Abstract
Background:
The posterolateral corner (PLC) of the knee is a complex anatomic region of the knee comprising the popliteus tendon, the popliteofibular ligament (PFL), and the fibular collateral ligament (FCL). Treatment of PLC injuries is based on the degree of varus and rotational instability on preoperative examination and is recommended in grade 3 injuries.1,6
Technique Description:
The key concept of this new surgical technique is to use adjustable loop cortical suspensory fixation implants for initial femoral fixation of the popliteus and the FCL grafts. This allows for individual tensioning of the grafts prior to definitive fixation with an interference screw. An anatomic fibular tunnel is initially created, followed by popliteus and FCL anatomic femoral socket drilling. A TightRope RT implant is attached to the popliteus end of the graft and secured through a bone tunnel on the medial femoral cortex initially. The popliteus end of the graft is then pulled into the femoral socket before tunneling the graft through the fibular tunnel. A second TightRope RT implant is secured to the FCL end of the graft before securing it to the medial femoral cortex through a bone tunnel and pulling the graft into the drilled femoral socket. An interference screw is first used to secure the graft at the fibular tunnel. The knee is then taken to 60° where the TightRope device is used to remove residual creep and optimize final tensioning prior to final fixation with interference screw in femoral socket. With the knee at 30° of flexion, neutral rotation, and slight valgus, the same steps are repeated for FCL graft. A posterolateral capsular shift is completed for additional stability.
Results:
Residual posterolateral corner instability or failure after reconstruction surgery has been reported in 6% to 9.4% of surgical reconstruction case.2,3,5 The use of cortical suspensory devices in PLC reconstructions allows independent dialing of optimal graft tension for both the popliteus and FCL reconstruction before final interference screw fixation, therefore presumably limiting any residual creep and allowing for a double femoral fixation of the graft.
Discussion:
We describe a novel technique for posterolateral corner reconstruction which allows for optimal and individual tensioning of the structures of the posterolateral corner. Given the technique still uses well-described and validated graft choices and anatomic landmarks for reconstruction, we expect this new technique to allow for similar if not improved outcomes when compared with current gold standard.4
This is a visual representation of the abstract.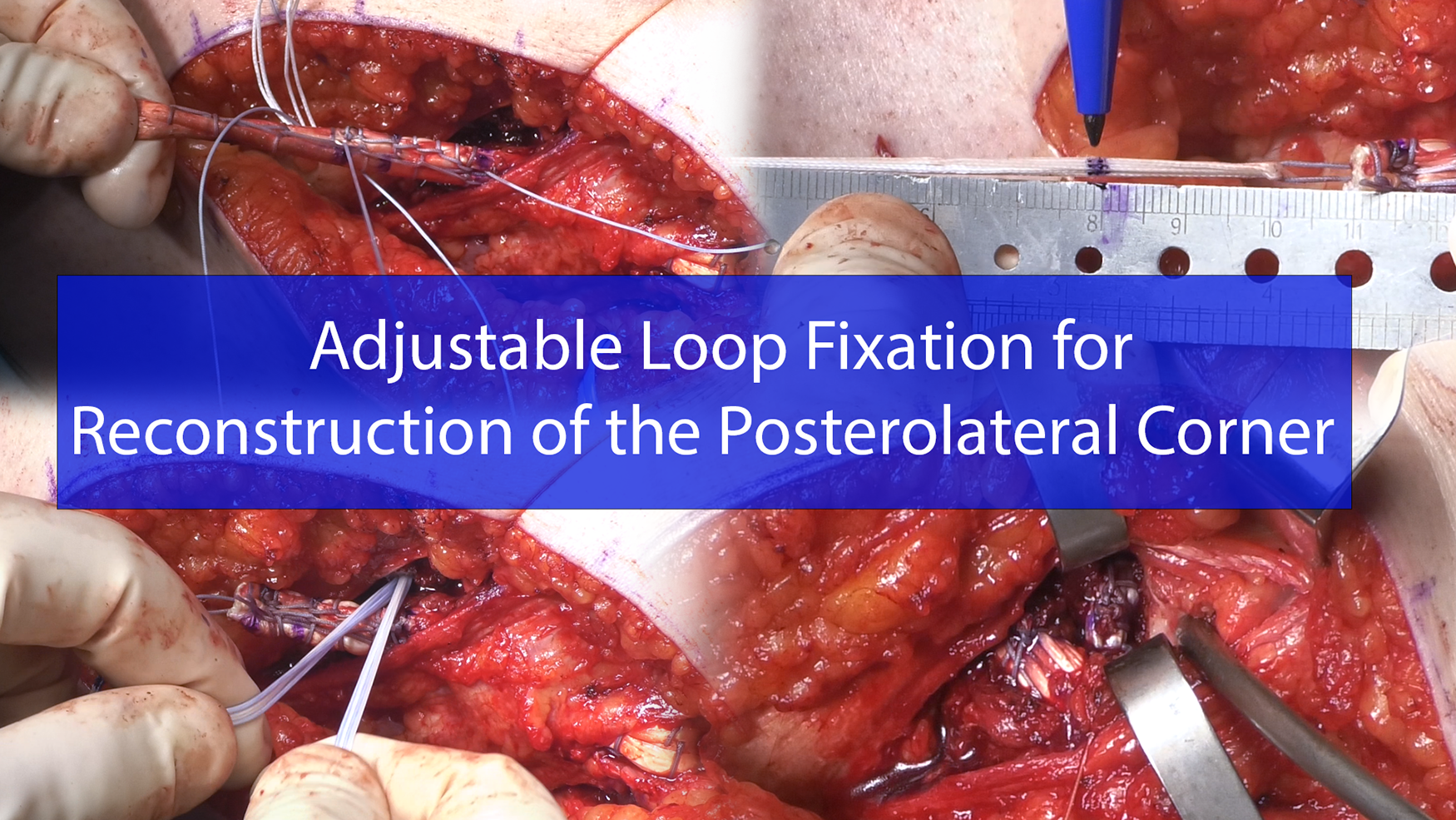
Video Transcript
This is a video presentation of a posterolateral corner reconstruction technique entitled adjustable loop fixation for reconstruction of the posterolateral corner.
