Abstract
Current treatments for gastroesophageal reflux disease (GERD) and other acid-related disorders either block stimulation of the parietal cell (H2-receptor antagonists) or inhibit gastric H+/K+-ATPase (proton pump inhibitors; PPIs). PPIs are usually very effective in healing esophageal erosions and improving heartburn in patients with GERD. However, the PPIs have a slow onset of action and do not provide complete symptom resolution in all patients. Therefore, there is a need for agents that offer a fast onset of action and sustained acid suppression to maximize both mucosal healing and symptom control. Potassium-competitive acid blockers (P-CABs) are a relatively novel class of acid suppressants showing potential for the management of GERD and other acid-related disorders. This article focuses on the likely role of P-CABs for treating GERD.
Keywords
Introduction
Gastroesophageal reflux disease (GERD) has an estimated prevalence of between 18.1% and 27.8% in the United States.1,2 In GERD, disruption of the gastroesophageal junction barrier results in excessive exposure of the esophagus to acidic gastric contents. The most typical symptoms of GERD are heartburn and regurgitation. GERD may also present with atypical features such as chronic cough, chest pain, dental erosions, laryngitis or asthma.3-5 GERD may be classified as erosive esophagitis (EE), non-erosive reflux disease (NERD), and Barrett esophagus (BE). 6 Of these 3 phenotypes, NERD is the most prevalent (60-70%), followed by EE (30%) and BE (6-12%).1,6,7
Proton pump inhibitors (PPIs), along with appropriate lifestyle modifications in specific patients, are currently the mainstay for the management of GERD. PPIs are widely available in prescription and over the counter formulations and have a generally favorable safety profile. However, among their limitations are a short pharmacokinetic half-life, acid-lability, a relatively slow onset of action, ability to inhibit only those molecules of proton pump (H+/K+-ATPase) that are in their active form on the membrane of the parietal cell secretory canaliculi rather than in their inactive form in the cytosol, and variable pharmacodynamic effects due to different genetically-determined polymorphisms of cytochrome P450 2C19, which is the isozyme responsible for most PPI metabolism.8,9
These unmet needs of PPIs have fostered the development of the potassium-competitive acid blockers (P-CABs) that block gastric H+/K+-ATPase by reversible and potassium-competitive, ionic (non-covalent) binding, thus inhibiting acid production. These features overcome some of the limitations of PPIs by delivering rapid, potent and sustained acid suppression; consequently, they address some of the unmet needs in patients with GERD. In this review, we provide an assessment of the P-CABs in the management of GERD.
History of Development of P-CABs
Gastric H+/K+-ATPase has a high affinity for potassium ions (K+) during the acid secretory process. Therefore, drugs that compete for K+ binding sites have the potential to inhibit acid secretion. P-CABs were initially developed based on this premise due to their fast, efficient and reversible blockade of H+/K+-ATPase. The prototypical P-CAB—SCH28080—was developed in the 1980s. It inhibited gastric acid secretion in humans through K+-competitive inhibition of gastric H+/K+-ATPase.10-12 However, its clinical development was stopped due to concerns about hepatotoxicity. This fostered research into several other P-CABs that can be further classified as imidazopyridines (eg, AZD0865 and BY841), imidazo-thienopyridines (SPI-447), imidazo-naphthyridine (eg, soraprazan), or quinolines (eg, SK&F96067 and SK&F97574), pyrrolo-pyridazine derivatives (CS-526), pyrimidine derivatives (revaprazan YH-1885), and pyrrole derivatives (vonoprazan).13-19 Table 1 summarizes the history and development of the P-CABs.
The history and development of P-CABs.
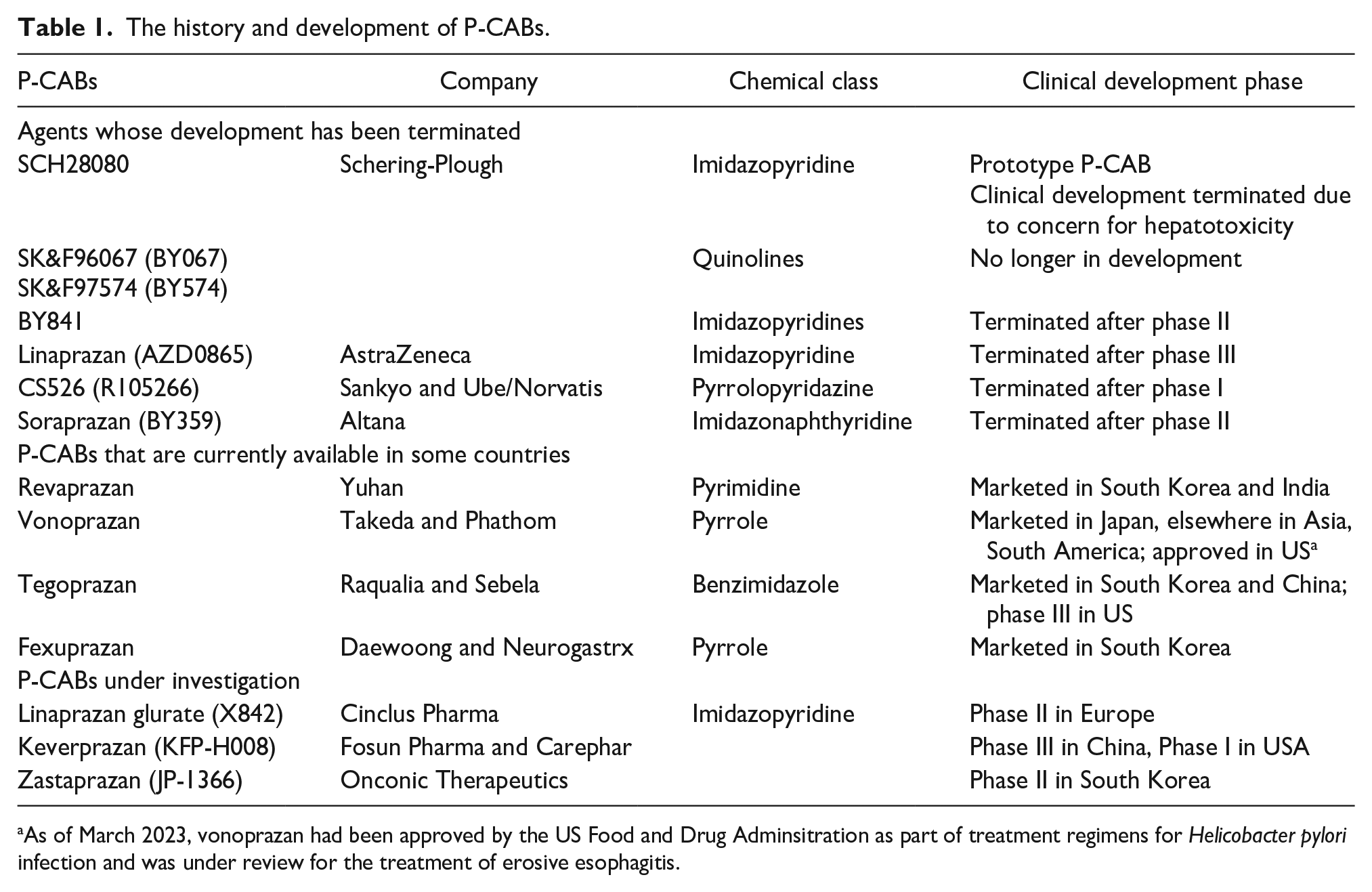
As of March 2023, vonoprazan had been approved by the US Food and Drug Adminsitration as part of treatment regimens for Helicobacter pylori infection and was under review for the treatment of erosive esophagitis.
