Abstract
Background:
Suppression of gastric acid secretion with proton pump inhibitors (PPIs) has been the mainstay of medical treatment for gastroesophageal reflux disease (GERD) for more than 30 years. Members of a newer class of medication, the potassium-competitive acid blockers (P-CABs), can inhibit gastric acid production faster, longer, and more potently than PPIs. In November 2023, vonoprazan became the first P-CAB to receive FDA approval for GERD treatment.
Methods:
The American Foregut Society (AFS) convened a 13-member panel of expert gastroenterologists and foregut surgeons to produce a white paper report on how clinicians might use P-CABs to treat GERD. After conducting a comprehensive literature review, panelists proposed 20 total statements on key aspects of P-CAB pharmacokinetics, use of P-CABs for erosive esophagitis and non-erosive reflux disease, and P-CAB safety, as well as 13 recommendation statements on how to use a P-CAB in clinical practice. Using RAND/UCLA Appropriateness Methodology, panelists independently voted to rank each statement for appropriateness, and, after panel review and discussion of first-round voting results, statements were accepted, discarded, or modified for a final round of voting.
Results:
Twenty-three statements were finally accepted (3 on P-CAB pharmacokinetics, 5 on P-CABs for erosive esophagitis, 3 on P-CABs for non-erosive reflux disease, 4 on P-CAB safety, 8 on how to use P-CABs in clinical practice).
Conclusions:
This AFS white paper report provides the statements accepted by the expert panel on the use of P-CABs in the treatment of GERD, and summarizes the literature review that provided the rationale for those statements.
Keywords
Introduction
Since 1989, the year omeprazole became the first proton pump inhibitor (PPI) to receive approval by the U.S. Food and Drug Administration (FDA), inhibition of gastric acid production with PPIs has been the mainstay of medical treatment for gastroesophageal reflux disease (GERD). 1 Although these medications have been highly effective in that regard, PPIs do not always heal erosive esophagitis when they are administered in conventional (once daily) doses or even in high (twice daily) doses, especially when esophagitis is severe. Furthermore, patients often complain of GERD symptoms that do not respond to PPIs. These unmet needs have spurred the development of a number of potassium-competitive acid blockers (P-CABs) that can inhibit gastric acid production faster, longer, and more potently than PPIs.2,3 In November 2023, vonoprazan became the first P-CAB to receive U.S. FDA approval, but the proper place of P-CABs in the armamentarium of GERD treatments is not definitively established. To address this issue, in April 2024, the American Foregut Society sanctioned a white paper report to be written by a panel of expert gastroenterologists and foregut surgeons on how clinicians might use P-CABs for the treatment of GERD. This report is the product of that panel. It should be noted that this work was supported by an unrestricted grant from Phathom Pharmaceuticals, which had no input or access to any aspect of the report during its formulation, and that 10 of the 13 physician panel members have served as consultants for Phathom Pharmaceuticals.
While the panel recognized that medication cost is a critical issue to consider when making clinical decisions, we did not conduct a formal cost-effectiveness analysis of PPIs vs. P-CABs. Such complex analyses were deemed beyond the scope of the current report. Clearly, the list price of vonoprazan (approximately $640 per month) presently exceeds that of PPIs in the United States, but health insurance organizations determine medication coverage and co-payments, which vary considerably among individual patients and institutions. The phrase “patient access to coverage of prescription drugs” might be preferable to “cost,” as the phrase better reflects the decision-making process for physicians and patients. Nevertheless, for the sake of simplicity, we have retained the less cumbersome term “cost” throughout the manuscript.
Methods
We used the RAND/UCLA Appropriateness Methodology, a modified Delphi Method, to establish the position of the expert panel on how to use P-CABs appropriately for the treatment of GERD. 4 Each panelist was assigned to one of four working groups in order to review pertinent literature and formulate statements on the following issues: (1) Mechanism of action and pharmacokinetics of P-CABS, (2) Use of P-CABs for erosive esophagitis, (3) Use of P-CABs for non-erosive reflux disease (NERD), and (4) P-CAB safety. Working groups conducted a comprehensive review of pertinent literature using the Medline (PubMed) database from inception through June 2024, using key words that included potassium-competitive acid blockers, P-CABs, vonoprazan, revaprazan, tegoprazan, keverprazan, fexuprazan, reflux esophagitis, erosive esophagitis, non-erosive reflux disease, and NERD. The working groups then proposed key factual statements about their assigned issues that they deemed to be supported directly by their literature review of published studies (Appendix A). Those factual statements were distributed to all panelists for an independent, online first round of voting. Working group leaders then proposed a list of recommendation statements on how to use vonoprazan in clinical practice (Appendix B), and those statements were also distributed to all panelists for an independent, online first round of voting. The latter statements were limited to vonoprazan and did not consider other P-CABs because vonoprazan presently is the only P-CAB available for clinical use in the United States, and the only P-CAB studied in Western populations.
Participants were instructed to rank each statement for appropriateness on a scale of 1 to 9 in which 1 meant that expected harms greatly outweighed expected benefits, and 9 meant that expected benefits greatly outweighed expected harms. Participants also were advised that: (1) Statements did not have to apply to any one specific patient, but rather to the overall care of patients, (2) Statements or measures should be considered “valid” if adherence is critical for providing quality care, irrespective of considerations of cost or feasibility, (3) Rankings should be based on the participant’s personal judgment, (4) Measures should be considered for the average patient presenting to the average community physician at an average hospital.
After the voting, participants were provided data on the median score, the range of scores, and the percent agreement for each statement. Statements were deemed appropriate when the median score was ≥7 and percent agreement was ≥80%. All participants then were invited to attend a virtual round table meeting in which all statements were discussed, especially those that did not meet criteria for appropriateness, and the statements were either revised or discarded. The revised statements were then distributed for a final vote (Round 2) and again independently ranked using a scale of 1-9. Table 1 lists all the final statements and highlights those for which agreement was met and the recommendation was deemed appropriate (ie, ≥80% of rankings fell within the 7-9 point range). All online voting was performed through Qualtrics (University of California, Los Angeles) and the process was moderated by an investigator with expertise in RAND methodology (JMK). The text below summarizes the literature review that supports these statements.
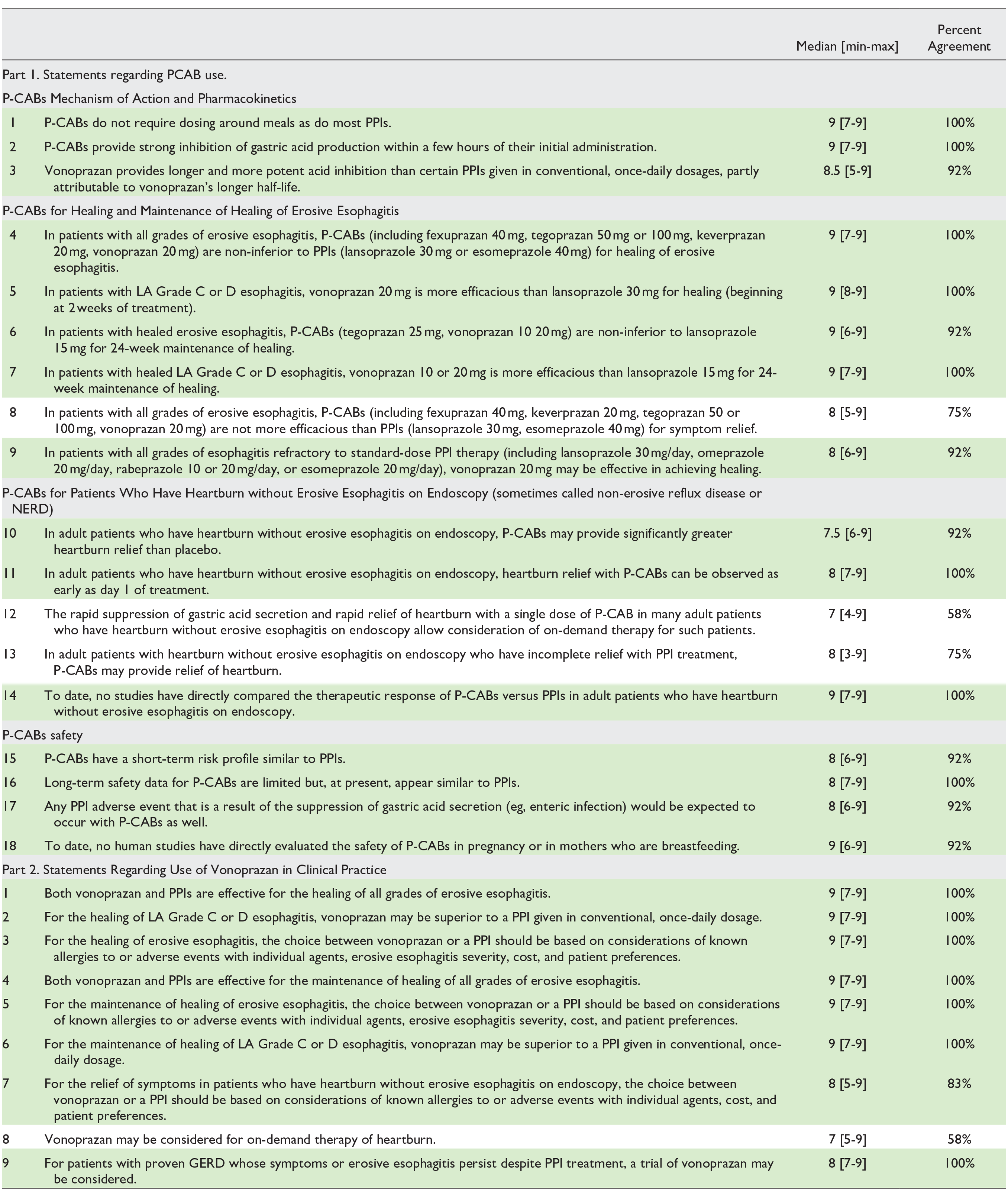
Results of Final Voting on White Paper Statements.
