Abstract
Endometriosis is a persistent inflammatory condition characterised by chronic pain, increasing the chances of infertility and adverse pregnancy outcomes. However, the existing evidence on this matter lacks consistency. The disease is associated with impaired folliculogenesis exerting a negative effect on egg quality and leading to defects during the luteal phase, decreased rates of fertilisation and abnormal embryonic development, all of which contribute to reproductive difficulties. As the number of endometriosis patients who become pregnant increases, there is a greater demand for comprehensive assessments to identify the possible negative impact of the condition on pregnancy outcomes, including the postnatal period. Patients with endometriosis might experience obstetric complications, including placenta accreta spectrum, fetal growth restriction and a higher likelihood of having to undergo a caesarean section, giving birth prematurely, and experiencing postpartum haemorrhage. The field of obstetrics often neglects to fully recognise and examine these complex issues, leading to the lack of thorough investigation. The impact of the disease extends beyond the conception stage, potentially influencing results in obstetrics and neonatology. Hence, it is imperative to carry out a targeted investigation to determine the most efficient strategies for managing and monitoring pregnant women who were diagnosed with endometriosis. An updated summary of the most recent research in this area is given in this review. An updated summary of the most recent research in this area is given in this review.
Keywords
Introduction
Endometriosis is a chronic inflammatory disorder defined by the occurrence of endometrium-like tissue outside the uterus. Approximately 10% of women of reproductive age and up to 50% of women with infertility are affected by the condition.1,2 Although endometriosis is an acknowledged risk factor of infertility, the precise processes by which infertility is induced have not been fully comprehended yet. Furthermore, a correlation was observed between adverse pregnancy outcomes and endometriosis. However, the available literature on this matter remains inconsistent.
Endometriosis is a complex disease, and, for better understanding and treatment, it could be divided according to the location of lesions, severity, morphological type and menopausal status. Pelvic endometriosis commonly presents in regions including the ovaries, uterine ligaments (especially uterosacral ligaments), the pouch of Douglas and fallopian tubes.3,4 Although less frequent, instances outside the pelvic region might manifest in different systems across the body. From the morphological perspective, endometriosis may be classified into three distinct types, indicated by available evidence. Red lesions are characterised by a high degree of vascularisation and antigenicity. Red lesions in endometriosis are distinguished by their enduring ability to regenerate. Upon the shedding of these lesions, an inflammatory reaction is initiated, leading to scar tissue formation and the subsequent darkening of the lesions. These lesions are suggestive of regions characterised by an increased vascularisation and proliferation. Subsequently, fibrotic processes result in the formation of areas with white opacification, which signifies a state of inactivity in the advancement of the disease. Black lesions indicate advanced development.5–7
Endometriosis was mainly diagnosed using surgical methods in the past. 8 Several problems arise when assessing its predictive usefulness in relation to symptoms, response to surgical therapy, postoperative pregnancy rate, symptom recurrence and illness relapse. The development of noninvasive technologies, including magnetic resonance imaging (MRI) and transvaginal sonography (TVS), have shown high accuracy in classifying endometriosis.9–11 Based on these available methods, there are different standard categorisation systems include, for example, the American Society for Reproductive Medicine (rASRM) classification, ENZIAN classification, endometriosis fertility index (EFI) and American Association of Gynaecological Laparoscopists (AAGL) classification.12,13 Every single one of these systems has its own advantages and disadvantages, and the fact that there are several classifications demonstrates the complexity of the illness and the difficulty in encompassing all its varied expressions within a single framework. We briefly review each of these classification schemes below.
Published in 1979 and developed over the years, the American Fertility Society (AFS) classification was finalised in 1996 as the revised American Society for Reproductive Medicine (rASRM) classification. 14 It serves well to assist clinicians in explaining the severity of endometriosis to patients using straightforward language. Moreover, the primary benefit of the rASRM categorisation is related to its extensive global recognition and increasing utilisation in recent times. Nevertheless, there are several drawbacks. There is frequently a difference between the histologically identified endometriosis and the visually diagnosed stage; the repeatability of the rASRM score is low; this classification is not intended for assessing deep infiltrating endometriosis (DE); and the severity of symptoms and infertility do not show an appropriate correlation with the rASRM stage. 15
The ENZIAN classification, which focuses on DE was first mentioned in 2003 and officially presented by the Scientific Endometriosis Foundation (SEF). It evolved over time to include more specific aspects of the disease, with the latest update being released in 2021.16,17 This classification offers a comprehensive evaluation of endometriosis, including preoperative assessment and symptoms, and contrasts them with intraoperative observations. The ENZIAN classification provides a thorough structure for assessing the intricacy and seriousness of DE, serving as a vital resource in clinical and research environments. 17
The ENZIAN classification was included in the recommendations of the working group consisting of the European Society of Gynaecological Endoscopy (ESGE), the European Society of Human Reproduction and Embryology (ESHRE), the World Endometriosis Society (WES) and the Consolidated Standards of Reporting Trials (CONSORT) based on numerous studies and current data. The guidelines of the German, Swiss, Czech and Austrian Societies for Obstetrics and Gynaecology advocated using the ENZIAN classification for classifying DE.18,19
In 2007, the AAGL initiated the development of a new classification system for endometriosis. This classification was created to focus on discomfort, infertility and surgical intricacy results. Surgical problems were categorised into four levels, ranging from a simple removal of surface implants to complex procedures, including intestinal resection and ureteral reimplantation. Despite being introduced long ago, the AAGL classification has not been fully validated and released. 20
The EFI system was created to predict the pregnancy rate in patients with surgically diagnosed endometriosis who have not undergone in vitro fertilisation (IVF) in an attempt to conceive. In 2010, Adamson and Pasta introduced an EFI system that utilised data from over 500 infertile patients diagnosed with endometriosis via surgery. 21 Since the minimum function score is assessed subjectively, the overall score may differ depending on the physician.
