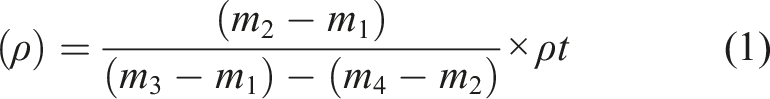Abstract
Recent advancements in composites involve integrating natural fibers into polymer matrices as a reinforcement or filler. This approach offers several benefits, including eco-friendliness, abundant natural materials, exceptional strength, cost-effectiveness, and simple extraction methods. Substituting synthetic materials with natural ones has environmental advantages, as some synthetics release toxins at high temperatures. Natural materials reduce the risk of toxic emissions, fostering a safer environment. Thus, our research centers on experimentally characterizing Fishtail Palm Leaf Stalk Fiber (FPLSF) through methods like Fourier transform infrared spectroscopy (FT-IR), X-ray diffraction analysis (XRD), thermogravimetric analysis (TGA), single fiber tensile test, and scanning electron microscope (SEM) analysis. These techniques unveil insights into composition, structure, thermal properties, strength, and morphology. Results highlight FPLSF’s attributes: 63.67% cellulose, 1473 kg/m³ density, 380.85 µm diameter, 31.37% crystallinity index, 249.23 MPa tensile strength, and 326°C thermal stability. In addition, the sound absorption properties such as sound absorption coefficient (SAC) and noise reduction coefficient (NRC) are found to be 0.34 and 0.39, respectively. FPLSF’s properties suggest its potential as an alternative reinforcement in composite manufacturing.
Keywords
Introduction
Exploration in biomaterials is increasingly focused on developing bio-based composites for various applications using sustainable options such as biopolymers, natural fibers, and bio-based materials. This trend coincides with a growing emphasis on implementing circular economy principles to improve the sustainability of green manufacturing practices. These developments mirror a broader trend in business towards environmental awareness and resource efficiency. Biomaterials hold enormous potential for the future of composite materials, driven by the need to lower carbon emissions and minimize environmental impact. In this context, natural fiber-reinforced composites have gained significant popularity in recent years due to their numerous advantages. Natural fibers offer several advantages over synthetic fibers, including lower cost, biodegradability, greater environmental sustainability, lighter weight, and wider availability. Their renewable nature provides additional benefits, as organic waste can be used for applications like electricity generation and construction materials. Recently, there has been a global shift towards natural fibers due to the drawbacks of conventional fibers, such as non-biodegradability, non-recyclability, high processing costs, and potential health risks. Studies have shown that natural fibers can effectively replace synthetic fibers because of their high specific strength, affordability, and non-toxic nature. These advantages have motivated researchers worldwide to explore and develop unique natural fibers for use as reinforcements in polymer composites. 1 The goal is to create innovative, high-quality bio-based fibers that are acceptable for industrial applications. Natural fibers typically consist of cellulose, the main building block, along with lignin and hemicellulose which act as binding agents. Cellulose, composed of long-chain molecules, contributes to the strength and stiffness of the fiber, maintaining its high structural rigidity and elasticity. These molecules form a branched polymeric structure, held together by microfibrils, with amorphous hemicellulose and lignin filling the gaps. However, natural fibers also have inherent limitations. Their hydrophilic nature, meaning they attract water, leads to poor moisture resistance. Additionally, the presence of non-cellulosic, waxy constituents can hinder adhesion to the matrix material, resulting in inefficient stress transfer and ultimately shortening the material’s lifespan. 2
To address these limitations, scientists and researchers are actively identifying novel, high-quality fibers from various plant parts like stalks, stems, roots, bark, and fruits. These fibers hold promise for widespread applications in textiles, marine, automotive, and aerospace industries. Recent research has explored the potential of novel natural fibers from various sources, including lavender stem, 3 Zingiber officinale, 4 beetroot plant, 5 reddish shell bean, 6 Alcea rosea L, 7 Sambucus ebulus L plant stem, 8 Ficus Benjamina L stem, 9 Sansevieria ehrenbergii, 10 Cymbopogon flexuosus stem, 11 Pandanus tectorius leaves, 12 and Mucuna atropurpurea. 13 Researchers have analyzed these fibers using physicochemical analysis, 14 FTIR spectroscopy, and XRD to assess their suitability as reinforcement candidates in polymer composites with distinct properties.