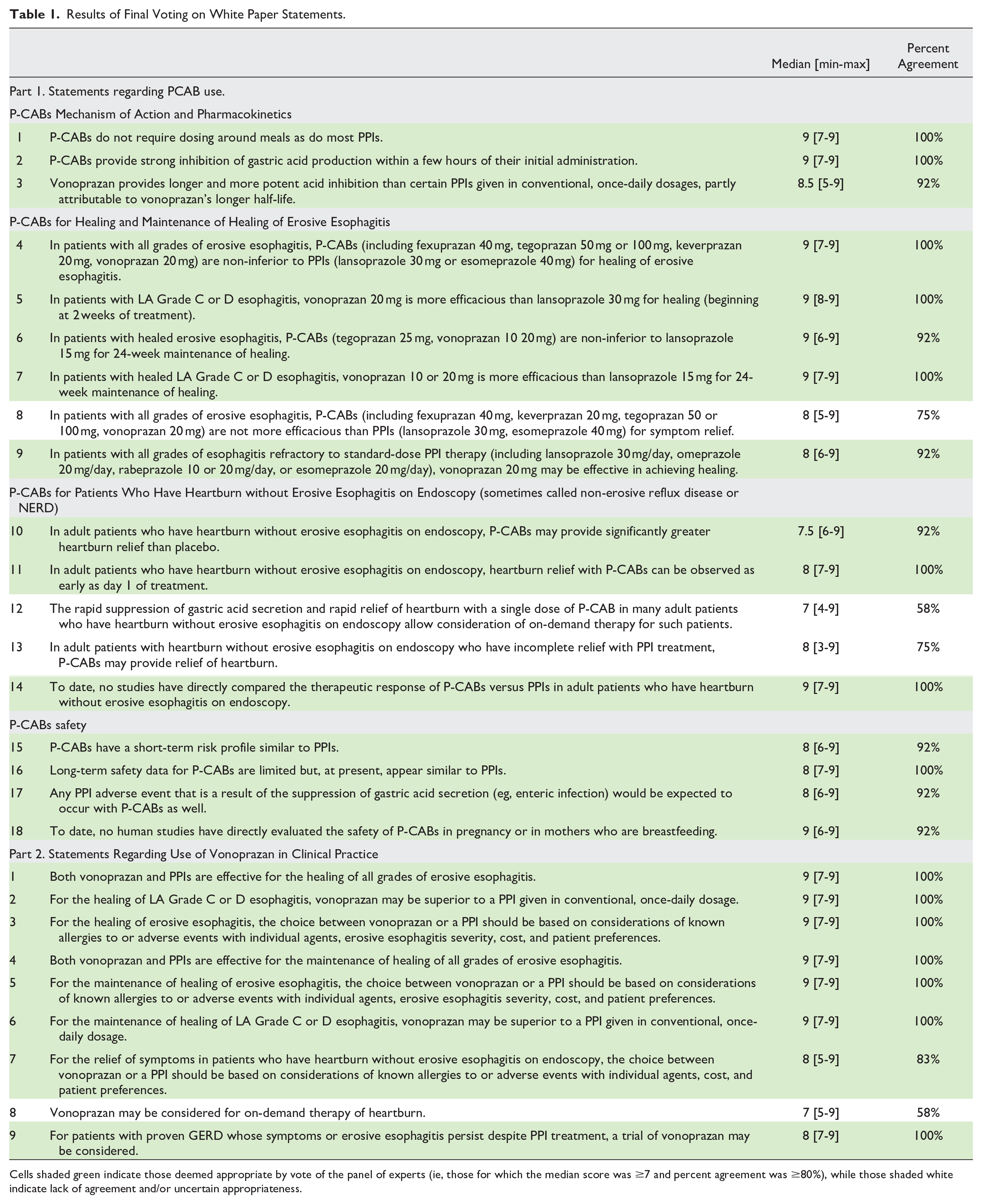
Cells shaded green indicate those deemed appropriate by vote of the panel of experts (ie, those for which the median score was ≥7 and percent agreement was ≥80%), while those shaded white indicate lack of agreement and/or uncertain appropriateness.
Mechanism of Action and Pharmacokinetics of PPIs and P-CABs
H+,K+-ATPase, the proton pump of the parietal cell
The gastric H+,K+-ATPase enzyme, the proton pump of the parietal cell, is a phosphorylation-type (P-type) ATPase, so called because phosphorylation of an aspartic acid residue within the enzyme is a key step in its activation (a feature common to all members of the P-type ATPase superfamily).5,6 The gastric H+,K+-ATPase uses energy it derives from the hydrolysis of ATP to pump hydrogen ions (protons) against a huge concentration gradient (>1 million-fold) from the cytoplasm of the parietal cell into the lumen of the stomach.
When activated, the H+,K+-ATPase enzyme spans the plasma membrane of the parietal cell in which it resides, such that the enzyme has both cytoplasmic and luminal domains. It is an α,β-heterodimeric enzyme comprised of a catalytic α subunit that has ten membrane-spanning segments, and a smaller, structural β subunit that has only one transmembrane segment. 6 The α subunit harbors both the phosphorylation site and the ATP binding site, and the β subunit functions to orient the enzyme in the apical (canalicular) membrane. 7 Under resting conditions, H+,K+-ATPase is stored in cytoplasmic tubulovesicles that have no extracellular access. 8 Acid secretion by the parietal cell is stimulated by secretagogues that include histamine (which binds the cell’s H2-receptor to activate adenylate cyclase and thereby increase intracellular cyclic AMP), gastrin, and acetylcholine (which bind the cell’s CCK2 and M3 receptors, respectively, to increase intracellular calcium). 9 In response to those secretagogues, the cytoplasmic tubulovesicles that contain H+,K+-ATPase fuse with the canalicular membrane, a process that gives the enzyme access to the gastric lumen to begin its proton pump function. 8
When ATP binds and phosphorylates a key aspartic acid residue in the cytoplasmic domain of H+,K+-ATPase, the enzyme undergoes a conformational change from a form that has high affinity for H+ ions in the cytoplasm (E1 configuration) to a form that has a high affinity for K+ ions in the gastric lumen (E2 configuration). 5 This conformational change opens a channel for the transport of cations, thus enabling H+,K+-ATPase to exchange H+ ions in the cytoplasm for K+ ions in the gastric lumen. When this exchange is completed, the enzyme again assumes its E1 configuration to restart the proton pump cycle. Regardless of the initial stimulus, the exchange of cytoplasmic protons for luminal potassium ions mediated by H+,K+-ATPase is the final step in acid secretion by the gastric parietal cell. Both proton pump inhibitors (PPIs) and potassium-competitive acid blockers (P-CABs) block that final step.
Proton pump inhibitors mechanism of action
The PPIs used in clinical practice are derivatives of the molecule benzimidazole. They are weak bases with pKa values ranging from ~4.0 to 5.0 and, in their unprotonated form, PPIs are permeable to cell membranes. 10 However, in the highly acidic environment of the canaliculus of the stimulated parietal cell, PPI molecules are rapidly protonated, which renders them far less membrane permeable, and they accumulate in the canalicular lumen where they reach concentrations 1,000-fold greater than their concentrations in the blood.10-12 Although acid is needed to trap and activate the PPI molecules in parietal cell canaliculi, PPIs themselves are acid-labile medications that can be degraded by acid in the stomach when taken orally. Consequently, most PPI formulations are given an acid-resistant enteric coat that delays their release until they are in the alkaline environment of the small intestine. 13
PPIs are prodrugs that must be activated by acid in the parietal cell canaliculus. In that acidic environment, PPI molecules are protonated and so converted into sulfenic acid or sulfenamide forms that are thiophilic (ie, that have affinity for ligands containing sulfur). These acid-activated, thiophilic PPI molecules bind covalently to sulfur-containing cysteine residues in the H+,K+-ATPase, thereby fixing the enzyme in its E2 configuration and permanently inactivating that proton pump. Since only proton pumps that are actively secreting acid have access to the activated PPI molecules in the secretory canaliculus, only actively secreting proton pumps can be affected by PPIs. In fasting individuals, fewer than 10% of gastric proton pumps are active, while approximately 70% are active when stimulated by meals. 12 Since PPIs have a short plasma half-life of only 30 minutes to 2 hours, 12 patients are advised to take their PPIs 30 to 60 minutes before meals so that the drug is in the blood in maximal concentrations available to the parietal cell when its proton pumps become active.
The H+,K+-ATPase enzyme has a half-life of approximately 2 days, and the stomach constantly synthesizes new proton pumps, replacing about 20% of them every 24 hours. Given the short plasma half-life of the PPIs, they will not be available to block acid secretion by the newly synthesized proton pumps throughout the whole day, even when PPIs are taken twice daily. All these factors contribute to delay the antisecretory effect of PPIs. It is estimated that 3 to 5 days of PPI therapy are required to reach steady-state inhibition of gastric acid secretion. 14 Thus, while PPIs provide substantial long-term acid suppression, they are not useful agents for treating acute or breakthrough symptoms in GERD patients.
The length of time that PPIs remain in the blood is a function of the rapidity of their metabolism by the hepatic cytochrome P450 system, especially by the cytochrome P450 2C19 (CYP2C19) enzyme. The CYP2C19 gene is highly polymorphic, and some of those polymorphisms profoundly influence the rate of PPI metabolism. 15 On the basis of polymorphisms in the CYP2C19 gene, individuals can be classified as normal, poor, or rapid metabolizers of PPIs. When treating patients with PPIs, therapeutic failure is especially likely in the rapid metabolizers who quickly clear the PPI from the blood. Since testing for polymorphisms in the CYP2C19 gene rarely is performed in clinical practice, clinicians do not know how rapidly any individual patient will metabolize a PPI.
In summary, PPI features that limit their speed of onset and/or efficacy in inhibition of gastric acid secretion include: (1) their need for an enteric coat, which protects them from degradation by gastric acid but delays their absorption, (2) their need to be converted from pro-drug into an active form by acid in the secretory canaliculus of the parietal cell, (3) their need to be dosed before meals (with the exception of dexlansoprazole) because activated PPIs in the canaliculi are accessible only to activated proton pumps, and proton pumps are activated primarily by meals, (3) their short plasma half-life, which limits their exposure to the actively secreting pumps that they can bind covalently, (4) their requirement for 3 to 5 days to reach steady-state inhibition of gastric acid secretion, and (5) their metabolism primarily by CYP2C19, which results in wide patient variability in the rate of PPI metabolism because of frequent polymorphisms in the CYP2C19 gene.
