Abstract
BACKGROUND
Fibrinogen is an important component of hemostasis and clot formation. Replacement therapy with a human Fibrinogen concentrate is the preferred therapeutic option for patients with congenital or acquired Fibrinogen insufficiency. To make the Fibrinogen available for treatment of such patients, Fibrogen-I®, a lyophilized product purified from pooled human plasma was developed and extensively characterized. The product quality attributes were assessed by different biochemical, functional and structural analytical methods.
MATERIAL AND METHODS
Functional activity of Fibrinogen in the Fibrogen-I® was demonstrated by determination of its clottable protein activity. Purity profiling of Fibrogen-I® was evaluated by SE-HPLC method. Activation and fibrinolysis markers like D-dimer, plasminogen was determined by automated coagulometer and fibrinopeptide A activity by ELISA. Far UV CD analysis was also performed by CD Spectrometer.
RESULTS
The product exhibited high purity 89.7% by SE-HPLC. Activation and Fibrinolysis marker proteins in the drug product were negligible. The high purity and integrity of Fibrogen-I® is underlined by the ratio of Fibrinogen activity by Clauss/clottable protein with mean calculated value of 0.9 which is in close proximity with theoretical value 1 indicating fully native Fibrinogen. The results of far-UV CD spectroscopy revealed that Fibrogen-I® exists mostly as a β-sheet secondary structure, which is in accordance with the three-dimensional structure of human Fibrinogen. Downscaling experiments for the two dedicated orthogonal pathogen inactivation steps, ie solvent detergent treatment and dry heat treatment, exhibited pathogen safety.
DISCUSSION
Fibrogen-I® exhibited specific functional activity, desirable quality attributes, pathogen safety and the ability to be made available to patients requiring fast and effective Fibrinogen replacement.
Introduction
Fibrinogen, a 340 Kda hexameric plasma glycoprotein produced in the liver, that is composed of two sets of three polypeptide chains—Aα, Bβ, and γ—joined by a disulfide bridge and is a significant structural component of the coagulation pathway. It creates a matrix and mesh network, both of which are necessary for clot strength. 1
Fibrinogen plays an important role in hemostasis by strengthening the platelet plug formed by fibrin polymer as a result of cleavage of Fibrinopeptide A and B by thrombin. Platelet aggregation is supported by Fibrinogen's interaction with platelet surface receptor molecules. The cross-linking of fibrin monomer takes place by activated factor XIII (FXIII) to create the clot's fibrin network. Thus, a fibrin stable plug is formed which traps red blood cells, platelets and bleeding is stopped from the site of injury.2,3
Human Fibrinogen is made up of two D domains that are joined by a centre E domain. Each D domain is made up of three polypeptide chains (α, β, and γ), which form a coiled structure. 4 A series of disulfide bonds connect the N-terminus of these domains to the core E domain. 4 Fibrinogen concentration in the blood range from 1.5 to 4.5 g/L when assessed as clottable protein and from 8.8 to 9.8 mmol/L when measured as total protein. 5 Low plasma Fibrinogen concentrations are associated with an increased risk of bleeding, due to impaired primary haemostasis (prolonged skin bleeding time in-vivo, decreased platelet aggregability in-vitro) as well as impaired secondary haemostasis. 5
Congenital Fibrinogen deficiencies can be either quantitative (afibrinogenemia and hypofibrinogenemia) or qualitative (dysfibrinogenemia). 6 Afibrinogenemia affects about one in every million people. 7 Clinical signs and symptoms differ depending on the type of insufficiency. In one Iranian study, 85% of patients with afibrinogenemia (total lack of Fibrinogen) had umbilical cord bleeding, 72% had muscle hematomas, and 54% had hemarthrosis. 8 Epistaxis, menorrhagia, and oral cavity bleeding were reported by ≥70% of patients in this study; central nervous system (CNS) bleeds occurred in 10% of patients. 8 Over half of people with dysfibrinogenemia have no clinical complications; however, bleeding affects around 25% of patients, and thrombosis (mainly deep vein thrombosis, thrombophlebitis, and pulmonary embolism) affects about 20% of patients - commonly at a young age. 9 Patients with hypofibrinogenemia (characterized by Fibrinogen levels <1.5 g/L) are usually asymptomatic, although they have a tendency to bleed when exposed to trauma or if they have a second associated hemostatic abnormality. 6 Hypofibrinogenemia is also associated with bleeding disorders and thrombotic complications.10,11