For instance, a recent study extracted a novel cellulosic fiber from the Areca palm tree and treated it with varying alkali percentages of 5%, 10%, and 15% by weight. The results showed that the 5% alkali-treated fibers exhibited the highest tensile strength of 486.41 MPa, demonstrating the potential for extracting useful natural fibers from palm trees. More research is needed to fully characterize and take advantage of such newly discovered natural sources. 15 Similarly, a new cellulosic fiber from Pogamia pinnata was modified using 5% sodium hydroxide (NaOH) for different durations of 15 to 75 min. The optimal treatment occurred with 5% NaOH for 60 min. Moreover, raw and alkali-treated fibers were compared through physicochemical analysis, FTIR spectroscopy, and XRD. 16 Likewise, a natural fiber from Elettaria cardamomum stems underwent chemical analysis, revealing higher cellulose content. 17 Additionally, studies on raw and alkali-treated Coccinia grandis fibers showed increased cellulose and decreased hemicellulose, lignin, and wax after alkali treatment. 18 To investigate this further, the researchers 19 fabricated composites by combining epoxy resin with untreated and treated Zanthoxylum acanthopodium bark fibers. They discovered that composites with 20% treated fibers exhibited the greatest strength (47.3 MPa). However, as they added more fibers, the composites absorbed more water.
There is limited research characterizing Fishtail Palm Leaf Stalk Fibers (FPLSFs) and evaluating their potential to substitute synthetic polymer reinforcements. This study aims to address that gap by investigating the physicochemical properties of FPLSFs. Specifically, FT-IR, XRD, TGA, and morphological analysis are utilized to assess properties including chemical composition, crystallinity, thermal stability, and microstructure. The results are benchmarked against common natural fibers to provide insights into the relative performance and feasibility of utilizing FPLSFs as reinforcements for polymer composites and other applications. Thorough material characterization will elucidate FPLSFs’ advantages, allowing informed decisions regarding their applicability as biodegradable, sustainable alternatives to synthetic fibers.
Materials and methods
Materials
Fishtail palms (Caryota mitis) are named for the shape of their leaves and belong to the Plantae kingdom, Arecales order, and Arecaceae family. Caryota mitis trees have clustered stems reaching heights up to 10 m (33 ft) and diameters of 15 cm (6 in). The leaves can grow up to 3 m (10 ft) long, as shown in Figure 1. The Fishtail Palm Leaf Stalks were obtained from Caryota mitis trees in the Coimbatore District, Tamil Nadu, India. Figure 1 illustrates the detailed procedure for extracting the