Symptoms related to the gastrointestinal tract may encompass the presence of haematochezia and sporadic bowel blockage. The involvement of the urinary system frequently results in haematuria, dysuria and pelvic pain. 13 This disorder may result in anatomical abnormalities such as adhesions and endometriomas, which interfere with the functioning of the fallopian tubes, impair the quality of oocytes and impede the retrieval of oocytes by the tubal fimbriae.3,4,22 More and more reports tackled the issue of the influence of endometriosis on endometrial receptivity.5,23 Progestogens, anti-progestogens, combined oral contraceptives, gonadotropin-releasing hormone (GnRH) agonists and antagonists, the levonorgestrel intrauterine system, danazol and aromatase inhibitors like letrozole are utilised to inhibit steroid receptors and enzymes, suppress ovulation and to prevent the development of endometriosis. However, this kind of medication should not be prescribed to women seeking to conceive.12,24–26
According to ESHRE guidelines and further reports ablation or excision is connected with an increase in spontaneous conception frequencies.12,27,28 According to the Cochrane analysis by Chen et al., 10 surgery followed by postsurgical medical care likely enhanced the pregnancy rate with relative risk (RR) of 1.64 (95%CI: 1.09–2.47) in pregnancy likelihood after surgery compared to diagnostic laparoscopy. Women who are unable or choose not to become pregnant may be administered hormone therapy right after surgery, which exerts no adverse effects on their fertility and enhances the immediate results of the surgery in terms of pain relief. 12
The pathophysiology of endometriosis may be significantly influenced by microbiome dysbiosis, which can lead to immunological dysfunction and persistent inflammation. The development of endometriosis is influenced by changes in the reproductive tract microbiota, which may also have an adverse effect on fertility and the success of pregnancies. 29 Gaining insight into these microbial imbalances provide a fresh perspective on the interaction between endometriosis and reproductive health.
Furthermore, endometriosis affects infertility not only through physical barriers but also through several risk factors that contribute to the development and progression of the condition.30–32 The risk factors of this disorder include early menarche, shortened menstrual cycles, heavy menstrual bleeding and a hereditary predisposition to the disease.5,7 The combination of these factors increases the likelihood of developing endometriosis and affects the severity and progression of the disease, influencing fertility. 6 Endometriosis negatively impacts both spontaneous and assisted conception. Recognising endometriosis as a condition affected by several causes highlights the importance of implementing a thorough approach in diagnosing and treating infertility linked to endometriosis. This strategy should take account of all aspects of an individual’s clinical features. 30 These frameworks frequently don’t accurately predict reproductive outcomes or pregnancy-related issues, even though there are a number of categorisation systems available that are intended to standardise disease description and guide therapy. Thus, it is necessary to conduct an up-to-date review that addresses the relationship among endometriosis, fertility and pregnancy outcomes.
In order to assist physicians in making accurate and patient-centred decisions in reproductive care, this study aims to provide an overview of the most recent data on the effects of endometriosis on female fertility and its correlation with pregnancy complications.
Conception and fertility
The intricate relationship between endometriosis and infertility has been unequivocally confirmed in the literature. Endometriosis is found in around 50% of women experiencing infertility, which is significantly higher than its incidence of 10% in the overall fertile population. 1 The primary obstacle to achieving natural pregnancy in endometriosis is believed to arise from a harmful pelvic environment that impairs the interactions between sperm and oocytes, hence affecting the processes of fertilisation. 33
It was demonstrated that severe endometriosis had a substantial negative impact on egg retrieval rates and subsequent fertilisation using assisted reproductive technology (ART).34,35 Variables such as adhesions in the pelvic region, abnormalities in the fallopian tubes, and an uncontrolled immune response were found to cause a decrease in fertility. All these problems affected the process of ovulation, the functioning of the corpus luteum and the implantation of embryos.34,36
Tubal occlusion in women diagnosed with endometriosis arises due to the presence of peritubal adhesions and hydrosalpinx. This obstruction of the fallopian tube and subsequent hindrance of gamete transportation is caused by this barrier, which serves a crucial function in reducing the likelihood of fertilisation and implantation and, thereby, substantially contributes to infertility.3,4 Apart from tubal occlusion, the occurrence of hydrosalpinx caused by endometriosis or other factors might lead to infertility and to an increased risk of extrauterine pregnancy. Hydrosalpinx, as an isolated pathological condition, exerts a detrimental effect on embryo implantation and pregnancy outcomes, particularly in ART, where its presence has been associated with significantly reduced success rates of embryo transfer. 37
In cases of endometriosis, the receptivity of the endometrium is impaired due to elevated levels of antibodies targeting the endometrial antigens. Luteinised unruptured follicle syndrome, a particular aberration, has a detrimental impact on fertility since it hinders the release of the egg despite typical hormonal signals expected to initiate ovulation. 34
Despite initial assumptions, research demonstrated that endometriotic women who underwent ART achieved reproductive outcomes similar to those without the disease.38,39 However, they did experience greater rates of cycle cancellation. It is noteworthy that the surgical intervention for endometriosis before ART demonstrated superior results and had no impact on the quantity of oocytes obtained.27,40
Endometriosis is distinguished from an immunological perspective by the presence of a changed peritoneal milieu that is richer in pro-inflammatory cytokines, reduced dynamics of natural killer (NK) cells and irregularities in T-cell function, playing a crucial role in connecting the innate and adaptive immune responses. 41 The decreased activity, which is more noticeable in the fluid surrounding the organs but is also found in the blood outside the organs, indicates a malfunction that may be influenced by inflammation caused by chronic diseases and an incorrect expression of receptors. This condition is characterised by atypical endometrial tissue that displays a distinct immune response, characterised by a heightened production of cytokines such as interleukin 6 (IL-6) and tumour necrosing factor alfa (TNF-α). These cytokines play a crucial role in orchestrating the inflammatory reactions that contribute to the development and persistence of endometrial lesions.42,43 In addition, the upregulation of killer cell immunoglobulin-like receptors (KIR) on these cells might play a role in their compromised functionality, likely elucidating the heightened autoimmunity reported in endometriosis. Furthermore, the existence of anti-endometrial antibodies in patients’ blood, together with increased levels of IgG and IgA antibodies, including autoantibodies that specifically target the endometrial tissue, provides evidence of autoimmunity in endometriosis. Autoantibodies have the ability to hinder the responsiveness of the endometrium, thereby decreasing the probability of achieving a successful implantation.41,44
The involvement of the oestrogen receptors (ER) in endometriosis is of significance since they exert influence over the suppression of apoptosis, inflammation and cell proliferation. In surgical models and murine investigations, the overexpression of the ER demonstrated its capacity to alleviate TNF-induced apoptosis and enhance interleukin-1 beta (IL-1β), resulting in improved cell survival and proliferation.45,46 The ER plays a crucial role in promoting the differentiation of the epithelial cells into the mesenchymal cells, influencing the endometriosis-linked infertility through the inhibition of decidualisation in the endometrial stromal cells. In contrast, ER inhibition was found to diminish the development of stromal cells.27,45 Furthermore, the dysregulation of progesterone receptors (PR), a defining feature of endometriosis, is intimately linked to the activity of ER. Reduced PR expression and changed PR isoform balance cause endometriotic lesions to demonstrate progesterone resistance, which hinders endometrial receptivity and the endometrium’s capacity to respond appropriately to hormonal signals necessary for implantation. 32 The relationship between oestrogen and inflammation creates a recurring pattern that worsens long-term inflammation in endometriosis, where the enzymes aromatase and cyclooxygenase-2 (COX2) maintain the production of estradiol and prostaglandin E2 (PGE2). These immunological abnormalities disrupt the process of follicle development, decrease the quality of oocytes and reduce the ability of the endometrium to facilitate the implantation of embryos. Moreover, the occurrence of oxidative stress, which is a consequence of endometriosis, has the potential to cause harm to DNA within gametes and developing embryos, thereby exacerbating the decline in fertility. 5
A recent study has shown that both adenomyosis and endometriosis cause genetic and epigenetic changes in the endometrium as a result of inflammation-related alterations. These changes may result in the production of oestrogen by CYP-19 in specific areas and make it more difficult to conceive. 47 Figure 1 graphically presents the immunological and hormonal influence of endometriosis on fertility.
