Abstract
Background:
Subacromial balloon spacers have been introduced as a potential treatment option for patients with massive irreparable rotator cuff tears. However, it is important to comprehensively assess the clinical efficacy of this procedure in the context of an increasing amount of contemporary literature.
Purpose:
To perform a systematic review of the contemporary literature to understand the propensity for clinically meaningful improvements after subacromial balloon spacer implantation for massive irreparable rotator cuff tears.
Study Design:
Systematic review and meta-analysis; Level of evidence, 4.
Methods:
The PubMed, Ovid/MEDLINE, and Cochrane databases were queried in July 2022 for data pertaining to studies reporting clinically significant outcomes after subacromial balloon spacer implantation. Freeman-Tukey double arcsine transformation was used to quantify the pooled rate of clinically meaningful improvements in outcomes as evaluated using the minimal clinically important difference (MCID), Patient Acceptable Symptom State (PASS), and substantial clinical benefit (SCB). Qualitative analysis was performed when data were variably presented to avoid misleading reporting.
Results:
There were 10 studies included, all of which reported MCID achievement. The overall pooled rate of MCID achievement for the Constant-Murley score was 83% (95% CI, 71%-93%; range, 40%-98%), with 6 of 8 studies reporting rates equal to or exceeding 85%. One study reported a 98% rate of PASS achievement for the Constant-Murley score at 3-year follow-up. The rate of MCID achievement for the American Shoulder and Elbow Surgeons (ASES) score ranged between 83% and 87.5%. The rate of PASS achievement for the ASES score was 56% at 2-year follow-up, while the rate of SCB achievement for the ASES score was 83% and 82% at 1- and 2-year follow-up, respectively. At 1-year follow-up, 74% and 78% of patients achieved the MCID for the Numeric Rating Scale and Oxford Shoulder Score, respectively. At 3 years, 69% of patients achieved the MCID for the Numeric Rating Scale and 87% achieved it for the Oxford Shoulder Score.
Conclusion:
Patients who underwent isolated subacromial balloon spacer implantation for massive irreparable rotator cuff tears demonstrated a high rate of clinically significant improvement in outcomes at short- to mid-term follow-up. A paucity of literature exists to appropriately define and evaluate the rates of achieving the PASS and SCB after subacromial balloon spacer implantation, necessitating further study.
Massive irreparable tears of the rotator cuff remain a challenging clinical entity without a standard treatment approach. 25 Previous treatment options have included nonoperative management, partial repair, debridement, superior capsular reconstruction, and reverse total shoulder arthroplasty. In recent years, the implantable subacromial balloon spacer has been introduced as a novel potential treatment option. This device offers a relatively quick and low-risk alternative option for the treatment of massive irreparable rotator cuff tears by depressing the humeral head and reducing subacromial friction during shoulder abduction, thereby restoring glenohumeral biomechanics and potentially ameliorating pain.1,14,22 Despite plausible clinical advantages conferred by the biomechanical changes imparted by this device, it is imperative to better understand the clinical utility of balloon spacer implantation and the potential for improvements in patient-reported outcomes, given its recent introduction into clinical practice.
Preliminary clinical data have suggested that balloon spacer implantation has the potential to improve pain and function at short-term follow-up.2,26 Familiari et al 3 performed a retrospective review of 51 patients who underwent balloon spacer implantation for massive irreparable rotator cuff tears and reported that significant improvements were observed for the Constant-Murley score (CMS) as well as the 12-item Short Form Health Survey (mental and physical) at a mean 36-month follow-up. Additionally, 90.2% of patients reported satisfaction with their outcome at this follow-up time point. A recent double-blind randomized controlled trial involving 117 patients demonstrated that patients experienced significant improvements on the Oxford Shoulder Score (OSS) and CMS at 12-month follow-up, although there may not be a clinically meaningful difference between balloon spacer implantation and debridement for irreparable rotator cuff tears. 16 Early systematic reviews have corroborated that patients can experience significant clinical improvements and reductions in pain, on average, with this procedure in the short term9,11,23; however, it is imperative that an updated assessment of contemporary clinical outcomes be performed because of the increase in the overall number of publications on this topic since the time that these previous reviews were published.
Although implantable balloon spacers are now being increasingly used across health care institutions both in the United States and globally, the most recent collective assessments on the clinical outcomes of this procedure have restricted study inclusion time frames to earlier dates, and therefore, they do not reflect the most recent investigations on its use.9,11,13,23 Furthermore, none of these reviews assessed the propensity for clinically meaningful improvements in outcomes after balloon spacer implantation as evaluated using the minimal clinically important difference (MCID), Patient Acceptable Symptom State (PASS), and substantial clinical benefit (SCB). As such, the purpose of the current study was to perform a systematic review of the contemporary literature to understand the propensity for clinical improvements after subacromial balloon spacer implantation for massive irreparable rotator cuff tears. We hypothesized that the most recent literature would reflect continued clinical improvements in the context of increasing familiarity with the procedure and that a high rate of clinically meaningful improvement in outcomes would be observed.
