Abstract
Introduction
Chronic kidney disease (CKD) is a prevalent condition characterized by a progressive decline in kidney function over time. It is associated with various complications, including cardiovascular disease and mortality. Inflammation, as indicated by elevated levels of high-sensitivity C-reactive protein (hs-CRP), has been implicated in the pathogenesis and progression of CKD. Serum creatinine (S. creatinine) levels, on the other hand, are commonly used as a marker to estimate kidney function.
Aim
The objective of this study was to examine the relationship between hs-CRP levels and S. creatinine levels in CKD patients. By investigating this association, this study aimed to shed light on the potential interplay between inflammation and kidney dysfunction in CKD.
Methods
We conducted a cross-sectional study in the Parul Sevashram Hospital, Vadodara. The study consisted of 80 CKD patients with 51 males and 29 females. The hs-CRP and creatinine levels were estimated for the study and further evaluated statistically for finding the association between them.
Results
The study observed a gender distribution with 63.75% male and 36.25% female patients. Among males, 41.17% had hs-CRP levels >2 mg/L, with a mean ± standard deviation (SD) creatinine value of 4.63 ± 0.32 mg/dL. In females, only 13.79% had hs-CRP levels >2 mg/L, with a mean ± SD creatinine value of 4.62 ± 0.30 mg/dL. The statistical analysis using Fisher’s exact test showed a highly significant association between hs-CRP and S. creatinine levels (P < .01).
Conclusion
This study demonstrated a strong association between hs-CRP levels and S. creatinine levels in patients with CKD. As S. creatinine levels increased, there was a corresponding rise in hs-CRP levels. This suggests that monitoring hs-CRP levels could be valuable in assessing the progression of renal dysfunction and predicting cardiovascular complications in CKD patients. Further research is needed to fully comprehend the underlying mechanisms driving this association.
Keywords
Introduction
Chronic kidney disease (CKD) has been recognized as a leading public health problem worldwide. 1 According to recent research, between 700 million and 1 billion individuals globally (or 9.1%-13.4% of the population) are suffering from CKD, 2 a condition in which the kidneys stop working and are not able to remove waste and extra water from the blood or keep body chemicals in balance. 3 Renal impairment denotes the kidney’s inability to perform its job. 4 The most common form of renal disorder is CKD. CKD is a long-term condition that does not improve over time. 5 The most common risk factors for CKD include diabetes, hypertension, and cardiovascular disease (CVD). Major outcomes of CKD include progression to kidney failure and increased risk for CVD. 6 The health and economic burdens of CKD are high. Patients with CKD are at increased risk of CVD and end-stage renal failure. Adverse changes in nutrition are prevalent with decreasing renal function and are a strong indicator of adverse outcomes in patients with CKD. 7
Serum creatinine (S. creatinine; a blood measurement) is an important indicator of kidney health, because it is an easily measured by product of muscle metabolism that is excreted unchanged by the kidneys. Creatinine itself is produced via a biological system involving creatine, phosphocreatine (also known as creatine phosphate), and adenosine triphosphate (ATP, the body’s immediate energy supply). 8 A creatinine level of greater than 1.2 mg/dL for women and greater than 1.4 mg/dL for men may be an early sign that the kidneys are not working properly. As kidney disease progresses, the level of creatinine in the blood rises. 9
Accumulating clinical evidence has demonstrated that inflammation is one of the major causes of poor outcomes in patients with renal failure. For example, the elevated level of C-reactive protein (CRP) is indicative of an inflammatory response and it is now widely accepted as a marker of atherosclerosis. 10 A simple blood test can be done to check your CRP level. A high-sensitivity C-reactive protein (hs-CRP) test is more sensitive than a standard CRP test. That means the high-sensitivity test can detect slight increases within the normal range of standard CRP levels. 11 hs-CRP is a marker of inflammation that predicts incident myocardial infarction, stroke, peripheral arterial disease, and sudden cardiac death among healthy individuals with no history of CVD, and recurrent events and death in patients with acute or stable coronary syndromes. 12
We aimed to evaluate the relationship between hs-CRP levels with creatinine levels in CKD patients. In this study, we need to determine the correlation between creatinine levels and the highly sensitive inflammatory marker, that is, hs-CRP in CKD patients.