Antifibrinolytic amino acids (eg, ε-aminocaproic acid, tranexamic acid) may be used alone or in combination with replacement therapy as a treatment option. 9 50 to 60 mg/kg every 4 to 6 h for ε -aminocaproic acid and 25 mg/kg every 6 to 8 h for tranexamic acid are recommended doses to control bleeding in such patients. 12 Estrogen–progestogen preparations may also help to reduce menstrual blood loss in women with iron-deficiency anemia due to menorrhagia. 12 In congenital Fibrinogen diseases, fibrinogen replacement treatment is useful in treating bleeding episodes and also in secondary prophylaxis in patients with fibrinogen deficiency. Fresh frozen plasma (FFP), cryoprecipitate and fibrinogen concentrates are all viable replacement options. Due to its enhanced safety, early onset of effect, dosing flexibility, small infusion volume, and convenience of administration compared to alternative options, fibrinogen concentrates are widely considered the therapy of choice in congenital Fibrinogen insufficiency.6,12–14
Fibrinogen concentrates can also be used to treat trauma-related coagulopathy, in which rapid depletion of circulating coagulation factors due to consumption and blood loss is a major cause of death.4,15 In this context, the role of Fibrinogen concentrate administered as immediate treatment in trauma has been investigated recently.16,17 Goal-directed coagulation treatment employing thromboelastometry-guided administration of Fibrinogen concentrate coupled with prothrombin complex concentrate was retrospectively reviewed in a study undertaken by Schochl et al. which included 128 bleeding trauma patients. 18 The observed fatality rate in these patients was lower than that predicted by the Trauma Injury Severity Score (TRISS) and the Revised Injury Severity Classification (RISC) scores. 18
Fibrinogen concentrate has proved to be life-saving drug in treatment of Postpartum hemorrhage (PPH) also. 19 A study found that prescribing 1 to 4 g of fibrinogen reduces the need for blood transfusions considerably. 20 Fibrinogen administration helps reducing the need of blood transfusion. 21
To deal with such life threatening conditions related to the deficiency of Fibrinogen, we have developed a Fibrinogen concentrate, Fibrogen-I® from fresh frozen plasma involving a series of precipitation and chromatographic steps. Safety of the Fibrinogen concentrate was ensured by incorporating two orthogonal virus clearance steps. Fibrogen-I® was characterized with different biochemical and functional analysis wherein it was compared with market comparator, a product derived from human plasma.
Materials and Methods
Materials
Freshly frozen recovered plasma was obtained in polyvinyl bags from Indian blood banks that had been approved by the Drug Controller General of India (DCGI) and had been registered with the Government of India's National Aids Control Organization. Antibody testing for HIV-I, HIV-II, and HCV was done with Biorad and a Difpro HBV kit. Hepatitis-B virus (HBV), Hepatitis-C virus (HCV), and Human Immunodeficiency Virus-I (HIV- I) nucleic acid testing of plasma was performed using standard USFDA approved Roche, USA kits.
Clauss method was used to measure fibrinogen activity with a Fibrinogen C kit from Instrumentation Laboratory, USA. Reagents were of analytical grade for clottable Fibrinogen and total protein estimation by Kjeldahl.
Biorad supplied a standard protein molecular weight marker for SDS-PAGE (USA).
Fibrinopeptide A levels were determined using a competitive Enzyme Linked Immunosorbent Assay using Zymutest kit, France.
D-Dimer, Factor XIII activity, von Willebrand factor antigen, plasminogen activity were measured using respective D-Dimer, Factor XIII antigen,von Willebrand factor antigen and plasminogen kits supplied from Instrumentation Laboratory, USA.
High performance Size-exclusion column was from TOSOH Biosciences, Japan. Other chemicals required for preparing mobile phase-phosphate buffer saline were procured from Merck (Darmstadt, Germany).