Mechanism of Action
P-CABs competitively block the potassium binding site of the H+/K+ ATPase transporter on the luminal membrane of parietal cells.20,21 They are lipophilic, weak bases with high pKa values, and are stable at low pH. 21 These properties allow P-CABs to concentrate in parietal cell canaliculi and bind ionically to H+/K+-ATPase transporters to block hydrogen ion (H+) secretion. Once bound, P-CABs block access of K+ ions to H+/K+-ATPase.
PPIs are prodrugs that require chemical conversion within parietal cells to produce their active forms. P-CABs do not require chemical conversion and act more rapidly on H+/K+-ATPase. Since they are acid-stable, they do not require any enteric coating. Their antisecretory activity is independent of mealtimes.22-24 These characteristics aid their rapid onset of action and result in competitive, reversible inhibition of acid secretion.22,23 In addition, the more prolonged pharmacokinetic half-lives of P-CABs allow sustained inhibition of newly synthesized molecules of H+/K+-ATPase, thus providing longer durations of action than PPIs. 22 Table 2 details the main characteristics and differences between PPIs and P-CABs.
Characteristics of, and Differences Between P-CABs and PPIs.
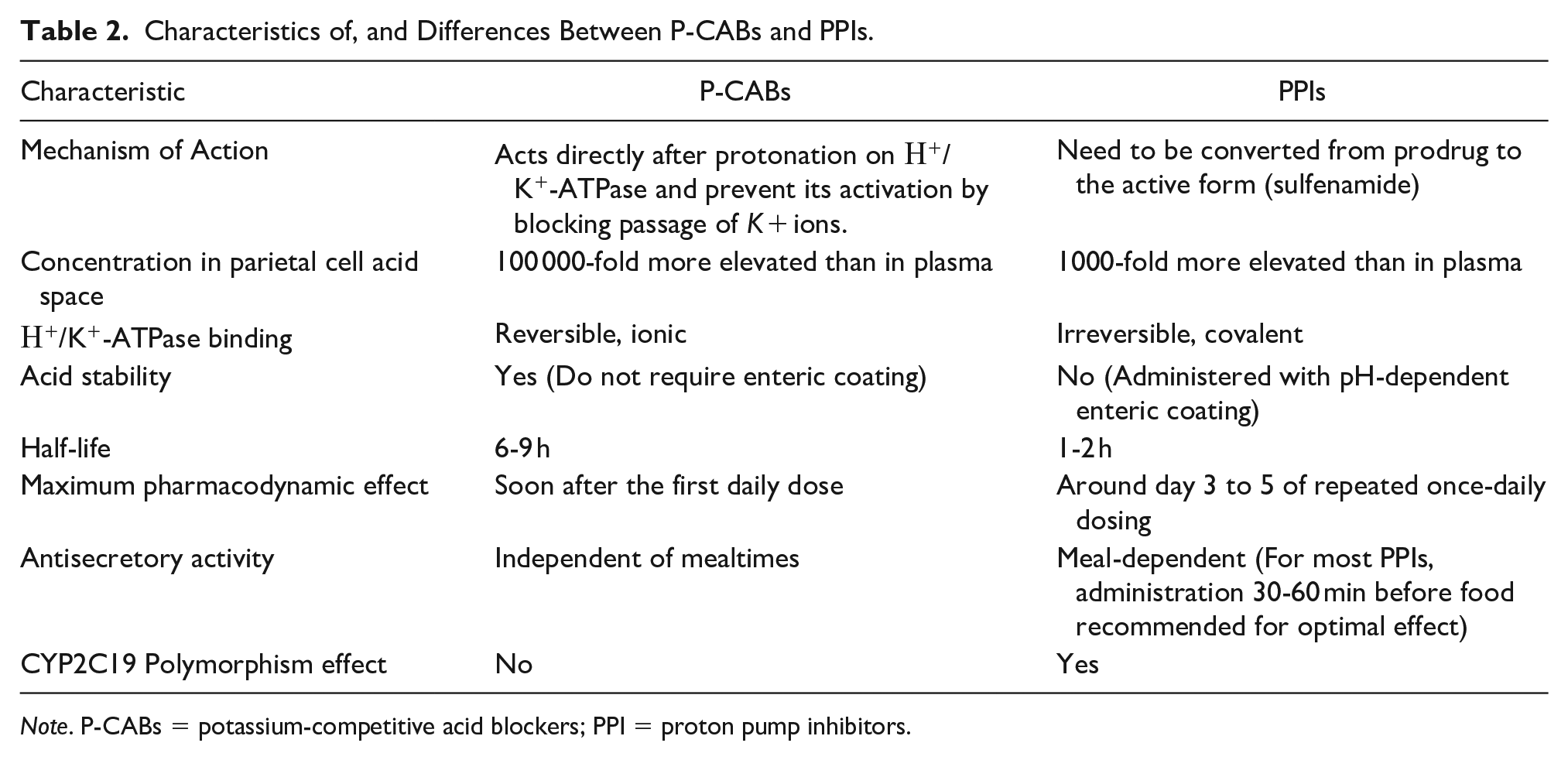
Note. P-CABs = potassium-competitive acid blockers; PPI = proton pump inhibitors.