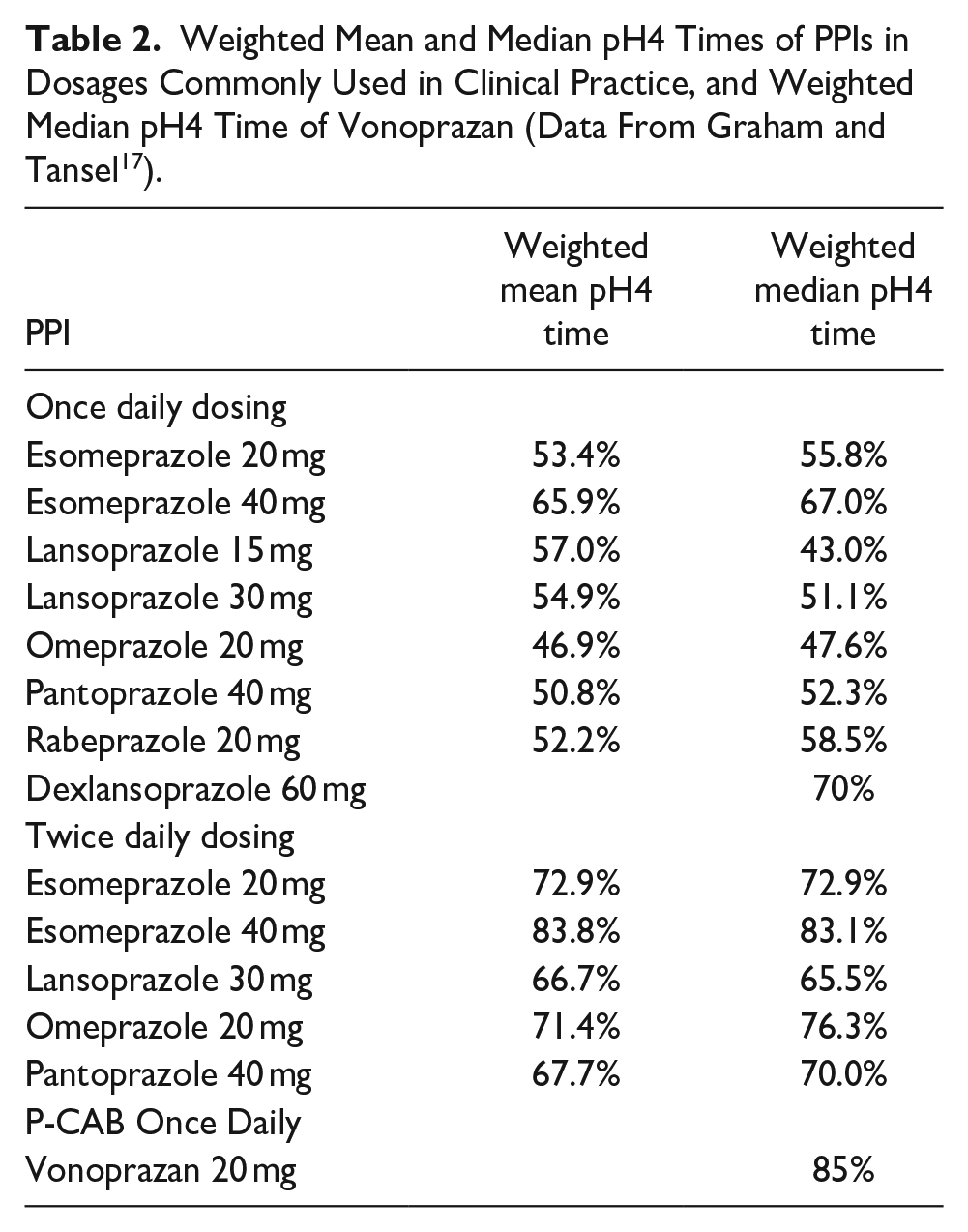
The time that a PPI formulation maintains gastric pH at a level ≥4 over a 24-hour period is called its pH4time (also called the pH4 holding-time ratio).16,17 This is an important metric to consider when treating GERD patients, because the success rate for healing erosive esophagitis with an antisecretory agent is directly related to its pH4time.18,19 The relative potencies of different PPI preparations have been estimated by comparing their effects on pH4time to that of omeprazole, yielding a value termed ‘omeprazole equivalents’ (OEs).16,17 Based on this comparison, Graham and Tansel estimate OEs for PPI preparations as follows: omeprazole 20 mg = 20 mg OE, pantoprazole 20 mg = 4.5 mg OE, lansoprazole 15 mg = 13.5 mg OE, esomeprazole 20 mg = 32 mg OE, rabeprazole 20 mg = 36 mg OE. 17 It should be noted that the predictive value of OEs for endoscopic healing and symptom control in GERD has not been established. Table 2 lists the weighted mean and median pH4times of different PPI formulations, given in dosages commonly used in clinical practice, based on an analysis conducted by Graham and Tansel of data from RCTs that performed pH testing for PPI formulations given for a minimum of 5 days. 17
Weighted Mean and Median pH4 Times of PPIs in Dosages Commonly Used in Clinical Practice, and Weighted Median pH4 Time of Vonoprazan (Data From Graham and Tansel 17 ).
Potassium-competitive acid blockers mechanism of action
Like PPIs, the P-CABs also inhibit the H+,K+-ATPase enzyme of the parietal cell, but through a somewhat different mechanism. P-CABs compete with K+ for binding to the proton pump and, in so doing, block its ability to pump H+ into the lumen in exchange for K+. 5 The first P-CAB, SCH 28080, was developed in the early 1980s, but SCH 28080 was never used clinically because of its hepatotoxicity.20,21 Currently marketed P-CABs such as vonoprazan feature a chemical structure distinct from first-generation P-CABs such as SCH 28080 and AZD0865. Those earlier compounds, characterized by a benzimidazole core, were linked to off-target hepatotoxicity. A number of candidate P-CAB compounds subsequently were evaluated, but many were withdrawn due to toxicity or short durations of action. 22 The P-CABs now available for use in some Asian countries include revaprazan, fexuprazan, keverprazan, tegoprazan, and vonoprazan, 23 and at least two other P-CABs are under development (linaprazan, zastaprazan). Vonoprazan has been used in Japan for the treatment of acid-related diseases including erosive esophagitis since 2015, and it is the most extensively studied of all the P-CABs. 23 In November 2023, the US FDA approved the use of vonoprazan for the healing of erosive esophagitis, maintenance of healing of erosive GERD, and relief of heartburn associated with erosive GERD. In July 2024, the FDA also approved vonoprazan for the relief of heartburn associated with non-erosive GERD. Presently, vonoprazan is the only P-CAB approved for clinical use in the United States.
P-CABs have several physicochemical properties that enable them to inhibit gastric acid production faster, longer, and more potently than PPIs. 24 Unlike the acid-vulnerable PPIs, P-CABs are acid-stable and administered in rapid-release formulations with no enteric coating to delay their absorption. Unlike PPIs, which are prodrugs that must be acid-activated to bind covalently to the proton pump, the P-CABs inhibit H+,K+-ATPase through rapid, reversible, ionic binding that requires no acid-activation. Most P-CABs have longer half-lives than PPIs, and animal studies have shown that P-CABs can bind both active and inactive proton pumps. 21 Thus, P-CABs do not have to be dosed around meals and, for all these reasons, P-CABs provide potent inhibition of gastric acid production within hours of their administration. 25 Furthermore, P-CABs are not metabolized primarily by CYP2C19, and so polymorphisms in the CYP2C19 gene do not appear to influence P-CAB efficacy.24,26 These features allow for greater flexibility in daily dosing, which has the potential to improve the low levels of patient adherence that have been reported to contribute to therapeutic failure for PPIs. 27
Vonoprazan, an acid-stable pyrrole derivative, is a weak base with a pKa of 9.3. 24 It has a plasma half-life of 7 to 9 hours (compared to PPI half-lives of <2 hours), and it is highly lipophilic and membrane permeable. 28 Vonoprazan moves rapidly from the blood into the acidic secretory canaliculi of the parietal cell, where protonation renders the molecule far less membrane permeable. 29 Consequently, vonoprazan rapidly accumulates in the canaliculi. These features account for its rapid onset and long duration of acid inhibition.
In a study of 20 healthy Japanese men given vonoprazan 20 mg, esomeprazole 20 mg, or rabeprazole 10 mg daily for 7 days, mean pH4 time on Day 1 was 71.4%-84.2% for vonoprazan, 23.9% for esomeprazole (difference between vonoprazan and esomeprazole 47.5%, 95%CI 35.6-69.4%), and 26.3% for rabeprazole (difference between vonoprazan and rabeprazole 58.2%, 95%CI 43.6-72.9%). 26 By Day 7, mean pH4time increased to 85.8%-93.8% for vonoprazan, 61.2% for esomeprazole (difference between vonoprazan and esomeprazole 24.6%, 95%CI 16.2-33.1%), and 65.1% for rabeprazole (difference between vonoprazan and rabeprazole 28.8%, 95%CI 17.2-40.4%). In another study, 28 healthy Japanese subjects were given four different regimens of vonoprazan or esomeprazole in a randomized, crossover fashion as follows: (1) vonoprazan 20 mg once daily, (2) vonoprazan 20 mg twice daily, (3) esomeprazole 20 mg once daily, and (4) esomeprazole 20 mg twice daily. 30 The pH4times for both vonoprazan doses (vonoprazan twice daily 100%, vonoprazan once daily 95%) were significantly greater than those for both esomeprazole doses (esomeprazole twice daily 91%, esomeprazole once daily 68%). However, the difference in pH4 time between vonoprazan once daily (95%) and esomeprazole twice daily (91%) was small. This is one of very few studies that have compared a P-CAB directly with a PPI given twice daily, although it should be noted that the pH4time for esomeprazole 20 mg twice daily in this Japanese study (91%) was considerably longer than that reported in some studies of Western subjects (~73%).31,32
Most studies of P-CAB effects on gastric acid secretion have been conducted in Asian subjects, whose gastric acid secretion rates might be lower than those of Western subjects due to differences in parietal cell mass and other factors. 33 In a study of 44 healthy US subjects (27% women) given vonoprazan 20 mg or lansoprazole 30 mg for 7 days, mean pH4 time on Day 1 was 62.4% for vonoprazan and 22.6% for lansoprazole (difference in least square means between vonoprazan and lansoprazole 39.0%, 95%CI 31.9-46.0%). 25 By Day 7, mean pH4time increased to 87.8% for vonoprazan and 42.3% for lansoprazole (difference in least square means between vonoprazan and lansoprazole 44.6%, 95%CI 37.6-52.7%). In both studies, vonoprazan provided faster and more potent inhibition of gastric acidity than PPIs. Graham and Dore estimate that 10 mg of vonoprazan once daily is approximately equivalent in intragastric pH control to 60 mg of omeprazole once daily, and that 20 mg of vonoprazan once daily is approximately equivalent to 60 mg of omeprazole or 40 mg of esomeprazole taken twice daily. 34 However, these estimates have not been confirmed in a head-to-head trial.