Structural analysis was performed using J-815 CD Spectrometer (Jasco), USA.
A GLP-certified qualified laboratory was selected for the virus validation study.
Methods
Antibody Testing
Every unit of plasma used to purify human fibrinogen was screened for antibodies to HIV-I, HIV-II, HBV and HCV, and those that tested negative for antibodies were then examined for viral nucleic acids by Polymerase Chain Reaction testing. Positive plasma units were discarded according to established procedures.
Nucleic Acid Amplification Testing
Each unit of Indian plasma was screened for HBV, HIV-I and HCV, and all units that were negative for the viruses were pooled and utilised to purify human fibrinogen. Nucleic acid amplification testing was performed in addition to the certification offered by other blood banks.
Manufacturing of Human Fibrinogen Concentrate (fibrogen-I®)
Manufacturing Process
Fibrinogen concentrate (Fibrogen-I®) is a sterile, heat-treated, lyophilized Fibrinogen (coagulation factor I) powder manufactured from pooled human plasma. Pooled human plasma was centrifuged to obtain cryoprecipitate which was dissolved in water for injection (WFI). Aluminum hydroxide gel was added to dissolved cryoprecipitate to adsorb prothrombin complex proteins. Clarified dissolved cryoprecipitate obtained after centrifugation was taken for glycine precipitation to precipitate Fibrinogen. The precipitated Fibrinogen after dissolution was treated with solvent detergent reagents for 6 h in order to inactivate the enveloped viruses present, if any. Clarified S/D treated material obtained after depth filtration was loaded onto anion exchange chromatography. Chromatography elute was then diafiltered followed by subsequent sterile filtration. This bulk is then formulated and lyophilized to obtain a sterile freeze-dried powder. For virus inactivation, the lyophilized product was treated to a 30-min terminal dry heat treatment at 100 °C. Pathogen safety is ensured by two orthogonal virus clearance steps viz. S/D treatment and dry heat treatment in the manufacturing process.
The product was filled 1 g per vial and before administration needs to be reconstituted with 50 ml of water for injection, yielding a 20 mg/ml fibrinogen solution. The Fibrogen-I® was characterized with different biochemical and functional analysis, wherein it was compared with market comparator derived from human plasma.
Characterization of Fibrogen-I®
Reconstitution of Lyophilizates and its Stability
The lyophilized Fibrogen-I® was reconstituted in 50 ml of water for injection to achieve a final concentration of 20 mg/ml. Within 10 min of reconstitution, the contents of the vial were totally dissolved. Physicochemical and functional assays were used to analyze the samples.
The reconstitution stability was tested for 24 h at room temperature. At each sampling time-point, the clottable fibrinogen activity was assessed by Kjeldahl method.
Fibrinogen Activity
Fibrinogen activity was determined in terms of clottable protein by Kjeldahl method as per European Pharmacopeia monograph. Fibrinogen activity was also measured according to Clauss method by a kit on an automatic coagulometer (ACL Elite Pro) from Instrumentation Laboratories. The value of fibrinogen in the calibration plasma was obtained using the WHO standard (09/264) which is used for the calibration.
Protein Concentration
Total protein was determined by the Kjeldahl method with market comparator for system suitability using semi-automated instrument from Gerhardt.
SDS-PAGE
Sodium dodecylsulphate–polyacrylamide gel electrophoresis (SDS–PAGE) was performed under reducing conditions using a 7.5 to 10% gradient separating gel. Biorad miniprotean tetra cell assembly was used to run the gel at a constant voltage of 200 volts for approximately 45 min, and the gel was silver stained using an in-house developed process.
Size-Exclusion High Performance Liquid Chromatography for Protein Purity
Chromatographic separation was accomplished on TOSOH column 30 cm × 7.8 mm I.D, particle size 5µ using mobile phase as phosphate buffer saline pH 7.0. Samples were standardised to 1 mg/ml, 100µ l of samples were injected, the flow rate was 0.5 ml/min and the absorbance was measured at 280 nm.