Methods
Article Identification and Selection Process
The current systematic review was conducted in accordance with the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) statement. 17 Using the following online databases, we performed a systematic review of the literature pertaining to the clinically significant outcomes after subacromial balloon spacer implantation for patients with massive irreparable rotator cuff tears: (1) Cochrane Database of Systematic Reviews/Cochrane Central Register of Controlled Trials, (2) PubMed (2000-2019), and (3) Ovid/MEDLINE. The query was performed in July 2022 using the following Boolean search and Medical Subject Headings terms: “rotator cuff tear” OR “rotator cuff injury” OR “irreparable rotator cuff tear” OR “massive rotator cuff tear” AND “subacromial balloon spacer” OR “subacromial spacer” AND “outcomes.” This review was registered with PROSPERO (registration No. 376280).
Articles were qualified for inclusion in this systematic review if their topics study pertained to any investigation on the clinical outcomes of subacromial balloon spacer implantation and they were published in the English language. Studies were restricted to levels of evidence 1 to 4. Exclusion criteria consisted of (1) articles concerning cadaveric, basic science, or animal models; (2) imaging and technique articles that did not report outcomes related to the intervention; (3) editorials, abstracts, case reports, and surveys; and (4) articles classified as level of evidence ≥5.
All articles identified via the search were screened by 2 independent reviewers (J.M., R.C.). Sequential screening of the articles was performed using the following systematic approach: assessment of duplicate articles, examination of the article title, evaluation of the content of the abstract, and full-text review. A full-text review was only performed during the study selection process if necessary to determine if the articles satisfied inclusion and exclusion criteria. Additionally, all references from the included studies were reviewed and reconciled to verify that no relevant articles were missing from the systematic review.
Data Extraction Process
All data were recorded into a custom spreadsheet using a modified information extraction table. 6 Categories for data collection for each full article included (1) article information; (2) indications for balloon spacer implantation; (3) clinical outcome measures; (4) rates of MCID, PASS, or SCB achievement; (5) measures of function (ie, strength and range of motion); and (6) adverse events pertaining to the procedure.
Assessment of Heterogeneity and Methodological Quality
The methodological index for non-randomized studies (MINORS) checklist 7 was used to evaluate the quality of all included studies. The checklist involves 12 items to assess quality, of which 4 are only applicable to comparative studies. The 4 additional criteria specific to comparative groups were used to assess the bias present in articles when selecting cohorts. The maximum MINORS score is 16 for noncomparative studies and 24 for comparative studies. There were 2 reviewers (J.M., R.C.) who assessed and graded each included study, and any discrepancies were resolved by a consensus agreement or excluded altogether. The interobserver reliability was excellent at 0.98 (95% CI, 0.97-0.99). Any discrepancies were resolved via a consensus.
Statistical Analysis
All data were qualitatively synthesized and reported in a narrative fashion and table format as well as quantitatively displayed in forest plots. Extracted data were presented as means and ranges when appropriate. Studies including patients derived from the same population but reporting clinical outcomes at different follow-up points were not excluded to present a more comprehensive assessment of a clinically significant achievement in outcomes; however, only the study with the longest follow-up was included in the formal meta-analysis. All studies considered a P value <.05 to indicate statistical significance. Where possible, the rate of clinically significant improvement in outcomes was pooled using a meta-analysis of proportions. To compensate for the heterogeneity of the data, we used an inverse-variance proportions model using Freeman-Tukey double arcsine transformation to quantify the pooled rate. 12 The 95% CI was used to report all pooled statistics. Heterogeneity was assessed via the P value of chi-square statistics and the I2 statistic using a random-effects model. We regarded heterogeneity as possibly unimportant when the I2 value was <40% and considerable when it was >75%. 8 All statistical analyses were performed using the computing software R (Version 3.6.1; R Foundation for Statistical Computing) and RStudio (Version 1.2.5033). Graphical depictions were generated using RStudio (Version 1.2.5033) and GraphPad Prism (Version 9.4.1; GraphPad Software).
Results
A total of 10 studies representing data obtained from 748 patients of which 379 underwent subacromial balloon spacer implantation were included (Figure 1). The pooled mean (±SD) age of all patients was 67.9 ± 3.3 years. Overall, 375/748 patients (50%) were male. Additional patient and study characteristics are described in Table 1. The mean MINORS score for noncomparative studies was 10.0 ± 0.53, indicating good methodological quality. For comparative studies, the mean MINORS score was 21.5 ± 2.00, indicating excellent methodological quality (Figure 2).
