Abstract
Background
The activity and purity of Fibrin Sealant Grifols are described in this article. This fibrin sealant contains two components (fibrinogen and thrombin) that mimic the final stages of clotting. Fibrin sealants are primarily used as an adjunct to hemostasis for mild to moderate surgical bleeding in adults when standard surgical techniques are ineffective or impractical.
Objective
The aim of these studies was to assess the biochemical and functional characteristics of Fibrin Sealant Grifols.
Methods
Total protein, plasma protein impurities, and molecular forms were analyzed for both components. Clottable fibrinogen and thrombin activity were also measured. Both components were analyzed by SDS-PAGE and Western blot. The fibrin sealant clot was functionally characterized.
Results
The components had minimal contamination by other plasma proteins and no aggregates or degradation products. The fibrinogen component was almost completely clottable fibrinogen. The thrombin component contains albumin. The fibrinogen component formed a fibrin polymer and clots with tensile strength similar to literature values. The application device applied the components in a 1:1 volume ratio. Clotting time was ∼ 5 s when applied with the drip tip and nearly instantaneous when applied with the airless spray apparatus.
Conclusion
This fibrin sealant was shown to contain highly purified fibrinogen and thrombin delivered in a 1:1 volume ratio. These properties contribute to the clinical safety and efficacy of this product for many applications.
Introduction
Fibrin generation is one of the final steps in the coagulation cascade that leads to clot formation. Fibrin is generated by the enzymatic action of thrombin on the glycoprotein precursor, fibrinogen. Soluble fibrin monomers formed from fibrinogen bind to each other to form insoluble fibrin polymers. An additional action of thrombin is to activate Factor XIII. Activated Factor XIII induces covalent cross-linkages within the fibrin polymer network and the formation of a stable fibrin clot. Calcium is essential for the generation of thrombin and Factor XIIIa. The fibrin clot is held in place by binding to proteins including fibronectin, collagen, cell surface receptors and other proteins, 1 as well as mechanical interlocking of the clot to the tissue.
All marketed fibrin sealants contain two major components (thrombin and concentrated fibrinogen). These sealants mimic and augment the final stage of the coagulation cascade when the two components are mixed, ie, the cleavage of fibrinogen into fibrin by the action of thrombin. Fibrin sealants are used in a wide variety of surgeries to control perioperative bleeding across a range of surgery types, including cardiovascular, orthopedic, thoracic, gynecologic, and urologic surgeries as an adjunct to hemostasis for mild to moderate bleeding when control of bleeding by standard surgical techniques (such as suture, ligature, and cautery) are ineffective or impractical. Additionally, fibrin sealants can adhere tissues together and to prevent post-operative seromas, hematomas, and hemorrhages.2,3 In these procedures, the function of fibrin sealants is to stabilize tissue anastomoses or junctions and to form liquid and airtight seals at incisions.
The components of fibrin sealants perform these functions through the formation of stable fibrin clots when the components are combined during application. One advantage of fibrin sealant over synthetic surgical adhesives (eg, cyanoacrylates) is that fibrin sealants are degraded and absorbed by the body via physiologic fibrinolysis. In addition, fibrin sealants are biocompatible, ie, they do not induce inflammatory responses, necrosis or scarring. 4 Plasma-derived fibrin sealants can also stimulate angiogenesis and tissue regeneration. 3
Although all the commercially available fibrin sealants have a similar mode of action and metabolism, they cannot be considered to be equivalent products for a number of important reasons. The concentrations of fibrinogen and thrombin differ and as a result the speed of hemostasis and clot formation may be affected. Moreover, the application device used can also impact clot formation (structurally and temporally). In regard to safety, the viral-inactivation and removal processes used in the manufacture of these products are different. In addition, some fibrin sealants use fibrinolysis inhibitors (eg, aprotinin) which may induce immunological reactions. 5
The Fibrin Sealant Grifols kit consists of two sterile syringes containing frozen solutions of human fibrinogen and thrombin with calcium chloride assembled into a delivery device. The Fibrin Sealant Grifols (marketed as VERASEAL or VISTASEAL) is a single-use device designed to deliver the two biological components simultaneously either by spraying or dripping without the use of pressurized surgical gas. The Fibrin Sealant Grifols syringe holder has two syringe caps which are removed when the component syringes are thawed, and the co-packaged dual applicator device can be connected to the syringe holder. Once attached the Fibrin Sealant is ready for use and the user can manually depress the plunger to either spray or drip (when the airless spray accessory (ASA) is removed).
In this article, the biochemical characterization of the two components of Fibrin Sealant Grifols is described as well as the clot formed after delivery.