Fibrinopeptide A
Fibrinopeptide A measurement was carried out using Zymutest kit which is a competitive Enzyme Linked Immunosorbent Assay. Calibration is performed using Fibrinopeptide A calibrator provided in the kit. The assay was carried out according to the manufacturer's instructions.
D-Dimer
D-Dimer activity is two-site enzyme immunoassay for specifically measuring the Fibrin degradation products (D-Dimer). The degree of agglutination is evaluated by measuring the decrease in transmitted light at 405 nm caused by aggregates, which is directly proportional to the concentration of D-Dimer in the sample (turbidimetric immunoassay). D-dimer activity was determined using D-Dimer kit as per manufacturer's instructions on plate reader. Quantification of D-Dimer is performed using calibrator provided in the kit.
Plasminogen Activity
The plasminogen chromogenic assay was carried out using a plasminogen kit and an automatic coagulometer (ACL Elite Pro) from Instrumentation Laboratories, according to the manufacturer's instructions. The value of plasminogen mentioned in calibration plasma package insert was used as reference for calibration.
Von Willebrand Factor Antigen
The assay was carried out using (IL) reagents on automatic coagulometer (ACL Elite Pro) from Instrumentation Laboratories, according to the manufacturer's instructions. The activity of vWF: antigen in calibration plasma was evaluated using WHO standard (07/316) for assay calibration.
Activity Analysis of Factor XIII
The F-XIII assay was performed on an automatic coagulometer (ACL Elite Pro) and kit reagents from Instrumentation laboratories. F-XIII qualified against WHO standard (02/206) was used for assay calibration.
Circular Dichroism (CD) Spectroscopy
Far UV-CD spectra were recorded at 25 °C with a scanning speed of 20 nm/min, a response of 4 s, a bandwidth of 1.0 nm, at a wavelength range of 205 to 260 nm on a J-815 CD Spectrometer (Jasco). The data acquired was an average of five scanning cycles to improve signal quality. A blank measurement was taken prior to the analysis to adjust the protein spectra using the buffer system.
Assessment of Virus Safety
To confirm the efficiency of the human Fibrinogen (Fibrogen-I®) manufacturing process to robustly inactivate/remove pathogens, spiking experiments with viruses were conducted. The scaled-down model was used to establish the capacity of the process to inactivate / remove the pathogenic viruses at a GLP certified laboratory. The virus validation investigation mainly used two orthogonal steps: solvent detergent (S/D) and dry heat treatment. The viruses used in the study were chosen in accordance with the ICHQ5A guidelines. 22 The S/D treatment step was evaluated for inactivation of enveloped viruses HIV-1, PRV, and BVDV, whereas both enveloped and non-enveloped viruses, such as HIV-1, PRV, BVDV, HAV and PPV, were investigated for assessing the dry heat treatment step.
To avoid matrix effects, cytotoxicity and interference tests were done to determine the appropriate dilutions. For endpoint titration and large-volume plating, a validated assay system was used.
The Tissue Culture Infectious Dose (TCID50) assay was used to assess virus titres based on endpoint dilution. The large volume plating (LVP) assay was employed to improve the limit of detection. The LVP assay was used to analyse the process samples that were expected to have no or only a few infectious viruses in them. The 95% confidence intervals and log10 reduction factors (LRFs) were calculated using the approach given in European regulatory guidelines. 23
The process intermediate after dissolution of glycine precipitates was spiked with individual viruses at a spike ratio (v/v, virus concentrate: process intermediate) of 10% to test the effectiveness of the S/D treatment step for inactivation of enveloped viruses (HIV-1, PRV, and BVDV). After mixing, a sample was withdrawn (Load). An aliquot was withdrawn and stored next to the process until the end of the process. Then, a sample was withdrawn (Hold). To achieve a final concentration of 0.3% (w/w) tri(n-butyl)phosphate (TNBP) and 1.0% (w/w) Tween 80, S/D reagents were added. After being spiked with virus, the material was incubated for 6 h at 25° ± 3 °C with stirring. Test samples were collected at pre-determined intervals during the process and studied using an endpoint dilution assay to evaluate the kinetics of virus inactivation. Sample collected after 6 h was additionally analyzed by LVP assay. Studies were performed in duplicates to show reproducibility.
