Abstract
Bio-based materials have attracted ample research interest due to their intrinsic beneficial impact on human society. Microcrystalline cellulose (MCC) extracted from a variety of bioresources is one of the most prominent candidate in this regard. Herein, the MCC was synthesized from jute fiber by a facile ammonium persulfate (APS) oxidation method resulting in carboxylic functional groups on the fiber surface. The carboxylated MCC was utilized to develop polyvinyl alcohol (PVA) composite films in a solution casting method. The surface chemistry, thermal properties, and surface morphology of MCC and MCC-PVA composite films were studied by FT-IR spectroscopy, thermogravimetric analysis, and scanning electron microscopy. The SEM images confirmed the rod-like MCC with average particle diameter 4.6 μm and length in 48.4 μm. The FT-IR spectroscopy suggested the complete removal of lignin and hemicellulose from jute fiber. A peak at 1730 cm−1 was introduced in MCC due to oxidation with APS. The MCC was readily dispersible in water, and chemical interaction with –OH group of PVA was spontaneous due to the carboxylation in MCC. The absorption peak of –OH groups in MCC-PVA composite films were shifted, somewhat disappeared and weakened due to the intra and intermolecular hydrogen bonding of MCC and OH groups in PVA. Thermogravimetric analysis expressed the thermal behavior of MCC and MCC-PVA composite films. The char formation at 500°C of the two different composite films was 15% and 16%, respectively. The stability of MCC-PVA composite films at elevated temperatures is the indication of potential application as flame retardant material.
Introduction
With the growing concern of the adverse effect of countless non-biodegradable and non-renewable materials, a great deal of momentum to counteract this effect is prevailing from all levels of stake-holders of the society. As of 2020, about three-fourth (73.8%) of the world textile fiber consumption is comprised of man-made fibers with a constant growing demand. 1 Since the synthetic fibers are non-biodegradable and coming from non-renewable resource, they lead an unsustainable cycle of production and consumption. In addition, the environment concern and long-term economic goal have raised an appreciable impetus to think about environment friendly composite materials. In this regard, cellulose based composite materials have attracted the ample attention towards the researchers of the world. Other biomass and organic materials such as grass, agricultural crops, and wood or wastes are significant renewable resources too. Among them, cellulose is the most abundant and ubiquitous natural polymer in the eco-system.2–4 The worldwide cellulose production is approximately 1012 tons per annum.5–7 Due to some novel attributes such as renewability, biodegradability, nontoxicity, and improved mechanical properties, low thermal expansion, low density and light weight cellulose can be applied in the field of pharmaceutical, medical, biomedical, construction, automotive, cosmetic and packaging industries.8–11 Traditionally, cellulose is extracted from the wood which contains only 45% cellulose.2,5 The other non-woody fibers such as hemp, sisal, flax, though contain higher amount of cellulose, are less important in terms of economy and world production. 12 In this context, jute fiber is the attractive and competent alternative for cellulose as it has higher amount of cellulose (∼75%) content and second most economical crop in the world next to cotton.12–15 Several attempts have been deployed to extract micro-nanocellulose from jute fiber. The prominent examples are acid hydrolysis, 16 steam explosion,17,18 TEMPO mediated oxidation, 19 and oxidation with ammonium persulfate. 12 Among them, APS oxidized cellulose contains carboxylic functional moieties due to the direct oxidation in the C6 position of the glucose ring of the cellulose chain.20,21
Polyvinyl alcohol (PVA) is water-soluble and colorless synthetic resin. Due to presence of hydroxyl groups, PVA shows excellent film-forming capability by creating intermolecular hydrogen bond.22,23 The barrier, mechanical, and thermal properties of PVA film can be improved by reinforcing filler materials like cellulose, 24 titanium, 25 collagen, 26 carbon nanotubes, 27 silica, 28 chitosan, 29 and so on. There are numerous examples of composite film preparation with highly functional materials like oligomeric and silica based materials.30,31 An overarching goal of this research is to develop a composite film of PVA reinforced with microcrystalline cellulose (MCC) obtained from jute fiber by single step APS oxidation method. Although there are many examples8,9 of composite films with micro or nanocellulose with PVA, the carboxylated MCC from jute fiber for composite film preparation with biocompatible PVA was still unexplored area of study. In this study, the surface topography, surface chemistry, and thermal properties of MCC and MCC-PVA composite films were investigated. The knowledge gathered from this study might lead the applicability of this MCC-PVA composite at different conditions.
