaire item: “Has a doctor or other health professional ever told you that you had uterine fibroids?” Respondents who answered “yes” were classified as uterine fibroid cases, and those answering “no” comprised the reference group without self-reported uterine fibroids. Respondents with refused or missed answers for either condition were excluded from the respective analyses. Both questionnaires were restricted to female participants aged 20–54 years.
Assessment of covariates
This study obtained core demographic variables through structured NHANES questionnaires. Baseline demographic characteristics included age, ethnicity, education level, poverty–income ratio, smoking history, alcohol consumption, and marital status. Ethnicities were categorized into 4 groups as follows: non-Hispanic White, non-Hispanic Black, Mexican American, and Other. Educational level was divided into three categories as follows: less than high school, high school or equivalent, and college or higher. 18 The poverty–income ratio is a quantitative variable ranging from 0 to 5, with higher values indicating better family economic status. Participants’ smoking history was classified as nonsmokers or smokers based on their responses regarding having smoked at least 100 cigarettes in their lifetime and current smoking behavior. 19 Alcohol consumption was categorized as nondrinkers or drinkers. 20 Marital status was categorized as married or living with a partner, never married, and single (divorced/widowed/separated). Disease-related covariates, including age at menarche, oral contraceptive use, female hormone use, and history of coronary heart disease, were derived from variables “RHQ010,” “RHQ420,” “RHQ540,” and “MCQ160C,” respectively.10,21–24
The flowchart of participant inclusion is shown in Figure 1.

The flowchart of participant inclusion.
Statistical analysis
NHANES uses a complex multistage sampling design; therefore, all data were weighted according to the NHANES scheme to ensure national representativeness. Baseline characteristics were evaluated using comparative analyses: (1) Wilcoxon rank-sum tests were used in continuous variables with skewed distribution; and (2) chi-square tests were used in categorical variables after adjusting the sampling weights. To describe statistics, continuous variables with skewed distribution were presented as weighted median (M) (Q1, Q3) and categorical variables as counts (n, weighted %).
We used a weighted univariate and multivariate Cox proportional hazard regression model to examine the association between gynecological diseases (endometriosis and uterine fibroids) and premature death. Three hierarchically adjusted models were constructed in the analysis: Model 1 (crude association, unadjusted hazard ratio [HR]), Model 2 (demographically adjusted, such as age and ethnicity), and Model 3 (fully adjusted). In the endometriosis analysis, patients diagnosed with uterine fibroids were excluded from the group (n = 4722) to avoid contamination from comorbidity. Similarly, in the uterine fibroids analysis, patients with endometriosis were excluded (n = 5033). This strategy ensured that the groups were mutually exclusive and that the risk estimates were not biased by the presence of the other disease. These exclusions were implemented to minimize potential bias that could arise from including participants with other gynecological conditions in the analyses. All analyses were performed using R software, and a two-tailed p value < 0.05 was considered statistically significant. Data processing and analysis were performed using R version 4.5.1 (Great Square Root) along with Zstats v0.90 (www.medsta.cn/software).
Results
Baseline characteristics of the study cohort
We calculated follow-up time from the examination date to either the date of death or December 31, 2019. The median follow-up time was 16.7 years (IQR 14.6–18.7 years). The mean age at death among deceased women was 55 years. Among the 5402 women included in the analysis, 369 (6.79%) and 680 (12.59%) reported a history of endometriosis and uterine fibroids, respectively. The endometriosis group had an older median age (41 years vs. 37 years), whereas the age difference was more pronounced in the uterine fibroids group (median age: 45 years vs. 36 years) (Table 1). Women without self-reported uterine fibroids were younger, with 73.84% (n = 3745) aged < 45 years, whereas women with this condition had a relatively higher proportion in the middle-aged group (53.76%) (Table 1). The prevalence of endometriosis among women with a college education or higher was 58.77% (Table 1). Women without self-reported endometriosis were mostly aged ≥ 13 years at menarche (51.42%), whereas those with self-reported endometriosis were mostly aged <13 years at menarche (56.10%) (Table 1). Women with endometriosis or fibroids were mostly married or living with their partners (68.14% vs. 71.14%). Most women had a history of oral contraceptive use, whereas most had no history of female hormone use. Regarding female hormone use, the endometriosis group had a significantly higher rate than the control group (41.95% vs. 13.37%), and a similar trend was observed in the uterine fibroid group (37.18% vs. 12.49%) (Table 1). The proportion of self-reported endometriosis patients with a history of oral contraceptive use was 91.53%. Women without endometriosis or fibroids accounted for a higher proportion of never-smokers (58.02% vs. 57.92%) (Table 1). No statistically significant differences were observed in the distribution of coronary heart disease (p value 0.282 vs. 0.058) or alcohol consumption (p value 0.204 vs. 0.771) between the two groups (Table 1).