Fishtail Palm Leaf Stalk Fibers (FPLSFs). First, the leaves were manually removed from the stalks. The stalks were then water retted by soaking in water for 3 weeks, allowing microbial degradation to loosen the gum-like materials between fibers. To extract Fishtail Palm Leaf Stalk Fibers (FPLSFs), the stalks were mechanically beaten with a wooden mallet. The separated fibers underwent an alkali treatment to remove surface impurities and waxes, which involved immersing the dried raw fibers in an aqueous sodium hydroxide (NaOH) solution (1% w/v) for 45 min followed by washing with distilled water. The fibers were then briefly immersed in dilute hydrochloric acid (0.1 N) to eliminate residual contaminants and subsequently washed again in distilled water. Finally, the treated fibers were dried at room temperature for 2 days prior to further analysis. This multi-step cleaning procedure helps ensure removal of non-cellulosic components to better assess the intrinsic chemical and physical attributes of the FPLSFs. Extraction of Fishtail Palm (Caryota mitis).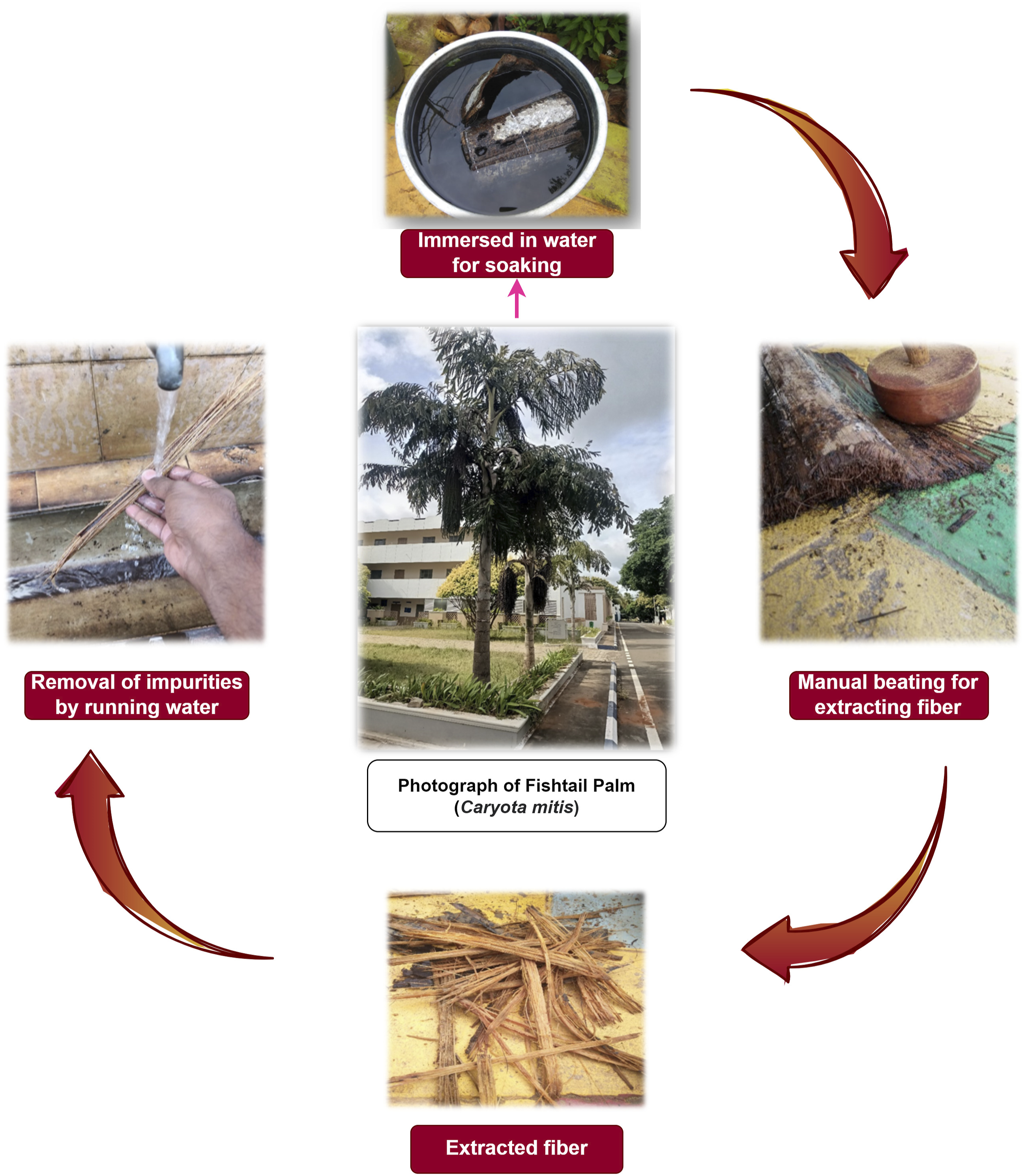
Physical analysis
The FPLSF fiber diameter was characterized via optical microscopy using a Carl Zeiss polarizing microscope (Germany). Given known variabilities, approximately 50 individual fibers were measured at two positions along their length (top and bottom) to quantify average diameter and variability between fibers. Density measurements were conducted based on ASTM standard method D578-89 using a pycnometer with toluene as the immersion solvent. Fibers were cut into 5–6 mm pieces, dried for 120 h in a desiccator with silica gel, and immersed in toluene for 2 h prior to testing to remove moisture and ensure full solvent penetration into the microstructure, respectively. Samples were transferred to the pycnometer for density measurements, calculated using equation (1), as defined in the ASTM standard:
Chemical analysis
The chemical compositions of the FPLSFs were determined through several standard test methods. Cellulose and hemicellulose contents were measured by Kushner’s method (ASTM D1106-56) and Hoffer’s method (ASTM D1107-56), respectively. The Klason technique per APPITA P11-78 was used to evaluate lignin percentage. Fiber moisture content was quantified with a Sartorius MA45 moisture analyzer, 18 and Ash percentage was calculated per ASTM standard method E1755-01. 20 Finally, the Conrad method determined wax content. Applying this array of standardized analytical techniques enabled the accurate characterization of FPLSF chemical constituents including cellulose, hemicellulose, lignin, moisture, ash, and wax. Thorough compositional analysis provides insights into the purity and reinforcing capability of the extracted fibers.
