Abstract
Background:
Adverse pregnancy outcomes (APOs) are strongly associated with an increased risk of long-term cardiovascular disease (CVD), with physiological adaptations during pregnancy, such as vascular remodeling and endothelial dysfunction, possibly contributing to an elevated lifetime CVD risk. This review explores the complex interplay between APOs and long-term CVD risk, emphasizing how racial disparities and socioeconomic status (SES) shape maternal cardiovascular health.
Methods:
A comprehensive review of existing literature was conducted to evaluate the association between APOs and long-term CVD risk. This review also examines the influence of SES and racial disparities on maternal cardiovascular health, highlighting their roles in modifying disease progression and health care access.
Results:
Approximately 29% of women experience pregnancy complications, including gestational diabetes and hypertensive disorders, which are associated with at least a twofold increase in subsequent cardiovascular event risk. Socioeconomic factors play a critical role in shaping prenatal care quality and treatment adherence, as women from lower SES backgrounds, due to systemic barriers, face a heightened risk of APOs. Racial disparities further exacerbate these risks, with Black women experiencing disproportionately higher maternal mortality and APO rates compared with White women.
Conclusions:
This review underscores the intersection of race and SES in shaping maternal health disparities and long-term CVD risk. Targeted interventions to address these inequities by addressing social determinants of health are necessary. Future research should focus on elucidating the mechanisms linking SES and race to CVD following APOs, developing strategies to mitigate disparities, and conducting longitudinal studies to track CVD progression in affected populations.
Keywords
Introduction
Numerous vascular, metabolic, and physiological changes occur during pregnancy, including decreased vascular resistance, deposition of adipose tissues, hypercoagulability, increased insulin resistance, and cardiac remodeling. 1 These adaptations are essential for supporting fetal growth and preparing the mother for the energy demands of labor, delivery, and lactation. 2 However, they can also lead to vascular dysfunction, inflammation, and abnormal placentation, potentially exacerbating subclinical cardiovascular disease (CVD) after pregnancy. 3 Research shows that adverse pregnancy outcomes (APOs) such as preterm delivery, small-for-gestational age (SGA) infants, preeclampsia, hypertensive disorders, and gestational diabetes are strongly associated with increased risks of CVD later in life. 3 Approximately 29% of parous women experience at least one pregnancy-related complication, correlating with a twofold increase in cardiovascular events. 4 For example, 3%–70% of women with a history of gestational diabetes mellitus develop type 2 diabetes within three decades postpregnancy, while those with preeclampsia are more likely to develop hypertension, coronary heart disease, stroke, and venous thromboembolism. 4
Socioeconomic factors including income, education, health care access, and social support are pivotal in shaping accessibility to antenatal care, quality of health care, and treatment adherence, ultimately influencing long-term cardiovascular outcomes. 5 These factors, in combination with the vascular and metabolic changes of pregnancy, significantly contribute to CVD risk postpregnancy. In the United States, maternal health disparities continue to worsen, with non-Hispanic Black women being three to four times more likely to die from pregnancy complications compared with non-Hispanic White women. 6 Low socioeconomic status (SES) has also been linked to APOs, underscoring the complex interplay of factors contributing to maternal health disparities. 7 The framework of intersectionality, acknowledging the link between social identities such as race, SES, and gender, is essential for understanding cardiovascular health disparities in maternal health. 8 While socioeconomic factors critically shape prenatal care access and health outcomes, analyzing them in isolation fails to fully capture the multifaceted nature of these disparities. Further investigation into how race and SES intersect is crucial for developing effective strategies to prevent and mitigate the risk of CVD in women.
Methods
A comprehensive narrative literature review was conducted to evaluate the association between APOs and long-term CVD risk, with a specific focus on the influence of SES and racial disparities in maternal cardiovascular health. To ensure the inclusion of the most relevant and up-to-date research, a systematic search was performed across multiple databases, including PubMed, Scopus, Web of Science, and Google Scholar. The search terms used included “adverse pregnancy outcomes,” “long-term cardiovascular disease risk,” “preeclampsia,” “gestational diabetes,” “hypertensive disorders of pregnancy,” “socioeconomic status,” “racial disparities,” and “maternal cardiovascular health.” Articles were limited to peer-reviewed studies published in English between 2000 and 2023 to capture contemporary trends and outcomes. Additionally, references from key articles were reviewed to identify further studies that may have been overlooked in the initial search.
