Abstract
Multiple myeloma associated with bleeding events secondary to von Willebrand syndrome is underdiagnosed. The management of this entity is highly complex, and aims to control the hemorrhagic syndrome and reduce plasma viscosity with plasmapheresis and multiple myeloma-specific treatment. The authors report the rare case of a young patient with high-risk multiple myeloma complicated by hyperviscosity syndrome and presenting an acquired von Willebrand syndrome with hemorrhagic manifestations, requiring urgent therapeutic management to save the patient's life.
Introduction
Symptomatic myeloma disease requiring treatment is characterized by renal failure, anemia, hypercalcemia, and/or lytic bone lesions and is often associated with a monoclonal paraprotein in the blood and/or urine. 1 Multiple myeloma (MM) is usually associated with a high risk of venous thromboembolism (VTE).2,3 Bleeding events in MM are observed with IgG, IgA, and IgM, with a rate of 13%, 33%, and 36%, respectively.3‐5
The acquired von Willebrand syndrome (AvWS) is a rare hemostasis disorder with a 1% to 3% prevalence in the overall population, featured by quantitative or qualitative von Willebrand factor (vWF) abnormalities, most commonly combined with diverse underlying diseases.6‐8 The clinical and biological presentation is similar to the hereditary form.6,8 In 47% of cases, AvWS is associated with an underlying lymphoproliferative or plasma cell syndrome including monoclonal gammopathy of undetermined significance (MGUS), MM, or Waldenstrom disease (WM).6,8 Furthermore, hyperviscosity syndrome (HVS) has been reported in 2% to 6% of patients with MM.9,10 The classic triad of hyperviscosity includes mucous hemorrhage, visual disturbances, and neurological complications. 11
Hemorrhage due to AvWS as a main symptom of the disease is uncommon in the setting of myeloma.12,13 Potential biological pathways in HVS as well as in AvWS involve paraprotein hypersecretion and inhibition or elimination by autoimmune inhibitors binding to vWF or platelet vWF receptors, and vWF adsorption by myeloma cells with aberrant GP Ib expression.6,7
The authors report the case of a young patient with high-risk MM presenting with plasma HVS combined with AvWS, requiring emergency therapeutic care.
Patient and Observation
Patient Information
A 37-year-old patient was diagnosed with IgG lambda MM in the context of a 6-week deterioration of the general condition with weight loss of more than 7 kg.
Clinical Findings
On admission, he presented with a spontaneous hemorrhagic syndrome consisting of epistaxis, gingivorrhagia, spontaneous hematomas, and decreased visual acuity.
Diagnostic Assessment
The blood count showed severe anemia with Hb at 4.5 g/dL and normal platelet count. The hemostasis test revealed prolonged prothrombin time (PT) and activated partial thromboplastin time (PTT), at 22.9 for a 13.5-second control and 55.7 for a 28-second control, respectively. Fibrinogen levels were normal. Factor V levels were low 45%, which may explain the PT and APTT. However, hemostasis tests should be analyzed with caution because of the major hyperprotidemia. Factor VIII level was 71%, vWF activity (vWF:RCo = 30%), and vWF antigen (vWF:Ag = 50%). Plasma HVS was evoked by the presence of signs of bilateral stasis retinopathy at the fundus, neurological signs with headache and vertigo, secondary to hyperprotidemia at 138 g/L, hence the suspicion of monoclonal gammopathy with a peak of monoclonal IgG Lambda at 114 g/L on serum immunoelectrophoresis and an excess of Lambda free light chains quantified at 488 mg/L. Bone marrow aspiration showed 10% plasma cell infiltration, confirming the diagnosis of MM (Figure 1); 99% of plasma cells had a malignant flow cytometry profile. The molecular cytogenetics by NGS on sorted plasma cells was high risk, showing hypodiploidy to 44 chromosomes, presence of monosomies 13 and 22, partial deletions 1p (including 1p32) and 14q and a translocation t(4;14), and presence of a clonal KRAS mutation.