Materials and Methods
Preparation of the Fibrinogen and Fibrin Components
Both components of this fibrin sealant product were isolated from human plasma obtained by a modification of the Cohn plasma fractionation process in which fibrinogen is purified from fraction I and thrombin from the prothrombin complex (PTC) obtained from Fraction I supernatant.6,7 Both components are subjected to two virus removal/inactivation steps: solvent/detergent treatment and double nanofiltration. 8
Fibrinogen purification includes a solvent/detergent treatment with tri-n-butyl phosphate (TnBP) and polysorbate-80 (Tween 80) to inactivate enveloped viruses. Fibrinogen is further purified through glycine purification steps and finally undergoes a series of nanofiltrations to remove enveloped and non-enveloped viruses. The thrombin purification process also includes a solvent/detergent treatment with TnBP/polysorbate to inactivate enveloped viruses and nanofiltration to remove enveloped and non-enveloped viruses.
Source plasma is collected from healthy donors in licensed plasma centers and submitted to strict testing according to regulations and safety standards as plasma derived therapeutic products, including viral removal / inactivation steps and plasma inventory hold period.
The stability of the frozen product is 2 years at a temperature of ≤18 °C, and the stability of the product once thawed is 24 h at room temperature or 48 h at 2-8 °C. 9
Purity of Biologic Components
Fibrinogen Component
Total protein in the fibrinogen component of the fibrin sealant product (Fibrin Sealant, Grifols, Barcelona, Spain) was measured by the Bradford method 10 (Bio-Rad, Hercules, CA, USA) based on colorimetric reaction with Coomassie blue. Absorbance at 595 nm wavelength was measured spectrophotometrically (Shimadzu, Kyoto, Japan).
Fibrinogen clottable protein was determined by clot formation in the presence of thrombin and calcium, digestion with sulfuric acid, and protein nitrogen determination by Kjeldahl analysis using a distillation-titration system.11,12
Turbidity of the component solution was determined by nephelometry (Turbidimeter, Loveland, CO, USA). Osmolality was measured by freezing point depression (Micro-Osmometer, Advanced Instruments, Norwood, MA, USA). pH was measured by potentiometry in a 1% protein solution.
In the fibrinogen component, the following product-related impurities, fibronectin, albumin, IgG, IgA, IgM, and C4 were measured by immunonephelometry (Siemens Healthineers, Erlangen, Germany). Factor XIII (FXIII) was measured by spectrophotometry (Berichrom FXIII kit, Siemens Healthineers). Plasminogen was measured by chromogenic method (Berichrom Plasminogen kit, Siemens Healthineers).
Distribution of molecular forms of fibrinogen was determined by molecular exclusion chromatography using a high-performance liquid chromatography system with an ultraviolet detector (280 nm) (Waters, Milford, MA, USA). A molecular exclusion column (GE Healthcare Europe GmbH, Breisgau, Germany) was used to separate the fibrinogen components based on molecular size. The relative contribution of different molecular forms was calculated by integrating the peak area for each form. Under the conditions of this assay, fibrinogen polymers were defined as those eluting in the void volume (approximate size range ≥5,000,000 Daltons).
Thrombin Component
As with the fibrinogen component, total protein in the thrombin component was measured by the Bradford method 10 (Bio-Rad, Hercules, CA, USA).
Thrombin activity was measured by determination of clotting time after the addition of fibrinogen (ACL Instrumentation Laboratory, Bedford, MA, USA) with a final reading at 405 nm. Turbidity, osmolality, and pH were measured as described above.
For the thrombin component, the following were measured as product-related impurities: IgG, IgA, IgM, Factor V (FV), Factor VII (FVII), Factor IX (FIX), Factor X (FX), proteins C, Z, and S, ceruloplasmin and C4. Measurement of IgG, IgA, IgM, and C4 were described in the previous section. FV, FVII, FIX and FX were measured by ELISA using FV BioAssay ELISA kit (US Biological, Location); Asserachrom VII:Ag Kit, Asserachom FIX:Ag kit, and Asserachrom FX:Ag kit; (Diagnostica Stago, Barcelona, Spain). Ceruloplasmin was measured by immunonephelometry (Siemens Healthineers, Erlangen, Germany).
The distribution of molecular forms of the thrombin component was determined by molecular exclusion chromatography using a high-performance liquid chromatography system with an ultraviolet detector (280 nm) (Waters, Milford, MA, USA). A gel filtration column (Tosoh Bioscience, Griesheim, Germany or, Waters Corporation, Cedex, France) was used. The result was graphically recorded, and the percentage of each peak calculated by integration.