Materials and methods
Materials
Jute fiber was purchased from Sirajganj district of Bangladesh. Polyvinyl alcohol (PVA, molecular weight: 1, 25,000 degree of polymerization: 1700–1800, Viscosity: 25–32 cps, hydrolysis (mole %): 98–99, volatile matter: max 5%, Ash: max 0.7%, pH (0.2% in water): 5.0–7.0) was purchased from Research-Lab Chem Industries, India. Ammonium persulfate (Extra Pure) was purchased from Daejung Chemicals & Metals Co. Ltd., Korea.
Extraction of microcrystalline cellulose from jute fiber and preparation of MCC-PVA film
The jute fiber was washed with water and ethanol twice to remove the dust and dirt. The fiber was chopped into pieces and oxidized with 1.0 M ammonium persulfate (APS) at 60°C for 3, 6, and 12 hours. The obtained cellulose powder was washed with de-ionized water for several time and vacuum dried. The powder form microcrystalline cellulose was stored and used for film preparation and characterization.
The MCC-PVA film was prepared by film casting method published previously. 32 Briefly, MCC powder and PVA were taken in different ratios such as 50:50, 60:40, 80: 20, and 90:10. The required amount of MCC and PVA was dispersed or dissolved in water. The two solutions were mixed homogenously by stirring 8 h at room temperature. The mixture of MCC and PVA solution was then poured into a Petri dish. The Petri dish was coated with silicone film which facilitated the easy release of the MCC-PVA film from the surface.
Characterization of MCC and MCC-PVA films
The surface chemical structure of MCC and MCC-PVA film were characterized by FT-IR spectroscopy (Shimadzu, Japan) with a spectral resolution 4 cm−1. The content of carboxylic group was quantified through conductometric titration using a conductometer Multi 9310 IDS, Germany as described elsewhere. 33 The surface topography was investigated with scanning electron microscopy (JEOL, JSM-6490LA, Japan). Thermogravimetric analysis (TGA) was examined using TGA measurement instrument (Shimadzu, TGA-50H, Japan.) at a heating rate of 10°C/min under N2 flow condition into the combustion chamber.