Materials and Methods
Study Design and Setting
This was a cross-sectional prospective study, conducted in Parul Sevashram Hospital (PSH), Vadodara for six months (Nov 22-May 23). The sample size was determined by nonprobability, characteristic, and convenient sampling method with a 95% confidence level, using the Open EPI online statistical tool.
Patient Selection
The institutional review board of PSH approved the study protocol, and informed consent was obtained from all participants. The methods were carried out in accordance with the Declaration of University ethical principles for medical research.
Patients aged 35 years and above were included in the current study. The weight of males and females included was around 65-75 kg and 55-65 kg, respectively. The type II diabetes mellitus patients were included.
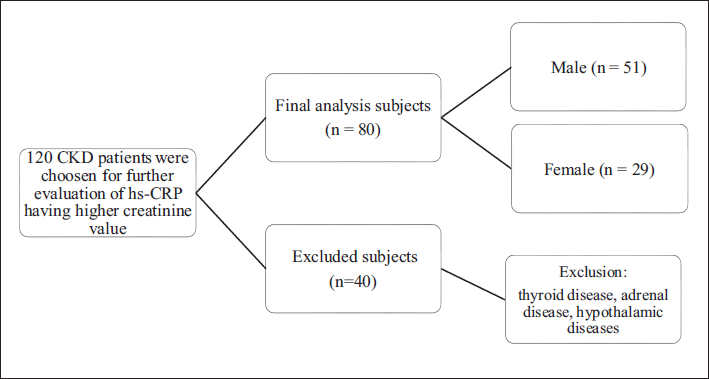
Patients suffering from any other disease other than CKD such as thyroid disease, type I diabetes, haemoglobinopathy, obstructive uropathy, and adrenal disease were excluded, Pregnant women were also excluded from this study. The study flowchart is shown in Figure 1.
Schematic Diagram of Study Flowchart.
Methods
Study participants were recruited and their medical history was recorded. Blood samples were collected from each participant using standard venepuncture techniques. The blood samples were immediately placed in plain blood collection tubes. The samples were then centrifuged at 3000 rpm for 10 minutes to separate the serum from the cells and gel separator. The hs-CRP immunoassay was performed using an automatic clinical chemistry analyser (FIA8000 Quantitative Immunoassay Analyser) and commercial kit (Getein Biotech, Inc.) The S. creatinine levels were excessed using Erba analyser and Erba kit. The hs-CRP and S. creatinine levels were reported in mg/L and mg/dL, respectively. The ranges for hs-CRP were as follows; low: hs-CRP level under 1.0 mg/L, average: hs-CRP level between 1.0 and 2.0 mg/L, high: hs-CRP level above 2.0 mg/L. The levels of S. creatinine were greater than 1.2 mg/dL for females and >1.4 mg/dL for males. Statistical analysis was performed to assess the relationship between hs-CRP and S. creatinine levels in the study population.
Data Entry and Analysis
Microsoft Excel 2019 was used for data entry. Study findings were explained through tables. In order to conduct the statistical analysis, SPSS 25.0 for Windows was used. Values were expressed as mean ± SD (standard deviation).
Results
The study consisted of 80 patients having higher creatinine values diagnosed with CKD, satisfying the inclusion and exclusion criteria. Out of total patients (80), 51 (63.75%) patients were male and 29 (36.25%) were female.