Thrombin and Fibrinogen SDS-PAGE
Thrombin and fibrinogen were analyzed by SDS-PAGE under reducing and non-reducing conditions after electrophoresis in a Bis-tris 4-12% gradient polyacrylamide gel using MOPS (3-morpholinopropane-1-sulfonic acid) buffer (20 × ) NuPAGE SDS running buffer and NuPAGE LDS sample buffer (Thermo Fisher, Waltham, MA, USA). For reducing conditions samples were treated with NuPAGE reducing agent and heat treatment. Fibrinogen gels were stained with Coomasie stain (SimplyBlue SafeStain-Thermo Fisher). Thrombin gels were stained with SilverXpress silver staining kit (Thermo Fisher).
Western Blot Analysis
After SDS-PAGE, Western blot analysis was conducted by transference to a polyvinylidene difluoride (PVDF) membrane (Millipore, Burlington, MA, USA) using NuPAGE Transfer Buffer and XCell II Blot Module (Thermo Fisher). A polyclonal fibrinogen antibody (DAKO/Agilent, Santa Clara, CA, USA) and a polyclonal thrombin antibody (DAKO/Agilent) were used as primary antibodies.
Viscosity
Dynamic viscosity of both components of the Fibrin Sealant Grifols was measured using a rolling ball microviscometer (Anton Paar, Graz, Austria).
Functional Testing
Fibrin Polymer Formation
The formation of fibrin chains was analyzed after treating the fibrinogen component of Fibrin Sealant Grifols with thrombin, in the absence or presence of calcium. Clots formed in the absence of calcium would be non-cross-linked. Those formed in the presence of calcium ion would be cross-linked. Clots were dissolved in urea and analyzed by SDS-PAGE under reducing conditions. SDS-PAGE analysis was conducted after electrophoresis in a Bis-tris 4-12% gradient polyacrylamide gel (Thermo Fisher GmbH, Dreieich, Germany) using standard materials and procedures (XCell-II MINI-CELL, Thermo Fisher-Novex). Coomasie stain (SimplyBlue SafeStain-Thermo Fisher) was used for staining.
Clotting Time
The rate of clot formation was measured after delivery of the components from the dual applicator device by drip application. The fibrin sealant was applied to an inclined glass surface (45 ± 3°) at room temperature (20-25 °C). Approximately 2 mL of the fibrin sealant was applied near the upper edge of glass surface and the distance and time from the point of application to the point at which the clot stopped was measured.
Mixing Test
Patent blue dye (Diagast, Loos, France) and Quinoline yellow dye (Diagast) were used to construct a color scale to be used as standards. The dyes were mixed in water in the following ratios (blue:yellow): 1:0, 3:1, 1:1, 1:3 and 0:1 to obtain a color scale ranging from dark blue, light blue, green, light green, and yellow as shown in the Results.
Mixing tests were conducted to determine the proportional combination of the two components of the fibrin sealant when delivered with the dual applicator using the ASA or drip tip. This was accomplished by adding yellow dye to the thrombin component and blue dye to the fibrinogen component in the same ratios as that of the standard control as shown in Table 1. Once the dyes were added, the syringes of thrombin and fibrinogen were assembled in the holder and the product was applied. Visual inspection of the mixture was used to match the color of the fibrin sealant clots derived using the dual applicator with the ASA and the drip tip. The color obtained was compared with the standard control of aqueous solutions shown in Table 1.
Different Volumes (0-6 µL/mL) of Patent Blue or Quinoline Yellow Dyes Were Added to the Components of Fibrin Sealant Grifols. These Solutions Were Used to Create Clots for Comparison to Aqueous Solutions to Assess Mixing When Fibrin Sealant Grifols Was Delivered with Airless Spray Accessory (ASA) or Drip Tip.

In addition, the absorbance of these color standards and the experimental samples were also measured over the range of 300-800 nm. Patent Blue has a peak absorption at 635–640 nm.13,14 Quinoline yellow has a peak absorption at 414 nm. 15 The height of the peaks at 414 and 640 nm should reflect the relative contribution of each component to the mixture delivered by the ASA or drip tip.
Tensile Strength
Tensile strength analyses were conducted by the Biomaterials, Biomechanics and Tissue Engineering group at Universitat Politecnica de Catalunya (Barcelona, Spain). Ten assays were performed for each Fibrin Sealant Grifols kit delivered using the drip tip.
Approximately 300 µL of each fibrin sealant kit (150 µL of each component) was applied to two circular pieces of synthetic fiber (Invitrogen, Thermo Fisher) attached to polyvinyl chloride cylinders (14.5 mm diameter). After application, the cylinders were compressed together with a force of 1-2 newtons (N) for 15 min. Then the clots were tested at room temperature with a device that applied a constant rate of axial movement (MTS Bionix 358 m, Eden Prairie, MN, USA). The maximal sustained load at failure was measured for each sample.