Results and discussion
MCC extraction and hybrid film formation
Microcrystalline cellulose (MCC) was extracted from jute fiber by simple ammonium persulfate (APS) oxidation method without pretreatment following the previous reports.12,20 The Figure 1 represents the schematic of MCC extraction and simultaneous MCC-PVA composite film preparation. Briefly, the mechanically ground jute fiber was placed in a reaction vessel with 1.0 M APS and heated at 60°C for 3, 6, and 12 hours. The complete removal of non-cellulosic carbohydrates from jute fiber was achieved within 3 h oxidation period. However, this oxidation time span was unable to disintegrate the cellulose from jute fiber. The further increase in oxidation time in 6 and 12 hours resulted the micro cellulose and complete powder from was obtained by 12 hours oxidation. The Figures 2(a) to 2(c) show the photographs of jute cellulose in different stages of oxidation. Schematic presentation of carboxylated MCC synthesis from jute fiber and consequent composite film preparation with MCC and PVA. Photographs of cellulose extracted from jute fiber by APS oxidation (a) 3h, (b) 6h, (d) 12 h and film formation with MCC at 12 h oxidation in different mixing ratio with PVA (d) 50:50, (e) 60:40, and (f) 80:20.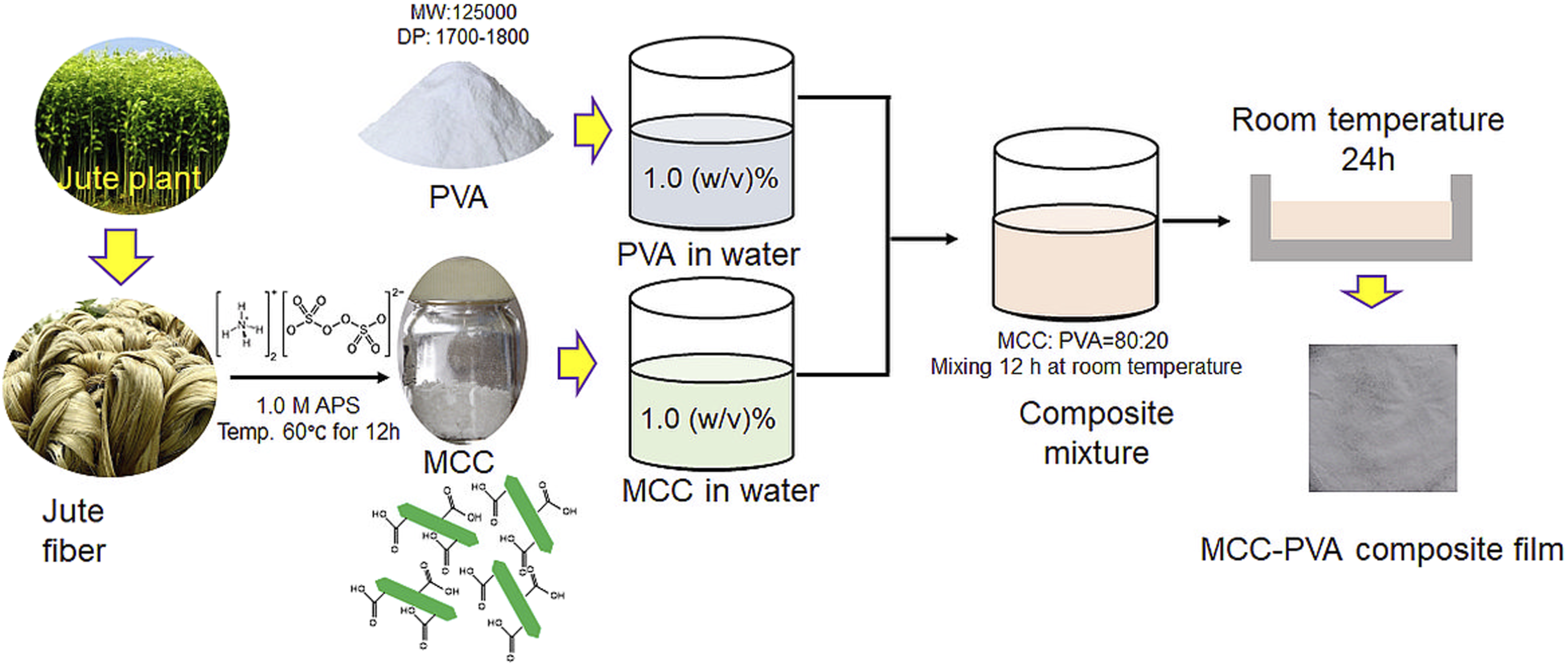

Microcrystalline powder cellulose was used to produce hybrid film with polyvinyl alcohol (PVA). Different mixing ratio of MCC and PVA was exploited to produce the hybrid film. The Figures 2(d) to 2(f) represent the photographs of 50:50, 60:40, and 80:20 film of MCC and PVA, respectively. The uniform film formation was obtained with the increase of the MCC content in the film. The lower proportions of MCC exhibited several cracks in the film and created difficulties in peeling of the film from the surface (figures 2d and 2e). The successful film was possible with 80:20 mixing ratio of MCC and PVA resulted the uniform and flawless film with easy removal from the surface.