According to the guidelines, the pre-lyophilized sample was spiked individually with the viruses viz. human Immunodeficiency Virus type 1 (HIV-1), Pseudorabies Virus (PRV), Bovine Viral Diarrhoea Virus (BVDV), Hepatitis A Virus (HAV), Porcine Parvovirus (PPV) at a virus spike ratio of 10% and tested for the robustness of the dry heat treatment step for their inactivation and/or removal. Load and hold samples were withdrawn. Lyophilization was performed on the pre-lyophilized viral spiked vials, followed by a dry heat treatment step at 100 °C for 30 min. At predetermined time intervals, samples were withdrawn for an end-point titration experiment. Samples after 20 min and 30 min of dry heat treatment were also analyzed by LVP assay for viral inactivation kinetics.
Additionally, virus safety of Fibrogen-I® was also determined by testing the product for antibodies against HIV-I, II, HBV and HCV as well as nucleic acid testing for viral antigens such as HBV, HIV-I and HCV, as required by the D & C act. 24
Results
Purification and Reconstitution Study of Human Fibrinogen (Fibrogen-I®)
The observed yield of Fibrogen-I® purification process was 0.6 vials/l plasma. The reconstitution study was performed for 1 representative batch at ambient temperature (25 ° ± 3 °C). The lyophilized powder dissolved completely with reconstitution time of 10 ± 5 min at ambient temperature (25° ± 3 °C). The reconstituted sample stability was determined by analyzing two replicates of clottable protein at every time–point till 24hrs at ambient temperature. Fibrogen-I® after reconstitution was found to be stable till 12 h exhibiting 86% retained activity at ambient temperature (Figure 1).

Reconstitution study of Fibrogen-I® at ambient temperature.
Results of biochemical and functional characterization of Fibrogen-I® are summarized in Table 1. Results are shown as mean obtained by averaging values from 2 replicates of 3 batches.
Biochemical Characterization of Fibrogen-I® (Results are mean average values from 2 replicates of 3 batches).

Fibrinogen Activity and Accompanying Plasma Proteins
The high purity and integrity of human Fibrinogen product is underlined by the ratio of Fibrinogen activity by Clauss/clottable protein with mean calculated value of 0.9, which is in close proximity with theoretical value 1 same as that in market comparator (1.0) indicating fully native Fibrinogen. Furthermore, the functionality of the protein was proved by clottable protein activity 23.7 mg/ml which was similar to that obtained in market comparator 23.2 mg/ml.
The product contains 89.8 ng/ml of fibrinopeptide A, which is cleaves off the Fibrinogen molecule after thrombin activation. The presence of a small amount of fibrinopeptide A indicates that no unwanted activation occurred during the processing of this product. The relatively low level of plasminogen activity (0.07 IU/ml) further supports this conclusion. D-Dimer was measured at a concentration of 209 ng/ml. Von willebrand factor appeared as high molecular weight protein in final purified preparation of human fibrinogen. Von willebrand factor antigen in purified product was observed to be 0.1 IU/ml which was lesser as compared to that in market comparator (3.0 IU/ml).
Factor XIII plays a critical role in the establishment of a stable clot. The fibrin strands are covalently crosslinked by activated FXIII as a last step in the fibrin polymerization process. This protein is co-purified to some level throughout the production process, with activity of 2.5 IU/ml, which is higher than the market comparator (0.2 IU/ml).
Purity by Size-exclusion Chromatography
High-performance Size-exclusion chromatography was used to determine the purity of Fibrogen-I® (SE-HPLC). Protein samples of 3 batches were analysed in duplicates using SE-HPLC, for heterogeneity in terms of distinct molecular weights, as well as probable denaturation, aggregates or fragmentation of the Fibrinogen molecule.
The chromatogram shows a major peak of Fibrinogen along with one co-eluting peak of high molecular weight species. The chromatographic profile of the market comparator was comparable to the purified human Fibrinogen product of 3 different batches as shown in (Figure 2). Fibrogen-I® is highly purified product as shown by its purity 89.7 ± 1.1% (1 SD) with high molecular weight impurities 10.3 ± 1.1% (1 SD) as compared to market comparator with 81.6% purity and 18.4% high molecular weight impurities.