Studies were included based on specific inclusion criteria: (1) epidemiological studies, cohort studies, systematic reviews, and meta-analyses that examined the relationship between APOs and long-term cardiovascular outcomes, (2) studies that explored the impact of SES or racial disparities on maternal cardiovascular health, and (3) studies that involved diverse populations to understand how various factors, such as income, education, health care access, and race, influence health outcomes postpregnancy. Exclusion criteria were studies that focused on small sample sizes (less than 100 participants), nonhuman studies, and studies that did not provide clear definitions of APOs or cardiovascular endpoints. Data extraction involved a systematic review of study design, population demographics, sample size, methodology, and key outcomes, with particular attention to how SES and race were measured and their relationship with cardiovascular risk postpregnancy. The findings were synthesized to assess the broader implications of maternal health outcomes, with a focus on identifying patterns in CVD risk as influenced by pregnancy complications, SES, and racial disparities. This review highlights both established knowledge and gaps in the literature to inform future research directions and potential policy interventions. A total of 1200 records were identified through database searches. After removing 240 duplicates, 960 records were screened by title and abstract, resulting in 144 full-text articles assessed for eligibility. Of these, 25 studies met the inclusion criteria. Screening and duplicate removal were conducted using Rayyan. Full details of the search strategy can be found in the Supplementary Table S1.
Association between APOs and cardiovascular complications
There is a significant delay in recognizing and predicting CVD in women due to its presentation differing from that in men. 9 Hence, efforts to identify women at increased risk for CVD have intensified to reduce the global disease burden. Increasing evidence links APOs with long-term cardiovascular risk, with Rich-Edwards et al. notably describing APOs as a physiological “stress test” for future CVD risk. 4 Meta-analyses consistently demonstrate that women with a history of pregnancy complications, such as gestational diabetes or hypertensive disorders, are more likely to develop metabolic and CVDs later in life. For instance, women with gestational diabetes are at a significantly higher risk of type 2 diabetes compared with those with normoglycemic pregnancies. 10 Similarly, women with gestational hypertension or preeclampsia are more than two times risk to develop chronic hypertension, 11 and those with elevated low-density lipoprotein and triglycerides during pregnancy show an increased risk for dyslipidemia years later. 12 Recent data from the Nulliparous Pregnancy Outcomes Study-Monitoring Mothers-to-Be Heart Health Study longitudinal cohort reinforces this association, revealing that women who experienced APOs in their first pregnancy have a markedly higher predicted risk of atherosclerotic CVD within 2–7 years postpartum. 13 While many studies suggest shared risk factors between APOs and CVD, further research is needed to describe the causative mechanisms linking pregnancy complications to future CVD.11–14 Table 1 shows a summary of the association between APOs and long-term cardiovascular risk.
Summary of Associations Between Adverse Pregnancy Outcomes and Long-Term Cardiovascular Disease

Risk ratio.
Odds ratio.
Hazard ratio.
Incidence risk ratio.
Adjusted for child characteristics and maternal characteristics.
Early preterm birth.
Risk when combined with maternal obesity.
CI, confidence interval; NA, not available; OR, odds ratio; RR, relative risk.
Socioeconomic disparities in APOs
Impact of SES on pregnancy outcomes
Pregnancy outcomes are greatly influenced by SES, with lower SES, despite stratification by race, being linked to APOs such as maternal mortality, low birth weight, preterm birth, and stillbirth.40,41 This impact is complex, but it is mostly caused by limited access to prenatal care and other health care services because of financial limitations and insufficient health insurance coverage. 42 The incidence of APOs is further increased by environmental triggers that are common among low SES communities, such as food shortages, pollution exposure, and insufficient housing.7,41 The barriers to accessing prenatal care such as lack of insurance, poor transportation, and provider shortages result in delays that compromise early detection and management of pregnancy complications, increasing the likelihood of poor outcomes. 43
Moreover, SES influences exposure to air pollution and poor housing, two environmental factors that raise the risk of low birth weight and gestational hypertension.44–46 Lead, particulate matter, sulfur dioxide, and ozone exposure are factors that cause oxidative stress, endocrine disruption, and vascular dysfunction. Preterm delivery and maternal depression are two APOs that are independently linked to the socioeconomic stresses associated with low SES, such as financial hardship and unemployment. 47 This complex interplay between socioeconomic disadvantages and maternal–fetal health underscores the urgent need for targeted interventions to mitigate these risks. Table 2 shows a summary of the association between SES and APOs.