Confocal Microscopy
Confocal microscopy to characterize clot structure after application was performed at the Institut de Recerca de l’Hospital de la Santa Creu i Sant Pau (Barcelona, Spain). Morphologic parameters measured in this analysis were fiber diameter, fiber branches, fiber density and pore size in the clots. The system used was a Leica TCS SP2 spectral confocal microscope with a high sensitivity multiband detection system and optical acoustic band splitter (Leica Microsystems, Wetzlar, Germany).
For confocal microscopy, clots were formed on 35 mm circular glass dishes. Clotting was allowed for at least 20 min in a moist atmosphere at 37 °C. The clots were washed with 500 μL of Tris-buffered saline (150 mM NaCl and 10 mM Tris-hydrochloride, pH 7.4) and 200 μL of gold suspension (5-nm-diameter) was applied (final concentration 2.5 × 1012 particles/mL). After application of the gold particles, the clots were washed out with 500 μL Tris-buffered saline to remove excess gold particles. Specimens were scanned with the confocal microscope using an argon-krypton laser (488 and 496 nm) at 50% power. One image was selected from each clot at 8 × and 16 × zoom. Ten optical sections were collected at 1 µm intervals on the z-axis. Three images and three series were collected for each sample.
Fibrin fiber diameter (nm) was measured in the images generated by image analysis software (Leica). Diameters were measured on randomly selected fibers. Branches were detected in single images and then visualized on the reconstructed images. Branch points were counted in five series and expresses as the number per µm3. Fiber density measurement and pore size were determined on two-dimensional reconstructions of the optical sections. These images were processed using National Institutes of Health Image Software (V1.62, public domain software, Dr Wayne Rasband, US National Institutes of Health, Bethesda, MD, USA). Fibrin fiber density and pore size were expressed as mm3.
Opacity Measurement of Fibrin Sealant
The Fibrin Sealant Grifols, was dispensed into a flat-bottom, polystyrene, 24-well microplate. The percent transmittance was measured using plate reader at 500 nm wavelength (Molecular Devices SpectraMax M3 with SoftMax Pro 5.4.1 Software, San Jose, CA, USA). Within two minutes after optical measurement, the plate was placed on a template containing the corresponding labels for the individual wells and an image was captured using a digital camera on macro setting from a distance of 5-10 cm.
Results
Purity of Biologic Components
Fibrinogen Component Composition
As shown in Table 2, the total protein in the fibrinogen component of the Fibrin Sealant was 83 ± 6 mg/mL. The protein in the fibrinogen component was almost entirely clottable fibrinogen (84 ± 8 mg/mL). The fibrinogen component was pale yellow or colorless when frozen or thawed and had a pH near the physiological range.
Characteristics of Fibrinogen and Thrombin Components of Fibrin Sealant Grifols.
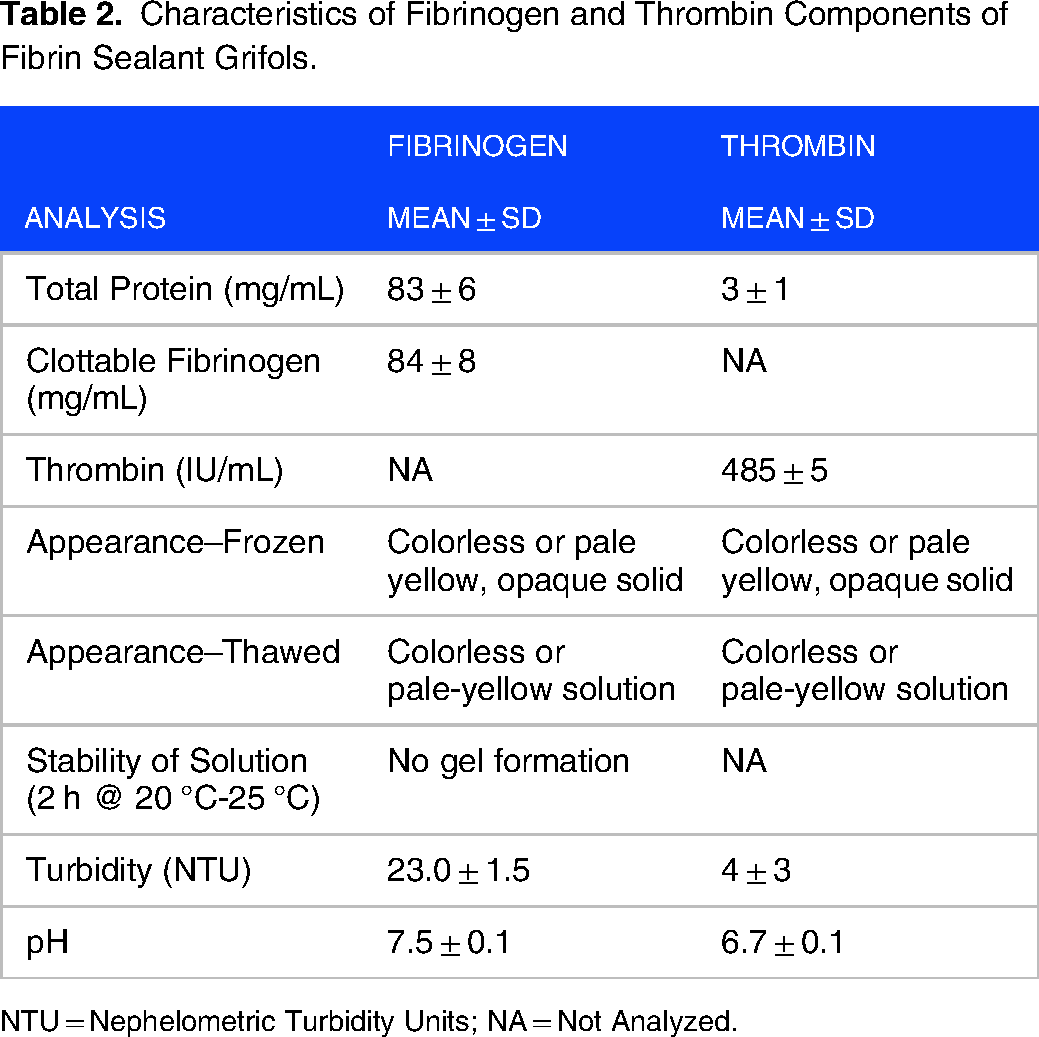
NTU = Nephelometric Turbidity Units; NA = Not Analyzed.