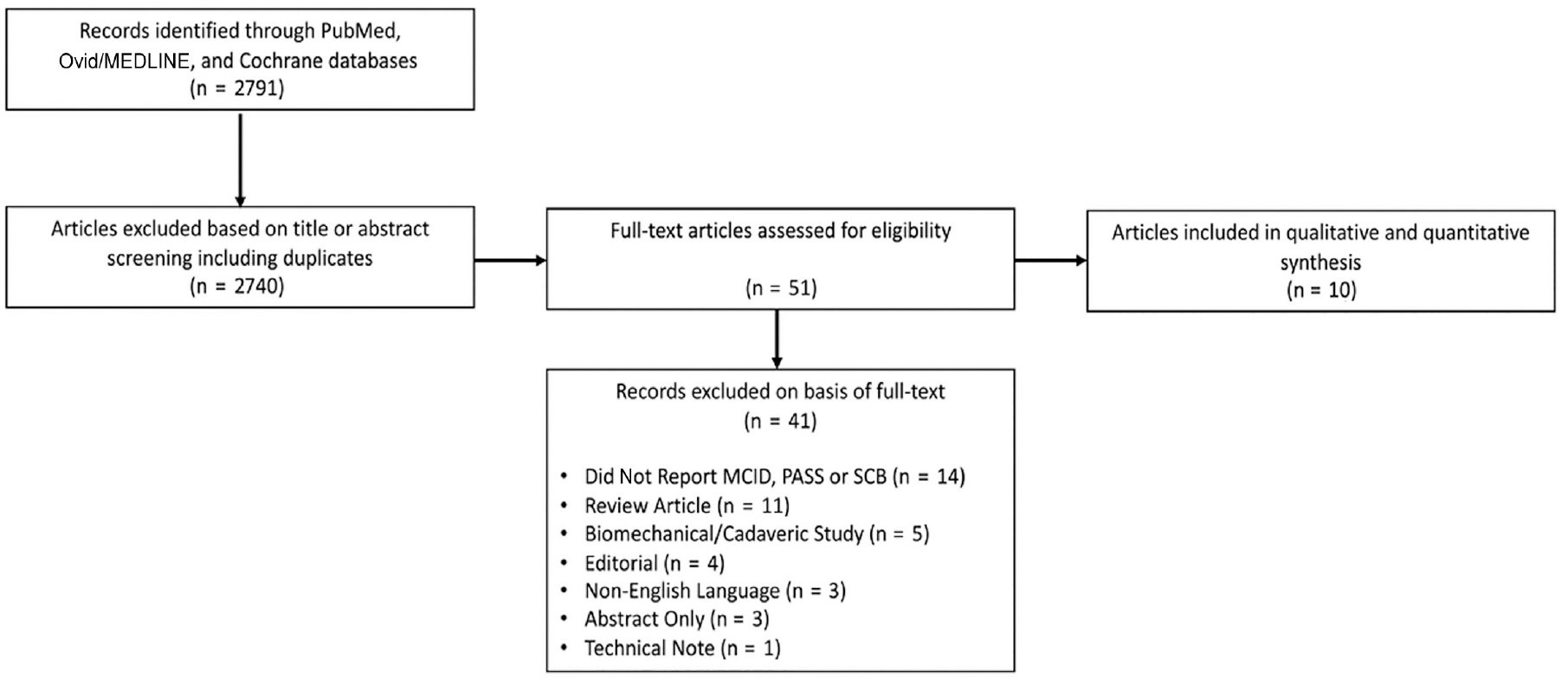
Study selection and eligibility flowchart for final inclusion. MCID, minimal clinically important difference; PASS, Patient Acceptable Symptom State; SCB, substantial clinical benefit.
Study and Patient Characteristics a
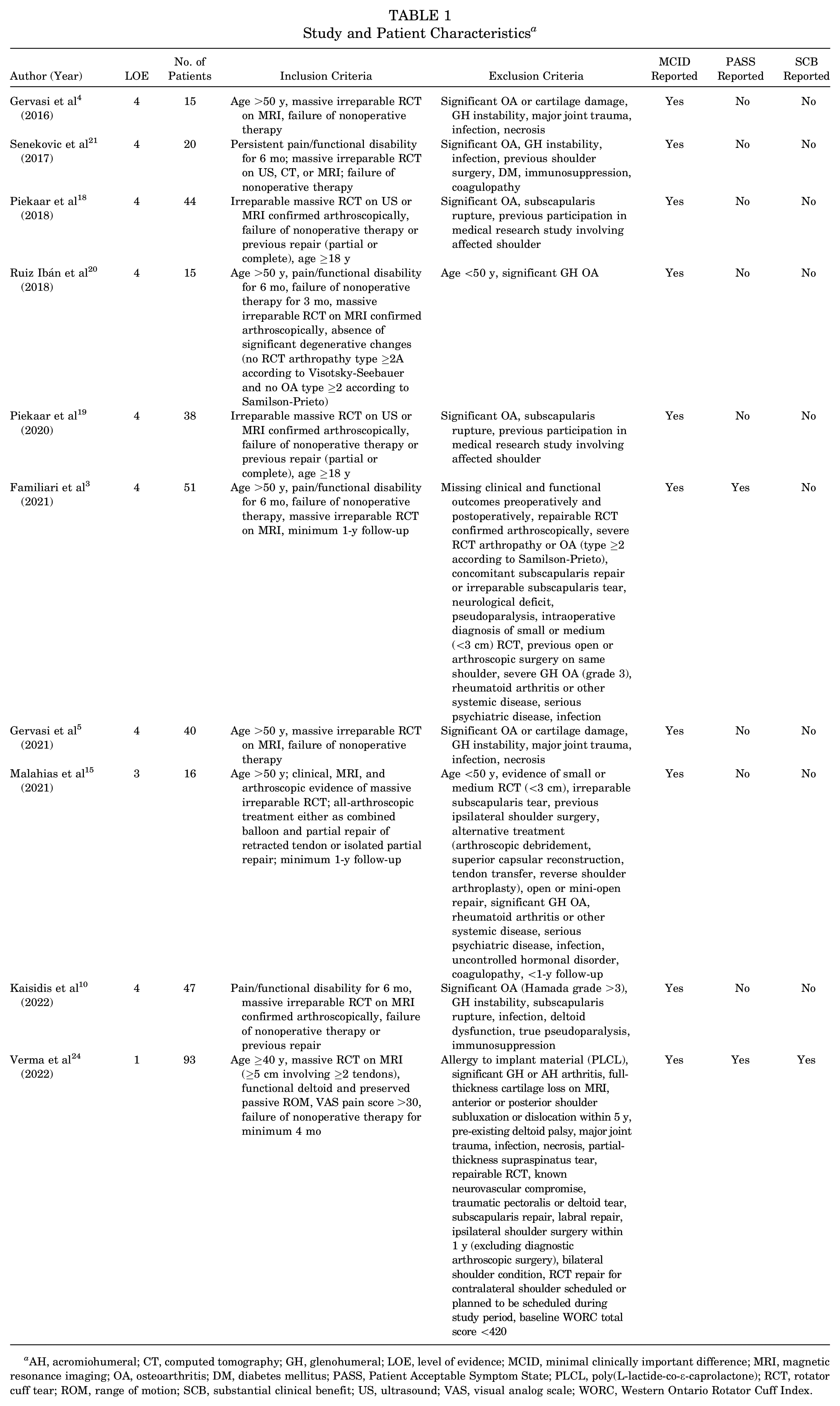
AH, acromiohumeral; CT, computed tomography; GH, glenohumeral; LOE, level of evidence; MCID, minimal clinically important difference; MRI, magnetic resonance