Abstract
Background:
Current joint-sparing treatment options for massive, irreparable rotator cuff tears aim to prevent superior migration of the humeral head and maintain the subacromial space. Although options such as superior capsular reconstruction and posterosuperior tendon transfer may appropriately restore function and reduce pain, these procedures harbor significant comorbidity and intense postoperative rehabilitation. Recently, studies have demonstrated excellent results from a subacromial balloon spacer composed of a biocompatible copolymer that degrades over the 12 months following implantation. This spacer enables a more anatomic position of the humeral head to restore normal shoulder biomechanics.
Indications:
The subacromial balloon spacer is primarily indicated in patients with massive, irreparable rotator cuff tears who demonstrate proximal humeral migration. Contraindications include subscapularis or teres minor tendon tears, deltoid dysfunction, and pseudoparalysis.
Technique Description:
Standard arthroscopic portals are established, and a diagnostic arthroscopy is performed to assess the glenohumeral joint space and subscapularis tendon. A subacromial bursectomy is performed without proceeding medial to the superior glenoid rim. The subacromial space is measured utilizing an arthroscopic measurement probe to ensure that the selected spacer will lie slightly medial to the glenoid. The rotator cuff is then reapproximated from the lateral to medial aspect of the tear and repaired via a side-to-side manner with high strength, nonabsorbable tape, and a self-capturing suture device. Following repair, the deployment device is inserted through the lateral portal, placed slightly medial to the glenoid, and inflated with saline.
Results:
Utilization of the described subacromial balloon augmentation provides significant pain relief by restoring glenohumeral biomechanics, improvements in range of motion, and an enhancement in quality of life.
Discussion:
This case report and video review of a technique for utilizing a subacromial balloon spacer demonstrates an effective, simple, and safe treatment for massive, irreparable rotator cuff tears. Positive results rely on appropriate inflation and secure fixation. Recent clinical trial data suggest that functional outcomes for the subacromial balloon spacer are noninferior to that of partial rotator cuff repair. These promising results suggest that the subacromial balloon spacer may prove an essential tool for joint-preserving surgery following massive, irreparable rotator cuff tears.
This is a visual representation of the abstract.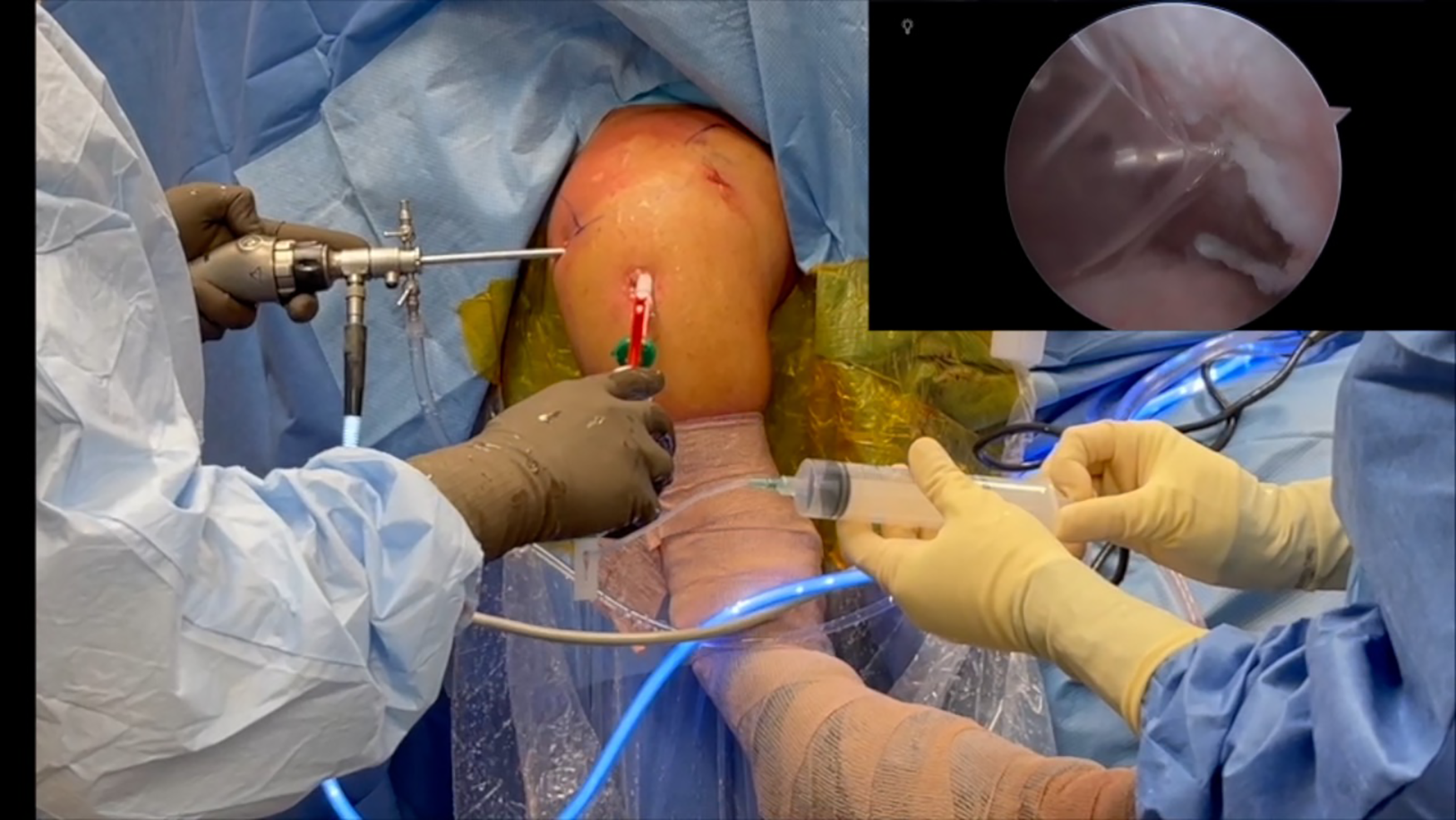
Video Transcript
The following video describes our surgical technique for the combined use of a margin convergence repair and absorbable subacromial balloon spacer in the treatment of massive rotator cuff tears.