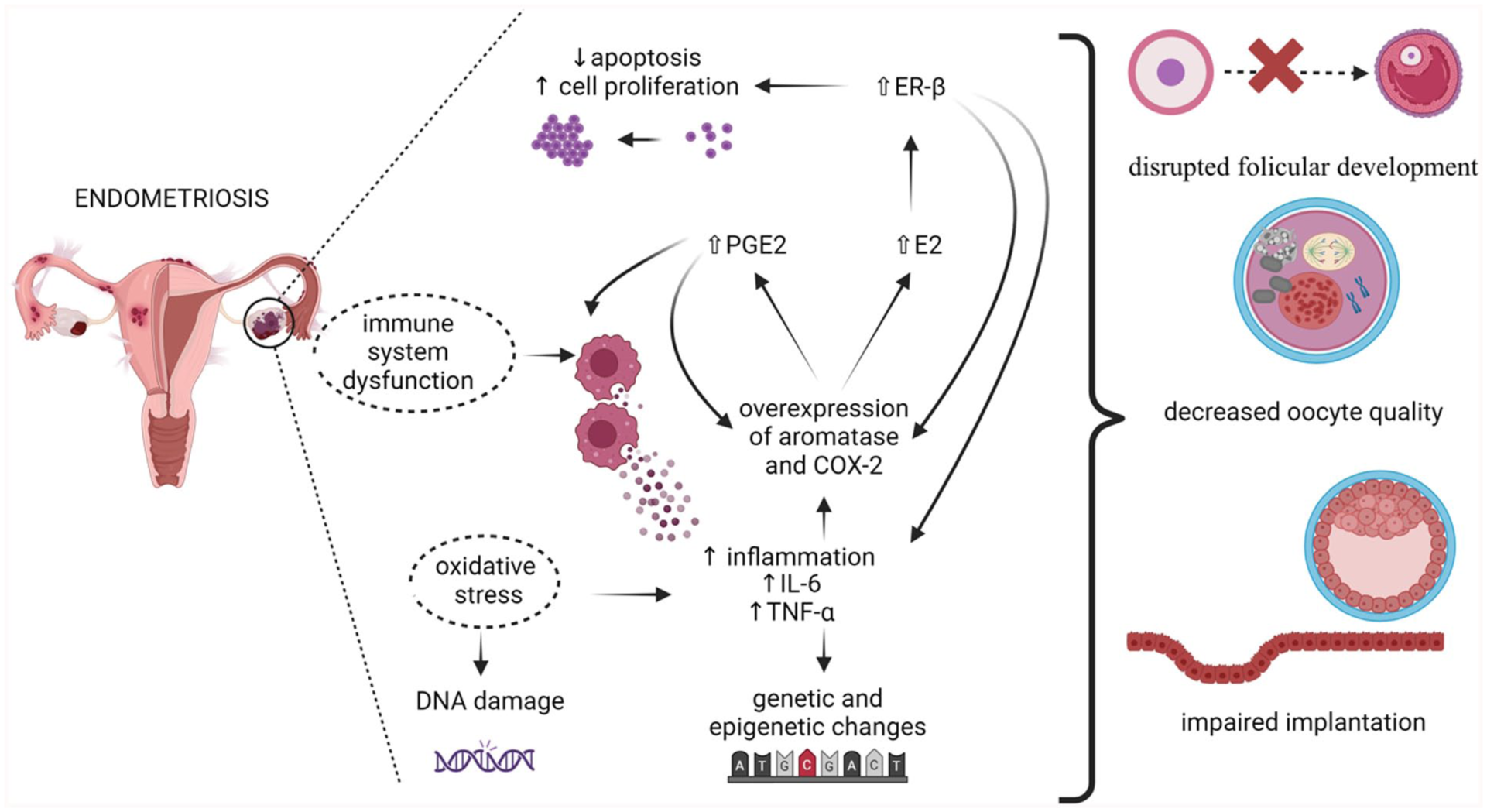
Impaired oestrogen metabolism, oxidative stress and the dysregulation of immune system function are associated with molecular events such as increased cell proliferation and inhibited apoptosis observed in endometriosis. Chronic inflammatory state results in increased levels of prostaglandins and estradiol but also genetic abnormalities, which, in turn, may lead to reproductive difficulties.
It is crucial to evaluate the influence of patients’ emotional states on their attempts to conceive. Challenges in conception are frequently correlated with decreased self-esteem, relational difficulties, strained familial interactions, diminished mood and the emergence of depressive symptoms or other psychological disorders, especially in individuals with a personal or family history of these conditions. Additionally, it significantly affects hormone balance, and endometriosis is typically thought of as a comprehensive illness that impacts several areas of the body. The clinical manifestations of endometriosis, characterised by chronic pain syndromes that are frequently underdiagnosed or diagnosed tardily, and thus treated late, were found to be linked to a heightened risk of psychological disorders.48,49 Research demonstrated that pain syndromes associated with endometriosis or other chronic pain diseases markedly diminished the quality of life, as they might obstruct the satisfaction of fundamental psychological requirements. 49
Furthermore, from the standpoint of needs hierarchy, the need to relieve pain usually takes precedence over procreation. The incapacity to or difficulty in conceiving affects the quality of life. Psychological issues, pain perception and the inability to fulfil basic living demands adversely influence conception.49,50 Such an effect is manifested not just in diminished libidos but also in distinct physiological influences on conception. 51 As a result, a complicated cycle of physical and psychological disorders arises, each potentially aggravating the other, further reducing conception success chances. Figure 2 graphically presents the psychological influence of endometriosis on fertility.
