Abstract
Background
Immune thrombocytopenia is an immune-mediated disorder with heterogenous presentation. It shows association with other autoimmune disorders with varying frequency.
Objective
The objective of this study was to establish the frequency of antinuclear antibody (ANA) in our population and to evaluate its association with severity of clinical presentation, disease remission and association with other autoimmune diseases.
Design
Cross-sectional study.
Methods
This was a descriptive cross-sectional study that included 160 patients with immune thrombocytopenia purpura (ITP) presented at National Institute of Blood Disease and Bone Marrow Transplantation from February 2021 to July 2022. Patients were screened for ANA and other causes of secondary ITP. ANA-positive patients were compared with ANA-negative patients in terms of bleeding manifestations and platelet counts at initial presentation, association with other autoimmune diseases and response to first-line treatment.
Results
In our study 43 (26.9%) patients were ANA positive and 117 (73.1%) were negative for ANA. The initial platelet counts, and bleeding symptoms were not found to be significantly different among the 2 groups. And 79.1% of the ANA-positive patients had other autoimmune markers as well compared to 20.9% ANA-negative patients, showing a significant association (P<.001). Response to treatment with steroid ± azathioprine assessed at 3 and 6 months were similar in both the groups.
Conclusion
Our results demonstrate that ANA positivity is not associated with severity of presenting symptoms, initial platelet counts, or response to treatment. However, the presence of ANA is associated with increased risk of other autoimmune diseases in patients with ITP.
Introduction
Immune thrombocytopenia purpura (ITP) is a bleeding diathesis affecting adult population of 1.6 to 3.9 per 100,000 patient-years, with slightly higher rates seen with increasing age and among women. 1 The severity of bleeding symptoms and platelet counts at presentation show marked diversity among patients with ITP.2,3 Multiple factors are involved in the pathogenesis of ITP involving both T–cell-mediated and B-cell-mediated immune mechanisms. 4 Conventionally, ITP was believed to be caused by immune destruction of platelets and megakaryocytes 5 by autoreactive antibodies. Recent studies have showed that hyper response of dendritic cells 6 as well as deficiency of T helper cells are implicated in the increased break down of platelets by CD8+ T cells 7 along with innate immunity. 8 This immune-mediated destruction of platelets in ITP may present as a primary disorder or secondary to an underlying disease.9,10
Secondary causes of ITP include autoimmune conditions, lymphoproliferative diseases, or chronic infections, for example, Helicobacter pylori, human immunodeficiency virus (HIV), or hepatitis C virus (HCV). 11 ITP is also reported to be associated with COVID-19 infection12,13 and its vaccination. 14 When ITP is associated with autoimmune diseases like systemic lupus erythematosus (SLE), Sjogren’s syndrome, antiphospholipid syndrome, immune thyroid disease, or Evans syndrome, it presents with distinct characteristics such as positivity for antinuclear antibodies (ANA) and other autoimmune markers. 15
High incidence of autoimmune markers has been reported in previous studies suggesting that ITP involves immune dysregulation beyond platelet destruction. 16 The clinical severity and disease response have also been postulated to be associated at some level to the presence of ANA among these patients. 17 Poor response to steroid therapy among ANA-positive patients with ITP has been observed by other studies. 18 Therefore, following study was aimed to determine the frequency of ANA positivity in adult patients with ITP and its relationship with severity of clinical presentation, disease remission with first-line treatment and association with other autoimmune diseases.
Materials and Methods
This was a cross-sectional study conducted at National Institute of Blood Disease and Bone Marrow Transplantation (NIBD) from February 2021 to July 2022. All patients diagnosed as ITP (on the basis of clinical history, general physical examination, and complete blood count (CBC) parameters), aged ≥ 12 years, who were tested for ANA, were included in the study. Nonconsenting patients and patients with missing data or alternate causes of thrombocytopenia (such as bone marrow failure, infiltration, or fibrosis; hematological or nonhematological malignancies or chronic liver disease) were excluded from this study. Ethical approval of the study was given by Institutional Review Board (IRB) of the NIBD, Pakistan Ethics Committee, IRB no. NIBD/IRB-228/17-2021.