These are our disclosures.
We will begin with a background on the treatment options for irreparable rotator cuff tears, followed by a case presentation, where we will discuss preoperative planning as well as surgical technique for the implantation of a subacromial balloon spacer. The video will then conclude with postoperative management guidelines, followed by a review of patient outcomes in the literature.
Massive, irreparable rotator cuff tears continue to remain a challenging pathology to treat. While reverse total shoulder arthroplasty (RTSA) may provide an excellent resolution for pain and disability in these patients, the RTSA construct limits motion and may not be a satisfactory option for younger, active patients. To delay or mitigate need for RTSA, either a superior capsule reconstruction or tendon transfer has historically been used as alternative options to arthroplasty. However, these procedures are either technically demanding or involve a prolonged postoperative rehabilitation period.
In recent years, an implantable subacromial balloon spacer has emerged with promising early results. The balloon is composed of a polylactide and ϵ-caprolactone copolymer. Although the balloon is believed to biodegrade within 12 months, studies have demonstrated long-term benefits for up to 5 years. The balloon acts as a barrier to subacromial impingement and lowers the humeral head closer to its anatomic position to restore normal shoulder biomechanics. Our technique describes the utilization of a subacromial balloon spacer in the management of a recurrent and irreparable posterosuperior rotator cuff tear.
Our patient is a 53-year-old woman who presented for the evaluation and management of ongoing right shoulder pain. She has a history of 2 prior rotator cuff repairs of the right shoulder, with the most recent being 4 to 5 years ago. Although she reports regaining 100% of function after her revision surgery, she has developed increasing pain and difficulty with activities of daily living in the last couple of years. Her pain is a 0/10 at baseline and 10/10 at its peak. At present, the patient reports having 25% of normal function in her right shoulder.
On physical examination, she has preserved active and passive motion in forward flexion and external rotation. Examination of the rotator cuff strength reveals 4+/5 supraspinatus and infraspinatus strength.
Anteroposterior and axillary radiographs reveal well-preserved glenohumeral joint space.
Coronal T2-weighted imaging revealed a full-thickness rotator cuff tear with medialization to the glenoid.
Sagittal magnetic resonance (MR) imaging (MRI) confirms the massive nature of this rotator cuff tear.
Last, on axial MR imaging, the subscapularis tendon is noted to be intact.
The subacromial balloon spacer is a good option for patients with massive irreparable rotator cuff tears who have preserved forward elevation and minimal arthritis. Those with deltoid dysfunction or tears of the subscapularis tendon or teres minor are not ideal candidates for this procedure. However, there is debate on whether the balloon spacer is an option in patients where either the subscapularis or teres minor tendons can be concurrently repaired at the time of surgery.
Our preoperative counseling with the patient included a discussion that the patient could ideally expect a significant pain reduction, with clinically significant improvements in function. In addition, compared with alternative strategies for the treatment of massive, irreparable rotator cuff tears such as a superior capsular reconstruction, the subacromial balloon spacer would allow an accelerated rehabilitation protocol.
We start with a diagnostic arthroscopy, which confirmed well-preserved glenohumeral joint space and a massive rotator cuff tear. Care is taken to ensure that the subscapularis tendon is intact. After completion of our intra-articular assessment and treatment of pathology, we then proceed to the subacromial bursectomy; care is taken to avoid too aggressive of a bursectomy medial to the superior glenoid rim, which will help prevent postoperative balloon migration. We then proceed to remove of the retained sutures from the prior rotator cuff repairs. In this patient, the torn rotator cuff tendon was confirmed to be chronic in nature without any significant excursion to the greater tuberosity. However, the posterosuperior rotator cuff was noted to have significant excursion and would benefit from a side-to-side repair.