imaging; OA, osteoarthritis; DM, diabetes mellitus; PASS, Patient Acceptable Symptom State; PLCL, poly(L-lactide-co-ε-caprolactone); RCT, rotator cuff tear; ROM, range of motion; SCB, substantial clinical benefit; US, ultrasound; VAS, visual analog scale; WORC, Western Ontario Rotator Cuff Index.
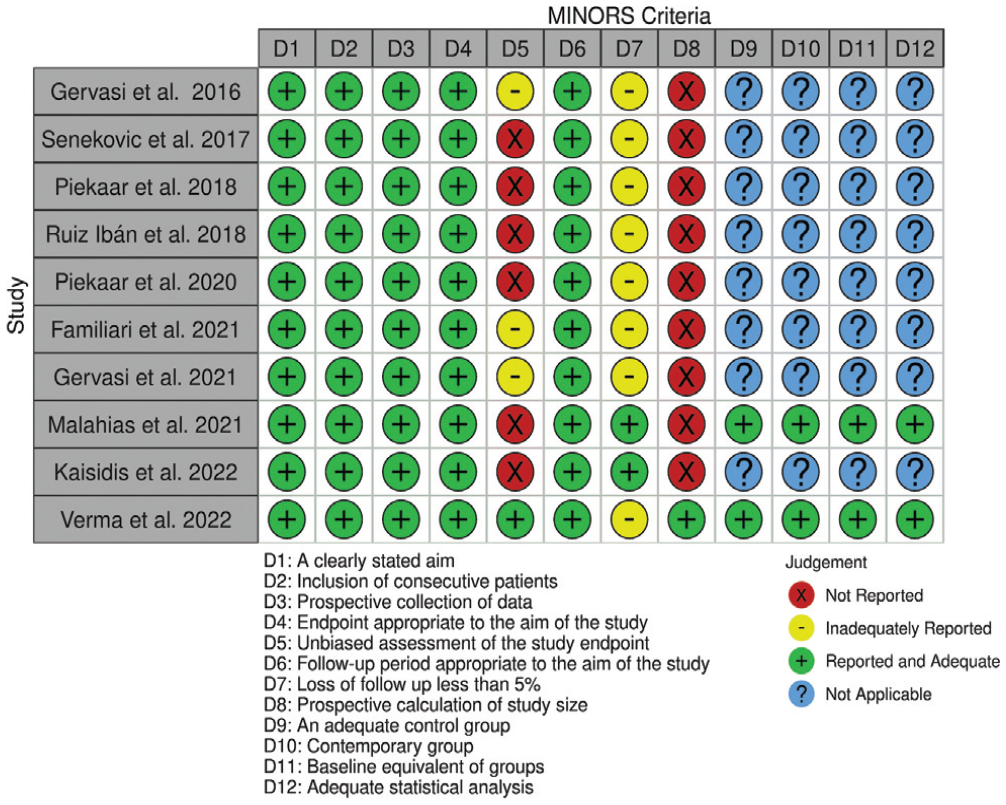
Traffic light plot demonstrating the risk of bias in each study as evaluated using methodological index for non-randomized studies (MINORS) criteria.