Baseline Demographic Characteristics According to Self-Reported Endometriosis and Uterine Fibroids Among 5402 Women

Data are presented as weighted % or weighted median.
Widowed/divorced/separated; bother Hispanic, other ethnicities, and multiracial.
M, weighted median; Q1, 1st Quartile; Q3, 3rd Quartile.
Results analysis
Among the 5402 women included in the analysis, a total of 273 (4.98%) deaths were reported (Table 2). Among the 369 women with self-reported endometriosis, 35 (8.84%) deaths were recorded compared with 238 (4.60%) deaths among 5033 women without endometriosis (Table 2). For uterine fibroids, 61 (7.68%) deaths occurred among the 680 women with the condition compared with 212 (4.54%) deaths among 4722 women in those without (Table 2).
The Mortality Data According to Self-Reported Endometriosis and Uterine Fibroids

%, weighed %.
The results showed that patients with self-reported endometriosis but without uterine fibroids had an increasing risk of premature mortality. In Model 1 (unadjusted for any covariates), there was an HR of 2.15 (95% confidence interval [CI] 1.35–3.42, p = 0.001) (Table 3). In Model 2 (adjusted for basic demographic characteristics), the association remained significant (HR = 1.68, 95% CI 1.08–2.60, p = 0.020) (Table 3). Model 3, which further adjusted for potential confounding factors (including coronary heart disease, oral contraceptive use, female hormone use, and age at menarche), showed an enhanced risk (HR = 1.77, 95% CI 1.15–2.75, p = 0.010) (Table 3).
Association Between Endometriosis/Uterine Fibroids and Premature Death
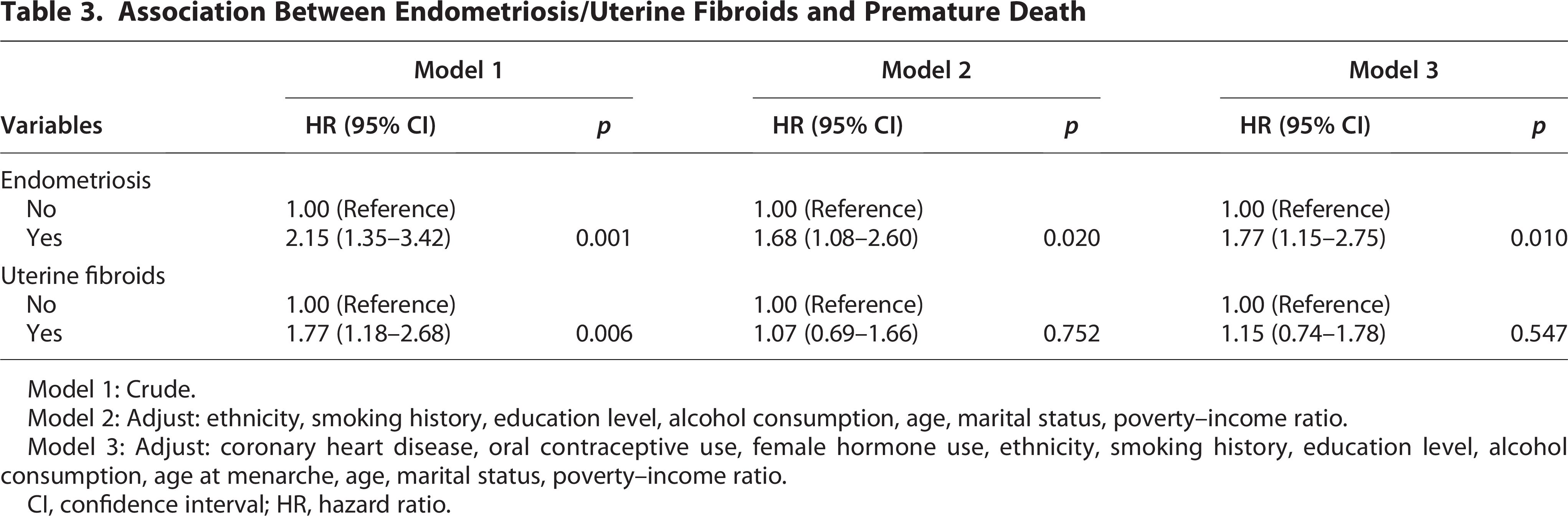
Model 1: Crude.
