Abstract
Background:
Secondary meconium-stained amniotic fluid (MSAF) is reportedly associated with adverse maternal and neonatal outcomes. In this study, we aimed to investigate the differences in maternal and neonatal outcomes between primary and secondary Grade III MSAF patients.
Methods:
This retrospective analysis enrolled women who experienced Grade III MSAF during childbirth between September 2020 and June 2023 in our hospital. We collected and compared the baseline characteristics, as well as maternal and neonatal outcomes, between the primary and secondary cases. For parameters with significant statistical differences and clinical significance, we performed multivariable logistic regression analysis to explore potential risk factors for secondary MSAF.
Results:
A total of 517 patients with Grade III MSAFs were included in the study. We found no significant difference in neonatal outcomes between the primary and secondary groups. More primary grade III MASFs occurred in the first stage of labor (53.1% vs. 28.8%, p < 0.001). A greater percentage of patients who underwent cesarean section (28.8% vs. 17.5%, p = 0.001) were in the primary group. More women with intrapartum fever were in the secondary group (19.8% vs. 28.1%, p = 0.027), but there was no significant difference in laboratory routine blood test results. This association remained significant after controlling for background confounders. However, when combined with an abnormal fetal heart rate (FHR) pattern, both the primary (9.0% vs. 33.3%, p < 0.001) and secondary (12.6% vs. 25.0%, p = 0.009) groups had increased rates of neonatal intensive care unit (NICU) admission.
Conclusions:
Primary Grade III MSAF is associated with an increased rate of cesarean delivery, while Grade III MSAF elevates the risk of intrapartum fever. Secondary Grade III MSAF alone does not increase the risk of NICU admission, but the presence of an abnormal FHR is a significant indicator.
Introduction
The term “Meconium” originates from the Greek word “mekoni,” meaning “poppy juice” or “opium-like.” Meconium, as the fetal colonic content, primarily consists of water (up to 80%), which also contains intestinal epithelial cells, bile pigments, and bile acids, giving it the characteristic greenish-yellow color. When meconium is passed into the amniotic fluid, it can cause staining, resulting in meconium-stained amniotic fluid (MSAF). 1 The prevalence of MSAF is approximately 15%, based on data from two large-scale studies.2,3 The possible risks for MSAF include prolonged gestation (especially post-term), placental insufficiency, gestational diabetes, hypertensive disorders of pregnancy, and oligohydramnios. 4
There are many types of MSAF. Like thin or thick MSAF, depending on the amount of meconium in the amniotic fluid, but practically, based on the practical experience of obstetricians and midwives. In addition, the MSAF is also commonly categorized into grades I to III, the definition of Grade I (translucent, light green or yellowish), Grade II (opalescent, deep green, and light yellow or brown), and Grade III (opaque and deep green). 2 A Grade III MSAF is often considered equivalent to a thick MSAF because it is associated with a greater risk of neonatal intensive care unit (NICU) admission, respiratory complications (such as meconium aspiration syndrome, MAS), and the need for neonatal ventilation.1,4,5 Based on data statistical analysis, approximately 3%–12% of the babies born through MSAF develop MAS. 6 But the morbidity of MAS is decreasing due to obstetric interventions performed before 41 weeks in cases of MASF. 7 Additionally, Grade III MSAF is also linked to a higher rate of cesarean section, intrapartum fever, and even intraamniotic infection. 1
However, in some studies, MSAF can also be classified as primary (presence of MSAF at the time of membrane rupture) or secondary (a change in the color of the amniotic fluid from clear to meconium). It was shown that secondary MSAF is associated with a greater rate of operative vaginal delivery, NICU admission, and composite neonatal outcomes.8,9
Therefore, the objective of this study was to collect and analyze recent three-year data on maternal and neonatal outcomes in pregnant patients with Grade III MSAF, differentiating between primary and secondary cases.
Methods
Data collection
This retrospective study was conducted in the obstetrics department of Jiangsu Province Hospital, the First Affiliated Hospital of Nanjing Medical University, covering the period from January 2020 to June 2023. The study included all pregnant women between 37 and 41+ 6 gestational weeks with Grade III MSAF, excluding cases of elective cesarean sections, inductions after stillbirths, incomplete data, and fetal renal abnormalities.
Baseline data collected included maternal age, body mass index (BMI), gestational age, primipara rate, use of assisted reproductive technology, and complications such as diabetes mellitus, preeclampsia, cord or placental abnormalities, positive group B Streptococcus, and premature rupture of membranes (PROM). Maternal outcomes were tracked from the discovery of Grade III MSAF to childbirth.