MCID for CMS
All 10 included studies reported the MCID for at least 1 patient-reported outcome measure. Of these studies, 8 reported MCID achievement rates for the CMS. 4 The overall pooled rate of MCID achievement for this outcome measure was 83% (95% CI, 71%-93%) (Figure 3). Among all studies, rates ranged between 40% and 98%, with 6 of 8 studies reporting achievement rates equal to or exceeding 85%. 4 The 2 studies reporting rates <85% reported an achievement rate of 40% at 2-year follow-up 20 and 66% at 1-year follow-up. 18
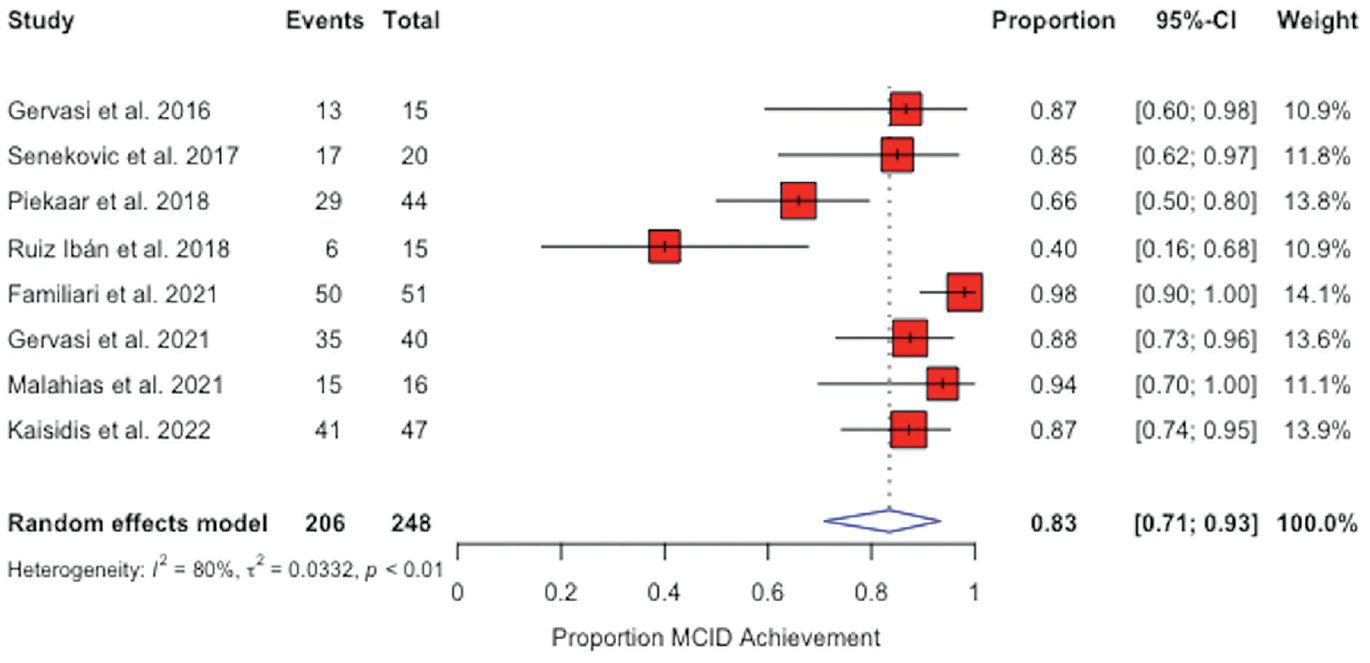
Forest plots for the pooled proportion of minimal clinically important difference (MCID) achievement for the Constant-Murley score across all follow-up time points. The diamond represents the pooled rate of MCID achievement. Black lines indicate 95% CIs. Squares indicate the weight of the individual studies. Black vertical lines within the red boxes indicate observed study effect sizes.
Piekaar et al 18 reported 1-year MCID achievement rates for a study population of 44 patients who underwent subacromial balloon spacer implantation for massive irreparable rotator cuff tears. They defined achievement of the MCID for the CMS as an 11-point improvement. A statistically significant improvement was reported in all 4 CMS categories, although only 66% achieved the MCID per their threshold. However, 11 patients (25%) underwent partial rotator cuff repair but were not separated in this analysis.
Ruiz Ibán et al 20 reported on the outcomes of subacromial balloon spacer implantation for irreparable posterosuperior rotator cuff tears in a small study sample consisting of 15 patients at 2-year follow-up. They reported that 5 patients required conversion to reverse total shoulder arthroplasty because of unsatisfactory outcomes between 6 and 16 months after balloon spacer implantation. In all 5 cases, no remains of the balloon spacer were found during surgery, although before 12 months, the presence of the implant was still to be expected. Another 4 patients did not demonstrate improvements at 2 years, defined as the absence of reoperations and no CMS improvement exceeding 10 points. Therefore, 6 of 15 patients achieved the MCID after index balloon spacer implantation. Notably, 3 patients in this cohort who did not achieve the MCID had previous failed rotator cuff repair.