Fibrinogen component was purified from Fraction I obtained from human plasma. In order to better evaluate purity, proteins present in Fraction I that could co-precipitate together with fibrinogen have been evaluated. Table 3 shows the results of analyses for product-related impurities in the fibrinogen component of Fibrin Sealant Grifols: fibronectin, albumin, IgG, IgA, IgM, activatable plasminogen, C4 protein and FXIII. All the analyses were below the limit of detection except fibronectin, albumin, C4 protein and FXIII. For albumin and C4 protein, only one of the three batches gave a value above the limit of detection. These results are in agreement with the fact that total protein concentration is mainly fibrinogen. Low levels of fibronectin and FXIII were found in all three batches.
Analyses for Product-Related Impurities in the Fibrinogen and Thrombin Components of Fibrin Sealant Grifols.

N = 3 different batches for all measures. NA = not analyzed.
Figure 1 shows example HPLC tracings for the fibrinogen (Figure 2A) and thrombin (Figure 2B) components of Fibrin Sealant Grifols. Figure 2A shows an example chromatogram with three peaks present. The smallest peak (0.6 ± 0.2% of the total area for all three batches) eluting after approximately 19.3 min reflects the polymer form of fibrinogen. The next peak eluting at approximately 22.8 min represents oligomeric forms of fibrinogen (5.92 ± 0.54 of the total). The largest peak eluting at approximately 27.3 min was the monomeric form accounting for 93.48 ± 0.68%. No additional peaks or fibrinogen degradation products were observed.


SDS PAGE
Thrombin Component Composition
The total protein for the thrombin component was 3 ± 1 mg/mL much lower than the fibrinogen component. Of this protein, 2.4 ± 0.1 mg/mL corresponded to albumin added as an excipient (Table 2). The thrombin component showed thrombin activity: 485 ± 5 IU/mL. This component was also pale yellow or colorless when frozen or thawed and had a near physiological pH.
Thrombin component was purified from prothrombin complex (PTC; also referred to as the prothrombin complex concentrate). In order to better evaluate purity, proteins present in PTC that could co-purified together with thrombin have been evaluated. For the thrombin component, Table 3 shows the results of analyses for product-related impurities: IgG, IgA, IgM, FV:Ag, FVII:Ag, FIX:Ag, FX:Ag, proteins C, Z and S, ceruloplasmin and C4 protein. All the analytes were below the limit of detection except for FIX:Ag.
Figure 2B shows an example HPLC chromatogram for the thrombin component of fibrin sealant. The peak at approximately 9.7 min was indicative of albumin aggregates. The next peak at approximately 12.9 min represented albumin intermediates and the peak at 15.2 min represented albumin monomers. The last peak at approximately 17.8 min was thrombin. The mean percentage of thrombin present in the three batches tested was 19.0 ± 0.7% with the remainder of peak area corresponding to the various forms of albumin. An additional peak not considered for integration was observed and corresponds to an amino acid added as excipient. No additional peaks or thrombin degradation products were observed.
