Abstract
Background
Understanding relationships between polypharmacy, treatment burden and other factors (e.g. age, knowledge of medicines), could inform interventions to reduce treatment burden.
Aim
To explore sociodemographic, clinical and medication factors associated with high treatment burden.
Design and setting
Secondary analysis of multi-centre UK primary care baseline clinical trial data.
Methods
Participants were ≥18 years, prescribed ≥5 repeat medicines with ≥1 indicator of potentially inappropriate prescribing (PIP). Survey data captured treatment burden (13-item Multimorbidity Treatment Burden Questionnaire; dichotomised with high burden ≥22), sociodemographic characteristics, quality of life, self-reported knowledge of medicines, and medication adherence. Electronic health records provided data on age, gender, long-term conditions, PIP, medicines use (last 3 months), and consultations (last 12 months). Associations between treatment burden and other factors were modelled using multivariable logistic regression.
Results
1711 adults from 37 general practices were included (mean age 72 years, 51% male). 381 (23%) reported high treatment burden. Multivariable analysis found high treatment burden was associated with younger age, being unemployed or in paid work, university or high education, multimorbidity, anxiety/depression, polypharmacy (≥8 medicines), lower medication adherence, knowledge of medicines, and quality of life. There was strong evidence (p<0.005) that the association between high treatment burden and polypharmacy was greater in people aged <60 years, those with poor knowledge of medicines, more multimorbidity, and those with low/moderate medication adherence.
Conclusion
High treatment burden was more common in adults with polypharmacy who were younger, had multimorbidity, poor medication knowledge, or low adherence. This should inform design of interventions addressing treatment burden.
Introduction
Treatment burden is the effort required for patients to look after their health conditions and the impact this has on their everyday life.1,2 This includes collecting and taking medication, self-monitoring health conditions, attending and co-ordinating appointments, actioning lifestyle advice and seeing different healthcare professionals. Treatment burden is particularly relevant to the growing number of people experiencing multimorbidity (multiple long-term conditions in the same individual), 3 which affects half of adults in the UK and a third of adults globally.4,5 Healthcare systems tend to be disease-centred rather than patient-centred, and patients with multimorbidity are often required to attend separate appointments with different healthcare professionals, and to take different treatments for each of their long-term conditions. Several studies have reported an association between high treatment burden and multimorbidity.6–8 High treatment burden is associated with poor health outcomes, including reduced quality of life.6,7
The cumulative complexity model conceptualises treatment burden as a balance between patient workload (healthcare demands) and capacity to perform the work. 9 Patients who have high workload (e.g. lots of appointments to co-ordinate and attend) but good capacity to manage the work (e.g. strong support from family or friends, no caring or work commitments etc) may report low treatment burden. Conversely, patients who have low workload, but poor capacity may report high burden. Treatment burden acts as a feedback loop between workload and capacity. Patients may become more complex over time as they accumulate long-term conditions and treatments. Some patients demonstrate relative resilience towards treatment burden and patients may have sufficient capacity to resist overburden through normalisation of health-related workload. 10 Accordingly, while it is possible to identify factors generally associated with higher treatment burden, there is significant individual variation.
Polypharmacy (prescribing of multiple medicines to one individual) is common in individuals with multimorbidity.11,12 For many patients, polypharmacy is appropriate - multiple medications are necessary to improve quality of life or increase longevity. 13 Problematic polypharmacy occurs when the risk of taking multiple medications outweighs the potential benefit.13,14 The impact of problematic polypharmacy includes an increased risk of adverse drug reactions, poorer health related quality of life and increased likelihood of emergency department attendance. 15 Polypharmacy, poor adherence to medicines and low health literacy are associated with increased treatment burden.8,16,17 However, the extent to which the association between polypharmacy and treatment burden might vary with other factors remains unclear.
Understanding the degree to which other factors (age, medication adherence and levels of multimorbidity) modify the association between treatment burden and levels of polypharmacy may provide a more nuanced understanding of these relationships. For example, groups of patients who would benefit most from interventions to reduce treatment burden. We hypothesised that greater levels of polypharmacy would be associated with higher treatment burden in younger individuals (for whom polypharmacy might be considered less typical or more difficult to manage), individuals with poor knowledge of their medicines, and individuals who find adherence to medication difficult.
The aims of this study were to examine the associations of high treatment burden with sociodemographic, clinical and prescribing characteristics, and to examine how the association between high treatment burden and greater levels of polypharmacy varied with age, medication literacy, multimorbidity, and medication adherence.