The assessment of amniotic fluid was conducted by senior obstetricians and midwives, who classified it as either “thin” (normal consistency with a greenish or yellowish color) or “thick” (viscous, tenacious, and opaque). The volume of amniotic fluid was estimated by a midwife or doctor.
Additional recorded variables included the rate of artificial rupture of membranes, labor mode, intrapartum regional analgesia rate, intrapartum fever rate (>37.5°C), abnormal fetal heart rate (FHR) based on the 2008 National Institute of Child Health and Human Development’s three-tier classification system, 10 delivery method, vaginal bleeding over 24 hours (measured by gravimetry), and routine blood tests conducted 24 hours postpartum. FHR abnormalities are diagnosed by clinicians, with at least 40 minutes of continuous fetal monitoring considered abnormal. FHR abnormalities are categorized into three classes. Class I Abnormal FHR: Requires simultaneous fulfillment of the following criteria: Baseline heart rate of 110–160 bpm; Moderate baseline variability; Absence of late decelerations and variable decelerations; Presence or absence of early decelerations; and Presence or absence of accelerations. Class III Abnormal FHR: Includes any one of the following: Baseline without variability, accompanied by any one of the following: recurrent late decelerations; recurrent variable decelerations; FHR bradycardia (FHR baseline <110 bpm); and Sine wave pattern. All other cases, excluding the first and third categories of abnormal FHR, are classified as Category II EFM.
Neonatal outcomes monitored included sex, birth weight, rate of congenital malformations, Apgar scores at 1 and 5 minutes, and NICU admission rate. The reasons for NICU admission are obtained from the electronic medical records.
Statistical analysis
Statistical analysis was conducted using the SPSS statistical package (version 29.0, SPSS Inc., Chicago, IL, USA). The normality of continuous variables was assessed using the Shapiro–Wilk test. Normally distributed data are presented as the mean ± standard deviation and were compared using Student’s t-test. Nonnormally distributed variables are reported as medians (interquartile ranges) and were compared using the Mann–Whitney U test. Categorical variables are presented as frequencies (percentages) and were compared using the chi-square test or Fisher’s exact test. Statistical significance was set at p < 0.05. For logistic regression, we used the SPSS statistical package. Potential confounding variables include maternal age and gestational age. We reported the results as odds ratios (ORs).
Results
Despite sharing the diagnosis of Grade III MSAF, the primary and secondary MSAF groups exhibited distinct maternal risk profiles and management patterns, culminating in different modes of delivery. A total of 517 patients with Grade III MSAFs were included in the study. A comparison between the primary and secondary groups revealed some notable findings. Maternal age was older, and the rate of preeclampsia was also greater in the primary group (Table 1). The secondary group exhibited a slightly prolonged gestation period, and a greater rate of cord or placental abnormalities and PROM (Table 1). Additionally, there was greater usage of oxytocin in the secondary group (Table 2). In terms of birthing outcomes, the majority of deliveries were via the vagina in both the primary and secondary MSAF groups, and the primary group had a lower percentage of vaginal births (55.8% vs. 66.2%) and a greater percentage of cesarean section deliveries (28.2% vs. 16.7%) than did the secondary group (Table 2). However, the primary group had fewer cases of intrapartum fever (16.7% vs. 27.2%) and a greater utilization of intrapartum regional analgesia. There were no significant differences observed between the two groups in terms of vaginal bleeding volume, routine blood tests conducted 24 hours after delivery, or neonatal outcomes (Table 2). Table 3 presents the independent associations of secondary MSAF and maternal outcomes after controlling for maternal age, pregnant gestation. These included artificial rupture of membrane (adjusted OR [aOR] = 0.421, 95% confidence interval [CI]: 0.301–0.589), intrapartum regional analgesia (aOR = 1.652, 95% CI: 1.154–2.363), intrapartum fever (aOR = 1.866, 95% CI: 1.264–2.754), and cesarean (aOR = 0.512, 95% CI: 0.346–0.757).
The Differences of Baseline Characteristics Between Primary and Secondary Grade III MSAF

ART, Assisted Reproductive Technology; BMI, body mass index; DM, diabetes mellitus; GBS, Group B Streptococcus; IQR, interquartile range; MSAF, meconium-stained amniotic fluid; PROM, Premature Rupture of Membranes.
The Differences of Maternal and Neonatal Outcomes Between Primary and Secondary Grade III MSAF

AF, amniotic fluid; CRP, C-reactive protein; FHR, fetal heart rate; NICU, neonatal intensive care unit; SD, standard deviation; WBC, white blood cell.