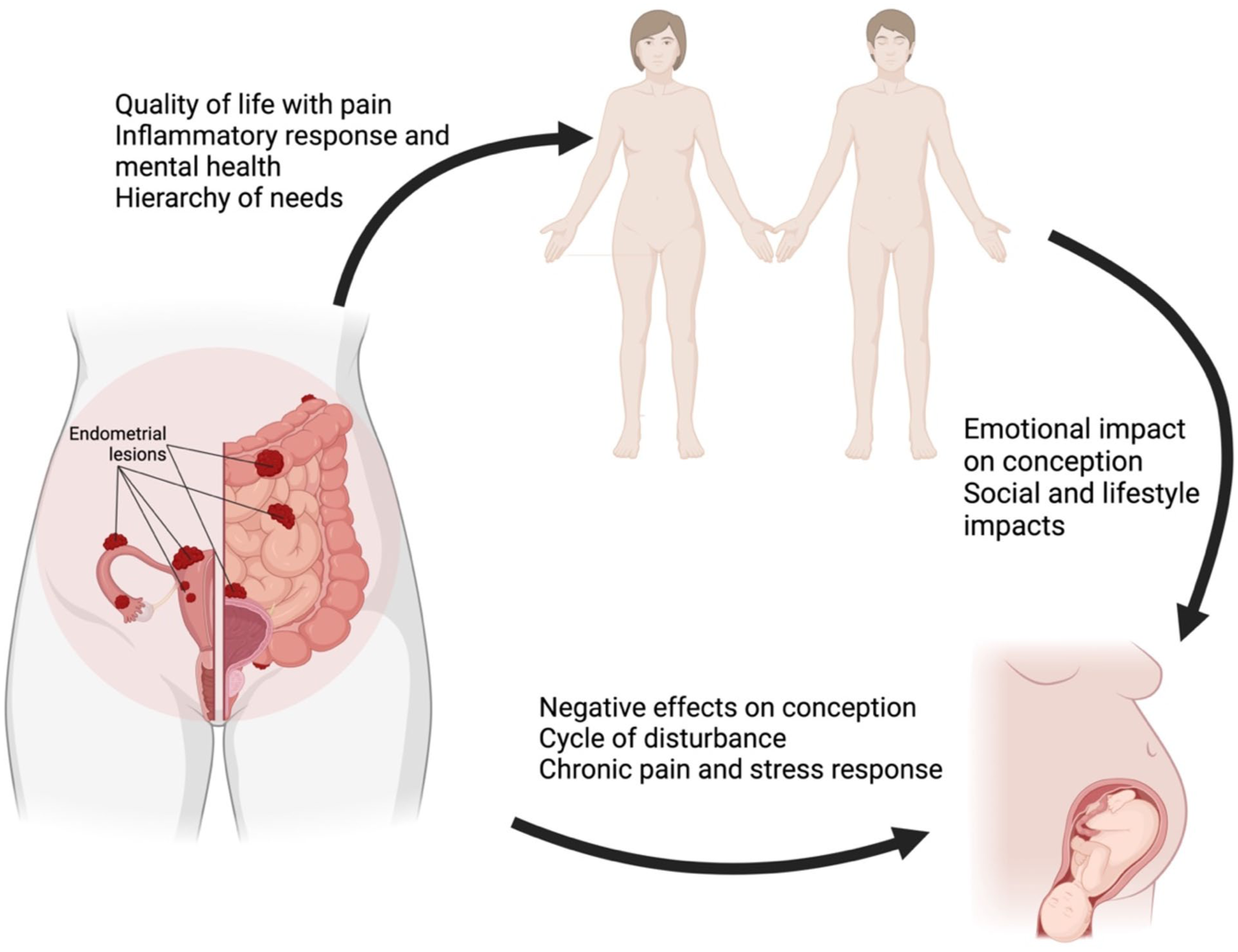
The psychological influence of endometriosis on fertility.
The effects of reproductive treatment in women with endometriosis are not similar in every study. Women with endometriosis often undergo surgical treatment, which may decrease ovarian reserve, increase pregnancy complications in the future and decrease chances of pregnancy. 34 Furthermore, in order to improve the likelihood of a natural conception, the ESHRE guidelines suggest that operational laparoscopy be used to treat infertility linked to stage I/II endometriosis as defined by the rASRM. The evidence, however, points to a limited benefit, and obtaining one more pregnancy might necessitate more surgical procedures. Thus, the choice to have surgery should be made on an individual basis, taking into account the dangers and resource implications of the procedure as well as any prospective reproductive benefits. 12 Moreover, it was confirmed that the postoperative period after endometriosis surgery presented the most favourable opportunity for attaining pregnancy. In over 90% of cases, surgery could lead to the alleviation of symptoms and generated an increase of over 65% in postoperative pregnancy rates.36,52 Notably, the optimal opportunity for conception was found to occur within 12 months after surgery. 53 There is controversy regarding the impact of different stages of endometriosis on ART outcomes. A study by Esmaeilzadeh et al. 35 showed no correlation between fertility parameters and the reproductive outcomes of severe compared to less severe endometriosis. However, a study conducted by Sharma et al. 54 revealed similar outcomes of infertility treatment in a cohort of younger women diagnosed with severe endometriosis compared to those without a diagnosis of endometriosis. Nevertheless, women above 35 who were diagnosed with endometriosis had a decreased probability of having a live birth (OR 0.47, 95%CI: 0.25–0.90) and a lower rate of pregnancy (OR 0.46, 95%CI: 0.22–0.95). Such an observation could be caused by damaged ovarian reserve linked to high stages of endometriosis. However, according to recent studies, lower ovarian reserves did not have to be associated with lower fertilisation and pregnancy rates in ART.55,56 This suggests that ovarian reserve markers alone may not be sufficient predictors of reproductive success. Additionally, it has been demonstrated that in certain situations, surgical intervention, such as the excision of endometriomas or deep endometriotic lesions, may improve the results of ART by increasing ovarian accessibility, lowering local inflammation, and potentially restoring pelvic structure. 40 These results emphasise how crucial it is to manage endometriosis-related infertility with a customised strategy that considers ovarian function as well as the possible advantages of surgery prior to ART.
Miscarriages
Although the correlation between the severity of endometriosis and infertility was not consistently established, some authors indicated that the existence of endometriosis did not necessarily exclude the possibility of normal pregnancy outcomes. 57 The ESHRE guidelines state that there is still inconclusive data on the miscarriage rate among endometriosis-affected women in comparison to controls. 12 The most recent systematic review indicated a higher risk of miscarriage in women with adenomyosis (OR 3.40, 95%CI: 1.41–8.65). 29 Although there is conflicting evidence and no clear correlation has been established, some studies indicate an increased risk of early pregnancy loss, particularly in women receiving ART. Because of this, endometriosis is not officially recognised as a risk factor for recurrent pregnancy loss on its own, and therapeutic care should take the patient’s overall reproductive profile into account. 12
Research findings indicated that couples who effectively managed mild endometriosis had a monthly likelihood of conceiving naturally ranging from 2% to 4.5%, in contrast to the 15%–20% likelihood observed in couples who did not have such a problem. Nevertheless, individuals with moderate to severe endometriosis had a probability of less than 2%, underscoring the significance of undergoing treatment before trying to conceive in order to enhance the likelihood of a successful pregnancy. According to Saraswat et al., 58 women with endometriosis were at a higher risk of miscarriage (aOR 1.76, 95%CI: 1.44-2.15).