Fibrinogen and Thrombin SDS PAGE and Western Blot
Western blot and SDS PAGE analyses of the fibrinogen component of fibrin sealant are shown in Figure 2. Figure 2A is the SDS PAGE conducted under reducing conditions which cause the separation of fibrinogen into the component polypeptide chains. A Western blot conducted using a polyclonal fibrinogen antibody under reducing conditions is shown in Figure 3B. Fibrinogen is composed of three sets of polypeptides: Aα, Bβ, and γ with masses of 46.5, 52, and 66.6 KDa. It has been described that there are many molecular forms of fibrinogen in blood. These forms can be visualized by variations in gel electrophoretic behavior resulting from alternative processing in both the Aα and the γ chain and phosphorylation of the Aα chain. 16

SDS PAGE
After electrophoresis under reducing conditions the fibrinogen component shows bands corresponding to the α, β and γ chains of fibrinogen. The same bands are detected by Western blot in Figure 2B showing the purity of the fibrinogen component. The α chain shows a triplet probably due to the heterogeneity of the molecule.
Under non-reducing conditions, fibrinogen appears as a single band with a mass greater than 191 KDa (data not shown).
SDS PAGE and Western blot analysis of the thrombin component of Fibrin Sealant Grifols was also performed under reducing conditions (Figures 3A and B). A single band corresponding to thrombin was visible in both analyses. Under reducing conditions, a band corresponding to albumin was visible on the SDS PAGE (Figure 4A).

SDS-PAGE analysis of fibrin chain formation ± calcium. Thrombin was added to fibrinogen in the presence or absence of calcium ion to form stable (crosslinked) or unstable (non-crosslinked) clots. Lane 1 shows the protein standards. Lanes 2, 4, and 6 show fibrinogen plus thrombin in the presence of calcium. Lanes 3, 5, and 7 show fibrinogen and thrombin in the absence of calcium. Lanes 2, 4, and 6 show the formation of the γ-chain dimer (94 kDa) and a high molecular weight α-chain polymer while the β-chain was unaltered. In the absence of calcium, the unstable clot formed was dissociated into the three native chains (α, β, and γ).
Clot Characterization and Functional Testing
Fibrin Polymer Formation
This test is specific to the fibrinogen component. Fibrinogen is an acute phase protein that is part of the coagulation cascade and is converted into the insoluble protein fibrin during the clotting process. If fibrinogen is in native condition, thrombin acts by cleaving the fibrinopeptides A (α chain) and B (β chains), to form fibrin monomers, which spontaneously polymerize to form a fibrin clot. The clot is subsequently covalently cross-linked by FXIIIa (activated by thrombin) in the presence of calcium ions. Links are formed between γ chains forming γ-γ dimers and α chains forming α-polymers. The formation of these covalent bonds makes the clot more mechanically stable and resistant to proteolytic degradation. To evaluate the capacity of fibrinogen component of Fibrin Sealant Grifols to form cross-linked fibrin clots, the fibrinogen was incubated with thrombin in the presence or absence of calcium ions to form a clot. These clots were dissolved in the presence of urea and subjected to SDS-PAGE.
In Figure 4, lanes 2, 4, and 6 on this gel are the three batches of the fibrinogen component incubated with thrombin in the presence of calcium. Lanes 3, 5, and 7 are the same batches in the absence of calcium. In the presence of calcium, a band appears at 94 KDa corresponding to a γ-chain dimer and a high molecular weight band appears at the top of the gel corresponding to α-chain polymers. In the absence of calcium, lanes 3, 5 and 7 show the three characteristic fibrinogen chain: α chain (64 kDa), β chain (56 kDa), and γ chain (47 kDa). These results show that the fibrinogen is functional and in the presence calcium ions a stable (cross-linked) clot was formed meanwhile, in the absence of calcium, unstable clots were formed that dissociate in the presence of urea.
Tensile Strength
To evaluate the adhesive properties of the product, the tensile strength of clots formed in vitro by drip delivery were measured. Two pieces of synthetic fibers were sealed with 300 µl of Fibrin Sealant Grifols and then submitted to a constant rate of axial movement. The mean tensile strength was 21 kPa for all batches indicating that the clot formed was able to sustain a load of 21 kPa before failure demonstrating the cohesive force of the sealant. Additionally, no significant differences were seen between different batches (p = 0.936 (by non-parametric one-way analysis of variance (Kruskal-Wallis test) Minitab Statistical Software, State College, PA, USA).
Confocal Microscopy
The clot structure and its properties are greatly influenced by the presence of activated platelets and erythrocytes that form a natural and very active environment for clot formation. Moreover, fibrin formation is also affected by external factors such as ionic strength, pH, and exogenous substances. The evaluation of the in vitro clot structure is a good indicator that the composition of the fibrin sealant components does not affect the ability of the product to form a fibrin networks with a regular distribution. Confocal microscopy allows for analysis of clot structure and does not require fixation, thereby limiting sample preparation artifacts (Figure 5).