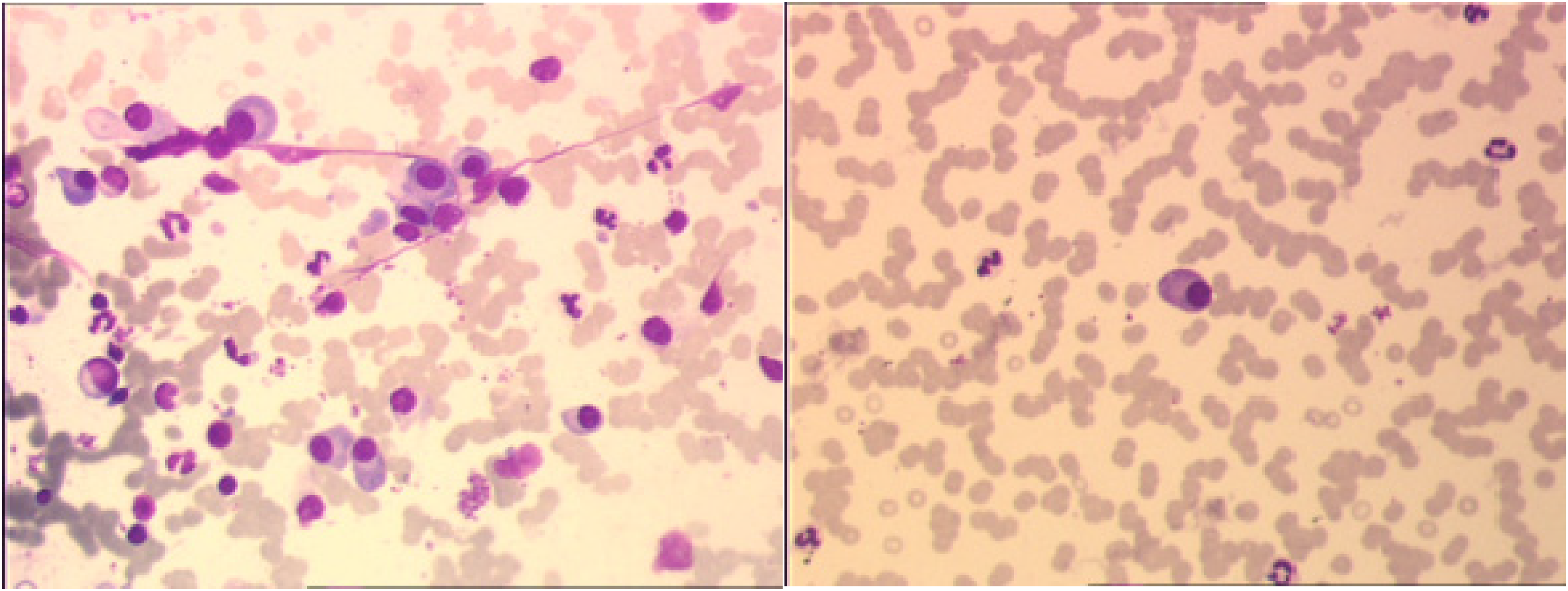
(A and B) Bone marrow sample of decreased richness with heterogeneous distribution of plasma cells. 10% of plasma cells, the majority of which are dystrophic (chromatin decondensation).
Therapeutic Interventions
The emergency treatment included four sessions of plasma exchange and a dexamethasone 40 mg/day course from day 1 to day 5, which reduced the serum protein level to 93 g/L and the monoclonal peak to 22 g/L. The patient received four induction cycles of daratumumab, bortezomib, thalidomide, and dexamethasone (Dara-VTD) with a very good partial response followed by a first autologous hematopoietic stem cell transplantation (AHSCT), then two consolidation cycles of daratumumab, bortezomib, lenalidomide, and dexamethasone (Dara-VRD) followed by a second AHSCT (neuropathy obliging to suspend thalidomide).
Follow-up and Outcome of Interventions
Currently, the patient receive an iberdomide-based maintenance therapy and myeloma is in complete response with undetectable residual disease in CMF at 10−5. Disease evaluations are summarized in Figure 2.