The heightened reproductive difficulty may be attributed to endometriosis, which impacts the growth of the endometrium, modifies immune reactions, impedes the production of new blood vessels and interferes with other associated biological mechanisms. Several variables have the potential to undermine the capacity of the endometrium to facilitate the implantation and the subsequent growth of embryos. 59
Aromatase is significantly upregulated in inflammatory disorders, particularly those impacting the endometrium, like endometriosis. This dysregulation may indicate an increased probability of implantation failure in previous ART cycles. The upregulation of this particular enzyme, together with the diminished functionality of the estradiol (E2) hydrolysing enzyme 17-β hydroxysteroid dehydrogenase II, leads to elevated concentrations of accessible E2. 45 The overstimulation of the oestrogen receptors may lead to the excessive growth of endometrial cells. Oestrogen suppresses the molecule b-3 integrin, which is essential for embryo attachment and invasion. Endometrial E2 metabolism regulation in endometriosis is controlled by intricate autocrine and paracrine signals related to inflammation. These signals involve the impacts of PGE2 and hypoxia-inducible factor-1, both of which are influenced by E2.45,60 However, there is a lack of agreement about the potential association between endometriosis and elevated levels of uterine natural killer cells, a frequently reported phenomenon in cases of infertility. 61
Ectopic pregnancy
The incorrect implantation of the embryo outside the uterine cavity, usually in the fallopian tube, but also in the cervix, abdominal cavity or within a scar after a caesarean section, is known as an ectopic pregnancy. The ESHRE classifies it as an abnormal pregnancy with a high risk of issues that require diagnostic and treatment to avoid major maternal morbidity or death. 12
Farland et al. 62 found that women with endometriosis had a far higher chance of having an ectopic pregnancy compared to those without the condition, with the risk that could be up to six times higher. In addition, Saraswat et al. 58 reported a higher occurrence of ectopic pregnancies in women with endometriosis (aOR 2.70, 95%CI: 1.09–6.72).
Endometriosis is associated with an increased vulnerability to ectopic pregnancy. 61 It was previously described that inflammatory reactions and bleeding due to histological and structural changes in the endometrial tissue could contribute to the development of lesions in the fallopian tubes, affecting the anatomy of the tubules and blocking the movement of the fertilised egg. The disruption was due to the proliferation of endometrium-like tissue in the fallopian tubes, resulting in obstructions or constricted passages that impeded the embryo’s progression towards the uterus. 63 Endometriosis might impair ciliary activity in the fallopian tubes, which is essential for facilitating the movement of the egg or embryo. 4 Endometriosis may lead to pelvic adhesions and inflammation, which can hinder the egg’s mobility and increase the risk of ectopic embryo implantation.3,4 The combined effects of impaired tubal functioning and the inflammatory environment commonly found in endometriosis are responsible for the increased incidence of ectopic pregnancy in women affected by this condition.5,41–43
Heterotopic pregnancy is another disorder that may be linked to endometriosis. Although it is uncommon, the co-occurrence of intrauterine and ectopic pregnancies may make it more challenging to diagnose and treat endometriosis in women because of changes in pelvic structure and inflammation. 64 According to this complication, endometriotic lesions can make surgical access more difficult and raise the possibility of treatment-related complications.
Pregnancy complications
According to the literature, the symptoms of endometriosis were reduced during pregnancy as the endometrial tissue was inhibited by pregnancy hormones. Nevertheless, Leeners et al. 65 reported that the behaviour of endometrial lesions during pregnancy varied, with some lesions disappearing completely after decidualisation, atrophy, fibrosis, or necrosis and others expanding. Endometrial lesions may occasionally indicate life-threatening complications necessitating a surgical intervention during the pregnancy. 66
Several perinatal complications were found to occur more commonly in patients with the diagnosis of endometriosis. 12 Complications in pregnancy, such as an increased risk of preterm birth, preeclampsia, and antepartum haemorrhage, ectopic pregnancy, were also associated with endometriosis. 63 The pathomechanism underlying these complications is not fully understood but it may involve the chronic inflammatory nature of the disease, which may affect placental development and function. Furthermore, individuals diagnosed with endometriosis frequently encounter challenges related to infertility, leading them to undergo ART interventions, as previously mentioned in this manuscript. An extended duration of infertility may also be linked to a later age at the moment of fertilisation, which, when combined with infertility treatment, further independently increases the risk of pregnancy complications. 67
Small for gestational age, fetal growth restriction and preeclampsia
Available information indicated a correlation between endometriosis and a heightened prevalence of growth restriction of foetuses. Nirgianakis et al. 68 observed that women diagnosed with endometriosis had an increased susceptibility to fetal growth restriction (FGR), regardless of the severity of the condition. An increased probability of small for gestational age (SGA) (OR 1.5, 95%CI: 1.4–1.6), congenital abnormalities (OR 1.3, 95%CI: 1.3–1.4) and higher rates of neonatal mortality (OR 1.8, 95%CI: 1.4–2.1) were observed in newborns born to mothers with endometriosis. 69 The hypothesised mechanisms underlying those growth issues included compromised blood circulation to the placenta, reduced placental dimensions, resistance to progesterone and the enduring inflammation intrinsic to endometriosis.27,58 Other possible factors that may contribute to such a situation include endometrial dysfunction that affects the exchange of nutrients and oxygen, abnormalities in angiogenic factors that are crucial for placental development, and hormonal imbalances that may impact fetal development. Furthermore, the presence of ectopic endometrial tissue in individuals with endometriosis may result in the release of compounds that interfere with the regular endocrine and paracrine signalling processes essential for foetal development. Additionally, the uterine environment may be affected by mechanical constriction caused by adhesions. The combination of these factors might have a negative impact on the environment inside the uterus, resulting in a more common occurrence of disorders like FGR in women with endometriosis.