Pharmacodynamics of P-CABs Compared to PPIs
P-CABs have generally shown greater reduction in intragastric acidity and more rapid onset of action than comparator PPIs. One outcome measure that has typically been reported is the pH4 holding time ratio (pH4 HTR), which is the proportion of a 24-hour recording period during which the mean intragastric pH was held above 4. Maintenance of intragastric pH above 4 may be an important determinant of healing of EE.25,26
Vonoprazan 20 mg was compared with esomeprazole 20 mg and rabeprazole 10 mg in healthy Japanese subjects. 27 Vonoprazan 20 mg once-daily for 7 days maintained intragastric pH above 4 for 85.8% to 93.8% of the 24-hour recording period. In a separate study in predominantly Caucasian subjects, 7 days of vonoprazan 20 mg once-daily maintained intragastric pH above 4 for 85.3% of the 24-hour period. 28
In a US-based study, healthy subjects received vonoprazan 20 mg once-daily and lansoprazole 30 mg once-daily in random order, with each given for 7 days and separated by a washout period. 29 On the first day of dosing, vonoprazan 20 mg raised mean intragastric pH to above 4 within 4 hours of administration. On day 7 of once-daily dosing, vonoprazan 20 mg maintained mean intragastric pH above 4 for 21.1 hours of the 24-hour recording period, compared with 10.2 hours with lansoprazole 30 mg (P < .0001).
Tegoprazan 100 or 200 mg was administered once-daily for 7 days to eight healthy Korean men and compared with esomeprazole 40 mg once-daily. 30 Intragastric pH rose substantially and rapidly with tegoprazan. The mean percent time with intragastric pH > 4 over 24 hours was higher on day 7 than on day 1, with mean values of 70.4% and 62.3% with 100 mg, and 94.6% and 76.8% with 200 mg. In Korean subjects, tegoprazan 50 mg once-daily and revaprazan 200 mg once-daily for 7 days maintained intragastric pH above 4 for 68.2% and 25.3%, respectively, of the 24-hour recording period. 31
Multiple once-daily doses of fexuprazan 40 mg maintained intragastric pH above 4 for 64.3%, 62.8% and 70.3% of the 24-hour recording period in Korean, Caucasian and Japanese subjects, respectively. 32
Although P-CABs have usually been administered in the morning, late evening dosing has been examined preliminarily. In a crossover study, healthy Korean subjects received single doses of tegoprazan 50 mg, vonoprazan 20 mg and esomeprazole 40 mg in random order and administered at around 10 PM. 33 Intragastric pH was monitored continuously for 12 hours after dosing. From approximately 10 PM until 10 AM on the following day, intragastric pH was above 4 for 66.0% of the time with tegoprazan 50 mg, 60.5% with vonoprazan 20 mg, and 36.1% with esomeprazole 40 mg.
P-CABs in the Management of GERD
GERD remains among the most common conditions seen by gastroenterologists and one of the most common gastrointestinal disorders managed by primary care physicians. Although PPIs have been the mainstay of treatment for GERD, they have a relatively slow onset of action and do not provide complete symptom resolution in all patients. P-CABs represent a new class of acid suppressants with potential for the pharmacologic management of GERD.
Erosive Esophagitis (EE)
The main treatment goals in EE are to alleviate symptoms, heal esophageal erosions, maintain healing, and improve overall quality of life. Treatment of EE comprises both initial treatment to heal the condition and subsequent maintenance for prevention of relapse.
P-CABs in the Initial Treatment of EE
Randomized controlled trials (RCTs) evaluating patients with Los Angeles (LA) grades A-D EE for assessing the safety and efficacy of vonoprazan have generally shown non-inferiority to a PPI (most typically, lansoprazole 30 mg) with 8-week healing rates above 90% and no difference in the rates of treatment-emergent adverse events (TEAEs)34-36 (Table 3). However, Ashida et al 34 reported significantly higher 8-week healing rates on vonoprazan than lansoprazole among patients who had LA grades C or D EE; these were 98.7% and 87.5%, respectively (P = .0082). Vonoprazan was also associated with more rapid healing of EE than lansoprazole as judged by higher healing rates after 2 weeks of treatment.