Surface morphology of MCC and MCC-PVA films
The surface topography of MCC and MCC-PVA composite films were investigated with scanning electron microscope (SEM). The SEM images of MCC confirmed the rod like structure of MCC (Figure 3a). The vigorous treatment of jute fiber with strong oxidizing agent APS resulted the removal of non-cellulosic substances such as lignin and hemicellulose. Surface morphology of MCC and PVA-MCCA composite films. SEM image of (a) MCC isolated from jute fiber, (b) and (c) diameter and length distribution of MCC respectively. Photograph of (d) 20/80 PVA-MCC composite film, (e) SEM image of composite film, and (f) magnification of the area indicating the MCC embedded in the PVA matrix.
As the oxidation time further proceeds, the jute cellulose was longitudinally decreased as well as reduced the diameter of the fiber. As a result, the aspect ratio (length: diameter) of the resultant fiber was increased. The distribution of diameter and length of the MCC is presented in Figures 3(b) and (c), respectively. The average diameter and length of the microcrystal was found 4.6 and 48.4 μm. The Figure 3(d) shows the MCC-PVA composite film with the corresponding SEM images of the film presented in Figures 3(e) and 3(f). The SEM image confirmed the random distribution of MCC in PVA matrix. In fact, the MCC was embedded in the surface of the film (Figure 3f). The uneven distribution of crevices and cracks were also observed over the surface with miscellaneous sized and shaped of MCC.
Surface chemistry of MCC and MCC-PVA composite film
The surface chemistry of MCC and MCC-PVA composite films was investigated by FT-IR spectroscopy and conductometric titration method. The Figure 4(a) depicts the typical FT-IR spectra of jute fiber and MCC oxidized with APS in different time span. The characteristic cellulosic peaks were clearly appeared in MCC indicating successful isolation of cellulose from jute fiber (Figure 4a). Surface chemical structure of jute fiber, MCC and PVA-MCC composite films. FT-IR spectra of (a) jute fiber and MCC of different oxidation time, (b) typical conductometric titration curve of MCC obtained from jute fiber by APS oxidation in 12 h time and (c) FT-IR spectra of PVA-MCC composite films of different composition.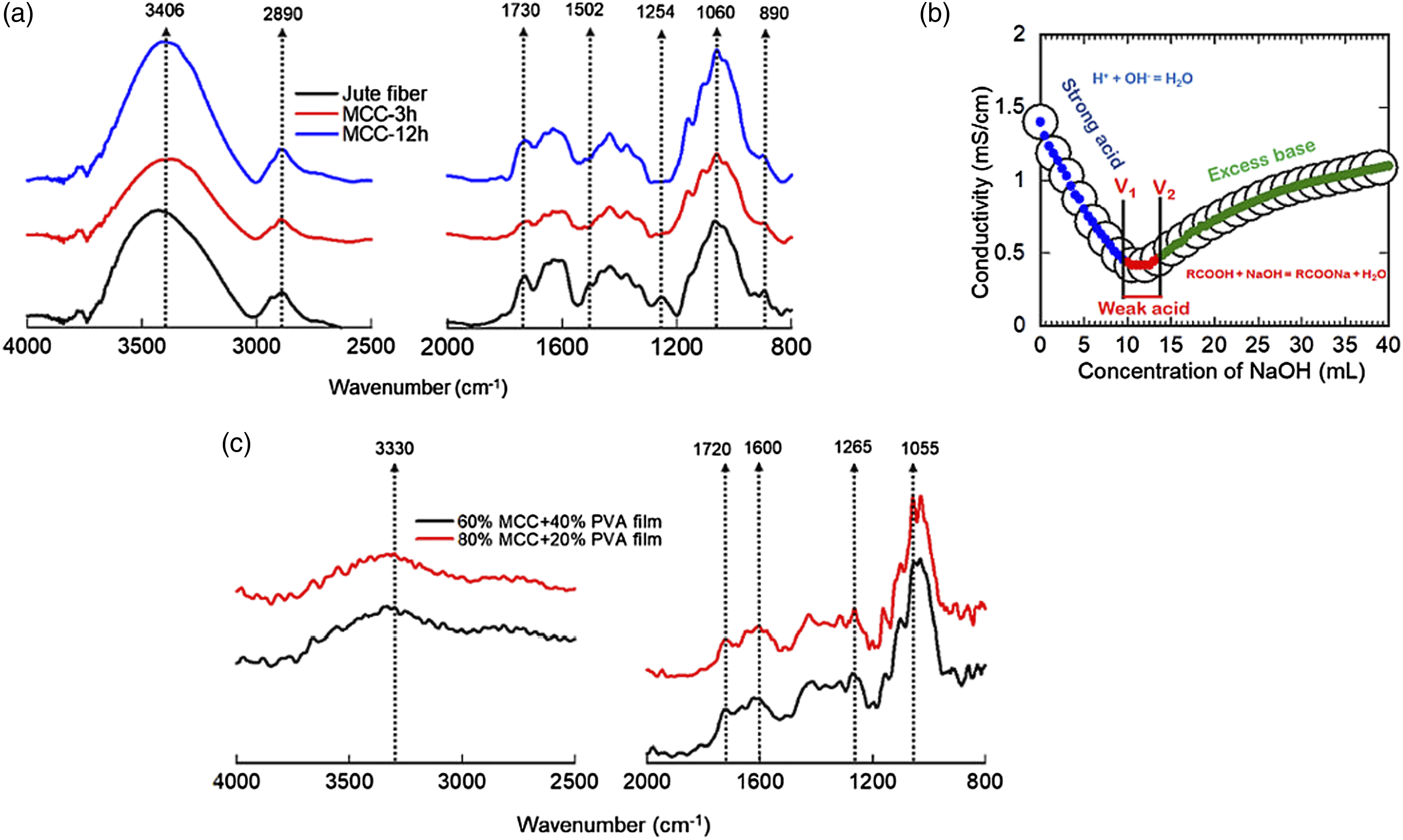