Results of hs-CRP Values
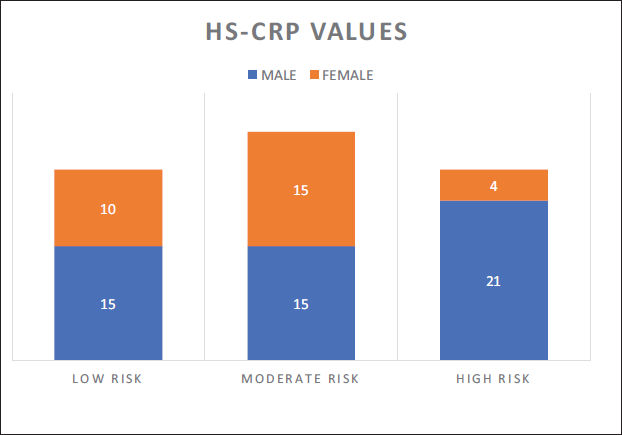
The hs-CRP levels were divided into three subcategories: hs-CRP = ≤1 mg/L (low risk), hs-CRP = 1-2 mg/L (moderate risk), and hs-CRP = >2 mg/L (high risk). In low-risk category, 15 (29.41%) patients were male and 10 (34.48%) were female. In moderate-risk category, 15(29.41%) patients were male and 15 (29.41%) were female and in high-risk category, 21 (41.17%) patients were male and 4 (13.79%) were females as shown in Figure 2.
Hs-CRP with Risk of Cardiovascular Disease in CKD Patients.
Results of S. Creatinine Levels
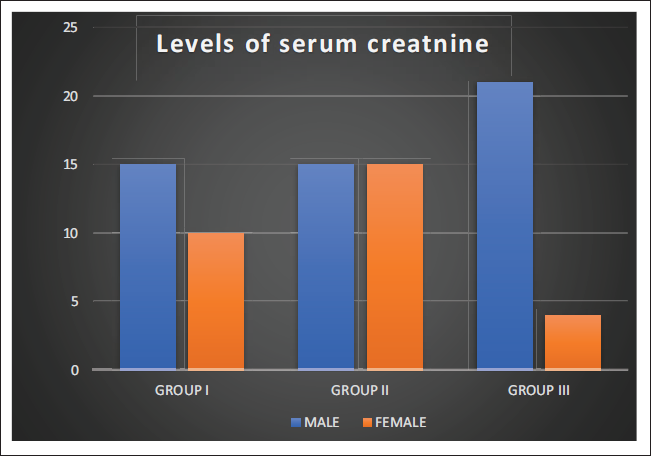
S. creatinine levels are divided into three subgroups: Group 1 (≥1-2 mg/dL), group 2 (≥2-4 mg/dL) and group 3 (≥4 mg/dL). In group 1, 15 (29.41%) patients were male and 10 (34.48%) were female. In group 2, 15 (29.41%) patients were male and 15 (29.41%) patients were female and in group 3, 21 (41.17%) patients were male and 4 (13.79%) were female as shown in Figure 3.
Number of Male/Female CKD Patients According to Groups of Serum Creatinine Levels.
Results of S. Creatinine Levels with hs-CRP Values
Notably, in this study with the increase in creatinine values, the hs-CRP values also increased respectively as shown in Table 1. In males, the mean S. creatinine levels for group 1, group 2, and group 3 were 1.62, 3.05, and 4.63, respectively, whereas, in females, S. creatinine levels for group 1, group 2, and group 3 were 1.51, 3.08, and 4.62, respectively.
hs-CRP Levels with Creatinine Levels in CKD Based on Study Subjects.
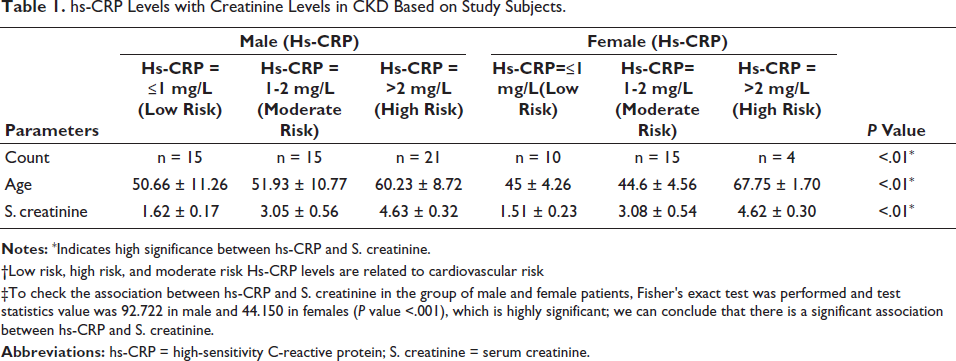
†Low risk, high risk, and moderate risk Hs-CRP levels are related to cardiovascular risk
‡To check the association between hs-CRP and S. creatinine in the group of male and female patients, Fisher’s exact test was performed and test statistics value was 92.722 in male and 44.150 in females (P value <.001), which is highly significant; we can conclude that there is a significant association between hs-CRP and S. creatinine.