Summary of P-CABs in the Initial Treatment of EE.
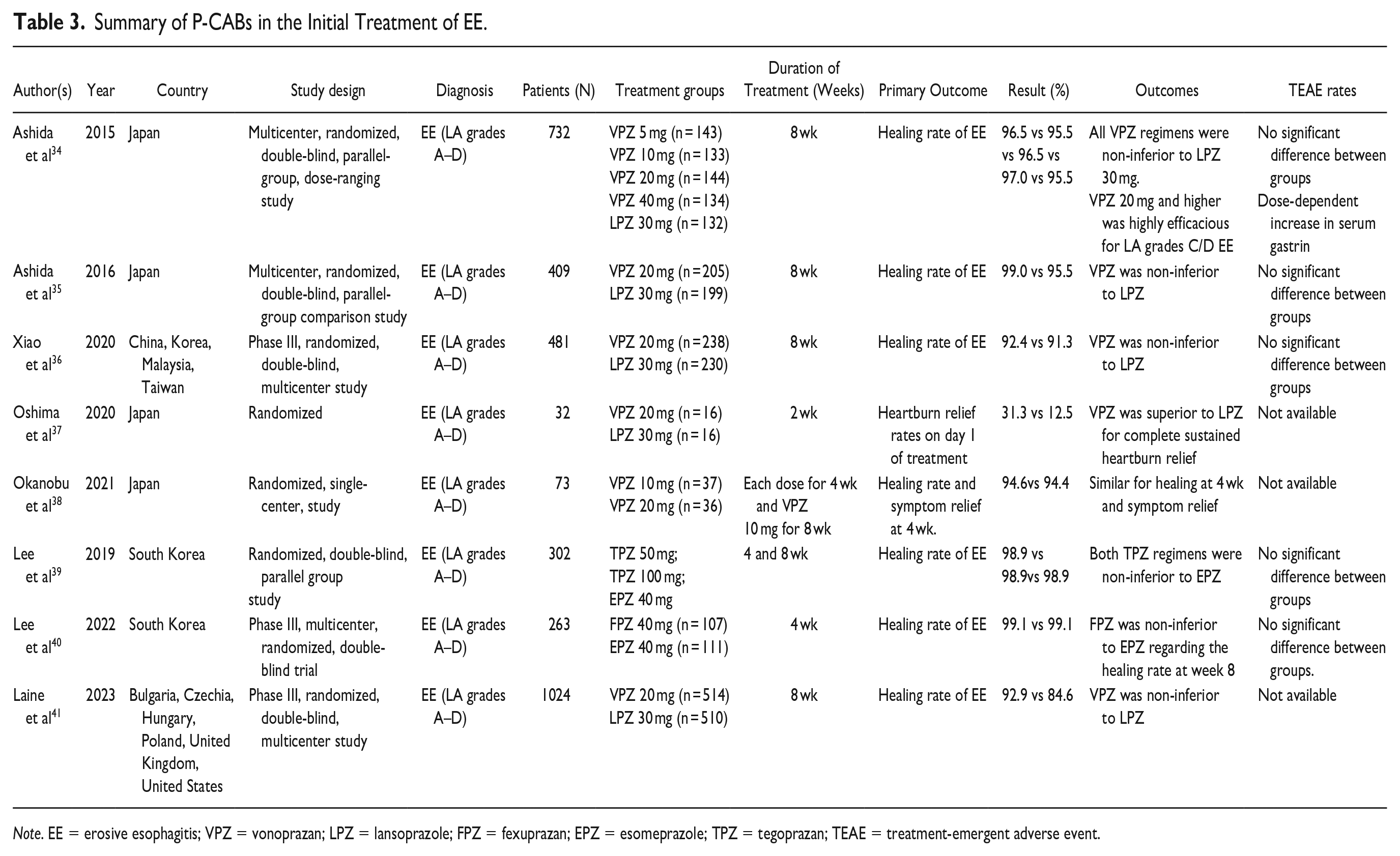
Note. EE = erosive esophagitis; VPZ = vonoprazan; LPZ = lansoprazole; FPZ = fexuprazan; EPZ = esomeprazole; TPZ = tegoprazan; TEAE = treatment-emergent adverse event.
Oshima et al 37 evaluated 32 patients with EE who had heartburn at least once a week and compared the safety and efficacy of vonoprazan to lansoprazole. There was complete heartburn resolution in 31.3% and 12.5% of patients on day 1 with vonoprazan and lansoprazole, respectively. During the first week of therapy (days 2-7), vonoprazan also provided more sustained complete heartburn relief and significantly more patients achieved complete heartburn relief with vonoprazan than lansoprazole (HR 3.58, 95% CI 1.16-11.08, P < .05).
In a randomized study, Okanobu et al 38 evaluated the efficacy of vonoprazan 20 and 10 mg in 73 patients with EE. After 4 weeks, healing rates with 20 and 10 mg were 94.6% and 94.4%, respectively. The frequency scale for GERD symptoms was reduced similarly in both groups.
Phase III, multicenter, RCTs from South Korea that have evaluated the safety and efficacy of once-daily administration of tegoprazan and fexuprazan compared to esomeprazole have shown non-inferiority with EE healing rates above 90% and similar tolerability.39,40
The phase III “PHALCON-EE” trial 41 evaluated vonoprazan 20 mg once-daily and lansoprazole 30 mg once-daily for up to 8 weeks in 1024 patients with EE in the United States and Europe. This large trial utilized central reading of endoscopic images for confirmation of the presence and grade of EE prior to patient enrollment, and subsequent confirmation of healing or non-healing at the end of treatment. Overall, vonoprazan was non-inferior to lansoprazole in the primary analysis of EE healing rates at 8 weeks; these were 92.9% and 84.6%, respectively (P < .0001 for non-inferiority). Vonoprazan was superior to lansoprazole in some pre-defined secondary or exploratory analyses. Healing rates at 2 weeks among patients with LA grades C or D EE were 70.2% and 52.6%, respectively (P = .0008). Overall, healing rates across all grades of EE at week 2 were 74.3% for vonoprazan and 68.2% for lansoprazole (P = .0174). Healing rates at 8 weeks between patients with LA grades C or D EE were 91.7% for vonoprazan and 72.0% for lansoprazole (P < .0001). While the efficacy of vonoprazan was similar across all grades of EE, its superiority over lansoprazole was established for grades C and D. For control of GERD symptoms, vonoprazan was non-inferior to lansoprazole in the number of heartburn-free days and was not superior to lansoprazole in sustained heartburn resolution by day 3 of treatment.
Further details of trials of P-CABs in the initial treatment of EE are given in Table 3. With the exception of “PHALCON-EE,” 41 all the other trials listed in Table 3 were conducted in Asia.
P-CABs in the Maintenance Treatment of EE
The study by Ashida et al 35 evaluated patients with EE during initial treatment and in the subsequent maintenance phase. Patients with healed EE were randomized to 52 weeks’ maintenance treatment with vonoprazan 10 or 20 mg once-daily. EE recurred in fewer than 10% of patients, and vonoprazan was well tolerated. Most TEAEs were mild, and did not increase in incidence over time. The most common TEAE was nasopharyngitis.
A randomized, phase III study by Ashida et al 42 evaluated the effectiveness of vonoprazan and lansoprazole for the maintenance of healing of EE. Recurrence rates were 16.8% for lansoprazole 15 mg, 5.1% for vonoprazan 10 mg and 2.0% for vonoprazan 20 mg.
A phase III, double-blind, parallel group, multicenter study from China compared the safety and efficacy of vonoprazan with lansoprazole for the maintenance of healed EE. 43 Recurrence rates at 24 weeks were 13.8%, 14.0%, and 25.0% for vonoprazan 10 mg, vonoprazan 20 mg and lansoprazole 15 mg, respectively. Both vonoprazan dose regimens were superior to lansoprazole in preventing recurrence.