Summary of Associations Between Socioeconomic Status and Adverse Pregnancy Outcomes

Hazard ratio.
GDM, gestational diabetes mellitus; IUGR, intrauterine growth restriction.
SES and cardiovascular outcomes
Low SES is a well-established contributor to increased CVD risk, associated with a higher prevalence of conditions such as diabetes, hypertension, coronary artery disease, congestive heart failure, and stroke.66–68 This relationship is explained by biological and socioeconomic mechanisms, mediated by key surrogates of SES, including educational attainment, income level, employment status, and environmental factors. 69 These factors lead to lower health literacy, limited access to healthy food, substandard health care, and financial barriers to prescription drugs. For instance, the Jackson Heart Study revealed that neighborhood-level socioeconomic disadvantage correlated with higher rates of coronary heart disease. 70 Similarly, a study in the Netherlands found that low-income patients were less likely to undergo interventions like percutaneous coronary intervention after acute myocardial infarction. 69 These disparities extend to cardiovascular health among pregnant women from lower SES backgrounds who face higher risks of developing conditions such as preeclampsia, gestational diabetes, and intrauterine growth restriction, as shown in Table 3, all of which predispose them to CVD later in life. 92 Limited access to quality health care, poor postpartum support, and ongoing socioeconomic problems are some factors that contribute to their increased vulnerability to long-term cardiovascular morbidity.7,93
Summary of Associations Between Socioeconomic Status and Cardiovascular Disease
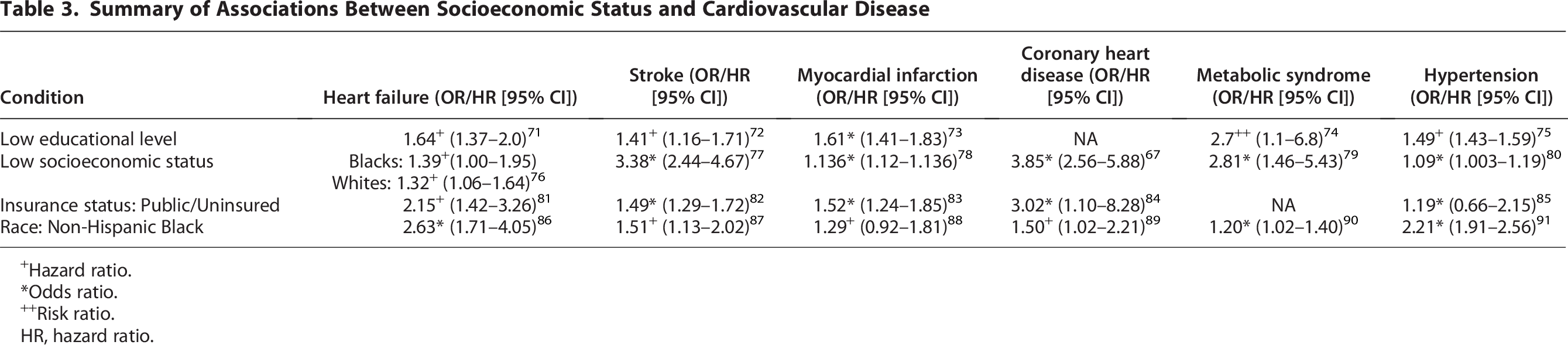
Hazard ratio.
Odds ratio.
Risk ratio.
HR, hazard ratio.