When stratifying MCID achievement for the CMS temporally, we found the rates ranged between 65% and 87.5% at 1-year follow-up, 40% and 87.5% at 2-year follow-up, and 85% and 98% at 3- to 5-year follow-up (Figure 4).
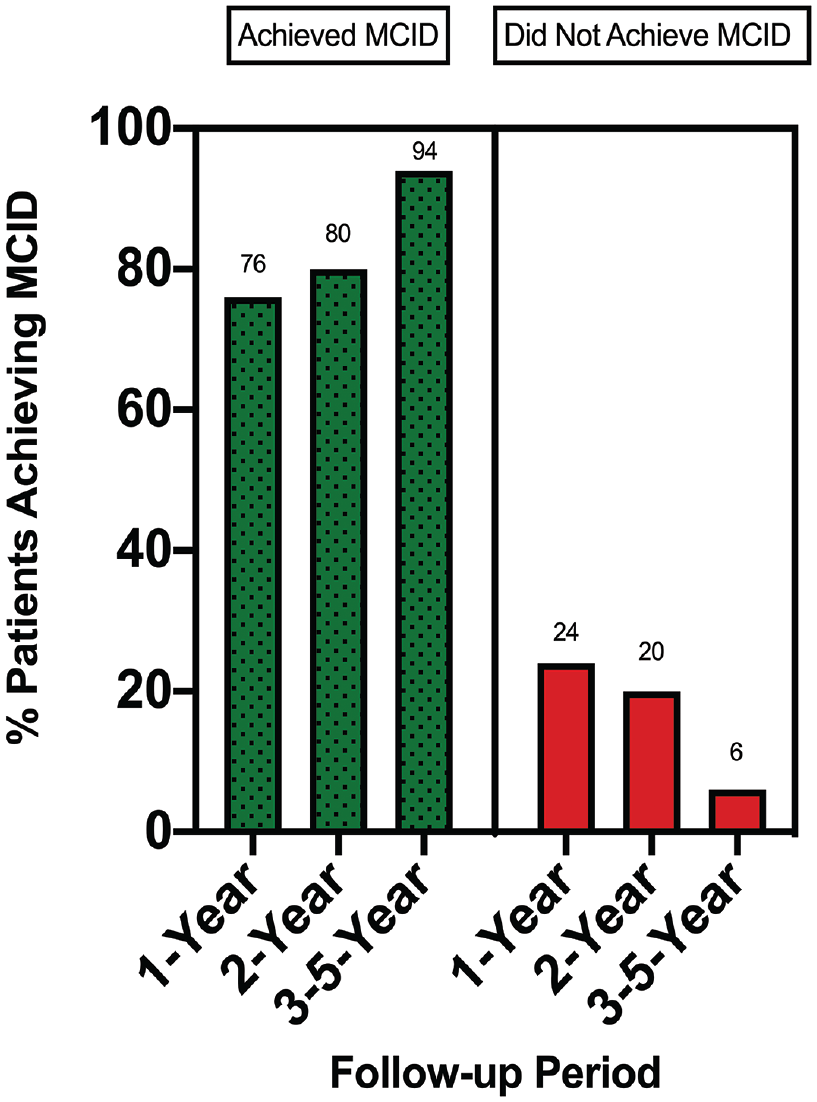
Nested proportions plot demonstrating temporal trends in minimal clinically important difference (MCID) achievement for the Constant-Murley score. The numbers above each bar represent the percentage of MCID achievement at each follow-up point (n = 3 studies at 1-year follow-up, n = 3 studies at 2-year follow-up, and n = 2 studies at 3- to 5-year follow-up).
MCID for American Shoulder and Elbow Surgeons Score
A total of 2 studies reported on the MCID for the American Shoulder and Elbow Surgeons (ASES) score.15,24 The overall rate of MCID achievement ranged between 83% and 87.5%. At 1-year follow-up, the achievement rate was 87% to 87.5% in these 2 studies. Malahias et al 15 performed a matched-pair study of 32 patients with massive irreparable rotator cuff tears who underwent partial repair with balloon spacer implantation (n = 16) versus partial repair alone (n = 16). They reported that the MCID, defined as an improvement of 17 points, was achieved by 87.5% of patients in both groups, although the overall mean ASES score in the repair with spacer group was 10 points greater than that in the repair only group.
Verma et al 24 performed a randomized controlled trial in which 93 patients were allocated to receive a subacromial balloon spacer only and 91 patients were allocated to undergo partial rotator cuff repair. The rate of MCID achievement was 87% in both groups at 1-year follow-up. Overall, 83% of patients in the subacromial balloon spacer group and 81% of patients in the partial repair group achieved the MCID for the ASES score at 2-year follow-up.