The phase III “PHALCON-EE” trial evaluated 878 patients with healed EE. Patients with documented healing on vonoprazan 20 mg once-daily or lansoprazole 30 mg once-daily were re-randomized 1:1:1 to double-blinded maintenance treatment with vonoprazan 10 or 20 mg, or lansoprazole 15 mg once-daily for 24 weeks. The primary endpoint was maintenance of remission at 24 weeks. Both vonoprazan doses were non-inferior to lansoprazole in the primary analysis (vonoprazan 20 mg: 80.7%; vonoprazan 10 mg, 79.2%; lansoprazole, 72.0%; P < .0001 for non-inferiority for both comparisons with lansoprazole). Both doses of vonoprazan were superior to lansoprazole for maintaining healing of EE among patients who initially had LA grades C or D EE. At 24 weeks, these were 77.2% with vonoprazan 20 mg, 74.7% for vonoprazan 10 mg and 61.5% for lansoprazole 15 mg (vonoprazan 20 mg v lansoprazole, P = .020; vonoprazan 10 mg v lansoprazole, P = .049) 41 (Table 4).
Summary of P-CABs in the Maintenance Treatment of EE.
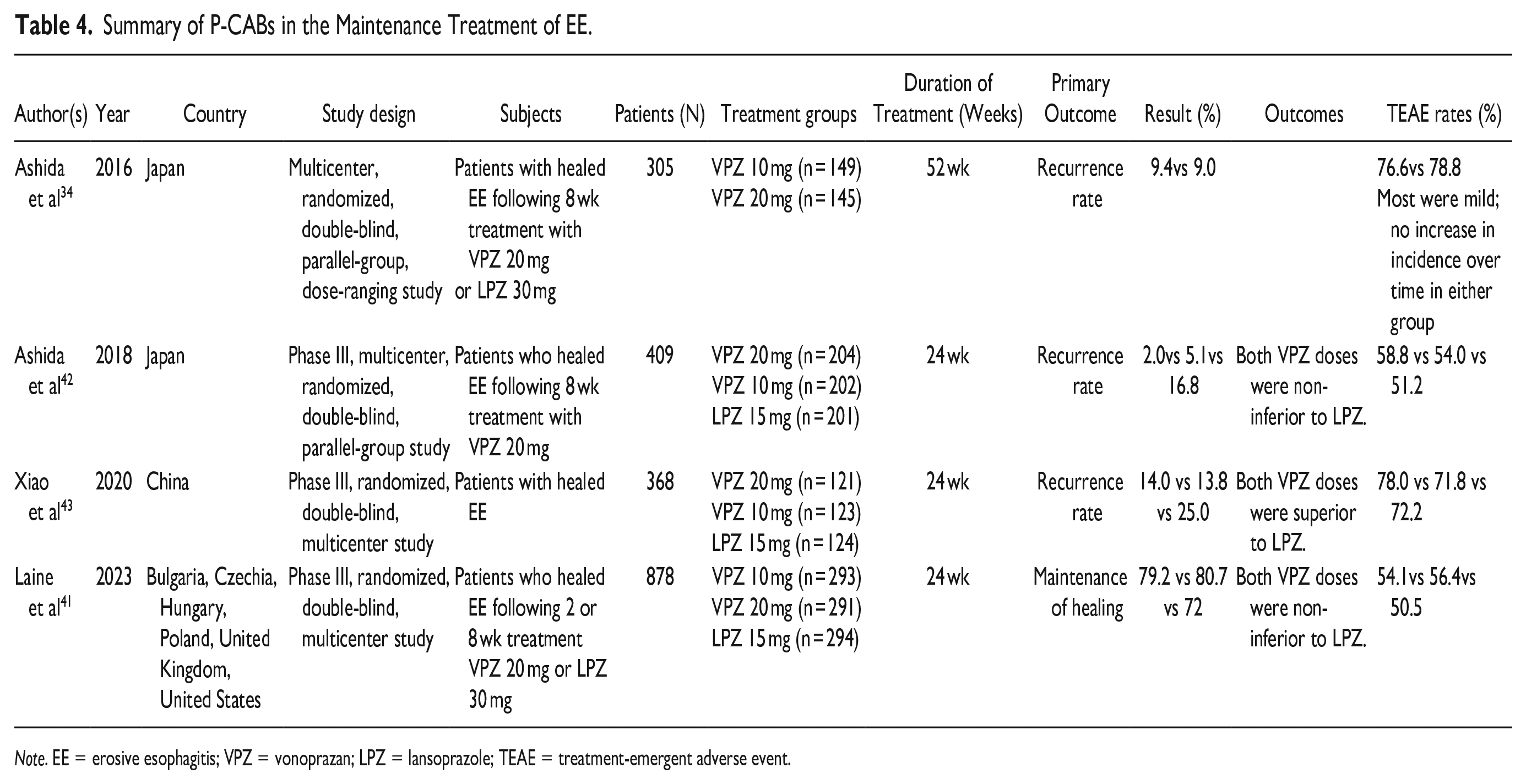
Note. EE = erosive esophagitis; VPZ = vonoprazan; LPZ = lansoprazole; TEAE = treatment-emergent adverse event.