The disclosures for the senior author are listed on this slide.
Injuries to the posterolateral corner are inherently unstable given the osseous biomechanics of the knee. Reconstruction techniques have shown improved surgical outcomes compared with surgical repair. With this in mind, biomechanical understanding is paramount when using reconstruction techniques for the posterolateral corner to prevent long-term instability and secondary knee injury. The patient presented is a previously healthy 42-year-old woman who sustained a fall from a horse onto a flexed knee. She had normal acquired brain injuries and no discernible neurovascular injuries on initial evaluation.
On physical examination, the patient was found to have a full range of motion from full extension to 140° of flexion. Notably, she had a grade 3 lachmans, a grade 3 posterior drawer test, instability with varus stress testing, and dial testing at both 30° and 90° of knee flexion. Examination was consistent with anterior cruciate ligament (ACL), posterolateral corner (PCL), fibular collateral ligament (FCL), and full posterolateral corner deficiency. Plain films demonstrated an avulsion type injury of the PCL noted on both the anteroposterior and lateral views with a red arrows indicator.
Coronal and axial magnetic resonance imaging (MRI) further characterized this substantial injury pattern showing the complete posterolateral corner injury, as well as a complete radial tear of the postmedial horn of the meniscus with significant medial extrusion. Sagittal MRI cuts demonstrated the PCL avulsion fracture injury as well as the degree of displacement, also shown is the abnormal appearance of the ACL. Three-dimensional reconstructions of computed tomography further demonstrated the bony avulsion injury of the PCL and its comminuted nature.
In summary, the injuries were as follows: a PCL avulsion fracture, a near complete ACL tear, a complete PLC injury, and a radial tear of posterior horn of medial meniscus.
Our plan was to address the injuries in 2 surgical stages: the first surgical stage addressing the PCL avulsion and the medial meniscal tear and the second stage including an ACL reconstruction with bone-tendon-bone allograft, an FCL, and PLC reconstruction with soft tissue allograft.
Fluoroscopic images demonstrate open reduction and internal fixation of the PCL avulsion injury. Arthroscopic images demonstrate the pre- and post-repair images of the complete radial tear, as well as the obvious incompetent nature of the native ACL. Final postoperative images for stage 1 are demonstrated. Varus stress x-rays obtained at the beginning of the case demonstrated asymmetric increased lateral joint space. Arthroscopic view demonstrates interval healing of the tibial plateau fracture. Subsequent pictures show ACL tunnel position as well as final graft localized in the tunnels. Fluoroscopic views then demonstrate positioning of the fibular and femoral tunnels for our PLC reconstruction. Final x-rays demonstrate suspensory fixation of the PLC reconstruction.
For postoperative rehabilitation, patient is maintained in full extension for 3 weeks and then was allowed to begin range of motion. Weight-bearing was delayed for 6 weeks while hamstring activation was delayed for 4 months. The goal was return to sport at 8 to 12 months. Prior literature regarding this topic was published by the senior author of this video. It was a retrospective review of 61 patients undergoing a similar, single-tailed fibular-based technique. They had a minimum 2-year follow-up, where they assessed International Knee Documentation Committee (IKDC) score, Lysholm score, range of motion, and stability. What they found was a mean IKDC score of 74.1, Lysholm 80.3, average range of motion of 0° to 126°, and notably 58 patients had grade 0 varus laxity in extension and 54 had grade 0 varus laxity in 30°. In conclusion, this single-graft, fibular-based technique resulted in satisfactory knee function and stability at a minimum of 2 years.