Multivariable Regression Analysis for Independent Associations with Secondary MSAF

Adjusted for: maternal age, pregnant gestation.
aOR, adjusted odds ratio.
The presence of an abnormal FHR delineated distinct risk profiles and clinical pathways between the primary and secondary MSAF groups. Regarding patients with abnormal FHR, the primary group had more patients with thick amniotic fluid but less volume (Table 4). Primary Grade III MSAF with an abnormal FHR was predominantly observed during the first stage of labor (Table 4). In the primary group, more patients required artificial membrane rupture and cesarean section for delivery when an abnormal FHR was present (Table 4). In the secondary group, primiparous women are more predisposed to FHR abnormalities (Table 4). The incidence of intrapartum fever is significantly higher among patients with abnormal FHRs in both the primary and secondary groups. Significant differences in C-reactive protein (CRP) levels and white blood cell (WBC) counts were observed in the primary group. Meanwhile, significant differences in neutrophil counts and neutrophil percentages were found in the secondary group (Table 4). A greater rate of NICU admission was observed in both groups with abnormal FHR (Table 4).
The Differences Between Grade III MSAF With and Without Abnormal FHR
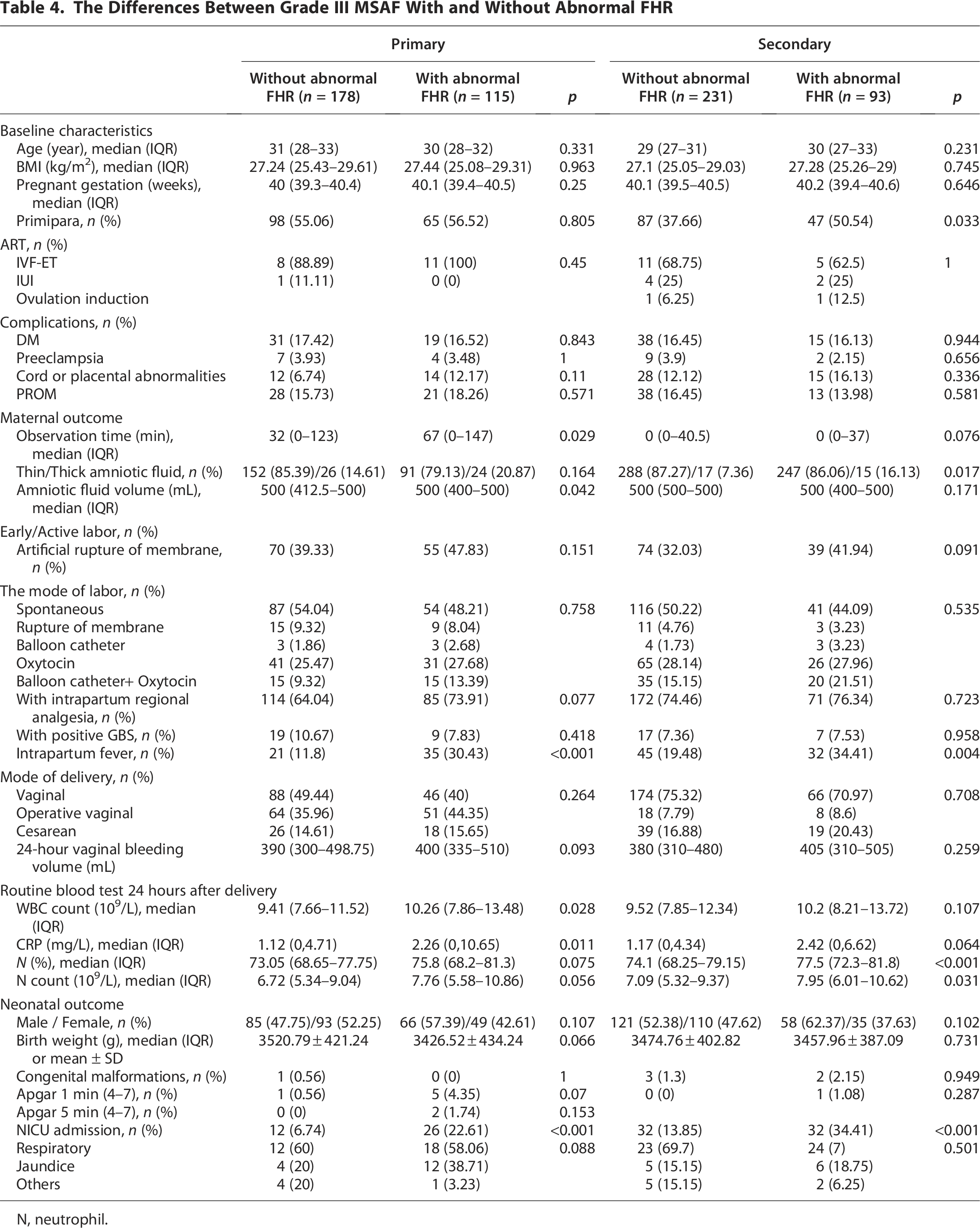
N, neutrophil.