Demographic data such as age, gender, and comorbid conditions were recorded at baseline. Detailed clinical history, physical examination, and laboratory parameters such as CBC, ANA, H. pylori testing and screening for hepatitis B surface antigen (HBsAg), anti-HCV and anti-HIV were noted at baseline. Titers of 1:80 or higher were taken as positive for ANA test. Additionally, ANA-positive patients were screened for extensive autoimmune profile including anti–double-stranded DNA (dsDNA), ANA profile, and ENA profile. Patients were followed for 6 months; bleeding symptoms, CBC, and treatment response were assessed at 3 and 6 months’ time points. Based on the presence of ANA, patients were segregated into 2 groups; ANA-positive patients were compared with ANA-negative patients for clinical characteristics and response to treatment at the designated time points.
Diagnosis of ITP was established in patients presenting with isolated thrombocytopenia with platelet count <100,000 × 109/L in the absence of any other causative etiology. 19 Primary ITP is diagnosed after exclusion of all other causes of immune-mediated thrombocytopenia. 19 Secondary ITP included all forms of immune-mediated thrombocytopenias except primary ITP, including thrombocytopenias that are associated with an underlying disease or drug exposure. 19 Response to treatment was assessed using the definitions of American Society of Hematology practice guidelines, 20 that are as follows: complete response—a platelet count ≥ 100 × 109/L measured on 2 occasions > 7 days apart and the absence of bleeding. Response— a platelet count ≥ 30 × 109/L or greater than 2-fold increase in platelet count from baseline measured on 2 occasions > 7 days apart in the absence of bleeding. No response—a platelet count < 30 × 109/L or less than 2-fold increase in platelet count from baseline or the presence of bleeding symptoms.
Statistical Analysis
Normality of analytical variables was statistically significant on the Shapiro-Wilk test. On the basis of normality, nonparametric statistical analysis was performed. Descriptive analysis, that is, qualitative, categorical variables at ordinal scale included frequency in the percentage of age, gender, disease nature, type of disease, ANA profile, viral markers, ultrasound findings, and bleeding symptoms. Chi-square was applied for ANA profile with symptoms and treatment responses at different time points. Median (range) (the median value and the difference in the highest and lowest values) was applied on quantitative, continuous variables on the nominal scale, including age and laboratory parameters, that is, hemoglobin, total leukocyte count, and platelet count. Wilcoxon signed-rank test was applied to compare the baseline, 3 and 6 months platelet counts. Statistical analysis was done by using SPSS version 23.0. In line with this, the P-value ≤.05 was statistically significant.
Results
A total of 160 diagnosed cases of ITP were recruited in the study, comprising of 47(29.4%) males and 113 (70.6%) females with median age of 37.5 years. The median value of laboratory characteristics with range (the difference in the highest and lowest value) at baseline includes hemoglobin 12.1 (11.3) g/dL, total leukocyte counts 8.52 (20.0) × 109/L and platelet count 17(98.0) × 109/L. Whereas total of 85 (53.1%) patients were characterized as severe ITP with platelet count <30 × 109/L. The median (range) platelet counts at 3 months, that is, 66.5(690) were significantly higher from baseline median (range) platelet count, 17(98.0). However primary ITP was identified in 90 (56.3%) patients while 70 (43.8%) patients had secondary ITP.
Patients were divided into 2 groups based on the results of ANA test, that is, ANA positive 43 (26.9%) and ANA negative 117 (73.1%). And 30.1% females and 19.1% males were tested positive for ANA. The frequency of autoimmune markers is illustrated in Table 1. A significant association (P-value < .001) of both groups was observed with the expression of other markers of autoimmunity. Among the ANA-positive patients, 34 (79.1%) showed presence of other autoimmune markers including ANA, antimitochondrial antibodies, antismooth muscle antibody, antigastric parietal cell antibodies, anti-dsDNA, anti-autoantibodies to histidyl tRNA synthetase (anti-Jo1), anti-Sjögren's syndrome type A autoantibodies (anti-Ro/SSA), anti-Sjögren's syndrome type B antibodies, and extractable nuclear antigen antibodies, while 9 (20.9%) patients were negative. Median (range) platelet counts at baseline in the ANA-positive group was 20 (94) while 18 (98) in the ANA-negative group. No significant association (P-value >.05) in the median platelet counts during the study follow ups was observed between the 2 groups, as depicted in Figure 1. Secondary causes of ITP other than markers of autoimmunity were also screened. H. pylori was identified as a potential cause in 36 (22.5%) patients. Anti-HCV, anti-HIV, and HBsAg were positive in 11 (6.9%), 1 (0.6%), and 2 (1.3%) patients, respectively.

