Abstract
Multi-phase metal oxides nanocomposites (NCs) have attracted considerable attention due to their extraordinary properties and novel applications over monometallic ones. Hence, trimetallic oxides nanoparticles (NPs) are preferred because of their immensely improved optical, catalytic, and biological properties, but few materials have been reported. Besides, glycine is an excellent structure-directing agent for NPs production with tailored physicochemical properties. Thus, in this work, a novel tri-phase CuO–Fe2O3–MgO (1:1:1) NCs was prepared via a sol-gel method in the presence of glycine as a fuel. The obtained NCs were characterized by Fourier transmission infrared, X-ray diffraction (XRD), Scanning electron micrographs, and UV-Vis. The XRD analysis emphasized the formation of NCs with monoclinic CuO, cubic MgO, hexagonal Fe2O3, and tetragonal CuFe2O4 crystals. The average crystallite size (D) was in the order of 10th of nm as computed by Scherrer method, with ternary phase seemingly affect the straightforward influence of glycine fuel concentration on the final crystallite sizes. UV-Vis analysis indicates two optical energy bandgaps which increased as glycine concentration increase. The antibacterial test against Staphylococcus aureus and Escherichia coli bacteria revealed comparable activity to that of Azithromycin standard drug, which increased with glycine concentration increase. The glycine-based tailored structural, optical, and biological properties of such trimetallic NCs making them of considerable candidate for certain applications development, possibly electronics and antibiotics; a case that encourage further investigations.
Introduction
Metal oxide nanocomposites (NCs) have attracted great attention in recent years due to their unique properties such as optical, electrical, mechanical, photocatalytic, thermal, and structural.1–3 They could be of two, three or more oxides present in a nanometer scale and might be involved in many applications, including solar cell, battery materials, UV detectors, gas sensors, photovoltaic devices, and fuel cells.4–7 By combining different metal oxides, various properties of individuals could be improved and a new track of research for biological, optoelectronics, thermal, and electrical applications could open up.5,8–10 Adding a new phase to a composite material can alter its electronic properties. Metal oxide NCs can be prepared through several routes, like hydrothermal, 10 co-precipitation, 11 self-combustion, 12 ultrasonic-assisted method, 13 etc. Among these, sol-gel method 14 is popular one to fabricate metal oxide NCs. It is a low cost, fast, and facile to carry out with less expensive devices.
The metal oxide CuO is a p-type semiconductor with optical bandgap of 1.22 eV. It possesses great optical, structural, and electrical properties and, additionally, being nontoxic and uncostly prepared. It is involved in several applications such as supercapacitor, superconductor, catalysis, gas sensing, and photocatalytic activity.12,15–21 MgO is nontoxic and inexpensive as well, demonstrating a wider optical bandgap of 7.3 eV, high surface reactivity, high adsorption capacity, and unique physicochemical properties.1,2,22–24 Fe2O3 is a n-type semiconductor metal oxide with energy bandgap of 2.1 eV. It is chemically stable, naturally abundant, benefit several applications, and most importantly it is cheap and safe material.25–27
Globally, bacteria induced diseases are one of the leading causes of human illness. Food contaminated with bacteria like Staphylococcus aureus (S. aureus) or Escherichia coli (E. coli) can cause serious diseases. 28 Over the last two decades, many of the antibiotics utilized to cure infectious diseases have failed to work due to bacterial resistance. 29 To outdo this problem, the preparation of new inorganic antibacterial agents is critical for fighting bacteria resistance. Several articles have reported that metal oxide NCs possess promising antibacterial potential due to their large surface areas and generation of charged radicals that inhibit bacterial growth.11,30–33
According to literature,34,35 amino acids can serve as useful facilitator in term of nanoparticles (NPs) synthesis. They provide strong bonding with particles due to their zwitterionic nature expressed in their amine and carboxyl groups protonation-deprotonation as the pH changed. Adding of such surfactant to metal oxide solution decreases crystal growth and, according to the agent type as well as the synthesis conditions, crystallization rate, and growth direction could be fabricated, thus allowing production of target NPs sizes and shapes. Glycine, as the simplest stable amino acid, have been applied as a capping agent in the synthesis of iron oxide magnetic NPs,35,36 which proven possible NPs controlled morphology. However, different amino acids could induce different morphology. 37
In this study, glycine was applied as a structure-directing agent for obtaining a tri-phase CuO–Fe2O3–MgO NCs, and the effect of its concentrations on the end-properties of the synthesized NCs were investigated. Therefore, the structural, morphological, and optical properties as well as the biological activity against both Gram-positive and Gram-negative bacteria were studied.