Model 2: Adjust: ethnicity, smoking history, education level, alcohol consumption, age, marital status, poverty–income ratio.
Model 3: Adjust: coronary heart disease, oral contraceptive use, female hormone use, ethnicity, smoking history, education level, alcohol consumption, age at menarche, age, marital status, poverty–income ratio.
CI, confidence interval; HR, hazard ratio.
For uterine fibroids, unadjusted analyses showed a significant link between self-reported cases (without endometriosis) and premature mortality (HR = 1.77, 95% CI 1.18–2.68, p = 0.006) (Table 3).However, after adjusting for basic demographic factors (including age) in Model 2, the statistical significance was weakened (HR = 1.07, 95% CI 0.69–1.66, p = 0.752) (Table 3). In model 3, which is fully adjusted, there is an HR of 1.15 (95% CI 0.74–1.78, p = 0.547) (Table 3).
Subgroup analysis
To assess the robustness and heterogeneity of the endometriosis/uterine fibroids-mortality association, we conducted stratified analyses across key population subgroups. The population was stratified by age groups, smoking history, education level, alcohol consumption, and marital status. Each subgroup analysis was adjusted for all original covariates except the stratification variables.
Subgroup analysis revealed notable variations in premature mortality risk among endometriosis patients across different subgroups. Age group analysis showed that middle-aged women had a more significant risk increase (HR = 1.89, 95% CI 1.07–3.36) (Fig. 2). By smoking history, the risk was significantly elevated in never-smokers (HR = 2.64, 95% CI 1.33–5.23), whereas the HR of smokers was 1.25 (Fig. 2). Analysis by educational level revealed that individuals with a high school education had the highest risk (HR = 2.47, 95% CI 1.12–5.45), and the HRs for those with less than a high school and college/higher education were 1.44 and 1.26, respectively (Fig. 2). Alcohol consumption analysis showed a significant risk level in drinkers (HR = 1.72, 95% CI 1.02–2.93), whereas no significance was shown in never-drinkers (Fig. 2). When stratified by marital status, women who were married or living with a partner showed the highest risk (HR = 2.07, 95% CI 1.17–3.68), whereas single and never married individuals had HRs of 0.77 and 1.27, respectively (p for interaction 0.185) (Fig. 2).
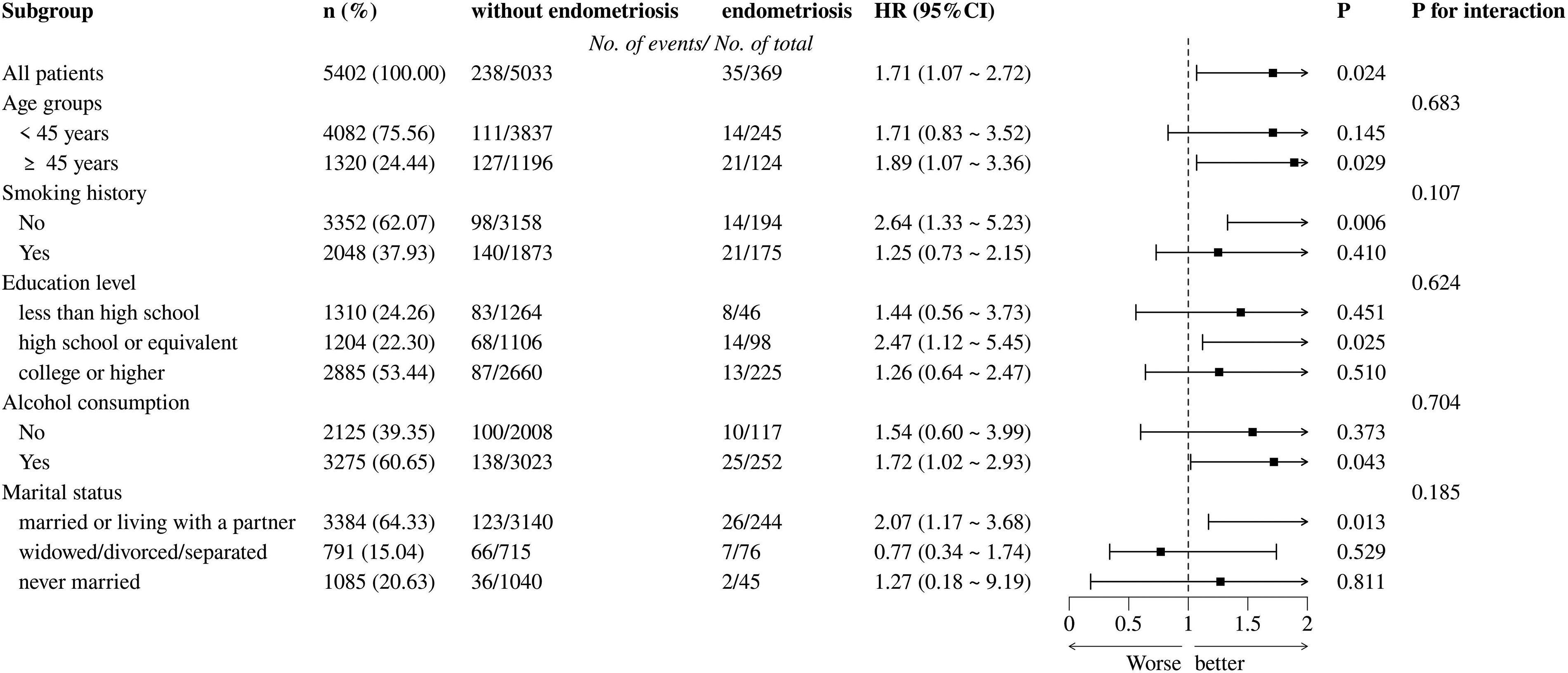