Trend of median platelet counts during follow ups in ANA-positive and ANA-negative groups. Abbreviation: ANA, antinuclear antibodies.
Frequency of Markers of Autoimmunity.

At initial presentation, 37 (23.12%) patients were asymptomatic, while 123 (76.8%) patients presented with bleeding manifestations. Baseline bleeding symptoms of the 2 groups are presented in Figure 2. Bleeding manifestations at baseline were more frequently observed in the ANA-negative group. Bruising was the most common presenting symptom observed in 17 (9.5%) ANA-positive patients and 49 (41.9%) ANA-negative patients. Gastrointestinal (GI) bleeding and hematuria were only observed in ANA-negative group. Comparison of bleeding symptoms between the 2 groups at different time points is shown in Table 2 with statistically significant difference observed in both groups for GI bleeding at baseline and 3 months follow up.

Bleeding symptoms at baseline in ANA-positive and ANA-negative groups. Abbreviation: ANA, antinuclear antibodies.
Association of Bleeding Symptoms With ANA Profile.

Note: P-value≤.05 is found to be significant.
Abbreviations: ANA, antinuclear antibodies; GI, gastrointestinal.
Response to treatment with steroid ± azathioprine was assessed at 3 and 6 months as presented in Table 3. Overall response rate (response + complete response) at 3 months was 77.5% in the ANA-positive group and 77.6% in the ANA-negative group. While at 6 months the overall response rates were 80% and 66.2% in the ANA-positive and ANA-negative groups, respectively. The difference in response rates was not statistically significant between the 2 groups at both time points.
Response to Treatment at 3 and 6 Months in ANA-Positive and ANA-Negative Groups.
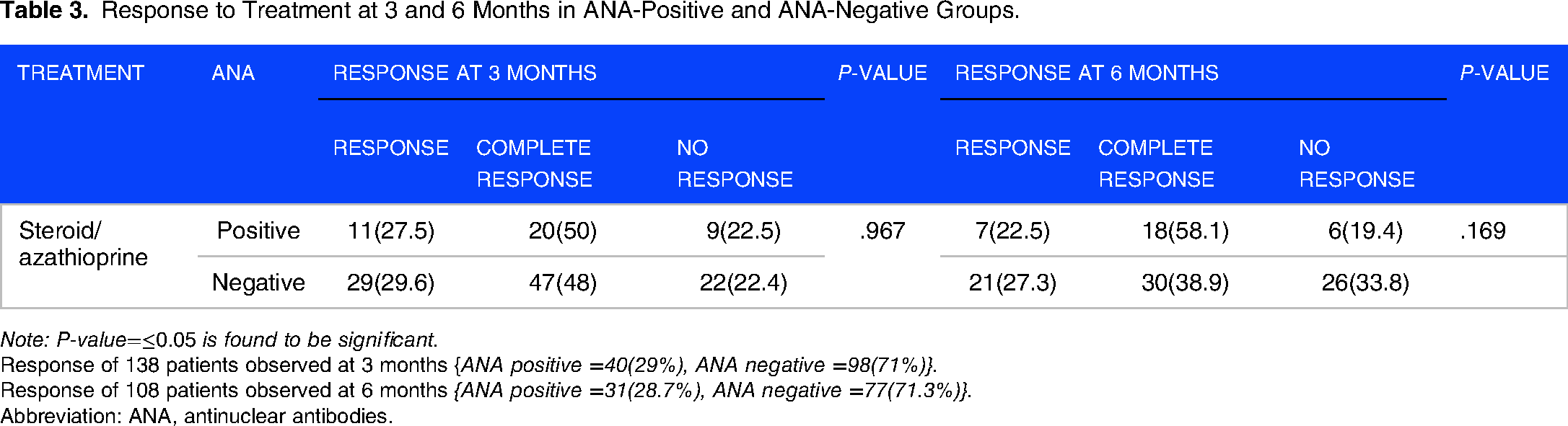
Note: P-value=≤0.05 is found to be significant.