Materials and methods
Materials
Iron (III) nitrate nonahydrate (Fe(NO3)3·9H2O; 97%) and magnesium (II) nitrate hexahydrate (Mg(NO3)2·6H2O; 97%) were obtained from BDH Chemical Ltd (Pool, England, UK). Copper (II) nitrate hexahydrate (Cu(NO3)2·6H2O; 98%) and glycine (≥98.5%) were procured from Sigma–Aldrich (Darmstadt, Germany). Sodium hydroxide (NaOH, 98%) was bought from Fisher Chemical (Loughborough, UK). All materials were used as received unless otherwise stated; and double distilled water was used wherever needed.
Preparation glycine-mediated tri-phase CuO–Fe2O3–MgO nanocomposite
Metal oxides NPs were obtained using sol-gel methods.
38
Therefore, salts solutions (0.7 M) of each Fe(NO3)3·9H2O (4.24 g), Cu(NO3)2·6H2O (2.54 g), and Mg(NO3)2·6H2O (2.69 g) were separately prepared in 15 ml distilled water, then mixed with stirring for 30 min at room temperature (termed solution I, SI). Subsequently, 20 ml of 1.0 M glycine solution (SII) and 15 ml of 6 M NaOH (SIII) were poured into SI, followed by heating at 90°C for about 60 min during which precipitate of CuO–Fe2O3–MgO started to form. The obtained dry mass was further calcined at 800°C for 90 min and termed G1. NCs G1.5 and G2 were prepared similarly as G1 using 1.5 and 2.0 M of glycine solution (SII), respectively. A schematic illustration for the experimental process is given in Figure 1. Schematic presentation of CuO–Fe2O3–MgO NCs synthesis, characterization, and application.
Characterization techniques
The X-ray diffraction (XRD) patterns of G1, G1.5, and G2 were recorded using an XD-2 X-ray diffractometer (Beijing Purkinje General Instrument Co., Ltd, Beijing, China), with CuKα1 radiation of λ = 1.54 Å, in a 2θ range of 10–80 and scanning rate of 0.02 min−1. Scanning electron micrographs (SEM) were imaged in a JSM-6360 LV scanning electron microscope (Jeol Ltd, Tokyo, Japan). Vibration spectra were analyzed using a Nicolet iS10 Fourier transmission infrared (FTIR) spectrometer from Thermo Scientific (Madison, WI, USA) which equipped with an attenuated total reflection (ATR, diamond crystal) accessory, over the range of 650–4000 cm−1, with 32 scans per spectrum and 4 cm−1 scanning resolution at room temperature. UV-Vis spectrophotometer (Hitachi-U3900; Tokyo, Japan) was used for recording electronic spectra over the range of 200–800 nm at room temperature.
Antibacterial test
In vitro antibacterial activity of CuO–Fe2O3–MgO NCs at concentration of 100, 200, and 400 mg/ml was carried out using the disc-diffusion method,39,40 against Gram-positive bacteria (S. aureus) and Gram-negative bacteria (E. coli) in reference to Azithromycin (AzM) as standard drug. Bacteria of the species S. aureus and E. coli were kind gift from Al-Jarfi medical Lab (Thamar, Yemen). The bacteria were cultured and swapped in Mueller Hinton agar medium, and discs having 6 mm diameter were prepared by punching Whatman filter paper. Solutions of various concentrations of NCs were fabricated singly. After that, paper discs were immersed in the test NCs solutions and positioned on the plates where bacteria lie. Simultaneously, discs for AzM (positive control) and DW (negative control) were also prepared and treated similarly as NCs. The plates were incubated for 20 h at 35–37°C. The susceptibility of bacteria to NCs was determined by measuring the zone of inhibition (ZOI) in millimeter. 41
Results and discussion
Structural properties
FTIR analysis
The synthesized trimetallic oxides NCs were characterized for their structure and composition using FTIR technique. The spectra of the fabricated three groups (G1, G1.5, and G2) are gathered in Figure 2. As can be seen, the broad band centered at about 3450 cm−1 is assignable for O–H stretching vibrations, for which the bending mode can be observed at 1643 cm−1, indicating metal–hydroxide structure.