Risk of premature mortality according to self-reported endometriosis among 5402 women (NHANES 1999–2006). The population was stratified by age groups, smoking history, education level, alcohol consumption, and marital status. Models were adjusted for age at menarche (continuous), poverty–income ratio (continuous), ethnicity, coronary heart disease, oral contraceptive use, and female hormone use.
In the subgroup analysis of uterine fibroids, there was an elevated risk of premature mortality in individuals with a less than high school education (HR = 2.99, 95% CI 1.53–5.84); however, significance was no longer observed in other education groups (Fig. 3). Nondrinkers exhibited an enhanced HR of 2.45 (95% CI 1.27–4.73), with drinkers showing an HR of 1.16 and a p for interaction of 0.105 (Fig. 3). No statistically significant interactions were found in other subgroups, including those stratified by marital status, age groups, and smoking history (all p > 0.05) (Fig. 3).

Risk of premature mortality according to self-reported uterine fibroids among 5402 women (NHANES 1999–2006). The population was stratified by age groups, smoking history, education level, alcohol consumption, and marital status. Models were adjusted for age at menarche (continuous), poverty–income ratio (continuous), ethnicity, coronary heart disease, oral contraceptive use, and female hormone use.
Sensitivity analysis
For covariates with less than 20% missing data, multiple imputation methods were performed. Then we conducted sensitivity analyses using the above datasets to determine the robustness of the models. The results showed that this method merely affected the correlation between endometriosis and premature mortality. In model 1 (crude), there is an HR of 2.15 (95% CI 1.33–3.42, p = 0.001) and a consistent significance in model 2 and model 3 (HRs 1.75 vs. 1.81) (Table 4). In the sensitivity analysis of uterine fibroids and premature death, there were also similar results compared with the unimputed datasets analysis. An original significance was observed in the crude model (HR = 1.77, 95% CI 1.18–2.68, p = 0.006), whereas the significance was weakened after adjusting for the covariates (an HR of 1.07 in model 2 and an HR of 1.12 in model 3) (Table 4). Therefore, the association between these two abnormal conditions and premature death would be effectively validated.
Association Between Endometriosis/Uterine Fibroids and Premature Death (Imputed Datasets)

Model 1: Crude.