Stratification by labor stages demonstrated that the risk profiles for NICU admission were distinct between primary and secondary MSAF, with the former being predominantly linked to markers of fetal compromise and the latter to maternal intrapartum fever and systemic inflammation. Stratifying the data by labor stages revealed significant differences in maternal and neonatal outcomes between NICU and non-NICU admission groups. During the primary group, neonates admitted to the NICU exhibited a higher proportion of thick amniotic fluid compared with those not admitted (31.58% vs. 14.9%, p = 0.011) (Table 5), alongside a markedly elevated rate of abnormal FHR patterns (68.42% vs. 34.9%, p < 0.001) (Table 5). Although maternal baseline characteristics such as age, BMI, and gestational age showed no significant differences, neonates admitted to the NICU had higher rates of low Apgar scores at 1 minute (13.16% vs. 0.39%, p < 0.001) and 5 minutes (5.26% vs. 0%, p = 0.016) (Table 5). In contrast, during the secondary group, NICU admission was associated with increased intrapartum fever (40.62% vs. 19.62%, p < 0.001) (Table 5), thicker amniotic fluid (20.31% vs. 7.31%, p = 0.002), and abnormal FHR patterns (50% vs. 23.46%, p < 0.001) (Table 5). Laboratory findings 24 hours postpartum further demonstrated elevated inflammatory markers in the NICU group, including higher WBC counts (median 12.73 vs. 9.45 × 10^9/L, p < 0.001), CRP levels (median 3.93 vs. 1.06 mg/L, p < 0.001), and neutrophil percentages (79.05% vs. 74.1%, p < 0.001) (Table 5). Notably, vaginal bleeding volume was significantly greater in the second-stage NICU group (median 424 vs. 377.5 mL, p = 0.001), while respiratory complications dominated the reasons for NICU admission (73.44% vs. 0% in non-NICU cases, p = 0.108) (Table 5).
The Differences Between Grade III MSAF With and Without NICU Admission

Discussion
This study provides a novel comparative analysis of maternal and neonatal outcomes in cases of Grade III MSAF, specifically stratified by the timing of its appearance as primary or secondary. The principal finding of our work is that while the overall incidence of composite neonatal morbidity did not differ significantly between the two groups, the underlying risk profiles and clinical pathways leading to adverse outcomes were distinctly different. This distinction underscores the value of classifying Grade III MSAF based on onset timing, as it reveals heterogeneous pathophysiological mechanisms that may be masked in broader analyses.
Previous studies have reported that adverse neonatal outcomes might be associated with secondary MSAF compared with primary MSAF, including NICU admission, the need for phototherapy, the need for a sepsis workup, 8 and a composite variable of neonatal morbidity encompassing various conditions. 11 However, it is important to note that the judgment of the need for phototherapy or sepsis workup was deemed subjective in these studies, which also included healthy women in their analysis. In the latter article, when comparing each item, only hypoglycemia showed a significant difference. Contrary to previous findings, our study, which involved 517 patients with Grade III MSAF, did not find any significant differences in neonatal outcomes. Our previous research on the impact of the MSAF on neonates revealed that a thick MSAF and abnormal FHR could be linked to NICU admission, 12 consistent with findings from another study. 13 Moreover, the long-term effects of the MSAF on neonates have been explored in other research, revealing a lower incidence of allergic diseases, 14 respiratory-related hospitalizations, 2 dermatitis and skin rash-related hospitalizations, 15 and total infectious morbidity rates, 16 suggesting a potential protective effect.