Confocal microscopy of the clot formed by application of Fibrin Sealant Grifols using the drip mode.
To assess the structure of clots formed after delivery by drip application, the following measures were evaluated: fiber diameter, fiber branches, density of fibers and size of pores. Fiber diameter was 353 ± 9 nm. The number of branching fibers was 19.1 ± 0.53 per µm3. Fiber density was 23.3 ± 0.9 mm3 and the pore size was 0.6 ± 0.2 mm3.
Viscosity
The dynamic viscosity of the two components of fibrin sealant was measured at different temperatures, (Figure 6). The viscosity of the fibrinogen component ranged from 65.23 to 17.23 mPa•s over the temperature range of 5 °C-37 °C. The dynamic viscosity of the thrombin component ranged from 1.55 to 0.71 mPa•s over the same temperature range. The relatively high viscosity of the fibrinogen component is due to the high protein concentration. The viscosity is low even at 5 °C which allows the application of the product as a liquid. This will provide ease of application of the product using either the drip tip or the ASA over a wide temperature range.

Dynamic viscosity (mPa•s) for the fibrinogen and thrombin components of Fibrin Sealant Grifols measured over the range of 5 °C-37 °C.
Clotting Time
When the ASA was used to deliver the fibrin sealant by spray application, the clotting time was nearly instantaneous. When the ASA tip was removed, and the drip mode was used to deliver the fibrin sealant, clotting time was 5.0 ± 1.0 s (range = 4-6 s; n = 4).
Mixing Test
To evaluate whether the mixing of fibrinogen and thrombin can be achieved at a1:1 (v/v) proportion, different proportion of dyes was added to the product. Clots formed from fibrin sealant with varied proportions of yellow (thrombin component) and blue dyes (fibrinogen component) provided a similar range of colors as those seen with the aqueous solutions (see Table 1 and Figure 7). Visual examination of the clots formed by delivery of the fibrin sealant produced a color range similar to the color seen with the aqueous solutions. This suggested that a 1:1 mixture of the fibrin sealant solutions was delivered by the device with either the drip tip or the ASA.

Comparison of aqueous solutions (A) with Fibrin Sealant Grifols clots formed after delivery with the drip tip (D) or the airless spray accessory (S).
In addition, absorbance was measured on the 1:0, 1:1, and 0:1 dye ratios for the aqueous solutions and clots formed after fibrin sealant was delivered by either the drip tip or the ASA. These data are shown in Table 4.
Absorbance Readings for Aqueous Solutions, Fibrin Sealant Grifols Clots Colored with Blue (Patent Blue) and Yellow (Quinoline Yellow) Dyes. Ratios of dye Concentration in the Aqueous Solution and the Fibrin Sealant Clots Were 1:0 (Blue), 1:1 and 0:1 (Yellow). in the Fibrin Sealant Tests, Blue Dye Was Added to the Fibrinogen Component Only and Yellow Dye Was Added to the Thrombin Component Only.

Opacity
The opacity of the Fibrin Sealant Grifols was determined. As shown in Figure 8, the percent transmittance had a mean value 73.6 and allowed observation through the fibrin sealant to the surface below the well containing the applied product. This clot transparency can be useful to evaluate in vivo if hemostasis has been achieved at the application site.

The opacity of fibrin sealant clots was assessed by measuring transmittance through clots formed in polystyrene culture plates. A. Shows the appearance and visual clarity of clots in three microplate wells. Part B shows the mean transmittance ± the standard deviation.
Discussion
Fibrin sealants have been shown to be useful in a variety of surgical settings.2,3 All marketed fibrin sealants contain two major components (thrombin and concentrated fibrinogen) which replicate and augment the final stage of the coagulation cascade, the cleavage of fibrinogen into fibrin by the action of thrombin, when the two components are mixed. Taking this into account, it can be considered that all the fibrin sealants have a similar mode of action and metabolism.
Commercially available fibrin sealants can be differentiated in their composition mainly by fibrinogen and thrombin concentrations. Besides fibrinogen and thrombin, some products may additionally include FXIII and fibrinolysis inhibitors.
The normal plasma concentration of fibrinogen is 2-5 mg/mL; however, it is present at a higher concentration in marketed fibrin sealant. Additionally, fibrin sealants contain human or bovine thrombin at a concentration that varies from 250-1200 IU/mL. The ratio thrombin (IU/mL)/fibrinogen (mg/mL) can determine the clotting time. Fibrin Sealant Grifols with a ratio of 6 IU thrombin/mg fibrinogen has demonstrated efficacy in clinical studies in cardiovascular, liver, and soft tissue surgeries.17–20 If compared with other marketed fibrin sealants (Table 5). The ratio of thrombin/fibrinogen for Fibrin Sealant Grifols is in the range (3-15 IU thrombin/mg fibrinogen) of other marketed fibrin sealants.