P-CABs do not require dosing around meals as do most PPIs.
P-CABs provide strong inhibition of gastric acid production within a few hours of their initial administration.
Vonoprazan provides longer and more potent acid inhibition than certain PPIs given in conventional, once-daily dosages, partly attributable to vonoprazan’s longer half-life.
P-CABS For Erosive Esophagitis (EE)
Clinical studies on the use P-CABs for erosive esophagitis have focused on four key outcomes: (1) healing of erosive esophagitis (EE), (2) maintenance of healing of EE, (3) relief of heartburn associated with erosive esophagitis, and (4) treatment of refractory EE. Data pertaining to P-CAB efficacy for each of these in endpoints is summarized below.
Healing of EE
All available P-CAB trials on EE patients have characterized the severity of EE using the Los Angeles A-D grading scale, 35 and included patients spanning this spectrum of severity. This is noteworthy because the first Lyon consensus on “The modern diagnosis of GERD” did not accept Los Angeles grade A or B esophagitis as definitive evidence of GERD, 36 arguing that LA grade A esophagitis is non-specific, found in 5%–7.5% of asymptomatic controls.37,38 With LA grade B esophagitis, it was opined that this was adequate evidence to initiate medical management, but not surgical management of GERD because of considerable inter-observer variability. This stance on Los Angeles B esophagitis softened in Lyon consensus 2.0, which now accepts LA-B esophagitis as conclusive evidence of GERD, but still does not accept Los Angeles A esophagitis without corroborative pH-metry findings. 39 With this degree of skepticism regarding Los Angeles A esophagitis, one might expect its inclusion into EE trials to somewhat dilute the findings.
Examining the eight EE RCTs thus far published comparing the efficacy of P-CABs to PPIs, the distribution of esophagitis severity among study subjects varies widely (Table 3),40-47 ranging from as few as 33% LA-A esophagitis subjects 46 to as many as 66% with LA-A esophagitis, 42 It is also noteworthy that only two of the studies in Table 3 used some method to adjudicate the endoscopic findings identified by local site investigators, thereby addressing the issue of inter-observer variability raised by the Lyon Consensus.40,46 In one of those studies that mandated central adjudication of endoscopic grading for study participation, 46 a subsequent, spin-off report found that local investigators and central adjudicators agreed on the LA grade in only 65% of cases. 48 This report highlights that there can be considerable disagreement among endoscopists in grading of EE by the LA classification system. Since most EE studies have relied on non-adjudicated endoscopy grading to assess study eligibility and treatment response, clinicians should consider that this is a possible source of error with potential to affect the outcome of clinical trials.
Randomized Controlled Trials of P-CABs versus PPIs for Healing EE. The Distribution of Esophagitis Severity Included in Each Study is According to Los Angeles (LA) Grade.
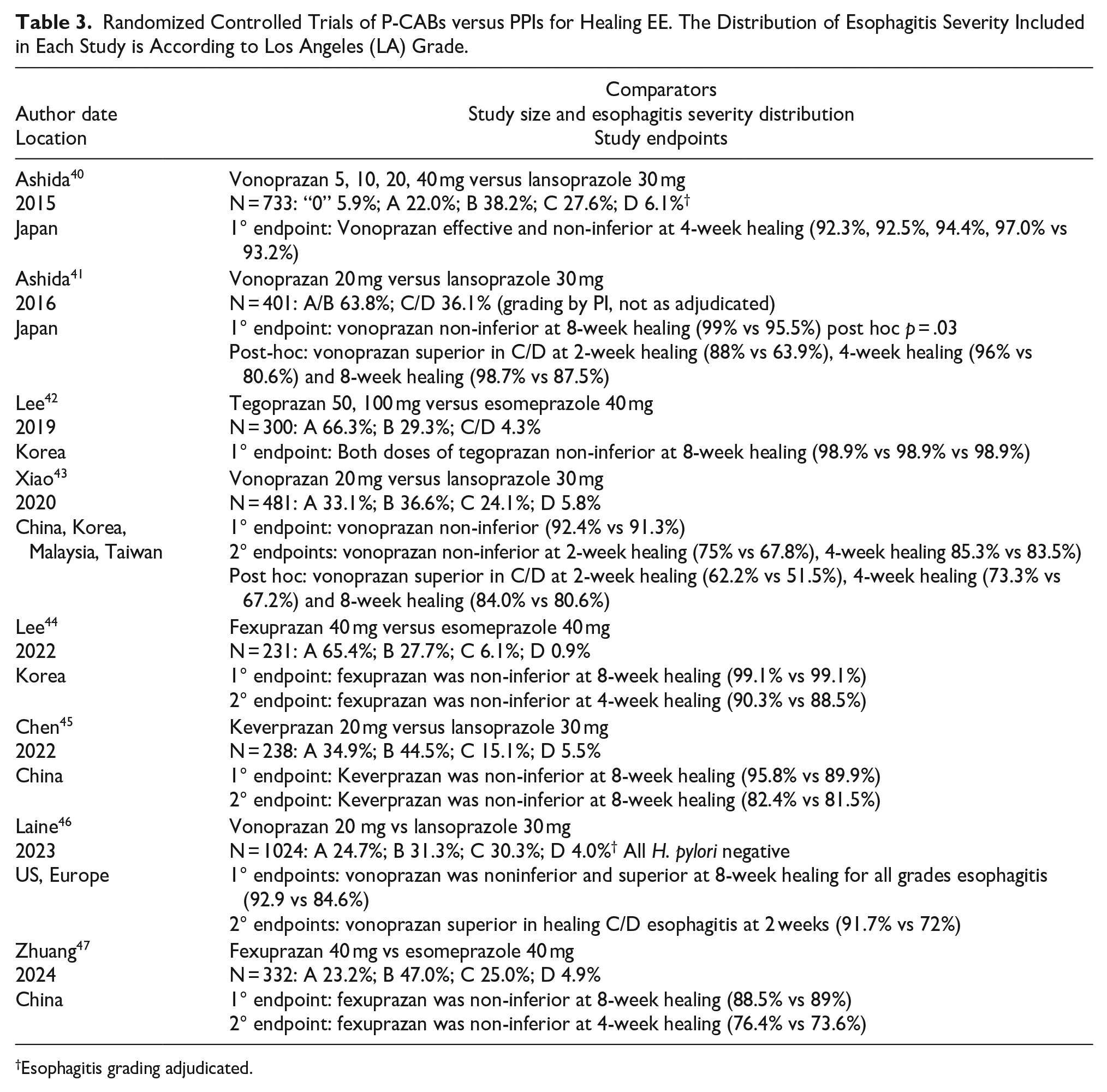
Esophagitis grading adjudicated.
All published P-CAB RCTs of EE have used FDA-approved healing doses of PPIs (lansoprazole 30 mg or esomeprazole 40 mg) as a comparator. Among the 8 studies in Table 3, 4 were of vonoprazan versus lansoprazole,40,41,44,46 and these were also the 4 largest EE trials, enrolling a combined total of 2,639 patients. All the trials showed non-inferiority of the P-CAB to the comparator PPI for EE healing, be that at 2, 4, or 8 weeks. Three of the trials demonstrated superiority of vonoprazan 20 mg over lansoprazole 30 mg for healing high-grade (LA-C/D) esophagitis either as a secondary endpoint 45 or in post hoc analyses.41,43 The one trial that showed superiority of vonoprazan 20 mg over lansoprazole 30 mg for healing all grades of esophagitis at 8 weeks was the largest study overall, which enrolled 1,024 patients. 46
Statements regarding healing of erosive esophagitis
In patients with all grades of erosive esophagitis, P-CABs (including fexuprazan 40 mg, tegoprazan 50 mg, or 100 mg, keverprazan 20 mg, vonoprazan 20 mg) are non-inferior to PPIs (lansoprazole 30 mg or esomeprazole 40 mg) for healing of erosive esophagitis.
In patients with LA Grade C or D esophagitis, vonoprazan 20 mg is more efficacious than lansoprazole 30 mg for healing (beginning at 2 weeks of treatment).