A greater rate of NICU admission was observed in patients with abnormal FHR in both the primary (6.74% vs. 22.61%) and secondary groups (13.85% vs. 34.41%), but there was no difference in respiratory cause. The rate of abnormal FHR among MSAF patients is notably greater, at 43.2%, than that among healthy women. 17 In another study focusing on women with category II FHR tracings, approximately 21.3% of women were found to have MSAF. 18 Notably, the presence of a thick MSAF in conjunction with abnormal FHR tracings may indicate a heightened risk of perinatal or neonatal morbidity, with ORs ranging from 1.67 to 2.97 depending on the severity of the abnormalities. 19 A large-scale study involving 580,000 women highlighted that the combination of abnormal FHR patterns and MSAF was associated with a 15-fold increase in the composite neonatal outcome. 20 Additionally, our findings showed that the population with an abnormal FHR had a greater incidence of intrapartum fever (primary group: 11.8% vs. 30.43%; secondary group: 19.48% vs. 34.41%). The primary group with abnormal FHR demonstrated abnormalities in routine blood tests, including WBC count, CRP, and the secondary group demonstrated abnormalities in routine blood tests, including neutrophil count, and percentage. While the presence of intrapartum fever, including chorioamnionitis, has been linked to increased fetal acidemia, it was not associated with composite perinatal morbidity. 21 Once membrane rupture occurs during labor, midwives should focus on the FHR pattern, the status of the meconium, and the maternal condition and conduct maternal perineal care in case of adverse neonatal outcomes.
The elevated incidence of low Apgar scores at 1 and 5 minutes in the primary NICU group highlights acute perinatal stress, which may be aggravated by prolonged in utero exposure to meconium. In contrast, in the secondary group, the observed association between intrapartum fever, elevated inflammatory markers (CRP, WBC, neutrophils), and NICU admission suggests a systemic inflammatory response, potentially linked to chorioamnionitis or meconium-triggered cytokine release. The correlation between increased vaginal bleeding volume and NICU admission in this stage may indicate uteroplacental insufficiency or delivery-related trauma, necessitating further mechanistic exploration. Although respiratory complications were the primary reason for NICU admission, the lack of statistical significance (p = 0.108) implies complex etiologies, such as meconium aspiration syndrome or neonatal sepsis. These findings contribute significantly to the existing literature, which has largely treated MSAF as a homogenous entity or combined different meconium grades. By focusing exclusively on the most severe grade (Grade III) and differentiating its onset, we demonstrate that the blanket management strategies often applied to “thick meconium” may be inadequate. Instead, our results advocate for a nuanced, phenotype-specific approach. For the primary MSAF phenotype, intensified and continuous FHR surveillance from the onset of labor is paramount to promptly identify signs of worsening fetal compromise. For the secondary MSAF phenotype, vigilance should extend to maternal condition, with close monitoring for signs of infection or inflammation, as timely management of intrapartum fever may be critical in preventing neonatal sequelae.
The present study focused on examining the differences among women with Grade III MSAF, which has not been investigated previously. The published studies included all cases of MSAF and did not differ based on the grade, leading to variations in the approaches used for different grades. Our study, however, had certain limitations. This was a single-center retrospective study with a small sample size. To design a prospective study, a larger sample from multiple centers, including different countries, would be necessary.
In conclusion, our findings challenge the notion that all cases of Grade III carry a uniform prognosis. We demonstrate that primary and secondary Grade III MSAF represent distinct clinical entities with different risk factors and implied mechanisms of neonatal injury. Consequently, the decision for urgent delivery, particularly cesarean section, should not be based solely on the presence of thick meconium. Instead, it should be guided by a careful integration of the MSAF onset context, continuous FHR interpretation, and maternal clinical status. This tailored approach has the potential to optimize neonatal outcomes while avoiding unnecessary operative interventions.
Authors’ Contributions
Z.J. and J.W. contributed to the design and revision of the study. H.S., Y.T., and C.C. contributed to the data collection from the medical records. Y.T. and J.F. analyzed and interpreted the data and drafted the article. Z.J. revised the drafted article. Q.Z., S.H., R.F., X.Z., and Y.L. managed the study in the hospital, and all commented on the draft paper. All the authors reviewed the article and approved the final version.
Footnotes
Acknowledgments
The authors would like to thank all participants who took part in the study and enabled this research to be possible.
Author Disclosure Statement
No competing financial interests exist.
Funding Information
This work was supported by the Postdoctoral Research Foundation of Nanjing (2021BHS202) and the Postdoctoral Research Foundation of China (2021M691332).
Ethics Approval and Consent to Participate
The study received approval from the Ethical Board of the First Affiliated Hospital of Nanjing Medical University (Ethics Committee 2020-SR-256). As this was a retrospective study, the requirement for informed consent was waived by the Ethical Board of the First Affiliated Hospital of Nanjing Medical University.