Potential mechanisms linking SES and APOs
SES significantly influences the development of APOs and the risk factors associated with CVD. Despite social mechanisms being the primary pathways through which SES impacts these conditions, molecular mechanisms also mediate long-term effects. Dysregulation of protein synthesis and inflammation is one speculated mechanism despite the marked paucity of data on such mechanisms. Research indicates that lower SES is correlated with heightened inflammation and immune system activation during pregnancy. A retrospective cohort study found that women from low SES backgrounds had significantly higher median serum concentrations of interleukin-6 (IL-6) at 13–18 weeks of gestation compared with their high SES counterparts.71,72 IL-6 is a key protein associated with chronic inflammation and has been implicated in gestational complications, including preterm delivery. 94 Additionally, pregnant women from low SES backgrounds have been observed to have elevated oxidative stress. A pooled analysis across the United States evaluated urinary concentrations of 8-iso-prostaglandin F2α (8-iso-PGF2α)—an established biomarker of oxidative stress—at various times in the gestational period. The results indicated that women facing greater socioeconomic disadvantages exhibited higher levels of 8-iso-PGF2α in their urine. 95 This biomarker has been linked to preeclampsia, preterm birth, and SGA infants.95,96 Moreover, psychosocial stress and depression contribute to inflammatory imbalances, increased adiposity, and immune dysregulation. 95 These factors increase the risk of complications such as SGA births, preterm delivery, and adverse outcomes postpartum. 97
Race and APOs
Impact of race on pregnancy outcomes
Black and Indigenous individuals experience disproportionately higher rates of APOs compared with White individuals.6,98 Black women are over three times more likely to die from pregnancy-related complications than their White counterparts. 6 Additionally, Black infants are more likely to be delivered preterm and have low birth weight. 99 These disparities persist even after controlling for socioeconomic factors, 100 highlighting the social determinants linked to race that heighten the risk of APOs. 93 Several factors contribute to these disparities, including racial bias, female underrepresentation in clinical trials, a lack of appreciation for nontraditional risk factors, and the undertreatment of disease.6,93,99 Non-Hispanic Black infants experience higher rates of fetal death and infant mortality compared with White infants. 101 Moreover, women from marginalized groups, including Black, American Indian/Alaska Native, and Native Hawaiian and Other Pacific Islander populations, after controlling for SES, face significantly higher risk of pregnancy-related mortality and morbidity, as well as higher incidences of preterm births, low birth weight, and inadequate prenatal care. 100
While biological factors, such as genetic predispositions to specific health conditions, may influence APOs, research indicates that environmental and health determinants play a more substantial role in racial disparities.102–104 Some genetic polymorphisms are associated with preeclampsia; however, they are unlikely to account for all observed racial differences.102,104 Socioeconomic disparities encompassing income levels, educational attainment, access to health care, and discrimination are pivotal in shaping these health inequities. 105 Individuals from the most affected racial and ethnic groups encounter various barriers at individual, provider, and health system levels. On a patient level, factors such as nontraditional risk factors, financial constraints, health literacy, and genetic variability come into play. At the provider level, implicit bias, underrecognition of atypical presentations, and suboptimal risk assessment further exacerbate disparities. Systemic issues, including a lack of diversity in clinical research, fragmented care, and insufficient infrastructure to meet diverse cultural needs, compound these challenges. 106 Moreover, individuals from racial minority groups often experience heightened stress due to discrimination and racism. Evidence suggests that these perceived and internalized stressors contribute to chronic inflammation and cardiovascular health disparities among minority populations, particularly minority women.107,108 This chronic inflammation and toxic stress can lead to vascular damage, increasing the risk of inflammation-mediated APOs during pregnancy.11,106–108 Chronic stress may also promote unhealthy behaviors such as smoking and substance abuse, along with physiological disturbances like hypertension and hormonal imbalances, all of which contribute to APOs.
Intersectionality: How race and SES intersect to exacerbate maternal cardiovascular disparities
Intersectionality, as articulated by Crenshaw (1991), highlights how overlapping social categorizations such as race, class, and gender create interconnected systems of disadvantage. The combined effects of race and SES intensify disparities in pregnancy outcomes, producing disproportionately adverse results for individuals experiencing multiple forms of marginalization. Historical injustices including slavery, segregation, and discriminatory health care policies have entrenched inequities in access to quality care and economic opportunity, particularly among Black and Indigenous communities.109,110 For instance, Black women of lower SES face greater barriers to early and adequate prenatal care, fewer options for high-quality obstetric services, and higher rates of medical mistrust compared with White women of similar SES. 6
The physiological toll of chronic stress from racism and poverty further compounds these inequities, increasing risks of hypertension, preterm birth, gestational diabetes, and preeclampsia, all of which elevate long-term cardiovascular risk. 76 A multisite prospective study demonstrated that individuals from racially marginalized groups with low SES experience the poorest pregnancy outcomes, indicating a synergistic rather than additive effect of race and socioeconomic disadvantage. 111 Specifically, Black birthing individuals from low SES backgrounds exhibit the highest rates of preterm delivery, preeclampsia, and SGA infants compared with both higher-SES Black women and White women of any SES. 112
Geographic inequities further magnify these disparities. Rural areas report higher maternal mortality and cardiovascular risk factors than urban regions, with limited health care access, longer travel distances, and lower health literacy disproportionately affecting rural Indigenous, Black, and Hispanic women. 113 These factors collectively restrict opportunities for timely diagnosis and management of hypertensive and metabolic disorders during pregnancy.