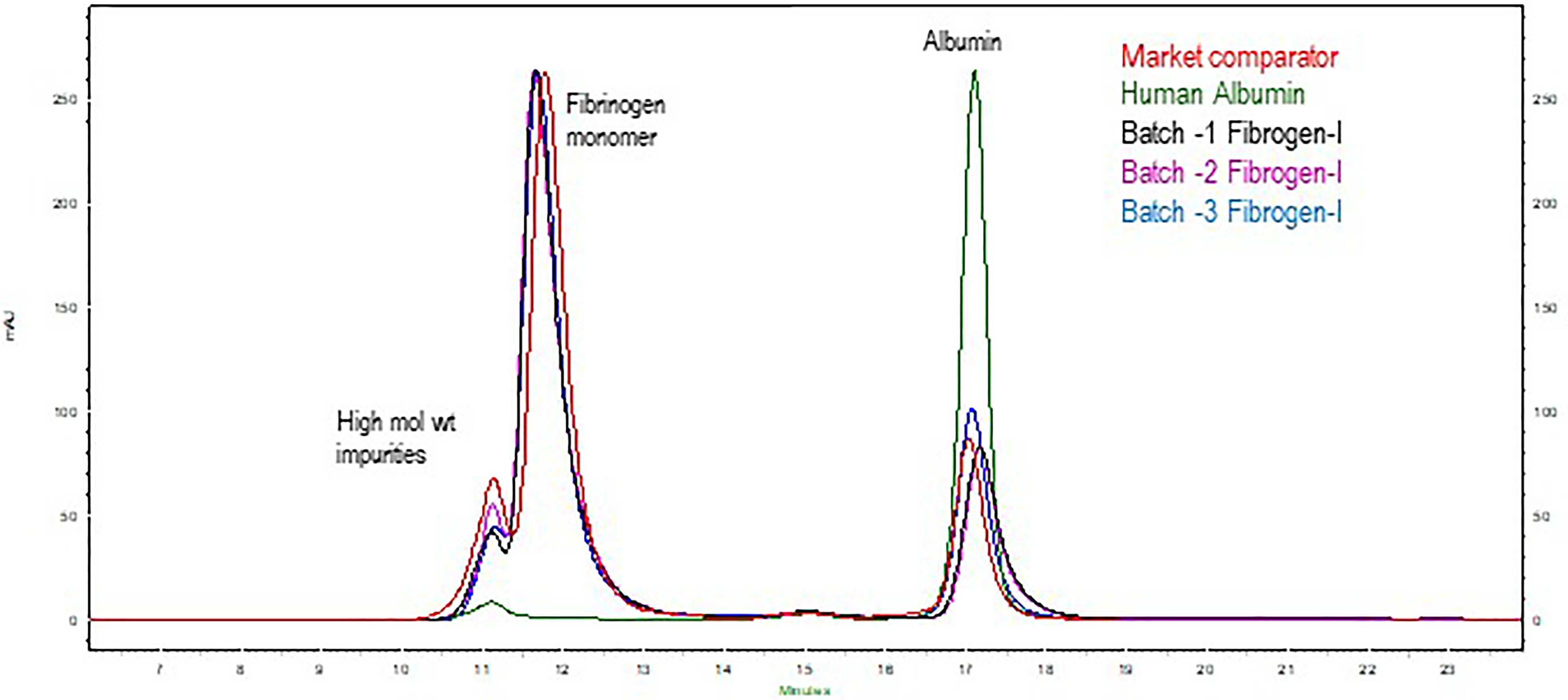
Overlay SE-HPLC chromatograms of Fibrogen-I® batches along with market comparator.