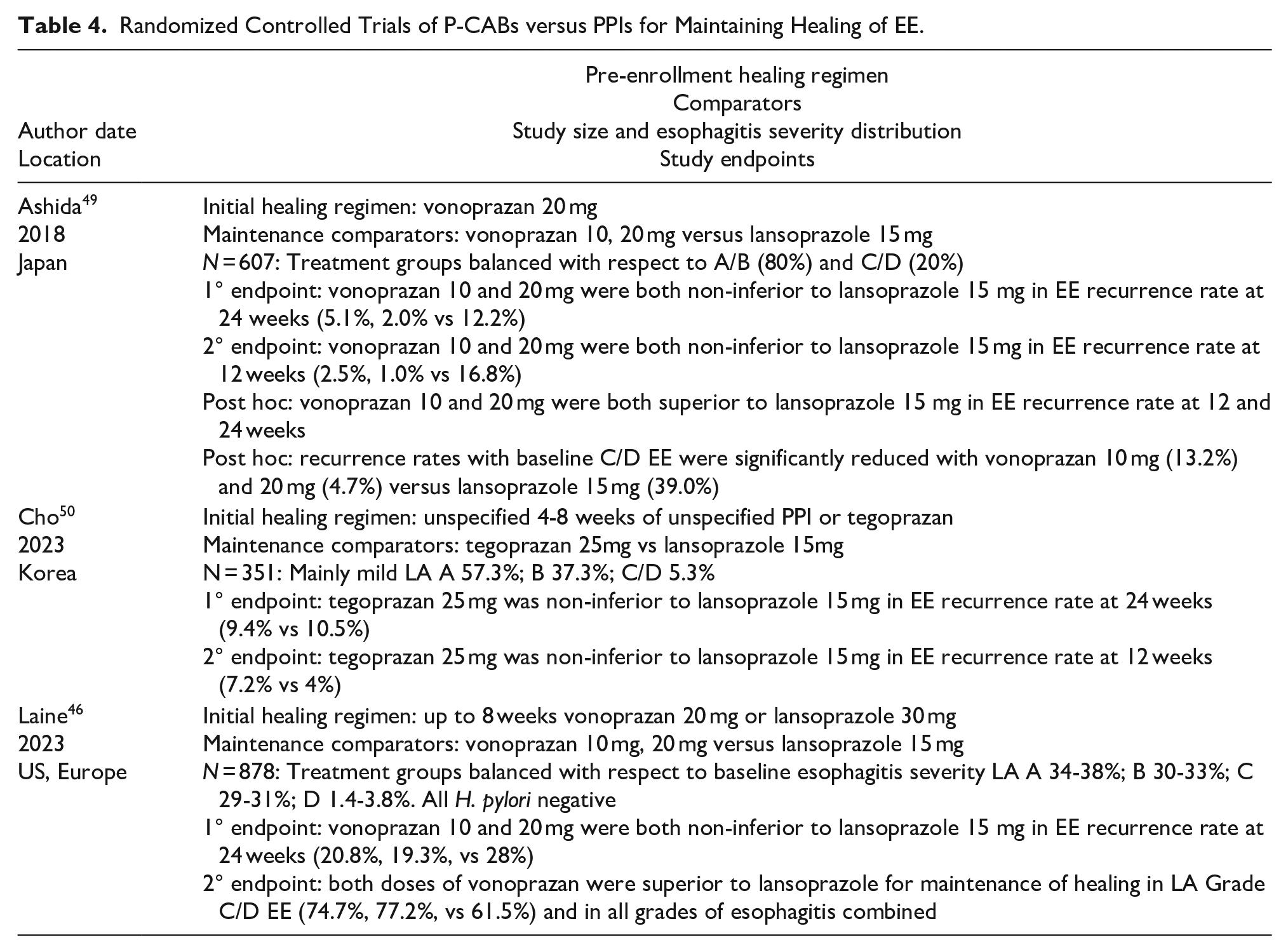
Maintenance of healing of EE
Erosive esophagitis is a chronic disease and, although it can be healed with sufficiently potent antisecretory medications, the underlying pathophysiology persists, and the esophagitis is likely to recur without maintenance therapy. Furthermore, the more severe the presenting esophagitis prior to healing, the more certain the recurrence with cessation of therapy. Consequently, maintenance therapy is often necessary and, in gauging the efficacy of maintenance antisecretory regimens, both the initial severity of esophagitis and the antisecretory regimen that had been utilized to achieve healing are important variables to consider. Examining the three RCTs thus far published comparing P-CABs to PPIs for maintenance of healing of EE (Table 4), these factors varied widely.46,49,50 The initial severity of esophagitis ranged from 95% mild (LA-A/B) disease in the Cho study, 50 to about one-third severe (LA-C/D) disease in the Laine study. 46 With respect to the initial healing of EE, this was specified in the two vonoprazan studies,46,49 but unspecified in the tegoprazan study. 50 All three trials showed non-inferiority of the P-CAB to the comparator PPI for maintenance of EE healing. Both vonoprazan trials showed superiority of vonoprazan 10 or 20 mg over lansoprazole 15 mg for maintaining healing in patients who had presented with LA-C/D esophagitis, either as a secondary endpoint 46 or in a post hoc analysis. 49 The one trial that showed superiority of vonoprazan (10 or 20 mg) over lansoprazole 15 mg at maintaining healing in all grades of esophagitis was the largest study overall, enrolling 893 patients, and the study with the highest proportion of LA-C/D disease. 46
Statements regarding maintenance of healing of erosive esophagitis
In patients with healed erosive esophagitis, P-CABs (tegoprazan 25 mg, vonoprazan 10 20 mg) are non-inferior to lansoprazole 15 mg for 24-week maintenance of healing.
In patients with healed LA Grade C or D esophagitis, vonoprazan 10 or 20 mg is more efficacious than lansoprazole 15 mg for 24-week maintenance of healing.
Randomized Controlled Trials of P-CABs versus PPIs for Maintaining Healing of EE.
Relief of heartburn associated with EE
Irrespective of PPI efficacy in healing EE, it has been a common observation in both clinical trials and clinical practice that a substantial percentage of GERD patients do not achieve complete symptom relief on PPI therapy.51,52 Conceptually, this could be attributable to inadequate acid suppression, be that because of the limited potency of the drug itself, CYP2C19 polymorphisms that result in rapid PPI metabolism, or poor compliance with respect to timing of PPI administration around meals; all these shortcomings potentially could be improved by substituting a P-CAB for the PPI. Alternatively, the refractory symptoms might be attributable to mechanisms other than acid reflux, as is frequently the case in patients thought to have non-erosive reflux disease. Among the P-CAB versus PPI erosive esophagitis trials, most included symptom improvement endpoints,40-47,53 but only one EE trial used a symptom endpoint as its primary outcome measure, 53 and that 32-patient, 14-day RCT of vonoprazan 20 mg versus lansoprazole 30 mg by Oshima et al. was clearly designed to leverage the superior pharmacodynamics of vonoprazan. The primary endpoint of that study was the first day of complete (day and night) heartburn relief that then persisted for 7 consecutive days; the severity of daytime and nighttime heartburn was scored on a 5-point Likert scale each day. The primary endpoint was achieved sooner with vonoprazan (31.3% on day 1) than with lansoprazole (12.5% on day 1), and significantly more patients achieved complete heartburn relief with vonoprazan on days 2-7 (HR 3.58, 95% CI 1.16-11.08, P < .05 vs lansoprazole).
The study by Oshima et al. just described suggests that the onset of sustained heartburn relief occurs faster with vonoprazan 20 mg than with lansoprazole 30 mg. 53 However, the much larger (1,024 patients) US/European RCT, which had a greater percentage of patients with LA-C/D esophagitis, used the same comparator medications but failed to confirm the finding that vonoprazan provides faster sustained relief of heartburn. 46 Rather, in the latter study, the percentage of subjects with the onset of sustained heartburn resolution (7 consecutive days without heartburn) by day 3 did not differ significantly between the vonoprazan and lansoprazole groups (34.4% for vonoprazan 20 mg versus 32.2% for lansoprazole 30 mg). The one symptom endpoint that was better with vonoprazan treatment was the percentage of heartburn-free days in the LA-C/D subgroup (87.5% with vonoprazan 20 mg vs 80.0% with lansoprazole 30 mg). 46
The remainder of the P-CAB vs PPI erosive esophagitis trials that included symptom improvement endpoints demonstrated similar results for symptom endpoints between treatment groups, be that for 1-, 2-, 4-, or 8-week patient diary entries of heartburn and regurgitation severity,40,41,43-45,52 improvement in the frequency or severity of symptoms, 45 percentage of heartburn-free days in the first 7 days, 4 weeks, or 8 weeks,42,46,47 4- or 8-week improvement in RDQ scores,42,44 GERD health-related quality of life,43,44,47 or percentage of days without rescue medication. 43
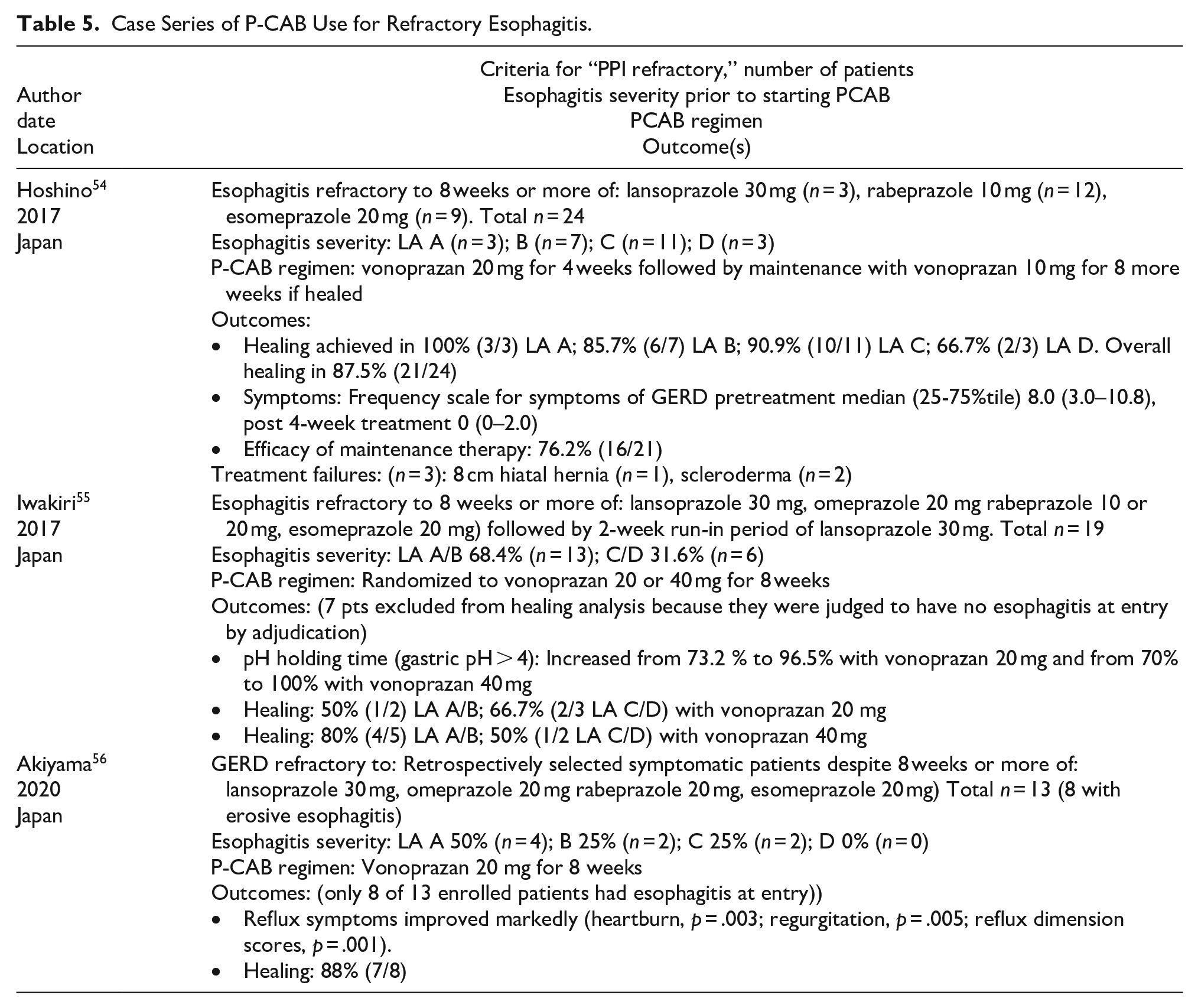
Treatment of refractory erosive esophagitis
With standard dose PPI therapy, refractory EE is sufficiently common in patients with high-grade (LA-C/D) esophagitis such that it is common practice to use twice-daily dosing, with PPIs taken before breakfast and dinner. Supportive of this practice are the data from Ashida, 41 Xiao, 43 and Laine 46 reporting healing rates for LA-C/D disease to be 87.5%, 80.6%, and 72%, respectively, after 8 weeks of treatment with lansoprazole 30 mg daily. Far fewer data are available for esophagitis refractory to twice-daily PPIs, but it clearly occurs, especially in challenging patient populations such as achalasia patients treated with LES destructive procedures (eg, POEM), patients with collagen vascular disease such as scleroderma, and patients with profound esophagogastric junction disruption. However, there are no published controlled trials addressing these challenging patients. What do exist are prospective and retrospective, uncontrolled case series of patients with esophagitis refractory to standard dose PPI, and these are summarized in Table 5.54-56 Healing was achieved with P-CABs in most of these patients, albeit the majority of them had low-grade esophagitis at entry.