After placement of an arthroscopic portal, we then used an arthroscopic measurement probe to evaluate the size of the subacromial space. The spacer used in this case comes in 3 sizes: small, medium, and large. These sizes vary in medial-lateral width by 40, 50, and 60 mm, respectively. Care must be taken to ensure that the spacer will lie slightly medial to the glenoid. For this patient, a medium balloon spacer was chosen.
We then proceeded to a routine acromioplasty using the lateral cutting block technique.
For the margin convergence repair, an ultra-high strength, nonabsorbable tape was used with a self-capturing suture device to perform a side-to-side repair. The rotator cuff was reapproximated starting at the lateral most extent of the tear, going medial. Final inspection of the repair confirmed that the ends were well-reapproximated.
All cannulas are removed from the lateral portal, and the deployment device is inserted. Care is taken to ensure that the end of the deployment device is slightly medial to the glenoid.
With the surgeon holding the device in place, an assistant can place a syringe filled with saline at the tip of the device. The sheath protecting the spacer is then retracted, and the final planned position of the balloon is confirmed. For the remainder of the steps, the surgeon should maintain control of the device while an assistant proceeds to inflate the balloon.
The amount of saline needed to inflate the balloon will vary by the size of the implant used. For this medium spacer, 24mL of saline was inserted to inflate the balloon, followed by removal of 8mL.
A clip on the top of the deployment device is turned, which then seals the balloon. The deployment device can then be removed.
With the spacer completely deployed, a final look in the subacromial space can confirm appropriate placement of the spacer. An intraoperative range of motion can also be performed to ensure that the balloon does not migrate.
Our tips for this procedure involve avoiding too aggressive of a bursectomy medial to the superior glenoid rim and maintaining a firm grasp on the deployment device during inflation to prevent inadvertent migration. We recommend drawing back the specified amount of saline after full inflation, so you do not overinflate the balloon.
Complications reported in the literature include infection, transient lateral antebrachial cutaneous nerve neuropraxia, balloon migration, and over-inferioralization or anteriorization of the humeral head.
We start a 3-phase rehabilitation postoperatively for our patients.
Phase 1 involves wearing a sling at all times except for therapy and home exercises. Passive range of motion is allowed, with active-assisted exercises after 2 weeks postoperative.
Phase 2 involves progressing toward achieving a full active range of motion out of the sling.
The final phase involves progressive strengthening exercises, including activity-specific routines.
In this systematic review of the available literature, the overall complication rate was low, with a 2.5% conversion rate to a reverse total shoulder at mean 22.9 months of follow up. Patients also had significantly improved patient-reported outcomes, with improvements in mean constant scores ranging from 18.5 to 49.6 points. In prospective, multicenter, randomized controlled trials, the balloon spacer has also been found to be noninferior to partial repair in the setting of massive rotator cuff tears for achieving minimal clinically important difference. In this study, specifically examining results following a rotator cuff repair protected with a subacromial spacer, patients had significantly improved patient-reported outcomes with MRI scans identifying that repair remained intact in 81.3% of patients a minimum 24 months postoperative.
In conclusion, the combined placement of a subacromial balloon spacer and rotator cuff repair can be an acceptable treatment for massive rotator cuff tears in improving function and limiting pain. This procedure may serve as a bridge to a RTSA, with high survivorship in short-term follow-up.
Here are the references cited for this video.
We would like to thank you for your time and watching this technique video for the combined use of a subacromial spacer and rotator cuff repair in the treatment of massive rotator cuff tears.
Footnotes
Submitted April 25, 2022; accepted June 9, 2022.
One or more of the authors has declared the following potential conflict of interest or source of funding: BF is a Board or committee member for the American Orthopaedic Society for Sports Medicine; he received research support from Arthrex, Inc; he received publishing royalties, financial, or material support from Elsevier; received stock or stock options from i-BrainTech; received stock or stock options from JACE Medical; received stock or stock options from Sparta Biopharma; is paid as a consultant and received research support from Smith & Nephew; is a paid consultant and received research support from Stryker; he is also on the Editorial or Governing Board of Video Journal of Sports Medicine. NNV is a Board or committee member of American Orthopaedic Society for Sports Medicine; a Board or committee member of American Shoulder and Elbow Surgeons; is a paid consultant and received research support from Arthrex, Inc; received research support from Breg; received stock or stock options from CyMedica; received stock or stock options from Omeros; received research support from Ossur; is on the Editorial or Governing Board of SLACK Incorporated; received IP royalties and research support from Smith & Nephew; is a paid consultant of Stryker; received publishing royalties, financial, or material support from Vindico Medical-Orthopaedics; and received research support from Wright Medical Technology, Inc. MH is a paid consultant of Moximed. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