Optimizing pregnancy outcomes is essential, as pregnancy serves as a cardiovascular “stress test” with lasting implications for maternal health. Although complications such as preeclampsia and gestational diabetes increase future CVD risk, studies adjusting for race and SES show attenuation of these associations, suggesting that structural and social determinants are primary drivers of disparities rather than intrinsic biological differences.114,115 Thus, inequities in health care access, neighborhood environment, income, and education sustain racial and geographic gaps in maternal cardiovascular outcomes. Addressing these disparities requires a comprehensive strategy that targets upstream social factors, mitigates structural inequities, and extends cardiovascular risk reduction beyond the postpartum period.
Addressing socioeconomic and racial disparities in cardiovascular health
Policy implications: Improving access to prenatal care, addressing social determinants of health
The research emphasizes how important social determinants of health (SDoH) are in influencing cardiovascular health and unfavorable pregnancy outcomes, especially in low SES and racially marginalized populations. The World Health Organization defines SDoH as the factors affecting people’s living situations, and it has a major impact on women’s health. Unfavorable pregnancy outcomes are increased when there is limited access to emergency services, wholesome food, suitable housing, and educational resources. Furthermore, to enhance maternal health and global public health outcomes, stressors such as racism, poverty, and unsafe living conditions call for immediate policy intervention.116,117 Effective SDoH screening and referral require collaborative dialogue between patients and health care providers, fostering understanding of the associated risks and benefits. This approach can reduce stigma and improve health-seeking behaviors. The NC Maternal Mental Health MATTERS toolkit emphasizes how crucial it is to screen for hazardous SDoH in order to identify people who are at risk. 118 Disparities in health care and resource access have been confirmed by recent studies, highlighting the necessity of policies that guarantee everyone gets safe SDoH.118,119 Improving maternal cardiovascular outcomes and attaining health equity require addressing the factors contributing to SDoH inequities. Promoting paid maternity leave can help improve women’s health by allowing proper recovery without the stressor of not earning an income, which is a pressing issue for families living by paycheck. 120 Policies that promote women’s education can also reduce unwanted pregnancies and short birth intervals, which reduces unsafe practices for pregnancy termination and practices leading to high-risk pregnancies, such as quick concession conception and sexually transmitted diseases, while also providing women with greater occupational opportunities through educational advancement. 121 In addition to improving access, universal health care and its expansion to neglected areas would advance gender equality and safer practices in prenatal care. 117
Clinical interventions: Tailoring postpartum care for high-risk populations
The United States faces a higher incidence of mortality during or within 1 year of pregnancy compared with other high-income countries, despite significant investments in the health sector.122–124 Approximately 83% of these pregnancy-related deaths are estimated to be preventable. 125 Alarmingly, the maternal mortality rate in the United States has risen from 7.2 per 100,000 live births in 1987 to 23.8 in 2020.126,127 In response to this trend, the American College of Obstetricians and Gynecologists recommends that patients engage with their health care providers within 3 weeks postpartum, whether in person or via telehealth.128,129 Following this initial contact, more comprehensive follow-up appointments should occur within 84 days postpartum. 130 However, a significant proportion of patients from low-income populations become disconnected from health care services after childbirth, and this further contributes to the disparities in health outcomes between low- and high-income groups.131,132 This disengagement often results in inadequate screening and detection of future CVD.