Although the ultimate outcome of preeclampsia (PE) is the successful delivery of the foetus and the removal of the placenta, some authors suggested that endometriosis did not necessarily lead to the development of the issue. Previous research conducted by Berlac et al. 69 and Zullo et al. 70 indicated that, although individuals with advanced endometriosis might exhibit a higher prevalence of PE symptoms, the overall elevation in the likelihood of developing PE in individuals with endometriosis did not reach the level of significance (OR 1.04, 95%CI: 0.83–1.29). Also, Stephansson et al. 71 observed an increased risk for preeclampsia in women with endometriosis (aOR 1.13, 95%CI: 1.02–1.26). The study by Bronses et al. 72 revealed a reduced risk of preeclampsia in patients with endometriosis (1%) compared to the control group (7%) (OR 6.6, 95%CI 1.2–37). The pathomechanism of PE development seems to be similar to the occurrence of growth restriction, as both complications of pregnancy often have a similar origin. 73 Despite the presence of abnormal placentation issues such as increased vascular resistance and endothelial dysfunction, which are believed to be associated with alterations in the uterine junctional zone, a region that is observed to be thicker in individuals with endometriosis, it is important to note that this condition does not necessarily result in a higher incidence of preeclampsia. Furthermore, there is a limited association between hypertension during pregnancy and endometriosis, indicating the lack of significant disparity in the likelihood of developing gestational hypertension among women affected by this condition (OR 0.90, 95%CI: 0.59–1.37).69,70
Patients deemed to be at risk of preeclampsia based on the Fetal Medicine Foundation guidelines should be advised to take preventive doses of acetylsalicylic acid (ASA) from the start of pregnancy until the third trimester. 74 This helps enhance placentation, thereby reducing the likelihood of preeclampsia and foetal growth restriction. The potential impact of the prophylactic administration of ASA in patients with endometriosis is intriguing. ASA injections proved to be successful in destroying the peritoneal endometrial tissue in experimental studies on rats. The ASA solution yielded similar results by either lowering growth or promoting more involution of endometrial implants when injected directly into endometriosis.75,76 Wang et al. 77 demonstrated that administering low-dose aspirin to rats with endometriosis, particularly during the implantation window, decreased progesterone resistance and increased the expression of leukaemia inhibitory factor. This may improve the responsiveness of the endometrium, boost the likelihood of pregnancy and provide a hopeful strategy for treating infertility linked to endometriosis. Hence, further research on humans is needed to corroborate this association.
Placenta praevia
Placenta praevia is a potentially serious obstetric condition that may have the connection to endometriosis. This complication of pregnancy frequently results in antepartum haemorrhage and necessitates caesarean birth, raising the rate of morbidity and mortality among mothers and newborns. Endometriosis may contribute to the development of placenta praevia through several interrelated mechanisms. Firstly, pelvic adhesions and altered uterine anatomy, commonly seen in endometriosis, can impair the normal upward migration of the placenta, increasing the likelihood of implantation near or over the cervix.3,6 In addition, defective decidualisation and abnormal vascular remodelling, driven by chronic inflammation and disrupted hormone signalling, can compromise the receptivity of the upper endometrium and promote abnormal placental attachment lower in the uterus.32,36,71 Moreover, persistent local inflammation and tissue injury may damage the endometrial lining, especially in the cervico-isthmic region, further facilitating implantation in suboptimal sites.36,78 The presence of abnormal uterine contractility is a possible explanation for this correlation. Furthermore, it has a negative impact on the placement of the blastocyst, together with an elevated resistance to progesterone. 66
Comprehensive systematic studies identified a significant association between endometriosis and placenta praevia (from OR 3.9, 95%CI: 3.5–4.3 to OR 15.1, 95%CI: 7.6–500).66,69 According to the findings by Jeon et al., 79 the occurrence of endometriosis increased the risk of placenta praevia (OR 4.04, 95%CI: 2.29–7.12). In addition, the deployment of ART in women who were diagnosed with endometriosis significantly increased the likelihood of developing placenta praevia (OR 5.54, 95%CI: 1.66–18.52). According to Gasparri et al., 80 pregnancies achieved using ART were linked to a higher likelihood of developing placenta praevia (OR 2.96, 95%CI: 1.25–7.03). Studies have shown that a history of caesarean birth is a known risk factor for placenta praevia, and that the risk may be further elevated if endometriosis coexists with it. When placenta praevia is diagnosed early in pregnancy, appropriate management is made possible, usually necessitating a scheduled caesarean birth. Nevertheless, these caesarean deliveries in individuals who have both endometriosis and placenta praevia are typically more complex with a higher risk of complications such as postpartum haemorrhage and surgical difficulties. 81
It is crucial to notice that, based on current knowledge, the ability of preventive surgical operations to reduce the detrimental effects of endometriosis on placenta praevia outcomes was not supported.
Preterm birth
The latest meta-analysis from 2022 provided evidence of a positive association between endometriosis and an increased occurrence of preterm birth (PTB) (RR 2.68, 95%CI: 1.19-6.02). 82 Berlac et al. 69 conducted a study which revealed that affected women had a twofold higher likelihood of experiencing such a result compared to women who were not impacted by endometriosis. Furthermore, the evidence indicated that women who were treated with ART and who had endometriosis were at a higher risk of experiencing premature delivery (OR 1.98, 95%CI: 1.09–3.62). 83 The conclusion was further supported by a comprehensive meta-analysis conducted in 2017 by Kim et al. 84 (OR 1.47, 95%CI: 1.22–1.79) for preterm birth in women diagnosed with endometriosis. Furthermore, previous studies conducted by Berlac et al 69 and Harada et al. 85 established a correlation between endometriosis and an increased likelihood of experiencing preterm delivery and premature rupture of foetal membranes. The latter condition exhibited the odds ratio of 1.7.
The causes of PTB are based on the increased production of proinflammatory cytokines, specifically interleukin 6, interleukin 1-β and TNF-κ.86,87 These cytokines are involved in the onset of labour and are also found in the endometrium of women with endometriosis who experience preterm labour. Endometrial lesions were found to have higher levels of prostaglandin E2, cyclooxygenase 2, and a range of cytokines in comparison with the healthy endometrium. Increased levels of progesterone and cytokines in the peritoneal fluid, heightened myometrial activity, and the subsequent duplication of preterm birth risk in individuals with endometriosis compared to a control group were linked to the upregulation of PGE2, COX-2, IL-6 and IL-1b. 38 Notably, the use of ART in cases of endometriosis did not seem to increase the likelihood of premature birth.85,88
Antenatal haemorrhage
The research conducted by Berlac et al. 69 provided insight into the increased likelihood of prenatal and postnatal haemorrhage (PPH) in women who underwent a surgical intervention for endometriosis (OR 2.3, 95%CI: 2.0–2.5). Moreover, a notable probability of placental abruption was reported in those patients (from OR 2.0, 95%CI: 1.7–2.3 to OR 14.5, 95%CI: 1.5–140).89,90
The heightened risk is associated with abnormal blastocyst implantation, which may be attributed to numerous causes, including a modified junctional zone, dysperistalsis, uterine fixity, localised inflammation and concomitant adenomyosis. Women with endometriosis were found to be more likely to experience retained placenta (OR 3.1, 95%CI: 1.4–6.6). 69
A study conducted by Nirgianakis et al. 68 revealed that women who had previously experienced endometriosis were more likely to experience haemorrhagic episodes after giving birth, regardless of whether the illness had spread. The primary factor was ascribed to the maturation of the junctional zone, an elevation in proinflammatory substances, impairment in the contractility of the myometrium, and the development of pelvic adhesions that intensified oxidative stress. Oxidative stress is a condition characterised by an imbalance between oxygen-free radicals and antioxidants, impacting the pathophysiology of endometriosis.