MCID for OSS
A total of 2 studies reported the MCID for the OSS.18,19 Piekaar et al 18 reported the rate of clinically significant improvement in outcomes at 1 year after balloon spacer implantation in 44 patients with shoulder pain due to irreparable massive rotator cuff tears confirmed initially using ultrasonography or magnetic resonance imaging and reconfirmed during arthroscopic surgery. They reported that the overall rate of MCID achievement for the OSS was 78% at this time point. This same research group published a 3-year follow-up study, reporting that among 38 patients who were not lost to follow-up, 87% achieved the MCID for the OSS at 3 years postoperatively. 19
MCID for Numeric Rating Scale for Pain
The rate of clinically significant reductions in pain, as determined via MCID achievement for the Numeric Rating Scale (NRS), was reported in 2 studies. Piekaar et al 18 reported that at 1-year follow-up, 74% of patients achieved the MCID for the NRS. At 3-year follow-up, this rate was reduced to 69%. 19
Patient Acceptable Symptom State
There were 2 studies that reported the PASS for ≥1 patient-reported outcome measure.3,24 Familiari et al 3 defined the PASS threshold as 44 points for the CMS and assessed outcomes at 3 years after isolated subacromial balloon spacer implantation. A total of 50 patients (98%) achieved the PASS at 3-year follow-up. Verma et al 24 reported that 56% of patients who received balloon spacers achieved the PASS for the ASES score at 2-year follow-up compared with 51% of those in the partial repair group.
Substantial Clinical Benefit
One study reported the rate of SCB achievement for the ASES score at both 1- and 2-year follow-up. 24 At 1-year follow-up, Verma et al 24 reported that 83% of patients in the balloon spacer group achieved the SCB while 81% in the partial repair group achieved the SCB. Overall, 82% of patients in the balloon spacer group and 79% of patients in the partial repair group achieved the SCB for the ASES score at 2-year follow-up.
Discussion
The main findings of the current study are as follows: (1) among the literature reporting on the rates of clinically significant achievement in outcomes after subacromial balloon spacer implantation, a high rate of achieving the MCID was observed for the CMS, ASES score, OSS, and NRS; (2) the current literature reporting on the proportion of patients meeting the PASS and SCB demonstrated varying levels of achievement depending on the outcome reported; and (3) a paucity of literature had defined PASS and SCB thresholds for clinically relevant outcome metrics after subacromial balloon spacer implantation.
Outcomes regarding the CMS were most frequently reported, with the overall pooled rate of MCID achievement being 83%, as determined via a meta-analysis of proportions. Furthermore, MCID achievement rates remained high across all follow-up periods when temporally stratified. All except 2 studies18,20 identified in this review reported MCID achievement rates equal to or exceeding 85% for the CMS, suggesting that MCID achievement for the CMS is reproducible in this population. Ruiz Ibán et al 20 reported the lowest rate of achievement of 40% at 2-year follow-up; however, their study exhibited several limitations potentially influencing this assessment. First, their study included the smallest sample size of 15 patients, which may lead to large fluctuations in overall achievement rates should a small number of patients fail to meet the MCID. Second, 3 of the patients not meeting the MCID had a history of failed rotator cuff repair, introducing heterogeneity into their study population and potentially raising the clinically important threshold. Finally, a substantial proportion of their study population (33%) converted to reverse total shoulder arthroplasty between 6 and 16 months but may still have experienced clinically meaningful improvements before conversion. Piekaar et al 18 reported the second lowest rate of MCID achievement at 66%. Potential reasons for their low relative rate of MCID achievement include the integration of patients who underwent partial rotator cuff repair with balloon spacer implantation. As suggested when considering the outcomes reported by Ruiz Ibán et al, this may introduce heterogeneity into the perception of what is considered clinically meaningful and falsely elevate the threshold for these patients. Although there exists some conflicting evidence regarding the propensity for clinically meaningful improvements in outcomes after subacromial balloon spacer implantation in the current literature, data from this study suggest that the majority of patients can expect to achieve clinically important improvements in function and symptoms after this procedure at short-term follow-up.
Studies reported MCID achievement for the ASES score, NRS, and OSS variably and less frequently relative to the CMS. However, in studies reporting on these outcomes, the majority of patients were determined to have achieved a clinically significant improvement postoperatively.15,18,19,24 Indeed, the overall rate of MCID achievement ranged between 83% and 87.5% for the ASES score and between 78% and 87% for the OSS, while the proportion of patients achieving a clinically significant reduction in pain (NRS) ranged between 69% and 74%. These findings suggest that improvements in pain and function cited by the previous literature, presumed to be the result of favorable biomechanical changes including restoration of the acromiohumeral interval, normalization of the glenohumeral center of pressure, and increased deltoid loads,1,14,22 may also translate into clinically meaningful improvements as perceived by patients.
The results of this study are an extension and advancement of previously published research examining the clinical outcomes of subacromial balloon spacer implantation by providing insight into whether favorable short-term outcomes may represent clinically meaningful changes in symptoms and function. Stewart et al 23 published a qualitative systematic review involving 12 clinical studies and 284 patients. In accordance with the current study results, the CMS was the most frequently used metric in 11 of 12 studies, with the mean improvement ranging between 18.5 and 49.6 points. These substantial changes provide plausibility for how a high rate of MCID achievement was observed in this study based on thresholds between 10 and 11 points. Likewise, Johns et al 9 performed a systematic review of clinical, biomechanical, and functional outcomes associated with subacromial balloon spacer implantation for massive irreparable rotator cuff tears. Their review included 19 studies with 337 patients. At a mean 33-month follow-up, the authors reported significant improvements for the CMS, the OSS, the ASES score, and shoulder range of motion. In conjunction with our results, the improvements noted in raw outcome scores across the existing literature appear to translate into clinically meaningful improvements based on the available data.