Model 2: Adjust: ethnicity, smoking history, education level, alcohol consumption, age, marital status, poverty–income ratio.
Model 3: Adjust: coronary heart disease, oral contraceptive use, female hormone use, ethnicity, smoking history, education level, alcohol consumption, age at menarche, age, marital status, poverty–income ratio.
Discussion
In this large, prospective cohort study, women with endometriosis faced a significantly elevated risk of all-cause premature mortality, whereas no such association was observed for uterine fibroids. The persistence of this association after comprehensive adjustment for all available confounders suggests that the underlying pathophysiology of endometriosis may contribute to long-term health risks beyond the reproductive system. Notably, HR varies with the degree of model adjustment, indicating that traditional risk factors, such as age, age at menarche, and female hormone use, the prevalence of which was significantly higher in both gynecological conditions, partially confound the true association. Furthermore, the varying risk across demographic subgroups, such as the pronounced risk among never-smokers with endometriosis, suggests potential effect modifications that warrant further investigation. By demonstrating this risk in a nationally representative sample and contrasting it directly with the null findings for fibroids, our study provides a robust contribution to the literature.
Current evidence on the association between endometriosis/uterine fibroids and premature mortality remains scarce, and our findings should be interpreted in the context of the existing literature. Our results align significantly with the large prospective cohort study by Wang et al. (2024) based on the Nurses’ Health Study II. 4 Both studies report a significantly elevated risk of all-cause premature mortality specifically associated with endometriosis, but not with uterine fibroids. This convergence is notable even considering methodological differences: while Wang et al. performed more rigorous, clinically confirmed diagnoses (laparoscopic, ultrasound, or hysterectomy), our study used self-reported data. 4 The magnitude of the association for endometriosis in our study (HR = 1.77) was somewhat higher than that reported by Wang et al. (HR = 1.31), a difference potentially attributable to variations in study population characteristics, follow-up duration, or confounder adjustment strategies. 4 In stark contrast, our conclusions directly oppose those of the nationwide Finnish registry cohort by Saavalainen et al. (2019), which found substantially lower all-cause mortality (MRR = 0.73) among women with surgically verified endometriosis. 17 Several fundamental methodological distinctions may explain this profound discrepancy. The Finnish study captures an inherently healthier cohort susceptible to the “healthy patient” effect and subject to extensive medical surveillance and intervention. Crucially, their finding of reduced mortality is severely confounded by the high rate of preexisting gynecological organ removals (38% in the endometriosis cohort vs. 3% in references), making it impossible to determine if the lower risk stems from the disease itself or the surgical interventions. 17 Meanwhile, unmeasured lifestyle confounders in their registry data likely lead to an underestimation of the true mortality risk. 17 In addition, the Finnish universal health care system could facilitate earlier detection and management of comorbidities, potentially offsetting the mortality risk associated with endometriosis itself. Ultimately, the convergence of findings from two large prospective cohorts (ours and Wang et al.) against the contrasting results from the administrative registry of Saavalainen et al. underscores the critical influence of methodology and highlights the specific, elevated risk faced by women with endometriosis in the populations we studied. Our study incorporates the following methodological enhancements to strengthen the analytical generalizability of the findings: when analyzing the association between uterine fibroids and premature death, hormone therapy history, a critical confounding factor, was systematically controlled.
The elevated mortality risk observed in endometriosis patients likely stems from the disease’s fundamental biological mechanisms, particularly its chronic inflammatory nature and associated hormonal perturbations. Prior research has confirmed that endometriosis is inherently a chronic inflammatory condition, marked by persistent increases in various pro-inflammatory cytokines (e.g., IL-6, CRP).25–29 These inflammatory agents contribute to the progression of atherosclerosis, in turn raising the likelihood of premature mortality from CVD.30–32 Furthermore, beyond its inflammatory profile, endometriosis—particularly certain subtypes—has been confirmed to be associated with an increased risk of ovarian cancer (especially Type I), which may represent another factor affecting the long-term survival of patients.14,33 In terms of treatment, gonadotropin-releasing hormone antagonists are an important treatment for endometriosis and relieve pain symptoms by inhibiting the menstrual cycle.34,35 However, their procedure induces a hypoestrogenic state, which may lead to adverse effects such as reduced bone density. 36 Notably, osteoporotic fractures not only severely impair quality of life but also are significantly associated with increased mortality. 37 In contrast, as benign tumors, uterine fibroids have biological properties that limit their aggressiveness and ability to trigger systemic inflammatory activation. Most patients exhibit mild clinical manifestations, and for symptomatic cases (e.g., abnormal menstrual periods), surgical resection effectively controls the condition. 6 This therapeutic responsiveness may explain the lack of significant association with premature death risk. Regarding confounding factors, age at menarche, a key indicator of reproductive health, exhibits a U-shaped association with CVD mortality, where both early (<12 years) and late (>15 years) menarche increase risk. 38 Similarly, all-cause mortality shows a nonlinear relationship with age at menarche. 39 Furthermore, oral contraceptive pills, a widely used contraceptive method, have dual health effects: short-term use may confer protective benefits while long-term use elevates the risk of thromboembolic disease.40–42 These factors were systematically controlled for in our study, enhancing the reliability of the results.
