Abstract
Minimal change disease (MCD) as a cause of nephrotic syndrome in the elderly is seen in around 15%–20% of patients. Also eosinophillic gastroenteritis (EGE) is a very rare disease. The association with minimal change nephropathy and EGE is even rarer and to the best of our knowledge there is only a single case report of the same. Theoretically atopy is a common factor in the pathogenesis of both diseases and both may be IgE mediated. Here we present the case of a gentleman with nephrotic syndrome who underwent abdominal paracentesis and was found to have eosinophillic ascitis. Renal biopsy including electron microscopy was suggestive of minimal change disease. He responded completely on starting steroids with resolution of ascitis, proteinuria and decrease in IgE levels.
Background
Nephrotic syndrome in the elderly is often secondary. Among primary causes of nephrotic syndrome among the elderly, around 15-20% of patients are seen to have minimal change disease (MCD). 1 It is not a usual practice to tap ascitis in nephrotic syndrome unless severe enough to cause respiratory compromise. There is therefore no available data on the prevalence of eosinophillic ascitis in nephrotic syndrome. EGE is also a rare entity, the prevalence of which has been reported by the US database to be around 5.1 per 100,000 of the general population including children and adults. 2 The association with minimal change nephropathy and EGE is even rarer and to the best of our knowledge there is only a single other case report of the same. 3 Here we report a 65-year-old gentleman with nephrotic syndrome and eosinophillic ascitis who responded to steroid therapy.
Case Report
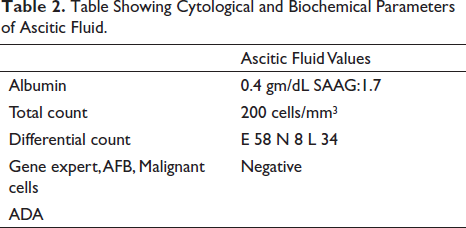
A 65-year-old gentleman was admitted with generalized oedema and abdominal pain. There was no history of allergies, rash, wheeze, fever, weight loss, diarrhoea or recent worm infestation. There was no lymphadenopathy or organomegaly on physical examination or by ultrasound. Physical examination was significant for bilateral pitting pedal oedema and massive ascitis. Preliminary laboratory values are detailed in Table 1. Of note, serum albumin level was 2.1 gm/dL and 24-hour urine protein of 7,677 mg with no active urinary sediment by light microscopy. Other laboratory investigations were remarkable for a serum creatinine of 1.9 mg/dL with normal bIlirubin, SGOT and SGPT. A renal biopsy was done. Meanwhile, as the patient had discomfort due to the massive ascitis, paracentesis was done. The ascitic fluid reports are as shown in Table 2; the predominant abnormality being a cell count of 200 cells with 58% eosinophils.
Table Showing Laboratory Parameters During Admission.
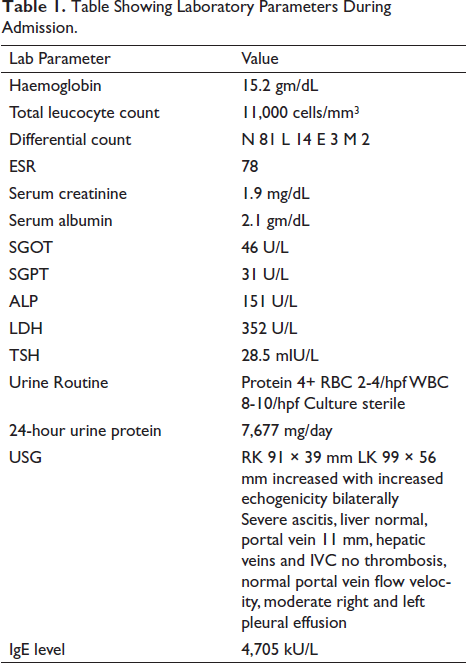
Table Showing Cytological and Biochemical Parameters of Ascitic Fluid.
Corresponding haematological values did not show any peripheral eosinophillia which was confirmed by peripheral smear. LDH was normal. Stool examination was negative for ova, cysts and parasites. The serum IgE sent was highly elevated at 4,705 IU/mL. Right pleural effusion was also present. This pleural fluid was also aspirated. Fluid was transudative and negative for eosinophils. Meanwhile, renal biopsy was reported as a minimal change disease by light microscopy which was confirmed by electron microscopy as well. Immunofluorescence did not show any uptake. The patient was started on steroids at 1 mg/kg/day. Within four weeks, he responded with urine protein creatinine ratio <0.2:1, serum albumin level of 3.2 gm/dL and serum creatinine level of 0.9 mg/dL. IgE values dropped to 1,350 IU/mL. On clinical examination, there is no oedema or ascitis. The patient is now on tapering dose of steroids in complete remission.
Discussion
Eosinophillic ascites is characterized by high eosinophil counts in the peritoneal fluid. The cause and aetiology are not known. It has been described in eosinophillic gastroenteritis (EGE), parasitic infestation, hypereosinophillic syndrome (HES) and rarely systemic lupus erythematosis, lymphoma and tuberculosis. In a review of cases of eosinophiliic ascitis, EGE was the most common at 74%. 4 EGE is an entity characterized by gastroenterological symptoms, histological demonstration of eosinophilic infiltration in the gastrointestinal tract and exclusion of parasitic infestation.5, 6 EGE usually presents with peripheral eosinophillia. However, among the types of EGE, the subserosal variety 7 is characterized by eosinophil-rich ascitis and is distinguished by the absence of peripheral eosinophilia in around 20% of cases. 8 EGE in association with nephrotic syndrome has rarely been reported in the literature. An Italian case report describes a patient with membranous nephropathy secondary to EGE. 9 Here the patient responded to steroids alone unlike primary membranous nephropathy. Another case report describes a patient with frequently relapsing nephrotic syndrome due to minimal change disease who developed EGE and relapse of nephrotic syndrome. 3 He also responded well to the reintroduction of steroids. Another case of EGE was reported with nephrotic syndrome secondary to immune complex-mediated disease. 10 The culprit antigen was thought to be bovine serum albumin (BSA) and the child responded to steroids with modification of diet. Our patient had a renal biopsy diagnosis of minimal change disease. He had no other evidence of atopy other than an elevated IgE level. On starting steroids, the patient responded dramatically and laboratory parameters also improved. Parasitic infection, malignancy and HES were excluded by appropriate investigations as detailed above. Endoscopy and biopsy were not done as EGE being patchy may be missed. Also, the treatment of EGE is steroids as in MCD so there would not have been any change in treatment offered. There is no consensus however of duration of therapy in EGE. There are some cases of relapse after stopping steroids and these patients may require continuation of steroids. 11 Minimal change disease itself is associated with elevated IgE. However, no direct association or diagnostic value has been attached to IgE values as of yet. 12
Conclusion
There may be an association of EGE with minimal change in disease. Both these conditions have underlying evidence of atopy and both these conditions usually respond to steroids.
Footnotes
Author Contribution Statement
Ranjani Ravi: original draft preparation; Vimala A: conceptualization, reviewing and editing, supervision; Jinsi N, Sreeja S Nair: reviewing and editing. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author received no financial support for the research, authorship and/or publication of this article.
Ethical Approval
Ethical committee clearance was not taken as no information pertaining to patient identity was included in this case report.
Informed Consent
Informed consent has been taken from the patient. In view of maintaining privacy, personal details of the patient have not been included within this manuscript.