Only 2 of the studies3,24 identified in this systematic review reported on the rate of PASS achievement after subacromial balloon spacer implantation, while only 1 study 24 reported on SCB achievement. Verma et al 24 reported on the rate of achieving both the PASS and SCB for the ASES score. The proportion of patients achieving the PASS for the ASES score at 2-year follow-up was low at 56%. However, when considering the SCB for the ASES score, a considerable majority of patients reported a substantial improvement on this measure at both 1-year (83%) and 2-year (82%) follow-up. This discrepancy may be a function of the mechanism of psychometric statistical analysis and threshold derivation. For example, the SCB can be reported as a change-based threshold (change between postoperative and preoperative scores), whereas the PASS is inherently an absolute threshold (single postoperative score used as threshold). Therefore, if patients began at a remarkably poor baseline state before balloon spacer implantation, they may have been more likely to experience a substantial improvement in their overall postoperative ASES score compared with the preoperative score. This may have exceeded the change threshold for SCB achievement while still falling short of the overall postoperative score needed to be considered an acceptable symptom state (PASS threshold). Interestingly, Familiari et al 3 reported that 98% of patients reached the PASS for the CMS at 3 years postoperatively, which may be explained in part by both the lower-level quality of the study and the nature of the outcome measure being used. Based on the current literature, there is inconclusive evidence to suggest whether subacromial balloon spacer implantation may result in clinically meaningful improvements greater than the MCID. However, this also suggests that in select patients, the PASS and SCB are possible to achieve, although more data are needed.
Until more higher-level studies are published examining more homogeneous patient populations, the propensity to achieve the PASS and SCB consistently after subacromial balloon spacer implantation will remain poorly understood. Likewise, although this review presents evidence for high rates of MCID achievement, specifically for the CMS, more data are needed on other clinically relevant outcome metrics used in the shoulder literature. To understand whether subacromial balloon spacer implantation results in meaningful clinical benefits, we recommend more consistent reporting of the MCID, PASS, and SCB in future studies examining the clinical efficacy of this procedure. Furthermore, it will be imperative that studies apply identical threshold values to define this achievement, as failing to do so will limit the generalizability of results across different populations.
There are several limitations that should be considered when interpreting the results of the current study. First, few of the included studies incorporated heterogeneous patient populations with previous failed procedures or partial rotator cuff repair in conjunction with balloon spacer implantation. Although this may introduce some degree of heterogeneity into compiling and analyzing reported data, the overwhelming majority of studies were in accordance with the observation that a high rate of clinically significant improvement in outcomes can be expected at short-term follow-up. Second, the potential for publication bias exists as a function of the quality of included studies. This limitation is inherent to the available literature and is an important consideration, as it highlights the need for additional high-quality studies examining the propensity for clinically meaningful improvements. Third, there exists some variation in the thresholds utilized to define MCID achievement for the CMS; such inconsistencies hinder meaningful comparisons across studies and warrant a consensus as to how to define and evaluate the MCID for future studies.
Conclusion
Patients who had undergone isolated subacromial balloon spacer implantation for massive irreparable rotator cuff tears demonstrated a high rate of clinically significant improvement in outcomes at short-term to midterm follow-up. A paucity of literature exists to appropriately define and evaluate the rate of PASS and SCB achievement after subacromial balloon spacer implantation, necessitating further study.
Footnotes
Submitted September 29, 2022; accepted December 6, 2022.
One or more of the authors has declared the following potential conflict of interest or source of funding: S.A.T. has received consulting fees from DJ Orthopedics, KCI USA, and DePuy Synthes and hospitality payments from Exactech. M.C.F. has received consulting fees from Stryker and Encore Medical, support for education from Smith & Nephew and Medwest Associates, and grants from Arthrex and Acumed. R.F.W. has received royalties from OrthoBio Therapeutics, Arthrex, and Zimmer Biomet. D.M.D. has received consulting fees from Zimmer, Merck Sharp & Dohme, and Trice Medical; publishing royalties and financial support from Thieme; other financial or material support from Biomet; and support for education from Gotham Surgical Solutions & Devices. L.V.G. has received royalties from Exactech and Zimmer Biomet; has received consulting fees from Biomet, DePuy Synthes, Exactech, Medical Device Business Services, and Responsive Arthroscopy; has received support for education from Gotham Surgical Solutions & Devices; has received speaking fees from Smith & Nephew; and holds stock or stock options in Imagen and Responsive Arthroscopy. J.S.D. has received royalties from Arthrex, Linvatec, Zimmer Biomet, Thieme, and Wolters Kluwer Health–Lippincott Williams & Wilkins; has received consulting fees and research support from Arthrex; has received support for education from Gotham Surgical Solutions & Devices; and holds stock or stock options in ViewFi. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