SDS-PAGE
Furthermore, SDS-PAGE analysis was used to evaluate the purity of Fibrogen-I® (Figure 3). The human Fibrinogen revealed a typical band pattern on a silver-stained gel of SDS-PAGE analysis: α-chains at a molecular weight of approximately 63.5 Kda, β-chains at 56KDa, and γ-chains at 47 kDa under reducing conditions. The SDS-PAGE band profile of human Fibrinogen was compared with market comparator. High molecular weight impurities are less as compared to market comparator. This result is in accordance with SE-HPLC data with both exhibiting high molecular weight impurities higher in market comparator than in human Fibrinogen batches.

7.5 to 10% Silver-stained SDS-PAGE profile of Fibrogen-I® under reducing conditions. Lane 1. M.W. Markers KDa (1.2
Far-UV CD Spectroscopy
The spectral analysis of Fibrogen-I® exhibited a similar structure as that of market comparator (Figure 4).

Far UV CD spectra of Fibrogen-I® and market comparator.
Virus Safety
Virus safety is required for the production of any biological product, including human Fibrinogen derived from human plasma. Besides delivering a high-purity plasma-derived concentrate, safety in terms of reducing the risk of viral transmission must also be addressed. The product's safety was assured using nucleic acid testing for viral antigens and ELISA testing for antibodies against specific viral markers. 25 Additionally, virus validation study was performed in the dedicated orthogonal steps in the purification process including S/D treatment for 6 h at ambient temperatures, and dry heat treatment at 100 °C for 30 min indicating that the process is sufficiently robust for inactivation of any unknown viruses that could possibly be present in the plasma. Based on the lipid membrane destroying properties of TnBP (solvent) and Tween-80 (detergent), the S/D treatment used in the manufacturing process is known for its extremely fast inactivation kinetics, robustness, and superiority in terms of inactivating enveloped viruses, with log reduction factors (LRFs) of 4.82 for BVDV, 5.09 for HIV-I, and 5.34 for PRV. (Table 2). After lyophilization, a dry heat treatment step was used as a synergistically effective step for inactivating both enveloped and non-enveloped viruses 22. Dry-heat treatment step resulted in log reduction factors of 5.41 for BVDV, 3.05 for HIV-I, 4.19 for PRV, 5.91 for HAV, and 1.4 for PPV. Within 30 min of the dry-heat treatment HAV, a non-enveloped virus showed a significant virus inactivation (5.91) whereas PPV, a non-enveloped virus, proved resistant to possible virus inactivation. The enveloped viruses BVDV and PRV were found to be susceptible to the treatment. Even non-enveloped viruses that are resistant to solvent/detergent treatment have been reported to be inactivated with this step (Table 2).
Log Reduction Factors for Viruses During Fibrogen-I® Manufacturing.

NA: Not applicable
Note: Each value is the average of 2 runs of 1 batch
In addition to the virus safeguarding steps in the Fibrogen-I® manufacturing process, the overall safety strategy includes selecting qualified donors, testing plasma donations and plasma pools for serological and viral markers (eg HIV, HBV, and HCV) in accordance with current plasma-derived medicinal product guidelines. 23 The fresh frozen plasma used to purify Fibrogen-I® was tested and determined to be free of blood-borne viruses such as HBV, HCV, and HIV.
Discussion
Fibrinogen replacement therapy is very essential in the management of patients with acquired hypofibrinogenemia, afibrinogenemia, excessive bleeding disorders. 6 In the past, patients with similar clinical circumstances were treated with cryoprecipitate, which is a rich source of Fibrinogen, FVIII, von Willebrand factor, Factor XIII, and fibronectin and is made by controlled thawing of frozen plasma. During major bleeding, the clotting protein fibrinogen was the first to be reduced to extremely low levels. To cater to the requirements of fibrinogen replacement therapy, we have developed and characterized Fibrinogen concentrate (Fibrogen-I®) from recovered human plasma.
Fibrogen-I® is a lyophilized preparation made from pooled human plasma that must be reconstituted in water before injection. The reconstituted product can be stored for at least 12 h at room temperature. Due to less reconstitution time required, this product can be made readily available to the patients in case of emergency. This provides flexibility in treatment regimen.
The biochemical and functional analysis of Fibrogen-I® revealed that the manufacturing process results in a consistent product composition with preserved Fibrinogen functionality, as evidenced by functional activity and a Clauss/clottable protein ratio of 0.9 ± 0.01, which is close to theoretical value 1 and comparable to that observed in market comparator product.