Statements regarding PPI refractory esophagitis:
In patients with all grades of esophagitis refractory to standard-dose PPI therapy (including lansoprazole 30 mg/day, omeprazole 20 mg/day, rabeprazole 10 or 20 mg/day, or esomeprazole 20 mg/day), vonoprazan 20 mg may be effective in achieving healing.
Case Series of P-CAB Use for Refractory Esophagitis.
P-CABs for Patients Who Have Heartburn Without Erosive Esophagitis
The term “non-erosive reflux disease” or “NERD” is applied to patients who have heartburn without erosive esophagitis. While it is often assumed that such patients have heartburn caused by reflux, for many such patients the heartburn sensation is in fact due to an extra-esophageal disorder (eg, heart disease, biliary tract disease), a non-GERD esophageal disorder (eg, achalasia, eosinophilic esophagitis), or a disorder of gut-brain interaction (ie, functional heartburn). 57 We feel that the term “NERD” should be applied only to patients who have heartburn without erosive esophagitis on endoscopy (performed with patients off PPIs), and who also have evidence of abnormal acid reflux by esophageal pH monitoring. In the absence of reflux monitoring, we prefer to call the condition “heartburn without erosive esophagitis.” Unfortunately, very few studies of patients diagnosed with NERD have included abnormal reflux monitoring as an entry criterion. For the sake of simplicity and to avoid confusion, we will use the term “NERD” as used in the studies described below, none of which included reflux monitoring as an entry criterion.
Four RCTs have compared P-CABs to placebo in patients with NERD, defined as heartburn (and/or regurgitation) occurring at least 2-4 days a week in the absence of advanced grades of erosive esophagitis on endoscopy;58-61 one of those studies 61 also enrolled patients with moderate to severe symptoms occurring at least 1 day a week. Three of the RCTs compared vonoprazan to placebo,58-60 while one compared tegoprazan to placebo. 61 In the U.S. trial comparing two vonoprazan doses (10 and 20 mg) to placebo, there was a significantly higher proportion of heartburn-free days at 4 weeks with both vonoprazan doses (44.8% and 44.4%) compared to placebo (27.7%, p < .0001). 59 In a trial performed in South Korea comparing two tegoprazan doses (50 and 100 mg) to placebo, the proportion of days without heartburn was 56.7% with placebo and 67.6% and 66.5% with 50 and 100 mg of tegoprazan, respectively (p ≤ .002). 61 In contrast, there were no significant differences in the proportion of heartburn-free days in two RCTs performed in Japan comparing vonoprazan at various doses to placebo.58,60 In the first of those RCTs, median proportion of days without heartburn in the 4-week treatment period was not significantly different between placebo (7.4%) and vonoprazan 10 mg (10.3%, p = .23) or 20 mg (12.0%, p = 0.05), although heartburn intensity was lower in the vonoprazan than in the placebo cohorts. 58 In the second RCT, vonoprazan 10 mg resulted in 72.6% heartburn-free days compared to 61.5% with placebo (p = .06) in an intention-to-treat cohort. 60
In the U.S. RCT, 772 patients with heartburn ≥4 days a week and normal endoscopy were randomized to receive placebo, vonoprazan 10 mg, or vonoprazan 20 mg for 4 weeks. 59 As noted above, the proportion of heartburn-free days was significantly higher with both vonoprazan doses (44.8% and 44.4%) compared to placebo (27.7%, p < .0001) at 4 weeks. With vonoprazan 10 mg, there was an 8.3% and an 11.6% therapeutic gain over placebo on days 1 and 2, respectively; corresponding proportions for vonoprazan 20 mg were 18.1% and 23.2%, indicating fast onset of symptom relief. Subsequently, patients on vonoprazan 10 and 20 mg were followed (on the same vonoprazan doses) for an additional 20 weeks. Those receiving placebo were re-randomized to receive vonoprazan 10 or 20 mg at 4 weeks, and were also followed for an additional 20 weeks. At the end of the study, the mean percentage of days without heartburn for the vonoprazan groups ranged from 61% to 63%, irrespective of dose and prior placebo administration.
The aforementioned Japanese RCT published in 2016 enrolled 827 patients who had ≥2 days of moderate or greater severity heartburn during a 1-week run-in period in which they received oral antacid therapy, and who had either normal or minimal changes on endoscopy. 58 These patients were randomized to receive placebo, or vonoprazan 10 or 20 mg for 4 weeks. The primary endpoint of proportion of heartburn-free days was low overall, with no significant difference between placebo (7.4%), vonoprazan 10 mg (10.3%, p = .23 compared to placebo) or vonoprazan 20 mg (12.0%, p = .050 compared to placebo) groups. Severity of heartburn, a secondary end point, was significantly higher with placebo than with vonoprazan. The study’s requirement for heartburn despite the use of antacids likely skewed the study population toward non-GERD mechanisms for symptoms, which may account for the low proportions with heartburn-free days despite treatment with vonoprazan. 58 In the RCT from Japan published in 2019, 483 patients with heartburn ≥2 days a week and either normal or minimal changes on endoscopy were randomized to receive placebo or vonoprazan 10 mg for 4 weeks. 60 The difference between the vonoprazan and placebo groups in the primary endpoint of proportion of days without heartburn at 4 weeks (vonoprazan 72.6%, placebo 61.5%) did not reach statistical significance (p = .064) However, the secondary endpoint of cumulative improvement rate of heartburn was greater in the vonoprazan group than the placebo group (p = .0003), and separation between study groups appeared to start on the first day of therapy.
In the RCT from Korea, 324 patients with heartburn and/or regurgitation ≥2 days a week (≥1 day a week for moderate to severe symptoms) and normal endoscopy were randomized to receive placebo, tegoprazan 50 or 100 mg for 4 weeks. 61 The proportion of days without heartburn was 56.7% in the placebo group, versus 67.6% and 66.5% for patients treated with 50 and 100 mg of tegoprazan, respectively (p ≤ .002). Complete symptom resolution was reported by 24.2%, 42.5%, and 48.5% of patients, respectively during the last week of the 4-week treatment period (p ≤ .005). Complete heartburn relief rates were significantly higher in the tegoprazan groups than in placebo-treated patients, but rates of improvement in regurgitation did not differ significantly among the groups.
As noted above, all of the aforementioned RCTs utilized a non-stringent definition of NERD (i.e., symptomatic heartburn and/or regurgitation in patients with either a normal endoscopy or an endoscopy showing only minor, reflux-related esophageal mucosal changes), a definition that did not require demonstration of abnormal acid reflux by esophageal pH monitoring. Rome IV now recommends that ambulatory reflux monitoring should be used to define NERD in order to exclude patients with symptoms due to functional disorders. Although some reports have described poorer symptomatic response to acid suppression in patients with NERD than in those with EE, relief of heartburn is considerably better when a diagnosis of NERD requires both a negative endoscopy and positive pH monitoring than when only a negative endoscopy is required. A systemic review has concluded that heartburn relief with PPI therapy in patients with NERD defined both by abnormal pH monitoring and normal endoscopy is 73.5% (95% CI 69.1%-77.6%), 62 a rate nearly identical to that of patients with EE (72%, 95% CI 68-74%). Importantly, no RCTs have compared P-CABs head-to-head with PPIs in patients with NERD.