Methods
Study setting
This study is a secondary analysis of data collected in the multi-centre cluster-randomised trial, “Improving Medicines Use in People with Polypharmacy in Primary Care” (IMPPP; ISRCTN 90146150). 18 The IMPPP trial assessed whether a complex intervention to optimise medicine use reduced potentially inappropriate prescribing (PIP) in adults with polypharmacy (prescribed ≥5 repeat medicines). General practices were recruited from two regions in England – the South-West and West Midlands. The current analysis uses baseline pre-randomisation data from the trial. The trial aimed to recruit 1,850 participants from 37 GP practices, powered to detect a clinically meaningful reduction of 0.5 in PIP indicators. 18
Study population, eligibility criteria and recruitment
Patients were eligible if they were aged ≥18 years, prescribed ≥5 current long-term (i.e. “repeat”) medicines, and exposed to at least one indicator of PIP. 18 Potentially eligible patients were identified pre-randomisation via an automated search of the electronic health records (EHR). A clinical member of the practice screened search results against exclusion criteria, excluding those patients (1) receiving end-of-life care; (2) deemed inappropriate to contact by practice staff; (3) unable to complete study questionnaires or medication review appointments; or (4) planning on moving general practice within the six-month follow-up period.
Patients were invited by post prior to randomisation. Those who wished to participate completed a consent form and baseline survey and returned both via post to the research team. Only participants who consented to their data being used for further research were included in the current analysis.
Survey data
The baseline survey captured socio-demographic characteristics, including ethnicity (seven categories based on census classification), English Index of Multiple Deprivation (IMD) 19 ; determined from the participant’s postcode), education level, employment status and whether participants lived alone.
Treatment burden was measured in the baseline survey using the 13-item Multimorbidity Treatment Burden Questionnaire (MTBQ), a simply worded measure that has been validated in two studies of adults with multimorbidity in the UK.6,20 Each question was scored as follows: 0 (not difficult, does not apply), 1 (a little difficult), 2 (quite difficult), 3 (very difficult) and 4 (extremely difficult). A global MTBQ score was calculated as the average score from the questions answered, multiplied by 25 to give a score from 0 to 100. Participants were excluded if more than 50% of responses to the MTBQ were missing. Treatment burden was categorised as no (0), low (0-10), medium (10-21) and high burden (≥22), as recommended by the validation study. 6
Health-related quality of life (HRQoL) was measured in the baseline survey using the 12-item Short Form Survey (SF-12), 21 a validated 12-item measure that assesses mental health status and physical functioning. The SF-12 was scored by calculating two summary scores: the Physical Component Summary (PCS) and Mental Component Summary (MCS). Higher scores indicated better physical functioning and mental health. HRQoL was also measured using a visual analogue scale adapted from the EQ-5D measure, asking participants to rate the question “Please tell us how good or bad your health is TODAY using the scale below” on a scale of 0 (worst health) to 100 (best health). 22
To assess knowledge of medicines, as part of the survey, participants were asked “Do you know what each of your prescription medicines is for?”(yes/no). Those answering “no” were asked to choose one of the following (i) “I am unsure what one or two of my prescription medicines is for”; (ii) “I am unsure what some of my prescription medicines are for”; or (iii) “I am unsure what most or all of my prescription medicines are for”. These questions were developed by the study team with input from the patient and public involvement (PPI) advisory group. For the logistic regression analysis, knowledge of medicines was dichotomised into “yes” (I know what each of my prescription medicines is for) and “no” (I do not know what each of my prescription medicines is for).
Adherence to medicines was measured in the baseline survey using the 10-item Medication Adherence Rating Scale (MARS-10).23,24 Each question was scored 1 if good adherence was indicated, with the sum of all questions providing a total adherence score, categorized as low adherence (0-4), moderate adherence (5-7) and high adherence (8-10).
Data from electronic GP records
The following anonymised data were collected from each participant’s electronic health record (EHR): age, gender, long-term conditions, PIP indicators, medicines prescribed in the preceding 3 months, and in-hours general practice consultations (phone and in-person) recorded in the preceding 12-months. Current medication count was the number of unique chemical substances issued in the 3-month period, irrespective of strength or dose. Long-term conditions were the list of 20 clinically important conditions as defined by the validated Cambridge Multimorbidity Score (CMS). 25 These conditions were the highest-ranking with regards to prevalence and effect size. PIP indicators were based on a series of clinical code-based rules, combining prescribing, diagnostic and test codes as listed in the IMPPP trial protocol. 18 Multimorbidity was measured using the 20-condition general outcome version of the CMS. 25
Analysis
The analysis was conducted using Stata (version 18.0). 26 Descriptive statistics were used to summarise the characteristics of trial participants. To explore associations between high treatment burden (outcome) and various exposure variables, treatment burden was dichotomised into no/low/medium burden (MTBQ score <22) and high burden (MTBQ score ≥22). Association with high treatment burden was modelled using univariable and multivariable logistic regression. The multivariable models additionally included the covariates age, gender, socioeconomic deprivation (IMD quintile), ethnicity, education, employment status, living alone, geographical location of GP practice (South-West England or Midlands), CMS, number of current medicines, adherence to medicines (dichotomised into low/moderate adherence (0-7) and high adherence (8-10)), and GP practice (random effect). These were chosen as they were considered to be clinically relevant with a biologically plausible association, as judged by clinical and topic experts on the research team. GP practice was included as a variable as it was a design characteristic of the IMPPP trial.