Quality attributes of the Fibrogen-I® meets the qualification criteria as mentioned in the European pharmacopeia. Lower amounts of high molecular weight impurities(∼10%) in human Fibrinogen demonstrates superior quality over market comparator (18.4%).Similar product profiling was observed in SDS-PAGE exhibiting high product purity as compared to market comparator. This demonstrates the effectiveness of the manufacturing process steps for removal of contaminating proteins.
The alignment of γ-chain pairs and subsequent covalent γ-γ - cross linking by activated FXIII facilitates functional features such as fibrin assembly, clot stability, and viscoelastic qualities. For in-vitro cross-linking, purified human Fibrinogen contains co-purified FXIII in adequate levels; so no external FXIII must be added. The amount of FXIII present in human Fibrinogen may be useful to patients in bleeding conditions because it promotes clot stability.
Fibrogen-I® predominantly exists in a β-sheet secondary structure, which is in accordance with the three-dimensional structure of human Fibrinogen according to far-UV CD spectroscopy findings. 26 Both structurally and functionally, the human Fibrinogen product matches the market comparator.
For a long time, the viral safety of human plasma-derived therapeutic protein products has been a major concern and comprehensive virus validation is required to ensure that these products are virologically safe for human patients. Precautions against viral transmission were undertaken at all stages of the process, such as the selection of plasma donors, screening of donations, the plasma pool and quality control measures of the final product.
Following the requirements of the “guidelines on plasma-derived medicinal products”, 23 the inactivation/removal of pathogens is accomplished by orthogonal and effective steps in the human Fibrinogen manufacturing process. Table 2 summarises log reduction factors for specific viruses for targeted steps and cumulative reduction factors. The viral safety portfolio included the standard process and robustness conditions of the human Fibrinogen production process steps, namely solvent-detergent (S/D) treatment and dry heat treatment. To assure the integrity of the test systems, cytotoxicity/interference study were done prior to these virus safety studies. Under conventional treatment circumstances, the S/D treatment confirmed its efficiency in inactivating lipid encapsulated viruses quickly and irreversibly. The robustness of this inactivation step was shown for all tested viruses. Furthermore, dry heat treatment step proved its effectiveness in inactivating both enveloped as well as non-enveloped viruses. Significant virus reduction obtained during the production process using a combination of S/D treatment and dry heat treatment was validated as per current international guidelines.22,23 For enveloped and non-enveloped viruses, the cumulative log reduction factors achieved by these process steps were ≥8.14 to ≥10.23 and ≥1.4 to ≥5.91, respectively. The total capacity of virus inactivation by these dedicated steps demonstrated their efficacy in virus inactivation, verifying the product's likely safety against unknown and unpredicted viruses.
Conclusions
In conclusion, Fibrogen-I® satisfies the safety, purity and functionality standards for the treatment of patients who require fast and effective fibrinogen replacement. Fibrinogen can be made easily available to the postsurgical patients during delivery or after child birth and having clinical indications of excessive bleeding or trauma due to injury.
Footnotes
Acknowledgements
We would like to thank the management of Intas Pharmaceuticals Ltd's for providing the opportunity and resources to complete this project. We are thankful to all donors who have contributed to the project by donating their blood/plasma.
Authorship Contributions
The Fibrinogen process development mentioned in this manuscript was performed by Sachin Verma. GMP batch was prepared under the supervision of Amit Pawar and Sachin Verma. Analytical assays were developed by Dr Sheetal Dolia. Dr Suma Ray provided continuous technical inputs and mentorship for this project.
Statement of Ethics
Freshly frozen recovered plasma was obtained in polyvinyl bags from Indian blood banks that had been approved by the Drug Controller General of India (DCGI) and had been registered with the Government of India's National Aids Control Organization. Blood was collected after receiving consent from the blood donors and after verification by medical practioner.
Declaration of Conflicting Interests
All the authors are employees of Intas and declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was funded by mangement of Intas and did not receive any financial grant from other funding agencies in the public or commercial sectors.