The rapid suppression of gastric acid secretion and heartburn relief provided by P-CABs has prompted consideration of using PCABs in ‘on demand’ fashion for patients with episodic heartburn. In a randomized, placebo-controlled, phase 2 study, 207 patients with heartburn ≥4 days a week, a normal endoscopy, and complete relief of heartburn during the last 7 days of a 4-week treatment period with vonoprazan 20 mg daily were enrolled in a study of on-demand vonoprazan. 63 These patients with NERD were randomized to receive placebo or vonoprazan in doses of 10, 20, or 40 mg on-demand. Significantly higher proportions of patients had complete relief of heartburn episodes within 3 hours of drug administration and no further heartburn during 24 hours after drug administration with all doses of vonoprazan (56.0%-70.0%) compared to placebo (27.3%). Symptom improvement compared to placebo was noted as early as 1 hour after vonoprazan administration.
Open-label, uncontrolled case series have reported that P-CABs can provide symptom improvement in patients with NERD who have symptoms that persist despite PPI therapy. In one such study of 62 NERD patients with PPI-resistant symptoms, those symptoms improved significantly after 4 weeks of treatment with vonoprazan 20 mg, with 76.8% reporting symptom improvement and 55.7% reporting symptom resolution. 64 In another study of 16 patients with PPI-resistant NERD, switching to vonoprazan 20 mg resulted in improvement in GERD symptom scores, increases in pH levels of the refluxate, and reductions in the percent times of intragastric pH<4.0 on pH-impedance monitoring. 65 While both acidic and weakly acidic reflux episodes decreased in patients who responded to vonoprazan, persistent weakly acidic reflux was noted in those who did not respond, perhaps indicating esophageal hypersensitivity as the underlying mechanism of symptom generation in the non-responders. 65 Finally, another study of 26 patients with NERD who had PPI-resistant symptoms found that switching to vonoprazan resulted in a significant decrease in GERD-Q scores, with 69% of patients reporting symptom improvement. 66 To date, no studies have directly compared the therapeutic response of P-CABs versus PPIs in adult patients who have heartburn without erosive esophagitis on endoscopy.
Statements Regarding P-CABs for Non-Erosive Reflux Disease
In adult patients who have heartburn without erosive esophagitis on endoscopy, P-CABs may provide significantly greater heartburn relief than placebo.
In adult patients who have heartburn without erosive esophagitis on endoscopy, heartburn relief with P-CABs can be observed as early as day 1 of treatment.
P-CAB Safety
The safety of P-CABs has been evaluated in randomized controlled trials and systematic reviews, and vonoprazan is the P-CAB with the most published safety data. Like PPIs, P-CABs are potent inhibitors of gastric acid secretion and, consequently, any adverse PPI effect due to inhibition of gastric acid secretion will likely apply to P-CABs as well. The prescriber information available for vonoprazan lists many of the same established and putative adverse effects described for PPIs (ie, acute tubulointerstitial nephritis, Clostridioides difficile-associated diarrhea, bone fracture, severe cutaneous adverse reactions, vitamin B12 deficiency, hypomagnesemia, increased chromogranin A, fundic gland polyps, and potential masking of the symptoms of gastric malignancy), 67 although some of these effects are not clearly related to gastric acid inhibition. 68 Due to renal and hepatic clearance mechanisms, patients with glomerular filtration rates <30 mL/minute and patients with Childs-Pugh Class B or C cirrhosis should receive vonoprazan in a dosage of 10 mg once daily both for healing and maintenance of healing of erosive esophagitis (ie, vonoprazan 20 mg should not be used).
Several recent randomized controlled trials have included safety data, including adverse events (AE) and serious adverse events (SAE). Much of the safety data is from short to medium-term studies, ranging in duration from 6 weeks to 6 months. In the previously mentioned study by Laine et al., patients were randomized to receive vonoprazan 20 mg daily or lansoprazole 30 mg daily for 8 weeks, followed by randomization to vonoprazan 10 or 20 mg daily or lansoprazole 15 mg daily for 24 weeks. 46 The most common AEs observed in this study were diarrhea and COVID19 infection. SAEs were uncommon, occurring in 0.6% of patients during the study’s initial phase. In the maintenance phase, 4.7% of patients in the vonoprazan 20mg group, 3.4% of patients in the vonoprazan 10 mg group, and 2.4% of patients in the lansoprazole 15 mg group had an SAE. There were no cases of Clostridioides difficile infection. Bone fractures were reported in 1.4% of the vonoprazan 20 mg group, 0.7% of the vonoprazan 10 mg group, and 0.3% of the lansoprazole group. Liver function abnormalities (AST or ALT >3 times the upper limit of normal) were uncommon, occurring in 0.3% of patients taking 20 mg of vonoprazan 20 mg, 1% taking vonoprazan10 mg, and 2% of patients taking lansoprazole. This study also assessed serum gastrin levels, which increased more with the 10 and 20 mg doses of vonoprazan than with lansoprazole. However, gastrin levels returned to near baseline 4 weeks after discontinuation of vonoprazan. Biopsies performed while patients were taking the P-CAB revealed a small increase in ECL cell hyperplasia in patients receiving 20 mg of vonoprazan, but no neoplasia was diagnosed in the vonoprazan or lansoprazole groups.
Short-term risk was also examined in a study by Fass et al., in which 458 NERD patients were treated with vonoprazan 20 mg daily for 4 weeks. 63 Compliant patients who had a good response to therapy were randomized to receive 6 weeks of on-demand therapy with either 10 mg, 20, or 40 mg of vonoprazan, or placebo. In this study, 3.5% of the treatment emergent adverse events (TEAEs) were related to the study medication. Abdominal distension (1.3%), diarrhea (1.5%), and nausea (1.3%) were the most common AEs reported. One case of anaphylaxis to vonoprazan was reported. During the on-demand portion of the study, 16.3%-18.4% of patients receiving vonoprazan and 21.3% of patients receiving placebo reported TEAEs. Baseline gastrin levels increased from a median of 19.5 to 128.5 pg/ml after 4 weeks of daily use, decreasing at the end of the 6 week on-demand period.
Short-term P-CAB safety data also have been examined in systematic reviews and meta-analyses.69-71 In a systematic review of efficacy and safety of P-CABs and PPIs used for acid-related disorders, safety data were extracted from 15 studies that included a total of 4,519 patients, of which 12 were studies of vonoprazan, 2 of tegoprazan and 1 of keverprazan. 69 The risk ratio for TEAEs comparing vonoprazan vs. PPI was 1.08 (95% CO 0.89-1.31). The risk ratio of TEAEs for tegoprazan vs. PPI was .084 (95% CI 0.6-1.17) and keverprazan was 1.37 (95% CI .92-2.03). The overall prevalence of TEAEs was 35.7% for P-CABs and 32.9% for PPIs, with no significant difference between groups. The longest study included had a duration of only 8 weeks, so the conclusions apply exclusively to short-term risks of PPIs and P-CABs.
A recent network meta-analysis compared the safety and efficacy of P-CABs and PPIs in patients with Los Angeles grade C and D erosive esophagitis, identifying 24 RCTs that used 3 different P-CABs and 6 different PPIs. 70 In this analysis, drugs were ranked in order of safety using surface under the cumulative ranking (SUCRA). For short-term safety, the network meta-analysis found similar incidence rates of AEs, SAEs, and medication cessation for daily vonoprazan 20mg and PPIs. Using SUCRA scores to rank safety, lansoprazole 30 mg had the highest short-term safety rating (SUCRA (S) = 0.73), followed by omeprazole 20 mg and pantoprazole 40 mg (S = 0.64), and vonoprazan 20 mg (S = 0.59). Esomeprazole 40 mg, rabeprazole ER 50 mg, and dexlansoprazole 60mg had SUCRA scores ranging from 0.42 to 0.52. Keverprazan had the lowest short-term safety SUCRA rating at 0.13, and no short-term studies of tegoprazan were included.
Longer-term data are starting to become available for P-CABs. In the above-mentioned network metanalysis, some of the P-CAB and PPI studies had durations of 24-52 weeks. 70 In pooled analysis of these longer studies, the incidence of AEs, SAEs, and drug cessation was similar for vonoprazan 20 mg and PPIs. Using SUCRA scoring for safety in this longer-term analysis, tegoprazan 20 mg ranked highest (S = 0.8), followed by lansoprazole 30 mg (S = 0.71). vonoprazan 10 mg (S = 0.58), and vonoprazan 20 mg (S = 0.49), with lansoprazole 15 mg and esomeprazole 20 mg showing SUCRA scores of 0.15-0.24.
The longest-term P-CAB safety study published to date is the VISION (Vonoprazan In patients with eroSIve esophagitis to evaluate lONg-term safety) study, which includes specific outcomes regarding P-CAB effects on the gastric mucosa. 71 VISION is a phase 4, RCT comparing 8 weeks of vonoprazan 20mg daily or lansoprazole 30 mg daily for healing of erosive esophagitis, followed by 260 weeks of daily vonoprazan 10 mg or lansoprazole 15 mg for maintenance of healing of erosive esophagitis. Endoscopy with biopsies of the gastric mucosa were performed at study start, and then annually during the 260-week maintenance portion of the study. Five-year safety data were reported for 135 patients who received vonoprazan and 67 who received lansoprazole. AEs were observed in 93.3% of the vonoprazan group and 95.5% of the lansoprazole group, with approximately half the AEs thought to be related to the study medication. AEs leading to drug discontinuation occurred in 6 (4.4%) patients taking vonoprazan and 1 (1.5%) of patients taking lansoprazole. The rate of serious adverse events potentially related to study medication was 2.2% with vonoprazan (leukopenia, cholangitis, abnormal liver function) and 0 in the lansoprazole group (1 patient died of myocardial infarction, not attributed to lansoprazole). Gastrin levels were monitored during the study and the median serum gastrin level was higher with vonoprazan (625 pg/mL) than with lansoprazole (200 pg/mL). At 260 weeks of treatment, parietal cell hyperplasia was more common in the vonoprazan group than the lansoprazole group (97.1% vs 86.5%), as was foveolar hyperplasia (14.7% vs 1.9%). G-cell hyperplasia was common in both groups, seen in 85.3% of patients taking vonoprazan and 76.9% of those on lansoprazole. ECL cell hyperplasia was seen in 4.9% of patients taking vonoprazan compared to 7.7% of patients taking lansoprazole. Foveolar-type gastric adenoma was identified in 1 patient taking vonoprazan (0.7%), and oxyntic-type gastric adenoma was seen in 1 patient taking lansoprazole (1.5%). There was no significant difference in the frequency of fundic gland or hyperplastic polyps between the groups. No patients taking vonoprazan or lansoprazole developed gastric dysplasia, cancer, or gastric neuroendocrine tumors.