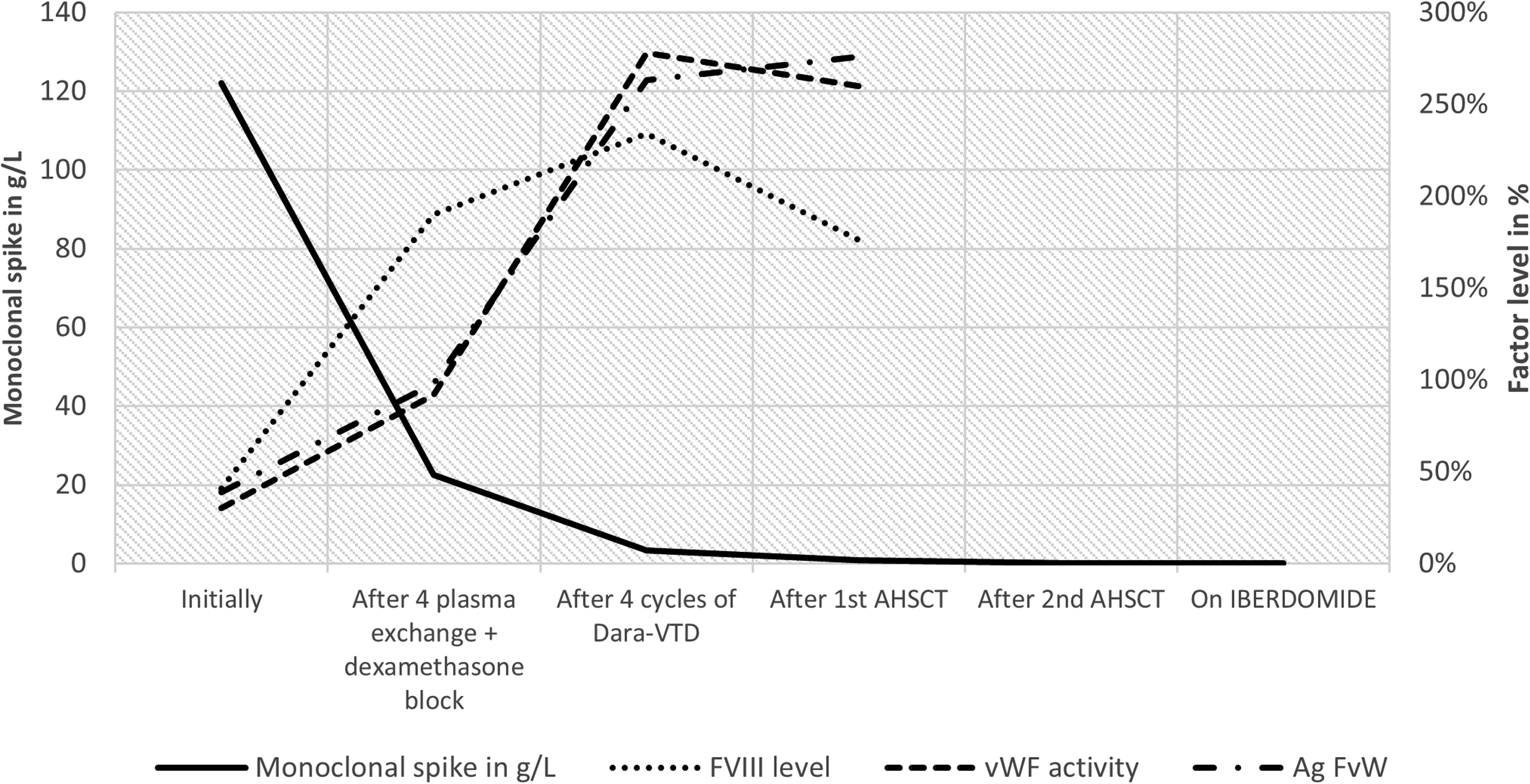
Evolution of the monoclonal peak and the level of FVIII and vWF according to the treatment administered.
Patient Perspective
During treatment, the patient was satisfied with the level of care provided to him.
Informed Consent
The purpose of the study was explained to the patient, and informed consent was received before samples were collected. The patient was made aware that her medical records would be kept confidential.
Discussion
To our knowledge, this presentation of MM complicated by plasma hyperviscosity and Willebrand syndrome remains misdiagnosed, and its therapeutic management has not been codified.