42
The presence of OH groups on the sample surface is due to a common sorption event of water molecules from the atmosphere.
31
According to literature, the strong band around 1350–1500 cm−1 is associated with carbonate ions weakly attached to the sample surface.31,43 Bands for oxygen–metal-oxygen (O–M–O) and metal-oxygen (M–O) are commonly traced in the region 430–1004 cm−1.1,13 Thus, the peak at 582–596 cm−1 is for Fe–O and Cu–O and 435 cm−1 for Mg–O. Overton stretching bands for metal oxides (M–O) were at higher frequencies; thus, the ones on the range 1004–864 cm−1 can be related to such overtone bands as reported elsewhere.
44
Meanwhile, no traceable bands for organic impurity in the FTIR spectra that possibly sourced by glycine fragments, few weak bands in the finger print region may indicate negligible quantity of carbons which could be of carbonate type, with no evidence of aliphatic fragments observed at 3000–2750 cm−1. FTIR spectra of CuO–Fe2O3–MgO NCs (G1, G1.5, and G2) synthesized at various glycine concentrations.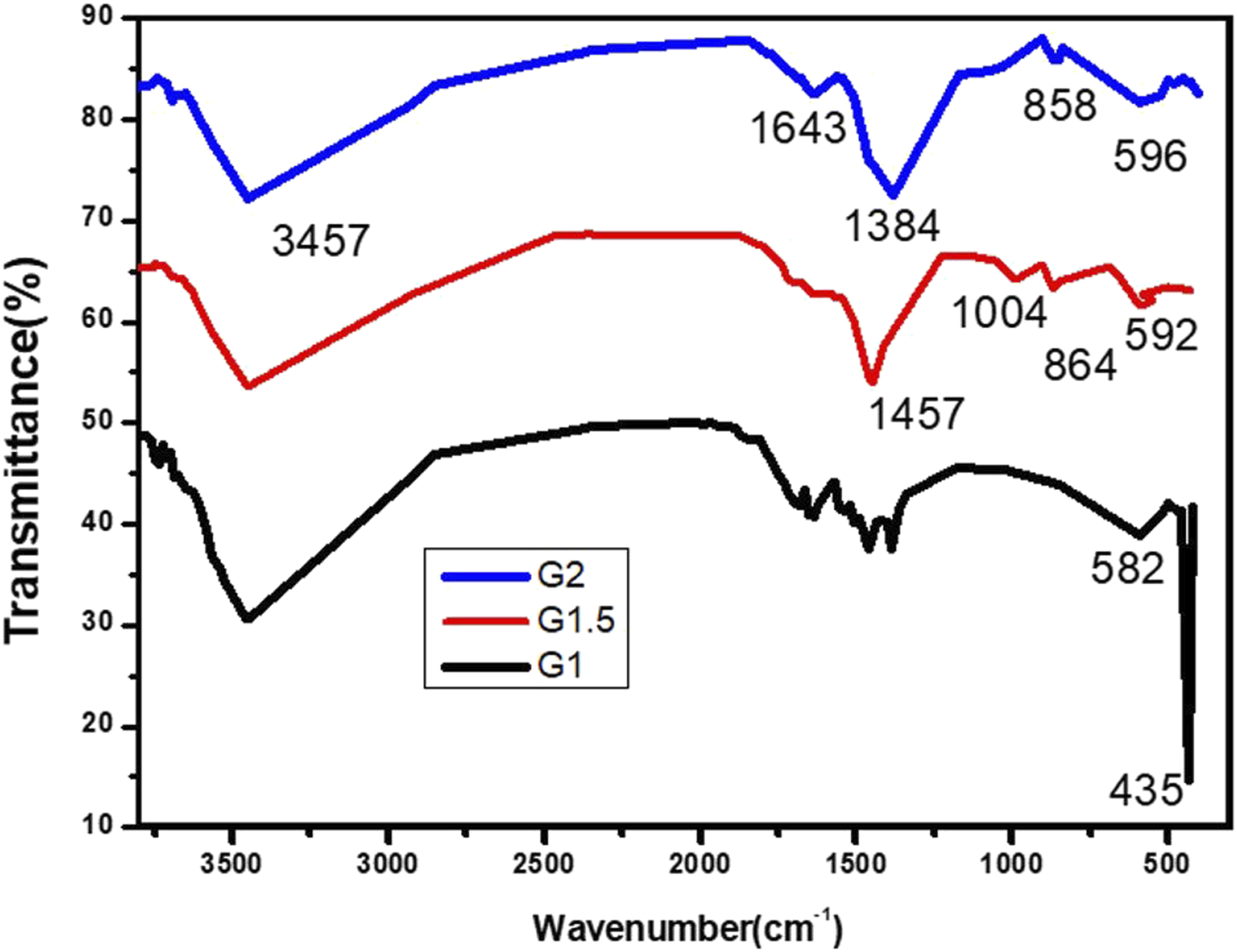
XRD analysis
The XRD patterns of CuO–Fe2O3–MgO NCs, fabricated at various concentrations of glycine, have displayed four identifiable phases for CuO, Fe2O3, MgO, and CuFe2O4 oxides as shown in Figure 3. The diffraction peaks were matched with JCPDS files of CuO (74–1021), Fe2O3 (76–1821), MgO (45–0946), and CuFe2O4 (72–1174) with monoclinic, hexagonal, cubic, and tetragonal crystals, respectively, indicating the polycrystalline nature of the synthesized materials. The average crystallite size (D, nm) of G1, G1.5, and G2 samples were computed by Scherrer’s law and Bragg’s law as expressed by equations (1) and (2)45,46 XRD patterns of CuO–Fe2O3–MgO nanocomposites at different glycine concentrations. Structural parameters of CuO–Fe2O3–MgO nanocomposites determined from XRD analysis.

It is clear that the averaged D of Fe2O3, CuFe2O4, CuO, and MgO were unsystematically changed with increasing of glycine concentration. This is due to the contribution of ternary phase (CuO, MgO, and Fe2O3 oxides) in aggregation process as a consequence of the ionic radii differences between Cu (0.073 nm), 47 Mg (0.059 nm), 48 and Fe (0.064 nm), 49 as well as the effect of electronegativity of metals Cu (1.90), Mg (1.31), and Fe (1.83) in production of metal oxide molecules. It is obvious that with increasing in glycine fuel–mediating synthesis have resulted in crystalline size decrease, the case that supposedly due to spacer abundance in the media, leading to large number of nuclei for crystal growth.