The typical O-H peaks of cellulose were visible at nearly 3400 cm⁻1, and antisymmetric and symmetric stretching vibrations of –CH2 in alkyl chain were observed at 2904 and 2890 cm−1, respectively. 34 A new peak appeared in 1730 cm−1 for C = O stretching vibrations of carboxylic group in MCC. The incorporation of this particular peak was due to the oxidation of C6 carbon of glucosyl residue of cellulose. 12 In contrast, pristine jute fiber also exhibited carboxylic peak at 1725 cm−1 region which was due to the uronic ester or acetyl groups of hemicellulose. 9 But, as the oxidation proceeds, the intensity of this peak decreased substantially indicating the removal of hemicellulose from the jute fiber surface during the early stage of oxidation. This indication is shown by the MCC after 3h of oxidation presented in red line. As the oxidation further proceeded the C = O stretching vibration newly appeared in the C6 carbon of cellulose chain. On the contrary, the MCC of different oxidation time was free from lignin as their FT-IR spectra did not exhibit any peak at 1254 and 1502 cm−1 responsible for lignin which is vividly present in jute fiber. 35 The other ordinary peaks of cellulose at 1606 cm⁻1 represents the absorbed water,36,37 1429 cm⁻1 expressed the CH2 bending 38 and the peak at 1060 cm⁻1 and 898 cm⁻1 were associated with saccharine structure 39 was common and found both in jute fiber and MCC. But in case of MCC, these cellulosic peaks are more exposed than the jute fiber which is very relevant as the raw jute fiber is cemented with several types of noncellulosic polysaccharides such as hemicellulose, lignins, and wax. Consequently, the MCC obtained from jute fiber was completely free from lignin and hemicellulose, and the relative amount of cellulose was higher in MCC than the jute fiber. The generation of carboxylic moieties at C6 position of cellulose chain was further quantified by conductometric titration. The Figure 4(b) shows the typical conductometric titration of MCC obtained from jute fiber. It clearly indicates three regions for strong acid, weak acid, and excess amount of alkali addition. As the mobility of H+ ion is faster, the conductivity decreased sharply up to the valley between volume V1 and V2. In this region, the change of conductivity is almost constant. The added OH− was consumed for the neutralization of weak carboxylic acid in cellulose chain generated through the oxidation with APS. The amount of carboxylic group charge density was found 320, 180, and 760 mmol/kg for raw jute fiber and MCC for 3 h and 12 h oxidation time, respectively. This investigation is well in agreement with the FT-IR spectroscopic studies of the jute fiber and MCC.