P-CABs in the Treatment of PPI-Refractory GERD
Between 10% and 40% of patients with EE do not achieve complete symptom relief and / or mucosal healing despite double-dose PPI treatment for 8 weeks.44-46 This may be related to inadequate acid suppression, CYP2C19 polymorphism, acid hypersecretory syndromes, or poor compliance. Some patients may have symptoms from causes other than GERD.44,47
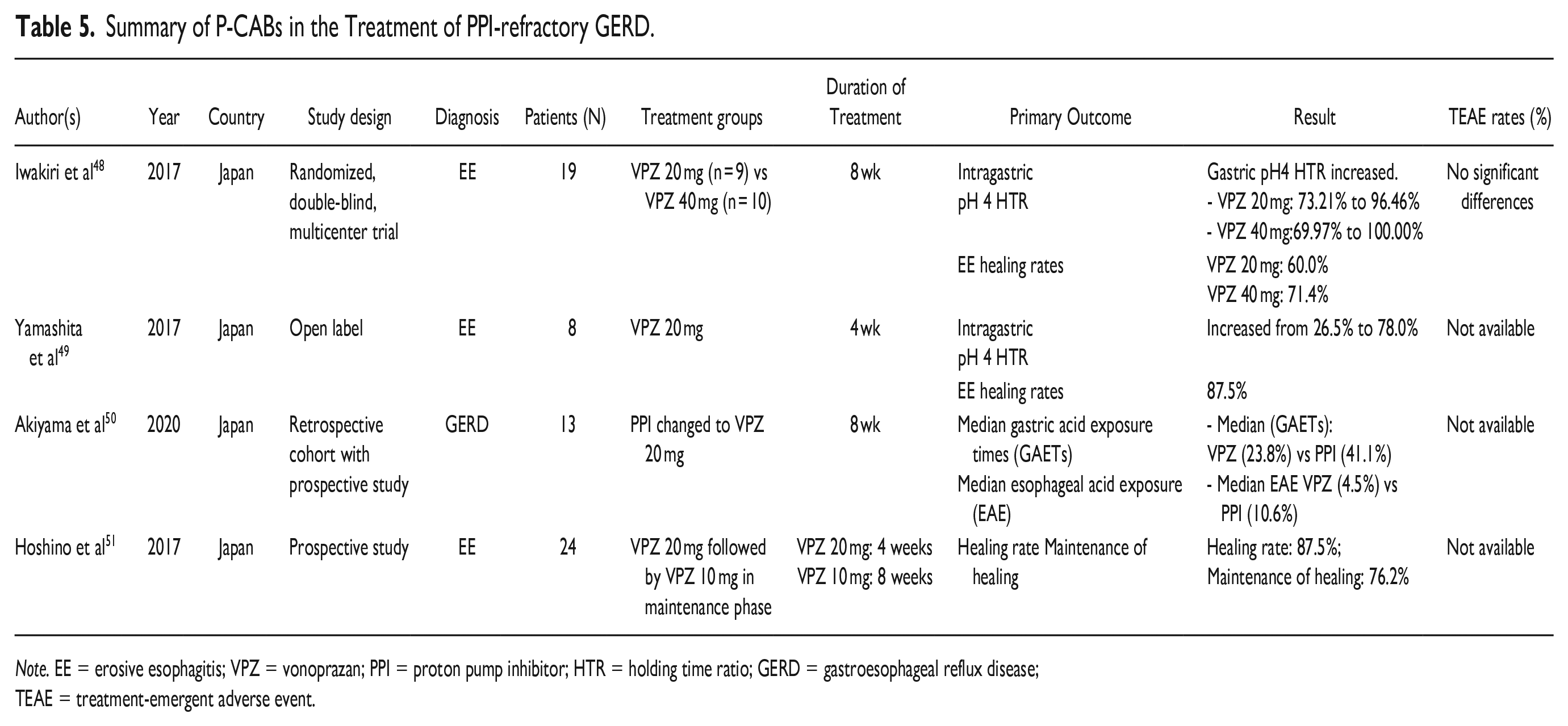
Iwakiri et al 48 evaluated 19 patients with PPI-resistant EE in a randomized, double-blind, multicenter trial. The authors studied changes in intragastric and intra-esophageal pH over 24 hours at pharmacodynamic steady state during the treatment period. They presented these as the percentage of time the pH was ⩾4, which was referred to as the pH 4 holding time ratio (HTR). It has been postulated that the persistence of EE despite PPI therapy is due to inadequate inhibition of gastric acid. They assessed the effects of vonoprazan 20 and 40 mg on 24-hour intra-esophageal and intragastric pH. Following initial treatment with lansoprazole 30 mg, 9 patients received vonoprazan 20 mg once-daily, and 10 received 40 mg once-daily for 8 weeks. There was a significant increase in the intragastric pH4 HTR; with vonoprazan 20 mg, this increased from 73.2% to 96.5%, and with vonoprazan 40 mg from 70.0% to 100%). In addition, inhibition of gastric acidity during the nighttime period (9 pm-9 am) by vonoprazan was maintained with pH 4 HTRs of 93% or greater. There was no difference in intra-esophageal pH 4 HTR between groups. The EE healing rate was 60.0% and 71.4% in the vonoprazan 20 and 40 mg groups, respectively. Similar findings were observed in studies of vonoprazan in healthy male subjects in Japan and the United Kingdom (UK).27,28
Yamashita et al 49 evaluated vonoprazan in 8 patients with PPI-refractory EE. Seven achieved healing after 4 weeks of treatment with vonoprazan 20 mg. Intragastric pH4 HTR increased from 26.5% to 78.0%.
Akiyama et al 50 investigated the effectiveness of vonoprazan on PPI-refractory GERD patients who had continued esophageal acid exposure. Thirteen of 124 patients underwent multichannel intraluminal impedance-pH monitoring at baseline (during PPI treatment) and after treatment with vonoprazan 20 mg. Both daytime and nocturnal median gastric acid exposure times were lower on vonoprazan (23.8%) than during PPI treatment (41.1%). Vonoprazan was superior to PPI treatment in suppressing gastric acidity, symptom relief, and healing of esophagitis in these patients.
Hoshino et al 51 evaluated 24 patients with PPI-resistant EE. Patients received vonoprazan 20 mg once-daily for 4 weeks, followed by vonoprazan 10 mg for an additional 8 weeks. The initial 4-week healing rate on vonoprazan 20 mg was 87.5% (21/24); healing was maintained over the additional 8 weeks in 76.2% (16/21) with vonoprazan 10 mg.
Additional details of studies of P-CABs in treating PPI-refractory GERD are given in Table 5.
Summary of P-CABs in the Treatment of PPI-refractory GERD.
Note. EE = erosive esophagitis; VPZ = vonoprazan; PPI = proton pump inhibitor; HTR = holding time ratio; GERD = gastroesophageal reflux disease; TEAE = treatment-emergent adverse event.
Nonerosive Reflux Disease (NERD)
As discussed above, the P-CABs have generally been found to be at least non-inferior to the PPIs in EE. On some comparisons, such as the speed of healing and the absolute healing rates of severe grades of EE, P-CABs have been shown to be superior to PPIs in EE. However, their benefits for GERD patients without visible mucosal injury (ie, NERD) may be less obvious.