Discussion
hs-CRP is a marker of chronic inflammatory conditions. Compared to a regular CRP test, a hs-CRP test is more sensitive marker of inflammation. hs-CRP is a biomarker used for risk prediction for CVD by assessing low concentrations of inflammation. 13 The hs-CRP test accurately detects lower levels of the protein than the standard CRP test. It measures CRP in the range from 0.3 to 10 mg/L. When hs-CRP levels are between 1 and 5, they are more likely to indicate cardiovascular risk. However, if the levels exceed 10, the elevation may suggest an acute infection, recent injury, or a noncardiac cause such as an autoimmune disease. 14
The researchers distributed questionnaires to the study subjects to collect demographic information, including age, gender, and clinical history, such as history of renal symptoms, common etiologies such as hypertension, diabetes mellitus, haemoglobinopathy, obstructive uropathy, and previous or family history of renal disease. The study consisted of 80 patients below the age of 70 years. They were having higher creatinine values already diagnosed with CKD and were+ satisfying the inclusion and exclusion criteria. Out of total patients (80), 51 (63.75%) patients were males and 29 (36.25%) were females.
The hs-CRP levels were divided into three subcategories: hs-CRP = ≤1 mg/L (low risk), hs-CRP = 1-2 mg/L (moderate risk) and, hs-CRP = >2 mg/L (high risk). In males, 41.17% of patients were having >2 mg/L of hs-CRP with reference to creatinine levels with mean ± SD were 4.63 ± 0.32 mg/dL (belonging to group 3). However, in females, only 13.79% of patients were observed with >2 mg/L of hs-CRP levels with mean ± SD creatinine values equal to 4.62 ± 0.30 mg/dL (belonging to group 3; Table 1).
Hence, it is noteworthy to say that males are more likely to show a higher hs-CRP value with an increase in S. creatinine as compared to females. At each stage—ranging from low to moderate and moderate to high—an increase in hs-CRP levels was observed, corresponding with a rise in serum creatinine i.e the increase in hs-CRP is observed at various stages alongside the increase in serum creatinine.
Elevated inflammatory markers are important risk factors for CKD. 15 Tonelli et al. and some other studies found that higher baseline CRP would accelerate kidney function failure in CKD patients.16, 17
According to clinical implications, low-grade inflammation level may partly explain the low incidence of cardiovascular events in patients with CKD. 18 Lee et al. 2020 findings suggested the importance of hs-CRP as a biomarker but also showed its limited value in risk stratification for adverse outcomes in this population. In this study, they showed that hs-CRP level was low in Korean patients with CKD, and this low-grade inflammation was associated with significantly higher risk of adverse cardiovascular outcome and mortality. However, it was observed that hs-CRP was not associated with CKD progression. The hs-CRP did not associate with adverse kidney outcome, and hence the predictive performance of hs-CRP was not strong. However, they observed that hs-CRP level was higher as eGFR was lower. 19
Tsai et al. also found that elevated serum hs-CRP levels were associated with reduced eGFR in their subjects. 20
The hs-CRP was found to be an independent marker of vast factors ranging from obesity, type 2 diabetes mellitus, metabolic syndrome, increased carotid intima-media thickness, stable coronary artery disease (CAD), first acute coronary event, and recurrent CVD events. The larger studies have mainly evaluated the association of hs-CRP and risk factors for CVD, diabetes mellitus, and glucose intolerance.21–24 A randomized controlled trial by Sabatine et al. 2007 suggests findings that the prognostic significance of hs-CRP in patients with stable CAD, elevated hs-CRP levels of >1 mg/L, leads to a greater risk of cardiovascular death, myocardial infarction, or stroke. 25
These findings suggest a relationship between hs-CRP and S. creatinine in patients with CKD. The results indicate that as creatinine levels increase, the hs-CRP levels also tend to rise. This association may have implications for understanding the inflammatory processes involved in CKD and its potential impact on cardiovascular risk.