MM, which represents 18% of hematologic malignancies, is defined by the proliferation of plasma cells of malignant phenotype producing monoclonal immunoglobulins responsible for many disease-related complications, including hypercalcemia, renal failure, skeletal lesions, and rarely HVS due to increased protidemia.14,15 Occasionally, plasma cell dyscrasias in MM can be responsible for bleeding events in about 15% of patients with IgG and more than 30% of patients with IgA and IgM monoclonal proteins.3,16 The classic triad of hyperviscosity includes mucosal bleeding, visual, and neurological disturbances. In this setting, a systematic fundus examination should be performed. 17 Hyperviscosity may explain, among other things, the pathogenesis of AvWS. Besides the evidence of rolling red cells on the blood smear, a thickening of the blood with rapid agglutination was observed at the time of collection, and the sedimentation rate was initially unmeasurable. 18 The bleeding and clinical symptoms of both entities resolved, and were associated with a decrease in the monoclonal peak after plasmapheresis and corticosteroid therapy. 19 Furthermore, circulating autoantibodies directed against functional or nonfunctional vWF domains have been reported in MM complicated by Willebrand syndrome.20,21
The diagnosis of suspected AvWS is similar to hereditary Willebrand disease.6,7 Characteristic features of AvWS include lack of previous clinical bleeding abnormalities, late onset, negative family history, and association with a variety of underlying diseases.6‐8 In this case, the primary hemostasis test (PFA-100) was abnormal. The prothrombin level is normal, and the activated PTT is prolonged only if factor VIII deficiency is present. Von Willebrand antigen (vWF:Ag) and vWF activity are both abnormally low.21,22
Regarded as a rare entity, AvWS secondary to MM with HVS is a major therapeutic emergency. Management is complex and aims to control the hemorrhagic syndrome and to reduce plasma viscosity. 23 Plasmapheresis may be an emergency option to eliminate excess paraproteins and immunoglobulins. Therefore, instead of albumin, fresh-frozen plasma should be used to avoid the depletion of fibrinogen and other coagulation factors, causing a hemorrhagic syndrome in patients with AvWS. In cases of severe hemorrhage, recovery of vWF activity can be accelerated by supplementation with a concentrate containing vWF or desmopressin. 24 A single plasma exchange cycle can reduce plasma viscosity by 30% to 50% and monoclonal peak of immunoglobulin by 60%. 17
In our case, we obtained an 80% decrease in the monoclonal peak after four plasma exchanges. Numerous other studies have shown that myeloma-specific therapies can control bleeding symptoms and restore vWF levels, inducing a favorable response to MM. Intensive melphalan-based chemotherapy followed by HSC autotransplantation has also been shown to be effective.25,26 Many studies have demonstrated that by controlling paraproteinemia, MM-specific treatment could stop bleeding symptoms and improve vWF levels.27‐29 In the setting of an AvWS related to MM, it is essential to initiate myeloma-specific therapy as soon as possible, plus treatments to rapidly control paraproteinemia.
Conclusion
Bleeding manifestations in the setting of myeloma patients with high proteinemia can be life threatening. This is a rare and interesting case involving a young patient with AvWS and hyperviscosity associated with high-risk MM. Urgent treatment with plasma exchange, corticosteroids, and myeloma-specific therapy should be administered in this setting to save the patient's life.
Declarations
Ethical Approval
Ethics approval is not applicable for this manuscript.
Informed Consent
Verbal consent was obtained from the patient himself for his participation in the case report via telephone. According to current legislation in France, written consent is not mandatory for this type of study (Jardé Law).
Author Contributions
Ouadii Abakarim, Condom Pauline, and Botin Teresa did conceptualization, data curation, formal analysis, funding acquisition, investigation, methodology, project administration, resources, software, supervision, validation, visualization, writing the original draft, writing—review and editing.
Voisin Sophie, Huart Antoine, and Perrot Aurore did conceptualization, data curation, formal analysis, methodology, project administration, software, validation, visualization, writing—review and editing.
Acknowledgment
The authors thank all clinical staff who participated in the treatment of the patient.
Availability of Data and Materials
All data is available on the patient's computerized file in our database.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