The surface chemistry of MCC-PVA composite films was also studied with FT-IR spectroscopy. The Figure 4(c) denotes the FT-IR spectra of 40/60 and 20/80 MCC-PVA composite films. In composite film, the stretching vibration of hydroxyl groups shifted to lower wavenumber at 3330 cm⁻1, implying the intermolecular or intramolecular hydrogen bonding with MCC and PVA or within PVA itself. A peak at 1720 cm−1 was observed in both the films confirming the stretching vibration of carbonyl functional group originated either from the ester linkage between –COOH groups of MCC and –OH group of PVA or residual acetate group remained in PVA due to the hydrolysis of polyvinyl acetate to polyvinyl alcohol during preparation and processing. 40 Another peak at 1265 cm−1 was found in the composite films ascertaining the ester linkage with MCC and PVA. However, the intra and intermolecular H-bonding with MCC and PVA in the composite films are predicted. The other cellulosic peaks at 1100–1055 cm⁻1 responsible for C–O–C, and 1153 cm⁻1 for C–O shifted to lower energy state than the MCC and became weaker in intensity.41,42 The more prominently, absorption peak of –OH groups were shifted, somewhat disappeared and weakened due to the interaction of MCC and OH groups in PVA. 43 These findings supported the formation of new intra or inter-molecular hydrogen bonds as well as conformation change within MCC and PVA. 44
Thermal properties analysis
Thermal properties of PVA, jute fiber, MCC, and PVA-MCC composite films.

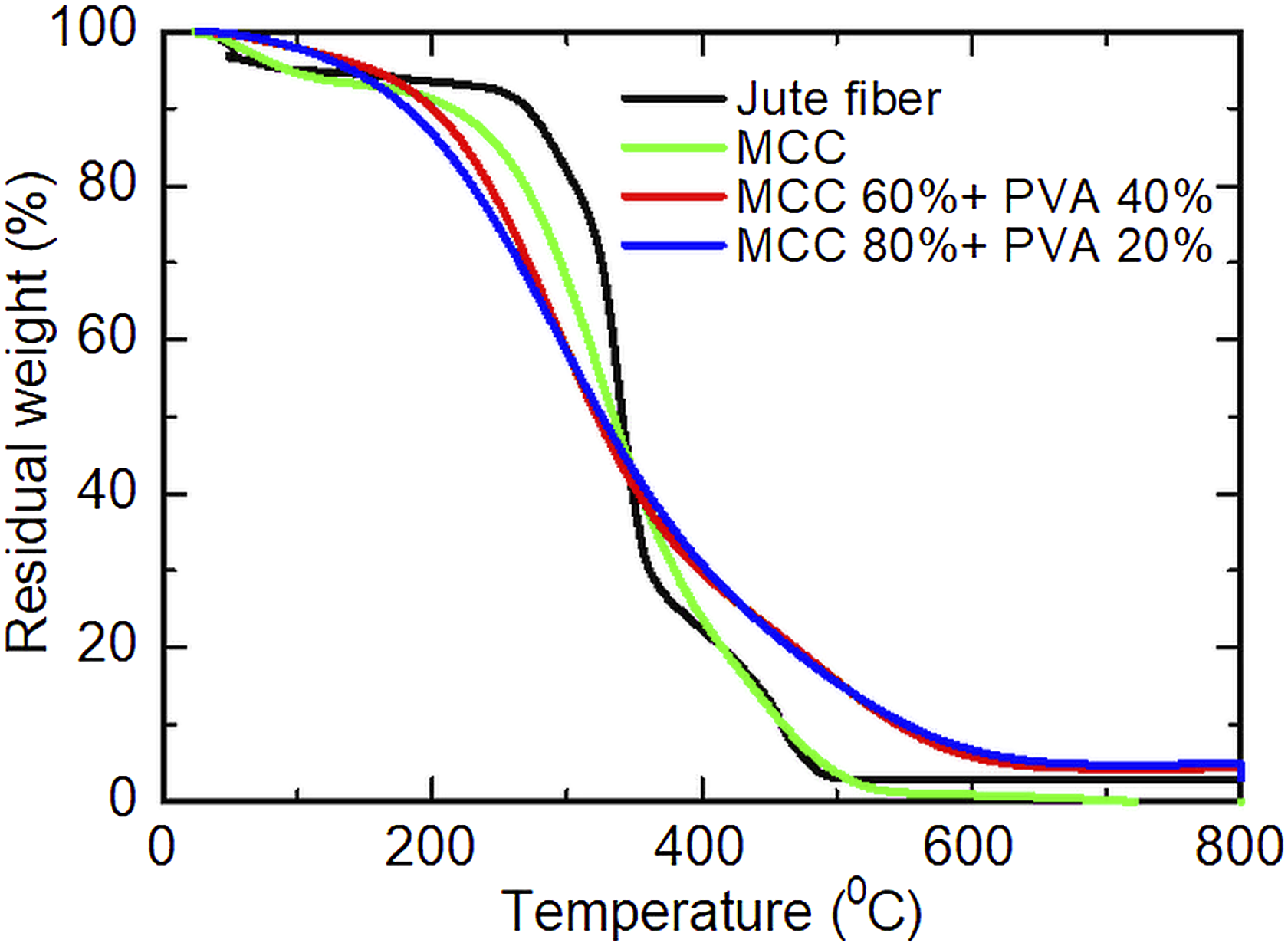
Thermogravimetric analysis (TGA) curves of jute fiber, MCC obtained by 12 h and PVA-MCC composite films of different composition.
Conclusions
This study validates the composite film formation of carboxylated MCC from jute fiber with water soluble PVA polymer. The maximum 80% MCC was incorporated into the hybrid film. The surface morphology of the composite film confirmed the rough surface and opaque film where MCC was found embedding in the PVA matrix. The surface chemistry of the MCC showed