Single fiber tensile test
The tensile strength of FPLSFs was evaluated using an INSTRON universal testing machine equipped with a computer interface and 1 kN load cell. Testing followed ASTM D822-07 specifications, including a crosshead speed of 5 mm/min, 50 mm gauge length, 65% relative humidity conditions, and ambient temperature control at 30°C. This standard test method provides quality-controlled determination of tensile properties, enabling quantitative comparison to other natural and synthetic fiber types under defined conditions.
Fourier transform infrared spectroscopy
Fourier transform infrared spectroscopy (FT-IR) was utilized to analyze the chemical composition of the alkali-treated FPLSFs. Testing was conducted using a PerkinElmer Spectrum instrument (Model: Diamond UTAR). Powdered fiber samples were mixed with potassium bromide (KBr) at a 1:10 ratio and pressed into pellets for analysis. Infrared spectra were obtained over a wavenumber range of 4000 to 400 cm−1 at 2 cm−1 resolution, with a 32-scan per minute signal-to-noise ratio. This consistent methodology provides reliable chemical characterization of the extracted and treated FPLSFs across the detectable IR absorption wavelength range for various functional groups.
X-ray diffraction
X-ray diffraction (XRD) analysis was conducted to determine the crystalline size (CS) and crystallinity index (CI) of the Fishtail Palm Leaf Stalk Fibers (FPLSFs). The analysis was performed using a PANalytical X’pert PRO spectrometer operated at room temperature. The spectrometer settings were 40 kV and 150 mA with CuKα radiation (wavelength 0.154 nm). Scans were performed from 10° to 80° 2θ at a rate of 5°/min and step size of 0.05° to acquire satisfactory diffraction patterns for analysis. The crystallinity index (CI) was calculated using equation (2):
The crystalline size (CS) was evaluated using the Scherrer equation (equation (3)):
Thermogravimetric analysis and differential scanning calorimetry
Thermogravimetric analysis (TGA) characterized the thermal stability of FPLSFs using a Netzsch STA 449 F3 Jupiter simultaneous thermal analyzer. Samples were prepared by placing 10 mg of powdered fibers in aluminum crucibles. Tests were conducted under a 20 mL/min nitrogen atmosphere, with weight loss of samples monitored continuously as temperature increased from 30°C to 550°C at 10°C/min.
Differential scanning calorimetry (DSC) (Make: Mettler Toledo 822) provided additional thermal analysis through determination of required heat flow to raise FPLSF sample temperature. Samples of 5 mg were placed in individual aluminum pans and heated from 30°C to 550°C at 10°C/min to acquire DSC traces. 21
Morphological characterization
Scanning electron microscopy (SEM) visualized the morphology and topography of alkali-treated FPLSF surfaces using a CARL ZEISS SIGMA instrument operated at 15 kV. To prevent electrostatic charging and ensure conductivity, fiber surfaces were sputter coated with gold prior to analysis. SEM provided microscopic examination at various magnifications, with images capturing finer details of fiber structure and surface features indicative of mechanical interlocking capacity. Understanding micro-scale topography and the effects of fiber extraction and alkali treatment can help tailor surface properties and predict composite reinforcement performance.
Sound absorption properties
The sound absorption properties of natural fibers were measured through a two-microphone impedance tube method following ASTM E1050-19 standards. An Impedance Tube Kit (Type 4206, 50 Hz–6.4 kHz) was used along with matched microphones and a data acquisition system for signal processing. After system calibration with a standard material, FPLSF fiber samples were mounted within the tube, and the transfer function between microphones was measured across the designated frequency range (63 to 6300 Hz). The sound absorption coefficient (SAC) and noise reduction coefficient (NRC) as shown in equations (4) and (5) will quantify the sound energy absorption by the fibers, providing valuable insights into their acoustic performance.