Our incision is marked out as well as its relation to the proximal fibula. The incision is taken through skin and subcutaneous tissue, followed by blunt dissection as well as bovie until biceps fascia and the posterior border of the distal biceps femoris tendon is exposed. We then bluntly dissect just posterior to the biceps tendon looking for the “soft spot” where the peroneal nerve can readily be identified. When we have localized the nerve and a safe plane has been established, fascia can be released in proximal direction. A vessiloop can then be placed around the nerve to help to free the nerve distally; the neurolysis can then be carried distally around the fibular neck releasing the nerve until it take a course deep into the anterior compartment.
One can appreciate the tight fibrous band as the nerve enters the anterior compartment. It is important to identify and release the fibrous band, as shown, until muscle is seen. Attention is then turned to locating the biceps bursa, incising the bursa, and identifying the FCL. It is of great importance the nerve is adequately released around fibular neck and at the entrance to the anterior compartment due to risk of compression at that site from postoperative swelling.
We can then trace the FCL to its fibular head insertion. Subperiosteal dissection is carried out posterior to the fibular head such that the exit point of the fibular tunnel can be reached.
A pin is then drilled into fibular head centered upon the FCL insertion. Fluoroscopy is used to confirm adequate trajectory of the tunnel. Care must be taken to protect the peroneal nerve at all times during the drilling of the fibular tunnel. The tunnel is then overreamed with 7-mm reamer.
Attention is then turned to the femoral attachments of the FCL and the popliteus. We can use the tagged FCL to help find the femoral attachment and guide location of our iliotibial (IT) band incision. An arthrotomy is made to localize the remnant of the native popliteus which can then be tagged. We can then localize the femoral attachment of the popliteus which should be located approximately 18.5 mm distal and anterior to the FCL attachment, in the anterior 1/5 and proximal half of the popliteal sulcus, just posterior to the articular cartilage margin. A guide pin can then be drilled into the popliteal femoral attachment, with confirmation on fluoroscopy and overreamed again with 7-mm reamer.
The ideal depth of tunnel is approximately 25 to 30 mm, the spade tip blade will give you the entire osseous length which will be marked on the suspension button system.
Marks on the graft are made at 20 and 30 mm in order to confirm adequate bone-graft interface. The TightRope (Arthrex; Naples, FL) device is pulled through the tunnel to the far cortex, and the graft is then pulled into the femoral tunnel to the 20-mm mark. Passing sutures are then used to pull the graft along proper course, through popliteal hiatus, then through the fibular tunnel from posterior to anterior. The Hughston suture passer can be very helpful for the passage of the sutures through the fibular tunnel.
The graft is then passed deep to fascial aponeurosis and iliotibial band in an extraarticular layer to the femoral attachment of the FCL. The FCL attachment can then be localized knowing its expected attachment is proximal and posterior to the lateral epicondyle and also 18.5 mm proximal and posterior to the popliteus insertion.
Utilizing fluoroscopy, the trajectory of the tunnel should be such that it is anterior to the ACL tunnel. Isometry of the FCL should be confirmed prior to reaming the FCL femoral tunnel. This is done by using mersilene tape and cycling the knee. Once the isometric point is confirmed, the femoral tunnel can be reamed to 30 to 35 mm with 7-mm reamer.
Desired graft length is confirmed and can then be whipstitched with #2 Fiberwire. The running stitch is secured through the second TightRope loop. A spade tip can again be used to measure the entire length of osseous tunnel to far cortex and mark the TightRope. The graft is then marked at 20 and 30 mm, before being pulled in the femoral FCL tunnel using the TightRope button. Grafts are approximately 20 mm deep in tunnel at this juncture, and a 7 × 20 mm fibular composite screw is used to secure the graft at the fibular head. This technique allows for independent retensioning of the grafts before final interference screw fixation of the popliteus and the FCL at the femur.
With the knee at 45° of flexion and neutral rotation, the popliteus graft is tensioned using the cortical button before final fixation with a biocomposite interference screw. Attention is then brought to the FCL’s final fixation with the knee at 20° of flexion in neutral rotation with a slight valgus load.
Final tensioning of the graft is done using the cortical suspension button before fixation with a biocomposite interference screw. The previously tagged native popliteus tendon can then be sutured to the reconstructed popliteus graft.
Closure can then begin in a layered fashion.
Final images can be obtained demonstrating position of the TightRope fixation.
Footnotes
Submitted January 17, 2022; accepted June 9, 2022.
Dr. Levy reports royalties and consulting fees from Arthrex, Inc. and consulting fees from Smith & Nephew. Dr. Stuart reports royalties, consulting fees, and research support from Arthrex, Inc. and research support from Strker and the USA Hockey Foundation. The authors declare that they have no other conflicts of interest in the authorship and publication of this contribution. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