Effective postpartum care depends on identifying people at risk using socioeconomic variables, including insurance status, employment, age, marital status, education, race, ethnicity, and proximity to health care facilities.133,134 Substandard prenatal care remains a significant risk factor for poor postpartum follow-up. 135 Research consistently demonstrates that patients with private insurance from higher SESs are more likely to attend postpartum appointments than patients from lower socioeconomic backgrounds, especially those with public insurance such as Medicaid. 135 Interventions should concentrate on guaranteeing proper prenatal care, promoting early and frequent prenatal visits, and teaching underprivileged groups the value of prompt follow-ups in order to increase postpartum care attendance. 130 High-risk pregnancies require close monitoring throughout gestation to identify and address potential complications. Culturally tailored interventions such as phone reminders, text messages, home visits, and telehealth appointments are likely to improve compliance with postpartum follow-up. 130 Ultimately, expanding access to health care services is paramount for improving postpartum care attendance.
Future research directions
Understanding mechanisms
The processes by which racial and socioeconomic factors affect cardiovascular health after unfavorable pregnancy outcomes (APOs) are not well understood in the literature. Future studies should investigate whether there are prenatal common risk factors for CVD and whether APOs themselves increase the likelihood of developing CVD in the future. This could entail investigating the ways in which marginalized racial and ethnic groups are disproportionately impacted by SDoH, such as access to high-quality prenatal care, nutrition, and mental health assistance.
Policies and interventions
Future studies must concentrate on creating and assessing interventions that address racial and socioeconomic inequities in cardiovascular health. Community-based programs that encourage lifestyle changes, such as physical activity, nutrition education, and smoking cessation, as well as legislative efforts to eliminate systemic disparities in health care delivery and access, could be examples of these interventions. Improving the efficacy of these interventions will require addressing biases in health care systems and placing a strong emphasis on culturally competent care.
Longitudinal studies
Long-term, prospective studies are essential for long-term follow-up of individuals with APOs, assessing the cumulative risk of future CVD. Such research can provide valuable insights into the trajectory of CVD development and identify critical intervention periods. Understanding how socioeconomic and racial factors interplay in the progression of CVD can inform targeted prevention strategies, particularly for high-risk populations.
Precision medicine approaches
Research must investigate precision medicine strategies designed to meet the requirements of different ethnic and socioeconomic groups. This involves determining how genetic, epigenetic, environmental, and social factors interact to affect treatment outcomes and CVD risk. We can create individualized treatment plans that successfully address health inequities by integrating socioeconomic data into precision medicine frameworks.
Implementation science
Future studies should concentrate on implementation science to understand how evidence-based interventions can be effectively translated into real-world settings and adapted to diverse socioeconomic contexts. This includes investigating strategies for overcoming barriers to access, such as transportation and financial constraints, and identifying facilitators that promote the equitable uptake of cardiovascular health interventions among underrepresented communities.
Health equity policies
It is essential to examine the effect of health policies aimed at promoting equity in cardiovascular care. Future studies should assess the efficacy of policies that address SDoH—including housing stability, educational access, employment opportunities, and income support—in reducing cardiovascular disparities. An understanding of how these policies influence health outcomes across different racial and socioeconomic groups can inform more effective advocacy and reform initiatives.
Community engagement and participatory research
Community engagement in the research process is vital for developing culturally sensitive and contextually appropriate interventions to address socioeconomic disparities in cardiovascular health. Employing participatory research approaches can empower communities, ensuring that interventions reflect their needs and preferences. This collaborative effort fosters sustainable change and enhances community resilience against health disparities.
Future research focusing on racial and socioeconomic disparities in postpartum cardiovascular health is essential to advancing our understanding of these differences. Such investigations will provide critical insights necessary for the development of targeted interventions aimed at improving health outcomes and promoting health equity across diverse populations, irrespective of SES or race.
Conclusions
APOs, which might result in future CVD, are more likely to occur in people from poorer socioeconomic backgrounds and marginalized racial groups. These differences have a significant impact on cardiovascular health after pregnancy. These disparities result from environmental factors, toxic stress, and poor access to high-quality health care. We need health care policies that recognize the intricate relationships between gender, SES, and race to address these problems. By encouraging collaboration between legislators and medical professionals, we can address systemic disparities and increase access to high-quality care. Further research is necessary to better understand these differences and identify practical ways to help all women achieve improved health outcomes.
Footnotes
Author Disclosure Statement
No competing financial interests exist.
Funding Information
No funding was received for this article.
Supplemental Material
Abbreviations
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