Response of 138 patients observed at 3 months {ANA positive =40(29%), ANA negative =98(71%)}.
Response of 108 patients observed at 6 months {ANA positive =31(28.7%), ANA negative =77(71.3%)}.
Abbreviation: ANA, antinuclear antibodies.
Discussion
ITP is a complex immune-mediated disorder with multiple factors playing role in its pathophysiology, 21 posing as possible predictors of disease characteristics.22,23 This study evaluated the frequency of ANA in adult patients with ITP and the association of ANA positivity with clinical severity and response to first-line treatment.
In our study, 26.9% (n = 43) patients tested positive for ANA. Previous data regarding the prevalence of ANA in patients with ITP is heterogenous.3,16,24,25 Hollenhorst et al, reported a higher rate of 65% ANA positivity compared with 13% to 30% reported in previous studies 16 and demonstrated an increased risk of thrombosis associated with positive titers of ANA. No thrombosis was reported in our study subjects, which is a peculiar finding as recent studies have demonstrated an increased incidence of thrombosis among patients with ITP. 26 However, the incidence of thrombosis was not a primary endpoint of our study hence a further study may be aimed to establish the association of thrombosis with ITP.
Although higher percentage of ANA positivity has been reported among healthy participants worldwide, data from previous Pakistani studies show incidence rates of 1% 27 and 0.4%. 28 Compared to this low rate of ANA positivity among healthy individuals, the rate of 26.9% among our patients with ITP represents a significant finding possibly related to disease pathophysiology. Recent studies have shown that although ANA positivity itself does not classify, a case as secondary ITP, it confers some distinct characteristics to the disease pathobiology. 29 Yuan Liu et al, demonstrated an increased risk of progression to connective tissue diseases especially systemic lupus erythematosus (SLE) in patients with primary ITP who tested positive for ANA. 30 Our study has postulated a positive association between ANA positivity and prevalence of other autoimmune diseases, as 79.1% of ANA-positive patients additionally reported one or more autoimmune markers. However, anti-dsDNA positivity (3.8%) and diagnosis of SLE were not a remarkable finding in this study. The multicenter Carmen registry3,31 evaluated the significance of ANA in primary ITP and failed to establish an association of ANA with clinical presentation, response to treatment, or development of connective tissue diseases. 31
The baseline platelet count was not significantly different among the 2 groups. While bleeding symptoms were more frequently seen in the ANA-negative group, the difference was not statistically significant. This contrasts with previously reported lower initial platelet counts in ANA-positive group.17,32 Similar clinical characteristics in these 2 groups of primary ITP have also been reported by prior investigators, 15 while some found ANA positivity to be associated with fewer bleeding symptoms. 33 The heterogeneity of clinical and laboratory characteristics in our study as well as these previous studies might be explained by the geographical and ethnic variability of the disease.
No significant difference in response to first-line treatment with steroids ± azathioprine was seen in our study groups, similar to the findings of the Carmen registry 31 but in contrast to the results of some other studies reporting better response rates for ANA-negative group.17,32 Our findings suggest that short-term treatment response in ITP is not altered by ANA positivity.
Presence of ANA has also been implicated in the response to treatment with eltrombopag 34 and rituximab. 35 Our study only encompassed the treatment duration of first 6 months from diagnosis, thus we could not annotate the durability of response to treatment beyond this time period. A longer duration of follow up will be required to elucidate long-term response and would enable us to include the response to second-line treatment which is usually added later in the course of the disease.
Conclusion
In conclusion, our results demonstrate that ANA positivity was not associated with severity of presenting symptoms, initial platelet counts or response to treatment. However, presence of ANA in ITP indicates autoimmune phenomenon beyond destruction of platelets and warrants further investigation of autoimmune diseases. Further research is needed to establish its effect on the severity of disease, long-term treatment outcomes, and association with other autoimmune diseases.
Footnotes
Ethics Approval and Consent to Participate
This study was approved by the Institutional Review Board (IRB) of the National Institute of Blood Diseases (NIBD), Pakistan Ethics Committee, IRB no. NIBD/IRB-228/17-2021 and written informed consent was taken from the study participants.
Author contribution(s)
Acknowledgements
Authors acknowledge the hospital staff and patients for their cooperation in collecting valuable data for the study.
Availability of Data and Materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