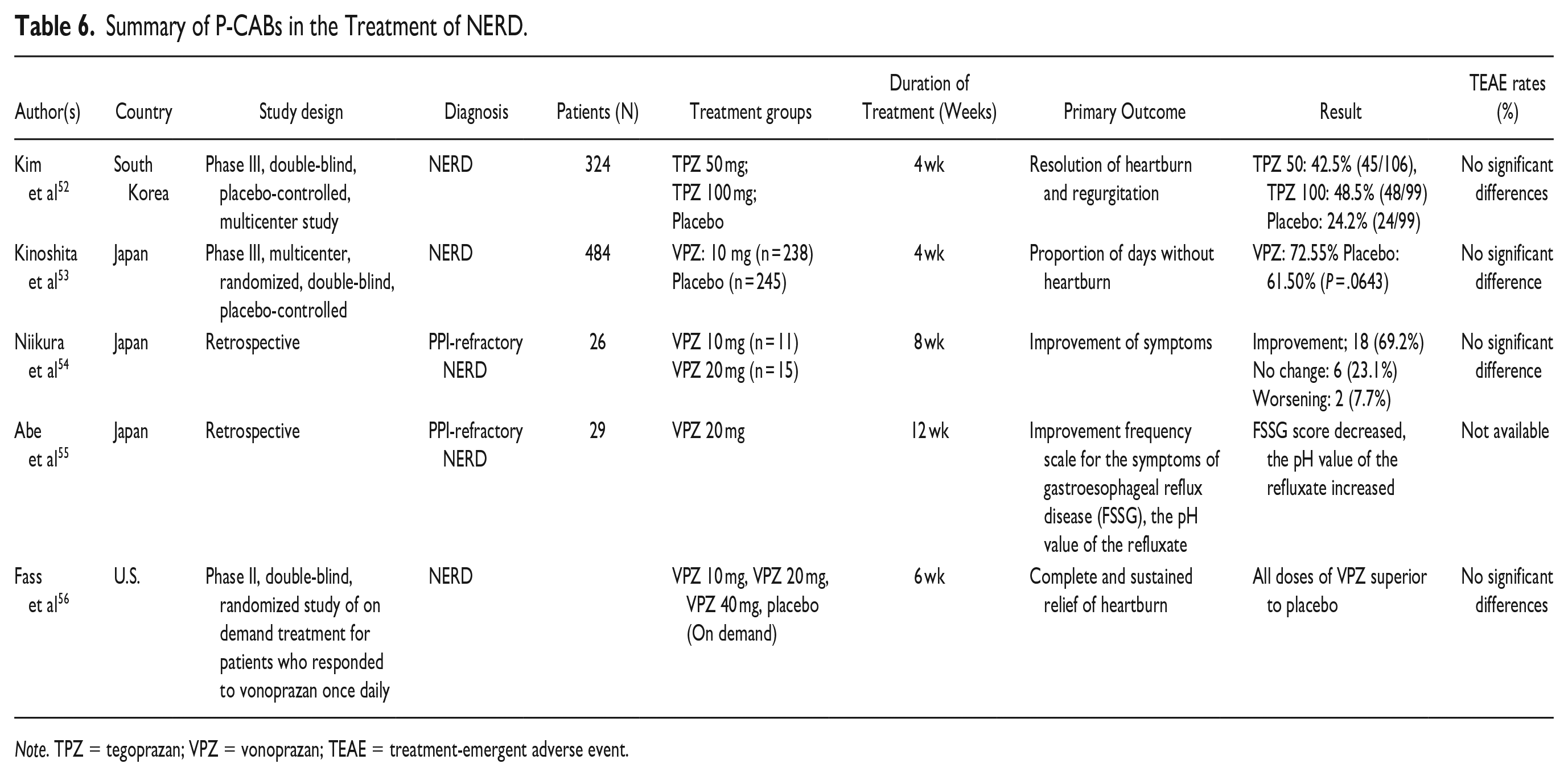
In a phase 3, double-blind, placebo-controlled, multicenter trial in Korea, 52 324 patients with NERD were randomized into 3 treatment groups (tegoprazan 50 mg, tegoprazan 100 mg and placebo). Both tegoprazan groups experienced higher rates of complete heartburn resolution at 4 weeks compared to placebo (42.5%-48.5% vs 24.2%, P < .05) with no significant difference in the incidence of TEAEs. 52
In a phase 3, multicenter, randomized, double-blind, placebo-controlled study of vonoprazan 10 mg daily compared with placebo in patients with NERD, the vonoprazan group experienced fewer episodes of heartburn at week four, but the difference was not statistically significant. 53 A retrospective study evaluated the efficacy of vonoprazan for GERD symptoms in patients with NERD who had an inadequate response to a PPI. After treatment was switched from a PPI to vonoprazan, 18 patients (69.2%) reported symptom improvement (P < .001). 54 Another retrospective study from Japan evaluated the efficacy vonoprazan 20 mg once-daily in patients with PPI-resistant NERD and showed inadequate symptom control with persistent reflux at pH 4 to 5. 55 These studies, which have been limited by small sample size, inclusion of patients with presumed NERD that was not confirmed by intra-esophageal pH testing, and restriction to Asian populations, have failed to show consistent benefits of P-CABs over PPIs in NERD. Further large scale RCTs need to evaluate the role of P-CABs for patients with confirmed NERD in non-Asian populations.
In the US, patients with presumed NERD who responded symptomatically to vonoprazan 20 mg once-daily for 4 weeks were then randomized to receive on-demand treatment with vonoprazan 10, 20 or 40 mg, or placebo once-daily for a further 6 weeks. 56 Each dose of vonoprazan was superior to placebo in achieving complete and sustained relief of episodes of heartburn. There was no significant difference in the rates of TEAEs among the groups—although the placebo group reported the most.
Details of individual trials are given in Table 6.
Summary of P-CABs in the Treatment of NERD.
Note. TPZ = tegoprazan; VPZ = vonoprazan; TEAE = treatment-emergent adverse event.
In a preliminary report of a phase 3 trial in NERD 57 once-daily vonoprazan 10 or 20 mg was superior to placebo in the proportion of 24-hour, heartburn-free days over 4 weeks (P < .0001 for each comparison with placebo). At the time of writing, that study had not yet been peer-reviewed.