Khodair et al. have studied the effect of glycine on the crystallite size (D) of MgO NPs 38 and reported that the NPs increased as the glycine concentration increased. The increase in the D was attributed to the small δ value due to the inverse relation between δ and D. According to the X-Ray diffraction data, an increase in the glycine concentration leads to the variation in the crystal lattice parameters and a decrease in the crystallites size as shown in Table 1. This result leads to the existence of additional distortions and vacancies in the structure.
SEM analysis
The surface texture of CuO–Fe2O3–MgO nanocomposite annealed at 800°C is displayed in Figure 4. It can be seen that the particles were almost in spherical shape ad agglomerated into visible dense structure. Meanwhile, particles in the nano range still observable, analyzed SEM image scale is hardly describing the nano structure. Nevertheless, the homogeneity of the surface may indicate advanced distribution facilitated by the glycine-media during the sol-gel production of multi-phase oxides which, in turn, could be reflected in the properties of the calcined final powders. Moreover, NCs produced at lower glycine concentration resulted in a more aggregate texture, as seen for G1 compared to G2. The result revealed concentration-dependent particle sizes of the synthesized NCs with countable particles in the order 100th nm. SEM image of glycine-mediated synthesized CuO–Fe2O3–MgO nanocomposite G1, G1.5, and G2.
Optical properties
Electronic spectra
Figure 5(a) displays the optical absorption spectra of CuO–Fe2O3–MgO NCs prepared at various glycine-mediated concentrations of 1, 1.5, and 2 M. All the spectra demonstrated the presence of at least two peaks (at about 300 and 370 nm), of which the ones at 373, 363, and 305 nm where the clearer peaks in G1, G1.5, and G2, respectively. The absorption was found to decrease as the wavelength increased, and that might be a result of internal electric fields or distortion of lattice due to strain. Moreover, the improved absorption spectra in the visible region could suggest photocatalytic activity enhancement toward certain pollutants, a property that previously noticed.