A retrospective cohort research conducted by Shmueli et al. included a total of 130 individuals diagnosed with endometriosis. The analysis revealed a statistically significant increase in the risk of postpartum haemorrhage (adjusted OR (aOR) 3.7, 95%CI: 1.6–8.5). In addition, Shmueli et al. 91 reported a reduction in postpartum haemoglobin levels to below 10 mg/dL (aOR 2.03, 95%CI: 1.31–3.14).
Some authors documented cases of spontaneous rupture of uterine arteries, which occasionally manifested as hemoperitoneum. The persistent inflammation associated with endometriosis might make the utero-ovarian vessels more susceptible to damage. Alternatively, adhesions caused by endometriosis might exert excessive pressure on those vessels when the uterus increased its volume during pregnancy. 92
The chronic inflammatory response observed in the pelvic region is distinguished by the presence of oxidative stress caused by oxygen-free radicals, which are generated as a result of oxygen metabolism. These radicals play a role in causing inflammation in cells, controlling cell growth, and increasing toxicity. Endometriosis is characterised by the amplification of oxidative stress through retrograde menstrual flow, which is facilitated by macrophages, erythrocytes, and ectopic endometrial tissue. The endometriotic foci produce proinflammatory cytokines, including IL-1β, IL-6, IL-8 and TNF. 93 These cytokines create a pathological environment that hinders the growth of follicles, alters the movement of the fallopian tubes, triggers a widespread inflammatory response, and promotes the infiltration of macrophages. The presence of this harmful environment has a negative impact on the survival of gametes and the macrophages in the peritoneal fluid. These macrophages actively engulf sperm, which affects fertility. Additionally, it negatively impacts the ability of the endometrium to receive and implant embryos. 93 Moreover, endometriosis in women leads to an elevation in the peritoneal fluid, which has a high concentration of macrophages, prostaglandins, and proteases. This fluid contains an inhibitor that hinders the contact between ova and fallopian tubes, making the implantation of ova, sperm and embryos more difficult. Placental abruption, associated with the premature rupture of the membrane, is more commonly observed in instances with extensive endometriosis.
Caesarean section
Women with endometriosis have an elevated likelihood of delivering via caesarean section, with studies indicating as much as double the risk compared to women without the condition (OR 1.49 (95%CI: 1.35–1.65) and 1.57 (95%CI: 1.39–1.78)).70,94 Further research by Ribot et al. 38 corroborated this significantly increased prevalence, showing that ca. 50% of women with endometriosis underwent caesarean deliveries, significantly higher in comparison with the 30% rate in those without endometriosis.
The higher occurrence of the caesarean section may be ascribed to the intricate interaction of various elements inherent in endometriosis, including the displacement of pelvic anatomy caused by adhesions and scarring, which may hinder the typical process of labour and impact the positioning of the foetus. Furthermore, the existence of endometrial lesions may elevate the probability of difficulties during the process of childbirth, hence increasing the likelihood of caesarean sections being performed in order to safeguard the well-being of both the mother and the child. Another probable reason for a higher incidence of elective caesarean sections may be the encroachment of endometrial lesions into the structures constituting the delivery canal, resulting in mechanical problems during delivery. Furthermore, issues secondary to caesarean sections, such as adhesions, may be more prevalent in patients with endometriosis, resulting in an elevated risk of bladder injury, intestinal injury and peripartum hysterectomy. 29 Nevertheless, no study has evaluated the increased occurrence of elective or emergency caesarean sections, and the exact pathogenesis of this condition remains unclear. While the presence of DE elevates the probability of caesarean section delivery, it should not be regarded as a definitive indicate for operational delivery.
The summarised influence of endometriosis on pregnancy complications is presented in Figure 3.
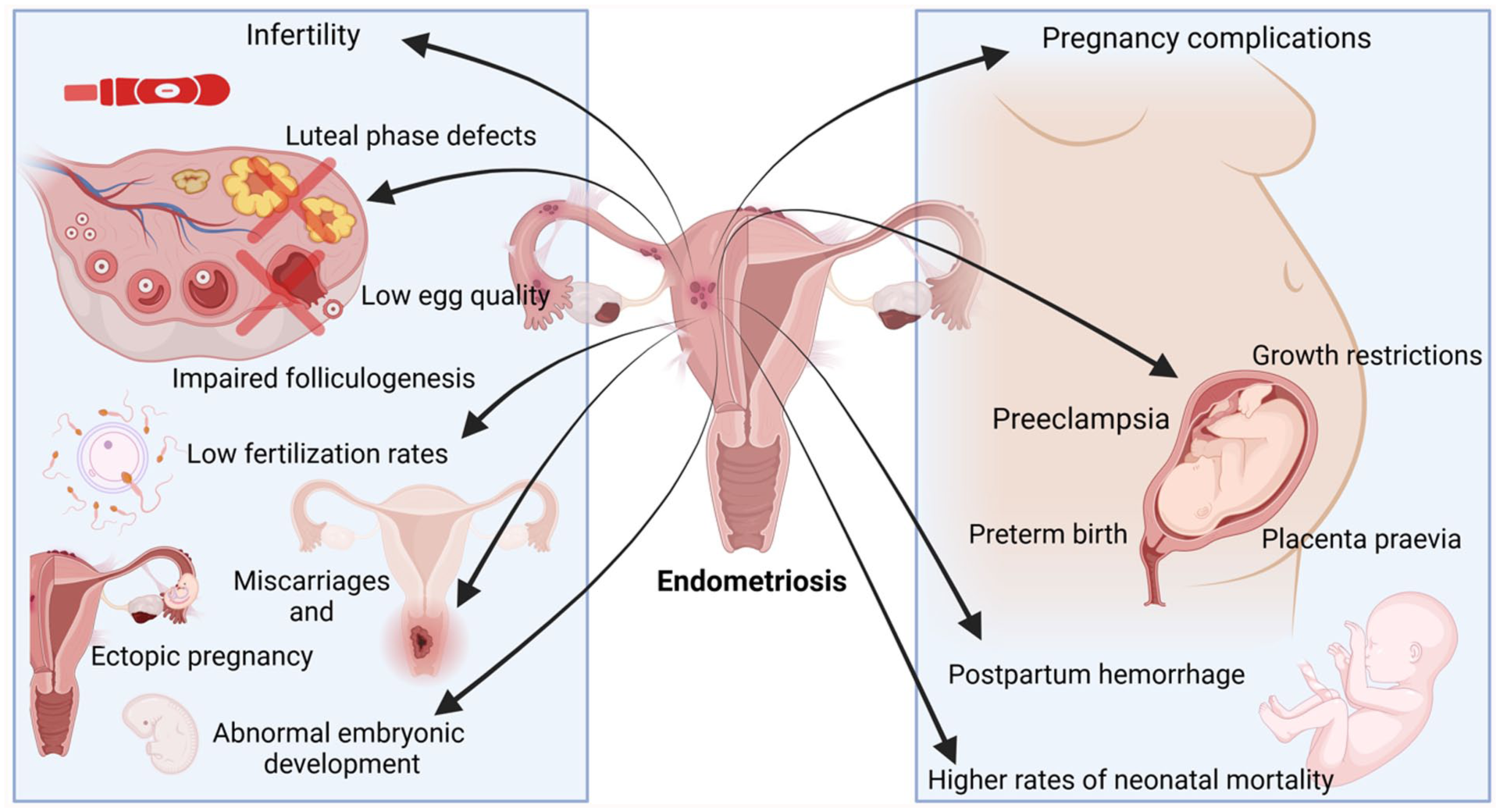
Key impacts of endometriosis on reproductive health, obstetric complications and potential neonatal outcomes.