Safety of P-CABs
Although PPIs have a favorable safety profile and have been used for decades, their long-term use has been associated with possible adverse effects (AEs) including Clostridioides difficile infection, community-acquired pneumonia, bone fractures, hypomagnesemia, chronic kidney disease, vitamin B12 deficiency, an increased risk of acquisition of Covid-19, and dementia.58-60 However, for virtually all of these suspected adverse consequences, supporting evidence is weak and inadequate to establish causality. 61 In the only RCT to report prospective safety data, 3 years of daily PPI use was not associated with higher rates of AEs than placebo except for a small but statistically significant increase in the risk of bacterial enteritis. 62
Less is currently known about the long-term safety of P-CABs and most safety data are from studies of vonoprazan. To date, this has shown excellent short- to medium-term safety that is comparable to that of PPIs. In a phase 3 multicenter clinical trial in Japan, the safety profiles of vonoprazan and lansoprazole were comparable, and nasopharyngitis was the most frequent AE in each treatment group. 42 A meta-analysis by Cheng et al 63 comparing vonoprazan and PPIs for GERD showed similar safety outcomes with a risk ratio of adverse events of 1.08 (95% CI 0.96-1.22). A multicenter, randomized controlled trial in Korea comparing tegoprazan and esomeprazole for EE showed similar rates of AEs with each treatment. 39 In a 4-week trial in patients with NERD, the rates of TEAEs seen with tegoprazan were similar to those on placebo. 52 Meta-analyses of trials of Helicobacter pylori treatment, and clinical trials of peptic ulcer prevention, have shown comparable rates of AEs between vonoprazan and PPI-based treatment regimens.64-67
However, since GERD is a chronic and potentially life-long condition, long-term safety data for the P-CABs are essential. The highly awaited “VISION” study has been investigating the safety of vonoprazan as maintenance treatment for patients with healed EE over 5 years. 68 A 4-year interim analysis of this study was presented at DDW in 2022. Approximately 80% of the enrolled patients had received either vonoprazan 10 or 20 mg, or lansoprazole 15 or 30 mg once-daily beyond 4 years. 69 No neoplastic changes had occurred in either group at 4 years. The incidences of gastric polyps, multiple white flat lesions, and black spots on the gastric mucosa were higher in the lansoprazole group; the incidence of “cobblestone mucosa” was higher in the vonoprazan group. Mean serum gastrin and chromogranin A levels were consistently higher on vonoprazan than lansoprazole; unfortunately, absolute values were not included in the abstract. The clinical relevance of these observations (if any) is uncertain. However, maintenance treatment with vonoprazan in these patients was generally well tolerated with few serious AEs, no alarming changes to the gastric mucosa, and no signal suggesting any augmented risk of gastric neuroendocrine tumors in comparison with lansoprazole. 69 Despite the excellent safety record of the P-ACBs to date, regulatory authorities including the US FDA will require monitoring of these agents—particularly when used long-term for such indications as maintenance of healing of EE. Although, as noted above, mean serum gastrin levels have been higher on P-CAB than PPI treatment, the magnitude of the mean difference has been relatively small, and this is unlikely to have major implications for their use. While we do not anticipate major differences in safety between Asian and non-Asian populations, this will require prospective confirmation.
As with PPIs, the long-term use of P-CABs may predispose patients to enteric infections by inducing dysbiosis and changes in the gut micribiota. 70 Studies on P-CAB effects on microbiota showed that vonoprazan causes changes that may decrease defense against enteric infections such as Clostridioides difficile. 71 Literature on P-CABs and their effect during pregnancy and lactating women is currently scant. In an animal study, vonoprazan exposure levels comparable to those in humans did not show any maternal or fetal toxicity. 72 Long-term safety data, including in non-Asian populations, are required. Based on the currently limited evidence, the overall safety of P-CABs during pregnancy appears comparable with that of PPIs.
Summary and Conclusions
P-CABs produce more profound inhibition of gastric acid secretion than PPIs and also offer other potential advantages including a more rapid onset of action, lack of a food effect, and lack of any influence on their effect due to CYP2C19 polymorphisms.
As of May 2023, different P-CABs had been approved for the management of GERD in multiple countries in Asia and Latin America. At the time of writing, vonoprazan was under review by the U.S. Food and Drug Administration (FDA) for the healing and symptom relief of EE and for the maintenance of EE healing.
The benefits of P-CABs over PPIs are most obvious in EE, where there is a defined relationship between suppression of acidity and the objective measure of endoscopically documented healing of esophageal erosions. 25 Trials conducted in Asian countries have, overall, shown non-inferiority of P-CABs to PPIs. However, this may reflect the fact that most patients in those trials had mild grades of EE. PPIs remain extremely effective for healing EE of LA grades A or B. Since the therapeutic gain of a P-CAB over a PPI in mild EE is small to non-existent, this would have diluted the overall efficacy of a P-CAB in EE. However, superior efficacy of P-CABs over PPIs has been documented in patients with severe EE (ie, LA grades C or D) with both higher absolute healing rates and a shorter duration of time to achieve complete healing. This is likely to be clinically relevant since complications of EE including bleeding and stricture formation are most likely to occur on a background of unhealed severe EE. Therefore, it is appropriate to effect healing of severe EE as reliably as possible. Currently, there are few options for patients with severe EE that heals incompletely on PPI treatment, apart from administering the PPI on a twice-daily schedule, which is not approved by the FDA. The more rapid healing of EE achieved by P-CABs in comparison to PPIs is an attractive therapeutic concept. However, of most importance in a chronic condition such as EE, is the ability of P-CABs to maintain healing of severe EE more reliably than maintenance doses of PPIs.
The advantages of P-CABs over PPIs in NERD are less obvious than in EE. Clinical trials in NERD vary with respect to inclusion and exclusion criteria. Trials that included all patients with apparently typical heartburn and normal endoscopic appearances may have been encumbered by the unintentional inclusion of patients with esophageal hypersensitivity or functional heartburn. The former would not be expected to have a marked response to more profound acid suppression, while the latter would be expected to have no response. This may have influenced the results and interpretation of at least some trials of P-CABs in presumed “NERD.” Trials that require documentation of pathological gastroesophageal acid reflux with or without significant symptom-association probability before recruitment more accurately identify patients who truly have NERD. However, they are difficult, cumbersome and expensive to perform and do not reflect typical clinical practice.
The potential for on-demand use of P-CABs by patients with NERD is of interest and worthy of further study. While on-demand treatment is generally inappropriate for patients with EE, it may be adequate for some patients with NERD if they obtain a level of symptom control that is acceptable to them. 73 On demand use may be more cost-effective for some patients with NERD than continuous daily maintenance treatment and may be especially appropriate for those with only intermittent symptoms.
To date, the safety profile of the P-CABs has been excellent with similar rates of TEAEs as PPIs in clinical trials. The final results of longer-term studies of patients on continuous maintenance treatment with a P-CAB (eg, the “VISION” study mentioned above) are awaited with interest. Assuming the FDA approval of vonoprazan (and, potentially, other P-CABs) for EE, long-term safety data will help to guide therapeutic decisions.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Howden is a consultant for Phathom Pharmaceuticals.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