31
(a) Absorbance and (b) absorption coefficient (𝛼) vs. wavelength for of CuO–Fe2O3–MgO NCs at different glycine concentrations.
The absorption coefficient (α) was evaluated using equation (4),
50
where A is the absorbance and t is the thickness
The variation of absorption coefficient for the three NCs with incident wavelength is shown in Figure 5(b). It was observed that the absorption coefficient exponentially decreased as the wavelength increased, a behavior that might be caused by distortion of lattice as a result of charge carrier’s inelastic scattering driven by phonons.
Extinction coefficient, refractive index, and optical conductivity
The extinction coefficient (k), refractive index (n), and optical conductivity (σ
opt
) were evaluated using the relations given in equation (5)–(7), respectively,51,52 and their values were depicted in Figure 6(a)–(c) (a) Extinction coefficient (k) and (b) refractive index (n) vs. wavelength (nm), and (c) optical conductivity (σ) vs. photon energy for CuO–Fe2O3–MgO NCs.



Extinction coefficient (k) is an essential optical constant that could express the fraction of light lost per unit distance in the testing material medium. 51 As shown in Figure 6(a), the extinction coefficient (k) increase in the visible region which specify good optical properties of the prepared tri-phase metal oxides at this region. The refractive index (n) gives information regarding ions polarization, phase velocity of light and local field inside the optical material. In Figure 6(b), the n was noticeably increased at the short lower wavelength, while decreased as the incident light approaching the UV region.
Optical conductivity σ opt is one of the basic properties of the material which depends on the 𝛼, n, k, and c. Figure 6(c) illustrates the changes in σ opt as a function of photon energy (eV) for all the prepared NCs. It possesses a maximum value in high energy regions which appoint that the prepared products can be utilized in optical applications.
Optical energy gap (E g )
The electronic data of the synthesized CuO–Fe2O3–MgO NCs can be employed to determine bandgap energy (E
g
) as well. The E
g
values for the direct transition can be computed via Tauc’s equation (8)6,51 Optical band gap plots for CuO–Fe2O3–MgO NCs at different glycine concentrations. Optical energy measurements of prepared samples.
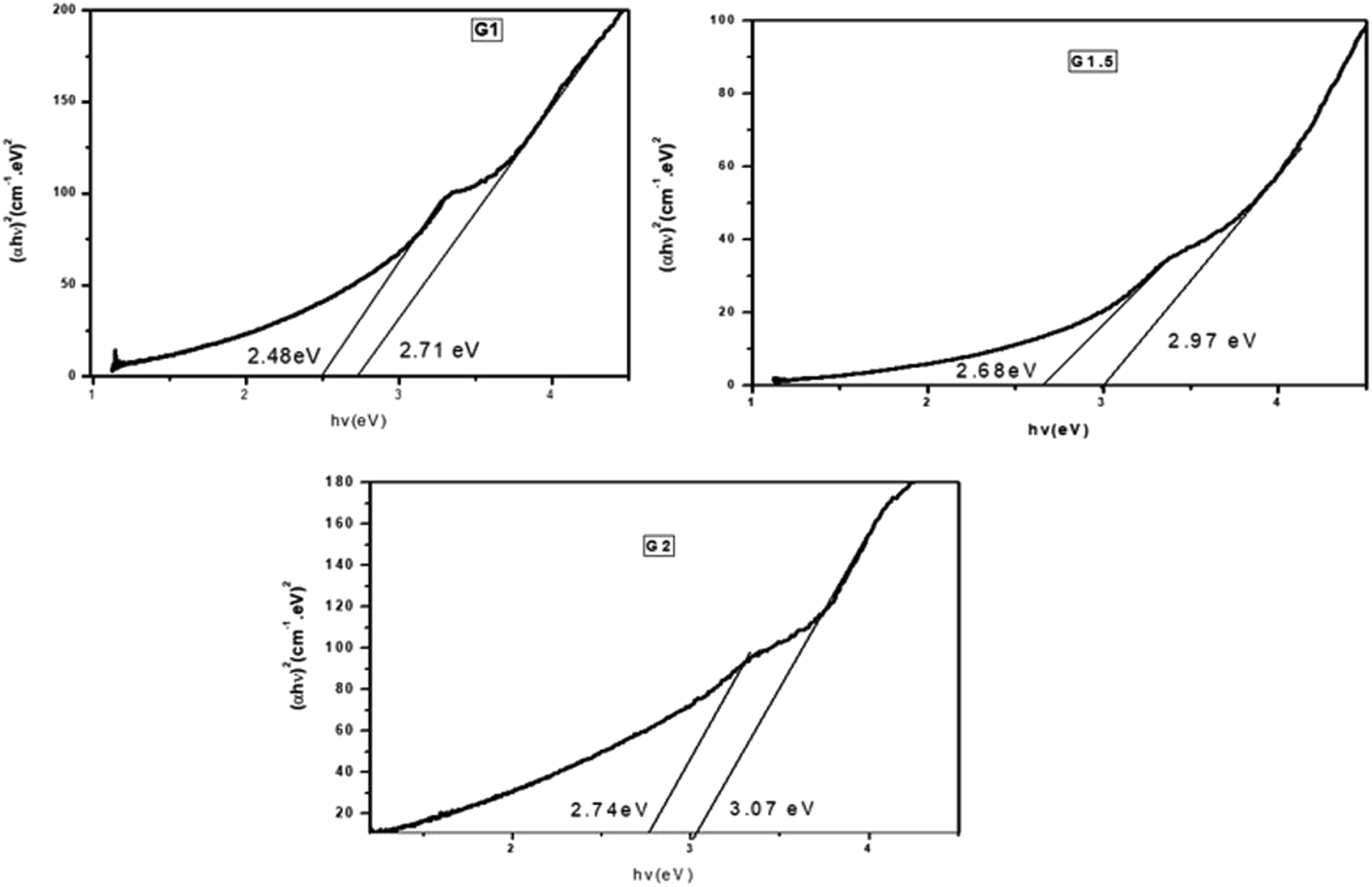