Microbiome dysbiosis in endometriosis and its implications for fertility and pregnancy outcomes
Recent research has highlighted the potential role of the microbiome in endometriosis and its impact on fertility outcomes. 95 The microbiome refers to the diverse community of microorganisms residing in the reproductive tract, including bacteria. Alterations in the composition and diversity of the microbiota have been observed in individuals with endometriosis, suggesting a possible link between dysbiosis (microbial imbalance and, particularly, the presence of anaerobic bacteria, Actinobacteria, Corynebacterium, Enterobacteriaceae, Flavobacterium, Fusobacterium, Pseudomonas) and the pathogenesis of the condition.95–97 Studies showed that changes in the vaginal and uterine microbiota might influence fertility outcomes by affecting sperm motility, embryo implantation and pregnancy maintenance. 98 Furthermore, dysbiotic microbiota might exacerbate the inflammatory response associated with endometriosis, exacerbating the symptoms and further impairing the reproductive function. 99
Several studies documented alterations in the composition and diversity of the microbiota in individuals with endometriosis compared to those without the condition. These changes involved the reproductive tract and other body sites, such as the gut. 100 Dysbiosis, or microbial imbalance, appears to be a common feature, with shifts in specific bacterial taxa and overall microbial richness observed in endometriosis patients. The presence of dysbiotic microbiota in the reproductive tract may contribute to the pathogenesis of endometriosis through various mechanisms. One proposed mechanism involves the disruption of the mucosal barrier integrity, allowing the translocation of bacteria and inflammatory mediators into the peritoneal cavity. 101 This process may trigger local inflammation and immune dysregulation, fostering the development and progression of endometrial lesions. 102 Furthermore, dysbiosis may exacerbate the chronic inflammatory state associated with endometriosis, perpetuating a feedback loop of tissue damage and immune activation. 102 Inflammatory cytokines and other mediators released in response to dysbiotic microbiota may promote angiogenesis, fibrosis and nerve sensitisation, contributing to the characteristic symptoms of pelvic pain and infertility in endometriosis patients. 42
The impact of the gut microbiome on endometriosis is also of growing interest.103–105 Alterations in gut microbial composition and function have been implicated in systemic inflammation and metabolic dysfunction, both of which may influence the development and progression of endometriosis. Dysbiosis in the gut may lead to increased intestinal permeability and the translocation of microbial products, which may exacerbate pelvic inflammation and pain.106,107 Understanding the role of the microbiome in endometriosis holds promise for novel diagnostic and therapeutic approaches. 96 Strategies aimed at restoring microbial balance, such as probiotics, prebiotics and dietary interventions, may help mitigate inflammation and alleviate symptoms in the individuals affected.108,109 Additionally, targeting dysbiotic microbiota could potentially serve as a preventive or therapeutic strategy for managing endometriosis-related infertility and other reproductive complications.96,107,110
Conclusion
Despite its widespread occurrence, endometriosis remains a subject of perplexity among professionals due to its implications for pregnancy. This condition has a negative impact on both natural and biomedical conception. The disease is associated with impaired folliculogenesis, resulting in negative effects on egg quality, defects during the luteal phase, decreased rates of fertilisation and abnormal embryonic development, all of which contribute to reproductive difficulties. With the growing number of endometriosis patients getting pregnant, there is a growing need for thorough evaluations to determine the potential adverse effects of the condition on pregnancy outcomes, including the postpartum period.
Endometriosis patients have a higher incidence of obstetric complications, such as placental adhesion concerns, growth restriction and an increased prevalence of CS, PTB and PPH. While the presence of DE elevates the probability of caesarean section delivery, it should not be regarded as a definitive indicate for operational delivery. The area of obstetrics frequently fails to completely acknowledge and investigate these intricate matters, resulting in their under-exploration. The precise incidence of endometriosis and the precise mechanisms by which it disrupts pregnancy are currently under investigation. The investigation of these mechanisms is of utmost importance in the development of more efficacious therapeutic interventions for endometriosis. Moreover, the microbiome appears to play a significant role in the pathogenesis and clinical manifestations of endometriosis. Further research is needed to elucidate the precise mechanisms underlying microbiome-endometriosis interactions and to translate these findings into targeted interventions that improve outcomes for patients with this debilitating condition.
The influence of the disease goes beyond the stage of conception, potentially affecting outcomes related to obstetrics and neonatology. Therefore, it is crucial to conduct a focused study in order to ascertain the most effective approaches for managing and monitoring pregnant women who have been diagnosed with endometriosis. Patient outcomes and related issues may be greatly improved by the establishment of specialised, multidisciplinary centres that offer integrated care from preconception through postpartum.
Footnotes
Author contributions
Conceptualisation, S.F.; methodology, N.Z.-L. and S.F.; formal analysis, M.A.; investigation, M.W.; resources, P.B.; data curation, J.K.-B.; writing—original draft preparation, S.F., N.Z-L., M.W., M.A. and P.B.; writing—review and editing, J.K.-B., M.C.; supervision, M.C. All authors have read and agreed to the published version of the manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Data availability statement
Research data is available upon request.