carboxylic group containing MCC without any non-carbohydrate polysaccharides. The strong ester linkage formation was plausible as well as various H-bonding interaction was confirmed in the composite films, and thus indicated the higher char formation of the films at higher temperatures. The stability of the composite film at elevated temperatures widens the scope of using MCC-PVA composite films at broader spectrum of applications such as a flame retardant material. As jute is widely cultivated in Bangladesh, it has profound socioeconomic impact for the jute farmers as well as the country’s economy. This study might open new areas of integrating jute in many different combinations of composite materials. Further study can be focused on the biodegradation, crystal structure, and mechanical properties of the composite films. A limiting point of this study was to develop larger film. In addition, the developed MCC-PVA film is water soluble in nature which facilitates an opportunity to incorporate the hydrophobicity by further research.
Supplemental Material
Supplemental Material - Poly-(vinyl alcohol) composite films reinforced with carboxylated functional microcrystalline cellulose from jute fiber
Supplemental Material for Poly-(vinyl alcohol) composite films reinforced with carboxylated functional microcrystalline cellulose from jute fiber by Md Zahid Hasan, Yeasin Arafat, M Mahbubul Bashar, Md Nabi Newaz Niloy, Md Imranul Islam, Shahjalal Khandaker, and AM Sarwaruddin Chowdhury in Composites and Advanced Materials
Footnotes
Acknowledgements
The authors are grateful to the Centre for Advanced Research in Science and Technology (CARST), University of Dhaka, Bangladesh.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors are thankful for the financial support from the University Grants Commission (UGC) of Bangladesh through the Research Cell of Mawlana Bhashani Science and Technology University for the fiscal year 2020-2021. The specific research grant number is 3631108.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