Based on published RCTs, systematic reviews, and meta-analyses, the short-term risks of P-CABS appear to be similar to those of PPIs. Fewer long-term data are available, with the longest RCT having 5 years of follow up, but the risk of AEs appear to be similar. P-CABs provide more robust acid suppression than PPIs, reflected by greater P-CAB-induced elevations in serum gastrin levels. The consequences of these greater gastrin elevations with long-term use of P-CABs are unknown and require further study. There are data to suggest that PPI usage after eradication of Helicobacter pylori may increase the risk of gastric cancer, 72 and one recent report has suggested that vonoprazan usage after eradication of H. pylori also might increase that risk. 73 The mechanism underlying this putative increased risk is not clear.
Few data are available on the use of P-CABs in pregnant and breastfeeding patients. The vonoprazan prescriber information recommends against its use during breast-feeding. 67 No publications are available about the use of P-CABs in pregnant humans. One animal study found no adverse developmental effects in rats given the highest vonoprazan dose (estimated to be >50-fold higher than human doses), while rabbit embryos had delayed growth at levels above 3.6 mg/kg/day (estimated 18-fold higher than human doses). 74 With numerous other options for treatment of GERD in pregnant persons, we feel that P-CABs should not be considered until all other avenues for GERD treatment have been exhausted, and in close consultation with the obstetrician.
P-CABs have a short-term risk profile similar to PPIs.
Long-term safety data for P-CABs are limited but, at present, appear similar to PPIs.
Any PPI adverse event that is a result of the suppression of gastric acid secretion (eg, enteric infection) would be expected to occur with P-CABs as well.
To date, no human studies have directly evaluated the safety of P-CABs in pregnancy or in mothers who are breastfeeding.
Panel Recommendations on How to Use Vonoprazan in Clinical Practice
The following statements on how to use vonoprazan in clinical practice are those deemed appropriate by vote of the panel of experts (ie, those for which the median score was ≥7 and percent agreement was ≥80%) after review of the above data. The statements are limited to vonoprazan because it is presently the only P-CAB available for use in the United States.
Both vonoprazan and PPIs are effective for the healing of all grades of erosive esophagitis.
For the healing of LA Grade C or D esophagitis, vonoprazan may be superior to a PPI given in conventional, once-daily dosage.
For the healing of erosive esophagitis, the choice between vonoprazan or a PPI should be based on considerations of known allergies to or adverse events with individual agents, erosive esophagitis severity, cost, and patient preferences.
Both vonoprazan and PPIs are effective for the maintenance of healing of all grades of erosive esophagitis.
For the maintenance of healing of erosive esophagitis, the choice between vonoprazan or a PPI should be based on considerations of known allergies to or adverse events with individual agents, erosive esophagitis severity, cost, and patient preferences.
For the maintenance of healing of LA Grade C or D esophagitis, vonoprazan may be superior to a PPI given in conventional, once-daily dosage.
For the relief of symptoms in patients who have heartburn without erosive esophagitis on endoscopy, the choice between vonoprazan or a PPI should be based on considerations of known allergies to or adverse events with individual agents, cost, and patient preferences.
For patients with proven GERD whose symptoms or erosive esophagitis persist despite PPI treatment, a trial of vonoprazan may be considered.
Finally, the panel discussed several special situations in which physicians might consider prescribing P-CABs because the gastric acid suppression needed might be greater than that achieved by PPIs, even when they are given in high dosage. One such situation is when intestinal metaplasia persists despite PPI treatment in patients undergoing endoscopic eradication therapy for Barrett’s esophagus. Ongoing acid reflux is a well-established cause of failure to achieve complete eradication of intestinal metaplasia, 75 and abnormal acid reflux persists in >20% of patients with long-segment Barrett’s esophagus who are treated with twice-daily PPIs. 76 The acid hypersecretion of Zollinger-Ellison syndrome can sometimes require PPIs administered in very high dosages, 77 a problem that might be obviated by treatment with P-CABs. Patients with severe esophagitis due to scleroderma or to achalasia treated by per-oral endoscopic myotomy (POEM) occasionally can have reflux disease refractory to PPI therapy,78,79 and there are data to suggest that acid reflux that persists despite PPI treatment can lead to pulmonary decompensation in patients with interstitial lung disease and lung transplants. 80 Although P-CABs might well have a role in these special situations, the absence of published data precludes specific panel recommendations regarding the prescription of P-CABs for these conditions at this time.
Footnotes
Appendix A
Appendix B
Abbreviations
AE: Adverse event
AFS: American Foregut Society
CYP2C19: Cytochrome P450 2C19 enzyme
ECL cell: Enterochromaffin-like cell
EE: Erosive esophagitis
G cell: Gastrin cell
GERD: Gastroesophageal reflux disease
OE: Omeprazole equivalent
LA grade: Los Angeles grade
NERD: Non-erosive reflux disease
P-CAB: Potassium-competitive acid blocker
PPI: Protein pump inhibitor
RCT: Randomized controlled trial
SAE: Serious adverse event
SUCRA: Safety using surface under the cumulative ranking
TEAE: Treatment emergent adverse event
Author Contributions
Stuart Spechler: Overall project leader, working group leader, review of pertinent literature, Delphi Method participant, analysis and interpretation of data, critical revision of manuscript, important intellectual content, primary drafter of manuscript
Jennifer M. Kolb: RAND/UCLA Appropriateness Methodology expert, Delphi Method participant, working group member, review of pertinent literature, analysis and interpretation of data, critical revision of manuscript, important intellectual content, drafting of manuscript
Kerry Dunbar: working group leader, review of pertinent literature, Delphi Method participant, analysis and interpretation of data, critical revision of manuscript, important intellectual content
C. Prakash Gyawali: working group leader, review of pertinent literature, Delphi Method participant, analysis and interpretation of data, critical revision of manuscript, important intellectual content
Reginald Bell: working group member, review of pertinent literature, Delphi Method participant, analysis and interpretation of data, critical revision of manuscript, important intellectual content
Ronnie Fass: working group member, review of pertinent literature, Delphi Method participant, analysis and interpretation of data, critical revision of manuscript, important intellectual content
Philip Katz: working group member, review of pertinent literature, Delphi Method participant, analysis and interpretation of data, critical revision of manuscript, important intellectual content
John Lipham: working group member, review of pertinent literature, Delphi Method participant, analysis and interpretation of data, critical revision of manuscript, important intellectual content
Felice Schnoll-Sussman: working group member, review of pertinent literature, Delphi Method participant, analysis and interpretation of data, critical revision of manuscript, important intellectual content
Rhonda F. Souza: working group member, review of pertinent literature, Delphi Method participant, analysis and interpretation of data, critical revision of manuscript, important intellectual content
Rena Yadlapati: working group member, review of pertinent literature, Delphi Method participant, analysis and interpretation of data, critical revision of manuscript, important intellectual content
Peter J. Kahrilas: working group leader, review of pertinent literature, Delphi Method participant, analysis and interpretation of data, critical revision of manuscript, important intellectual content
Declaration of Conflicting Interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Stuart Spechler has served as a consultant for Takeda Pharmaceuticals, Phathom Pharmaceuticals, Castle Biosciences, and Lucid Diagnostics.
Jennifer M. Kolb has served as a consultant for Castle Biosciences, and as research support for Exact Sciences.
Kerry Dunbar has no disclosures.
Prakash Gyawali has served as a consultant for Medtronic, Diversatek, Braintree, Phathom Pharmaceuticals; and as a speaker for Carnot Laboratories.
Reginald Bell has served as a consultant for Castle Biosciences, Intuitive, Ethicon, Medtronic, and Implantica.
Ronnie Fass has served as a consultant to Medtronic, Phathom Pharmaceuticals, GERDCare, Celexio, Syneos, GIE Medical, BrainTree Labs/Sebela and Renexxion; and as a speaker for Takeda, Eisai Pharmaceuticals, Medicamenta, Carnot, and Daewoong.
Philip Katz has served as a consultant for Phathom Pharmaceuticals, Sebella, and Medtronic.
Loren Laine has served as a consultant for Phathom Pharmaceuticals.
John Lipham has served as a consultant for Ethicon, Castle Bioscience, Phathom Pharmaceuticals, Laborie, and Implantica.
Felice Schnoll-Sussman has served as a consultant for Medtronic, Braintree/Sebela, Ethicon, Phathom Pharmaceuticals, and Implantica
Rhonda Souza has served as a consultant for Castle Biosciences, Phathom Pharmaceuticals, Capsulomics, Lucid Diagnostics, and has received research support from Sanofi.
Rena Yadlapati has served as a consultant for Medtronic, Phathom Pharmaceuticals, StatLinkMD, Braintree Pharmaceuticals, and Reckitt Benckiser Healthcare Ltd; she has received research support from Ironwood Pharmaceuticals; and she is on the Advisory Board with Stock Options of RJSMediagnostix.
Peter J. Kahrilas is on the Advisor Board for Reckitt; has served as a consultant for Implantica, Johnson & Johnson, and Reckitt; and is on the Speakers Bureau for Phathom Pharmaceuticals.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by an unrestricted grant from Phathom Pharmaceuticals, which had no input or access to any aspect of the report during its formulation.
Use of Artificial Intelligence
Artificial intelligence was not used in the drafting of this manuscript.