Results and discussion
Diameter and density analysis
The diameter of FPLSFs can vary based on factors including climate, plant age, soil properties, and extraction conditions. In this study, the measured FPLSF diameter was 380.85 μm, as shown in Figure 2. Fifty fiber samples were analyzed, and the density of the FPLSFs was measured as 1473 kg/m3, which is higher than some other biofibers like Parthenium hysterophorus (1251 kg/m3),
22
Musa paradisiaca leaf (1325 kg/m3),
23
and Cortaderia selloana grass (1261 kg/m3).
24
However, FPLSF density was lower than fibers such as Coccinia grandis stem (1517 kg/m3)
25
and Vetiver fiber (1498 kg/m3).
26
The relatively low density of FPLSFs may be advantageous for lightweight composite fabrication (Table 1). Diameter measurement of FPLSFs. Comparison of physical properties of FPLSFs with other natural fibers.

Chemical properties analysis
Comparison of chemical analysis of FPLSFs with other plant fibers.

Single fiber tensile strength analysis
Comparison of tensile strength of raw CRPLSFs with various plant fibers.

FT-IR analysis
Figure 3 indicates the FTIR spectrum for Fishtail Palm Leaf Stalk Fibers (FPLSFs) over the range of 4000 to 400 cm−1. The FPLSF spectrum exhibits characteristic peaks at 3341, 2918, 2854, 1580, 1456, 1026, and 658 cm−1. The broad peak at 3341 cm−1 corresponds to O-H stretching, indicating the presence of α-cellulose.
41
The peak at 2918 cm−1 arises from C-H stretching vibrations in cellulose and hemicellulose.
42
The peak at 2854 cm−1 represents CH2 stretching associated with organic compounds.
43
The peak at 1580 cm−1 corresponds to lignin (C = C bond).
22
The 1456 cm−1 peak indicates aromatic rings in polysaccharides.
44
The peak at 1026 cm−1 represents lignin vibrations.
40
Finally, the peak at 658 cm−1 indicates beta-glycosidic linkages between sugars in hemicellulose and cellulose.
45
Table 4 summarizes the functional group assignments for the key components of the FPLSFs based on the FTIR spectrum, and the references are cited to support the peak assignments. FTIR spectrum of FPLSF. FT-IR functional group for Fishtail Palm Leaf Stalk Fibers (FPLSFs).
XRD analysis
Figure 4 shows the X-ray diffraction (XRD) spectrum for FPLSFs. Two distinct peaks are observed. The first peak at 2θ = 15.27° corresponds to the amorphous region. The main peak at 2θ = 22.25° represents the crystalline region. The calculated crystallinity index (CI) for FPLSFs is 45.70%. This is higher than fibers such as Acacia nilotica L (44.82%),
35
Juncus effuses (34.4%),
48
Tridax procumbens (34.46%),
49
Phoenix pusilla L (42.6%),
42
and Phaseolus vulgaris (42.17%).
47
However, the FPLSF CI is lower than fibers like Aerva javanica (47.0%),
17
Coccinia grandis (45.09%),
25
Morus alba L (62.06%),
50
Shwetark stem (72.06%),
51
and Sesbania rostrata (69.11%).
38
The CI value strongly correlates to the tensile properties of FPLSFs. XRD spectrum of FPLSFs.
Comparison of CI and CS of FPLSFs with various plant fibers.

TGA and DSC
The thermal stability of FPLSFs was analyzed, as shown in Figure 5. The TGA curve indicates three different degradation stages for FPLSFs. The first stage of weight loss observed in the TGA curve between room temperature (RT) and 100°C is attributed to moisture evaporation (Raja, Senthilkumar, et al., 2021), while the second stage from 200 to 300°C corresponds to the decomposition of hemicellulose and cellulose components within the extracted FPLSFs.
43
The third stage from 300 to 400°C is associated with wax and lignin degradation.
55
The DTG curve shows two exothermic peaks. The first peak at 314.6°C results from the breaking of polymer chains in hemicellulose and cellulose.