It is important to note that the study was conducted on a specific group of patients, and further research is needed on larger scale to validate these findings in diverse populations.
Limitations
The sample size is limited, as a larger sample size is necessary to effectively study these markers in CKD patients. A time-dependent study could provide valuable insights into the progression of CKD in the same patients over time.
Conclusion
The results of the study suggest a significant association between hs-CRP levels and S. creatinine levels in patients with CKD. The findings indicate that as S. creatinine levels increase, there is a corresponding increase in hs-CRP levels. These results highlight the role of inflammation, as measured by hs-CRP, in kidney dysfunction, as reflected by elevated S. creatinine levels. This association suggests that inflammation may contribute to the progression of CKD and the increased risk of cardiovascular complications in these patients. Hence, hs-CRP levels may become an alarming tool for the progression of renal dysfunction and further advancement may lead to CVDs in future. An in-depth study is still essential for the exact mechanism involved related to this.
It is important to have reliable and cost-effective markers for assessing the severity of chronic kidney disease (CKD) in the community, given its prevalence. High-sensitivity C-reactive protein (hs-CRP) is one such marker that has been associated with CKD. However, as you mentioned, it is not a perfect indicator.
False negatives and false positives can occur with any diagnostic marker, including hs-CRP. A false negative result means that the marker fails to detect the condition when it is actually present. In the context of CKD, this could mean that some CKD patients may have normal hs-CRP levels, and therefore, relying solely on hs-CRP may lead to missed diagnoses.
Conversely, a false positive result means that the marker indicates the presence of the condition when it is not actually present. In the case of hs-CRP, if a patient has an underlying infection or a connective tissue disease, it may result in elevated hs-CRP levels, which could be falsely interpreted as CKD. To address these limitations, it is essential to use a combination of diagnostic markers and clinical assessment to evaluate the severity of CKD. Other markers, such as estimated glomerular filtration rate (eGFR), urinary protein levels, and imaging studies, can be used in conjunction with hs-CRP to improve diagnostic accuracy.
Furthermore, regular follow-up and monitoring of CKD patients are critical, as trends in hs-CRP levels over time can provide valuable information about disease progression or response to treatment. Healthcare professionals should consider the full clinical picture and multiple diagnostic tools to make an accurate assessment of CKD severity and tailor treatment accordingly.
Footnotes
Acknowledgment
This study was supported by grants from Parul Sevashram Hospital. The authors are also grateful to the administrative staff and the patients who participated in this study.
Authors’ Contributions
Dr Nivedita Priya conceived and designed the study. Shivani Goria collected the data and drafted the manuscript. Dr Nivedita analyzed the data, critically revised and approved the final version of manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Strict confidentiality was maintained regarding the information about the patients and the identity of the patients was not revealed.
Funding
The funds were obtained from CR4D (Centre of Research for Development), Parul Institute of Paramedical & Health Sciences, Parul University.
Informed Consent
All participants provided written informed consent before taking part in the study. They were informed about the purpose of the research, the procedures involved and their right to withdraw at any time. Confidentiality was assured, and the data collected were anonymized to protect participants’ identities.
Institutional Ethical Committee Approval Number
The study was conducted after obtaining approval (date:19/12/2022) from the Parul University - Institutional Ethics Committee for Human Research (PUIECHR) and the ethical approval number is PUIECHR/PIMSR/00/081734/5313.