Our study offers several methodological advantages: First, the utilization of NHANES data ensures that our results are generalizable to the noninstitutionalized U.S. population, significantly improving external validity compared with single-center studies; second, weighted COX proportional hazards regression models were implemented to properly account for the complex sampling design, ensuring the accuracy of statistical inference; in addition, a mutually exclusive grouping design effectively controlled for bias arising from coexisting diseases. These innovative statistical approaches provide a methodological foundation for the reliability of research conclusions. However, several limitations warrant explanation: First, disease identification in this study depends primarily on self-reported diagnoses, a method that may introduce misclassification bias. Second, due to limitations in the survey questionnaire, we were unable to access indicators of clinical severity, such as rASRM staging for endometriosis or symptomatic features of uterine fibroids.2,43 Third, despite controlling for numerous confounding variables (such as nutrition and dietary habits), residual confounding cannot be ruled out due to inherent constraints of the study design, especially the lack of critical clinical data like surgical histories and pain evaluation measures.4,44 It is worth noting that previous studies have confirmed that the risk of CVD and hypertension in patients with endometriosis is significantly increased, suggesting that endometriosis should be integrated into the female cardiovascular risk assessment system. 24 Future research directions include the following: (1) conducting prospective cohort studies to systematically evaluate the impact of different hormone therapy regimens on prognosis; (2) deeply exploring the predictive value of inflammatory markers (e.g., neutrophil/lymphocyte ratio); and (3) developing more accurate risk prediction models by integrating clinical indicators and biomarkers.45,46
Conclusions
Our findings clearly show that endometriosis is linked to a significantly higher risk of premature mortality, whereas the connection between uterine fibroids and early death remains to be confirmed through additional research. There were several limitations, such as the potential misclassification bias introduced by self-reported methods of disease diagnosis. Our results highlight the need for greater focus on endometriosis and uterine fibroids. We should make efforts to enhance patients’ physical and psychological well-being. Moving forward, developing more precise and accessible diagnostic tools for these conditions will be crucial to enabling timely clinical intervention.
Authors’ Contributions
X.G.: Data curation, investigation, writing—original draft, conceptualization, writing—review and editing, software, resources, and formal analysis. R.L.: Conceptualization, writing—review and editing, methodology, validation, and supervision. J.G.: Conceptualization, visualization, writing—review and editing, supervision, and project administration. All authors read and approved the final article.
Footnotes
Acknowledgments
The authors extend profound thanks to all participants from the NHANES.
Author Disclosure Statement
The authors declare that they have no competing interests.
Funding Information
This work was sponsored by the Natural Science Foundation of Shanghai Municipality (Grant No. 23ZR1408100).
Ethics Approval and Consent to Participate
The NHANES study protocol was reviewed and approved by the National Center for Health Statistics Research Ethics Review Board. All survey participants provided written informed consent. As this secondary analysis exclusively utilized de-identified, publicly available data, it was exempt from requiring additional ethical approval.
Consent to Publication
All authors have read and approved the final version of the article and consent to its publication.
Availability of Data and Materials
All NHANES data files and related documentation are available to the public through the NHANES website (www.cdc.gov/nchs/nhanes/), including demographic, examination, laboratory, and questionnaire data. Mortality outcomes were accessed via data linkage with the NCHS, and all datasets are publicly accessible through the NCHS official website (data release as of December 31, 2019; available at ![]() ).
).