17
The second peak seen at 340.5°C represents the decomposition of residual high-molecular weight compounds (Madhu et al. 2020). The TGA/DTG analysis indicates FPLSFs have comparable thermal stability to other natural fibers, withstanding temperatures up to ∼326°C. This is higher than many fibers, making FPLSFs suitable for low-temperature composite processing. Table 6 compares the initial and final decomposition temperatures of FPLSFs with other natural fibers, citing references. TGA and DTG curves of FPLSFs. Initial and the final decomposition temperature of different fibers.

Surface morphology analysis
Figure 6 shows SEM micrographs of the Fishtail Palm Leaf Stalk Fiber (FPLSF) surface morphology. The fibers have a multicellular structure composed of cellulose fibers surrounded and bounded by lignin and hemicelluloses. The SEM images reveal impurities present on the FPLSF surfaces that created improper dispersions Figure 6(a) and voids Figure 6(b). The fibrillar structure consists of fibrils connected along their length by non-cellulosic compounds like pectin. The FPLSF surface appears rough. The rough surface morphology revealed through SEM imaging suggests that the extracted palm leaf stalk fibers may form a strong interfacial bond with polymer matrices during composite fabrication Figure 6(c), owing to increased surface area and mechanical interlocking Figure 6(d). In summary, the SEM analysis provides insights into the multicellular composition, presence of impurities, fibrillar nature, and surface roughness of the extracted FPLSFs. SEM images of FPLSFs.
Sound absorption analysis
This analysis explores the sound absorption properties of Fishtail Palm Leaf Stalk Fibers (FPLSFs) based on the sound absorption coefficient (SAC) and noise reduction coefficient (NRC) as shown in Figure 7. An SAC value of 0.34 indicates that FPLSFs absorb 34% of the sound energy incident upon them at the measured frequencies. This value falls within the medium sound absorption range (0.20–0.60). This suggests that FPLSFs have a moderate ability to dampen sound waves, potentially making them suitable for applications requiring some level of noise reduction. The NRC value of 0.39 further supports the findings from the SAC. NRC considers the sound absorption across a specific range of frequencies relevant to human hearing. An NRC of 0.39 translates to an average absorption of 39% of sound waves across these frequencies. This again falls within the medium sound absorption range (0.35–0.60) for NRC. The sound absorption capabilities of FPLSFs can be attributed to several factors such as porous structure, fiber diameter, and density. Fibers like FPLSFs often possess a porous structure with air pockets. Sound waves traveling through the material can be trapped and converted into heat energy within these air pockets, leading to sound absorption. The diameter and density of the fibers can also influence sound absorption. Finer fibers with lower density tend to be more effective sound absorbers. Sound absorption properties of FPLSFs.
Conclusions
This study presents a comprehensive characterization of the fundamental properties of Fishtail Palm Leaf Stalk Fibers (FPLSFs). The key results are as follows: ❖ FPLSFs have a relatively low-density of 1473 kg/m3 and diameter of 380.85 μm, making them suitable as lightweight reinforcements to replace non-organic fibers in composites. ❖ Chemical analysis revealed FPLSFs have high cellulose (73.67 wt%) and moderate hemicellulose (14.59 wt%) and lignin (9.12 wt%) content compared with other natural fibers. ❖ XRD analysis found a crystallinity index of 45.70% and crystalline size of 16.92 nm for FPLSFs. ❖ TGA results showed FPLSF thermal stability up to ∼326°C, enabling potential processing into composites. ❖ FTIR confirmed the presence of cellulose, hemicellulose, and lignin, with peaks at 3341, 2918, and 1580 cm−1 representing O-H, C-H, and C = C bonds, respectively. ❖ A tensile strength of 249.23 MPa was measured through single fiber testing.
In summary, the extensive characterization of FPLSF chemical composition, structure, morphology, and mechanical properties demonstrates their potential as a sustainable reinforcement for green composites, offering advantages over synthetic fibers.
Footnotes
Acknowledgments
We gratefully acknowledge PSG TECHS: COE InduTech for providing acoustical testing and Centre for Machining and Material Testing (CMMT) lab at KPR Institute of Engineering and Technology, Coimbatore, for providing compression molding facilities.
Author contributions
All authors are equally contributed.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.