A Comparison of the Component Ratios of Marketed Fibrin Sealants.

The results presented in this paper have shown that the manufacturing process results in a product that contains only the relevant plasma proteins and reduces most contaminating proteins to levels below the limits of detection. The elimination of other plasma proteins results in a purified product with predictable efficacy and minimal adverse effects.17–20
One of the few plasma proteins remaining after the purification process for the fibrinogen component is Factor XIII. There has been disagreement in the literature regarding whether supraphysiological levels of Factor XIII are necessary for a fully efficacious fibrin sealant product.25,26 Clinical studies on this product have demonstrated clinical efficacy without supraphysiological levels of Factor XIII.15–18
Similar discussions on the role of fibronectin in fibrin sealant can be found in the literature 27 but the clinical efficacy of high purity fibrin sealants like FS Grifols does not confirm this hypothesis. Moreover, the in vitro assessment of fibrin chains showed that with small levels of Factor XIII, the fibrinogen component of Fibrin Sealant Grifols was able to form cross-linked clots.
Plasma-derived fibrin sealant products, such as the one described herein, have an advantage over synthetic surgical adhesives in being degradable by physiological processes leaving no residual foreign material in the body.
Adverse events have been observed with the use of fibrin sealant products. Some products contain non-human derived components, eg, aprotinin28–30 that present a risk for allergic reactions up to and including anaphylaxis. 31 The fibrin sealant characterized in these studies is derived solely from human plasma. The risk of allergic reactions to non-human components is therefore eliminated. Moreover, the high purity of both components reduces possible allergic-type hypersensitivity reactions to accompanying plasma proteins.
Application of Fibrin Sealant Grifols with the spray device resulted in nearly instantaneous clot formation while administration with the drip tip resulted in clot formation in approximately 5 s. The faster clotting time with spray application is due to better mixing by the static mixing element in the ASA tip.
The factors affecting fibrin polymerization and clot structure are numerous. 32 Increased concentrations of fibrinogen and thrombin have been shown to produce more fibers that are longer and thinner than those formed at lower concentrations.32,33 The supraphysiological concentrations of fibrinogen and thrombin in commercial fibrin sealants has been shown to produce clots with more numerous, thinner fibers. 34
Although difficult to compare with other studies similar bonding properties (around 30 kPa) for commercially available fibrin glue has been described. 35 The tensile strength of clots formed by administration of fibrin sealants has been shown to be comparable to physiological clots due to the high concentration of fibrinogen. 34 Furthermore, high degree of clot transparency provides better visualization in the surgical field.
In addition, the potential for an air or gas embolism has been reported when a fibrin sealant product is administered using compressed air or gas.36–38 Fibrin Sealant Grifols is provided with an ASA. This allows the product to be sprayed without the use of a compressed gas propellant when the surgical use necessitates spray application. In the current study, the ASA was shown to provide a homogenous 1:1 mixture of the fibrin sealant components that clotted immediately forming a rapid, adherent, and durable clot at the application site. The ASA provided with Fibrin Sealant Grifols is removable and thereby allows administration via drip application. Drip application also provided homogenous 1:1 administration of the fibrinogen and thrombin components.
In conclusion, the fibrin sealant product described in this paper is wholly derived from human plasma with multiple safeguards to disrupt or remove potential infectious agents. The studies reported in this paper show that this product contains highly purified fibrinogen and thrombin effectively delivered in a 1:1 ratio by an ASA or drip tip. In addition, the highly purified nature of the components of this product makes it a safe and useful product with many useful surgical applications.17–20
Footnotes
Abbreviations
Acknowledgments
Michael K James, PhD (Grifols) is acknowledged for medical writing and Jordi Bozzo, PhD, CMPP (Grifols) is acknowledged for editorial support in the preparation of this manuscript. Ashley DeAnglis, PhD (Ethicon), Salim Ghodbane, PhD (Ethicon), Sridevi Dhanaraj, PhD (Ethicon), and are acknowledged for the participation in the opacity test. Ashley DeAnglis, PhD (Ethicon) is also acknowledged for the technical support in the revision of the paper.
Author Contribution(s)
Availability of Data and Materials
The data underlying these studies are available from the corresponding author on reasonable request.
Competing Interests
All authors (IR, LL, MM, PM, ML, NM, and SG) are full-time employees of Grifols, the manufacturer of Fibrin Sealant Grifols.