Generally, the E g values increase as fuel (glycine) concentration increase because of the decrease in the crystallite size which emphasized the nanosized nature of the synthesized NCs. Also, these results confirmed the existence quantum confinement of the fabricated samples. The optical results indicated that the CuO–Fe2O3–MgO NCs are convenient substances for many applications such as optoelectronics photocatalysis and thermoelectric.1,6
Antibacterial activity
Antibacterial activity of CuO–Fe2O3–MgO nanocomposites at different concentrations.


Selected plate images of antibacterial tests of CuO–Fe2O3–MgO NCs for (from top to bottom) G1, G1.5, and G2 at concentrations of 100, 200, and 400 mg/ml (discs termed 1, 2, and 3, respectively; 4 and 5 discs are for azithromycin antibiotics (positive control) and distilled water (negative control)) against Staphylococcus aureus (left column images) and Escherichia coli (right column images). The histograms illustrate the zone of inhibition in mm.
The observed general trend in the antibacterial activity and the predicted particle size were to increase with glycine fuel concentration increase, that is G1 > G1.5 > G2, supporting the fact that the smaller the particles, the easier the interaction and thus penetration into inside the cell, which subsequently could cause the cell death.
A comparison of antibacterial analysis of CuO–Fe2O3–MgO NCs with other NCs.

Conclusion
The trimetallic oxide CuO–Fe2O3–MgO NCs were synthesized for the first time using the sol-gel technique and under different concentrations of amino acid glycine as a NPs–facilitating and shape–directing agent. Analysis indicates the decrease in particle size and increase in optical energy bandgap with glycine concentration increase. This NCs has a comparable antibacterial activity to its alike multi-phase oxides, more active against S. aureus than E. coli. Such introductory results making them one potential candidate for advanced applications, possibly to benefit catalytic, electronics and bactericidal fields. These findings could support the used approach for synthesis of multi-phase oxides NPs where specific particle sizes are required, by controlling, beside other conditions, the glycine concentration.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors are grateful to the Deanship of Scientific Research, King Saud University, for the support through the Vice Deanship of Scientific Research Chairs, Engineer Abdullah Bugshan Research Chair for Dental and Oral Rehabilitation.