Individual models were used to examine the association between individual long-term conditions and treatment burden, adjusting for other overall morbidity burden. Regression models thus included a binary variable for the specific individual long-term condition, and a modified CMS which excluded the corresponding individual condition. For example, to explore an association between diabetes and treatment burden, the weighting for diabetes was removed from the CMS for individuals who had diabetes.
Interaction between polypharmacy and age, multimorbidity, knowledge of medicines and adherence
Although the association between polypharmacy and treatment burden was expected, we hypothesised that this may nevertheless vary with other factors. For example, it’s widely accepted that younger people describe higher treatment burden but whether this modifies the association between polypharmacy and treatment burden has been hitherto unknown. We pre-specified four key effect modifiers, which we considered relevant based on the existing literature: age, level of multimorbidity, knowledge of medicines and adherence to treatment. We hypothesized that younger people were more likely to report high treatment burden due to work and childcare commitments. The 60-year age cut off was chosen rather than a 65-year cut off because a reasonable proportion (23%) of participants aged 60-64 years were retired, compared to 7% of participants aged 55-59 years.
To examine the interaction between levels of polypharmacy and age in relation to treatment burden, an interaction term was created between number of medicines (dichotomised using median; <8 medicines, ≥8 medicines) and age (<60 years and ≥60 years), resulting in four categories. A logistic regression model was then applied comparing non-high treatment burden (MTBQ score 0-21) and high treatment burden (MTBQ score ≥22). The regression models were adjusted for age, gender, individual deprivation, ethnicity, education, employment status, living alone, geographical location, CMS, and clustering by GP practice (random effect). This process was repeated to examine the interaction between levels of polypharmacy and other factors, including multimorbidity (dichotomised using median CMS; i.e. low multimorbidity <2.2, high multimorbidity ≥2.2), knowledge of medicines (knowledge of all medicines, unsure of medicines) and adherence to treatment (low/moderate adherence, high adherence), in relation to treatment burden.
Missing data
Multivariate analyses were conducted on complete cases with no imputation of missing data.
Results
A total of 1727 participants were enrolled in the IMPPP trial (Figure 1). Of these, 1711 consented for their data to be used for further research and were included in the analyses. Global MTBQ scores were positively skewed: 14% reported no burden, 38% low burden, 25% medium burden and 23% high burden (Table 1, Figure 2). Participant flow diagram. * i.e. excluded due to practices already meeting the target of 50 eligible participants during the 6-month intervention delivery period. Participant characteristics and outcome scores (N=1711). aUnemployed includes participants who selected: looking after the home; unable to work due to long-term illness or disability; and unable to work due to carer responsibilities. b20 clinically important long-term conditions included in the Cambridge Multimorbidity Score. cCambridge multimorbidity score calculated using weighted scores for each of the long-term conditions. dNumber of current medicines prescribed in the preceding 3 months recorded on the electronic health records. e“Do you know what each of your prescription medicines is for?” (yes/no). Those answering “no” were asked to choose one of the following: (i) unsure about one or two medicines; (ii) unsure about some medicines; (iii) unsure about most or all medicines. Responses (i) and (ii) were combined. fAdherence to medicines was measured using the 10-item Medication Adherence Rating Scale (MARS-10)24 and categorized as low adherence (0-4), moderate adherence (5-7) and high adherence (8-10). gNumber of primary care consultations include GP and nurse consultations recorded on the electronic health records. Abbreviations: standard deviation (SD), transient ischemic attack (TIA), chronic obstructive pulmonary disease (COPD). Distribution of global treatment burden scores among participants (N=1,671). A global Multimorbidity Treatment Burden Questionnaire (MTBQ) score was calculated as the average score from the questions answered, multiplied by 25 to give a score from 0 to 100.
6
The dotted vertical lines represent the cut off values between no treatment burden (0), low treatment burden (<10), medium burden (10-21) and high burden (≥22). MTBQ global score was missing for 40 (2%) of participants (1,671 included in the analysis for Figure 2).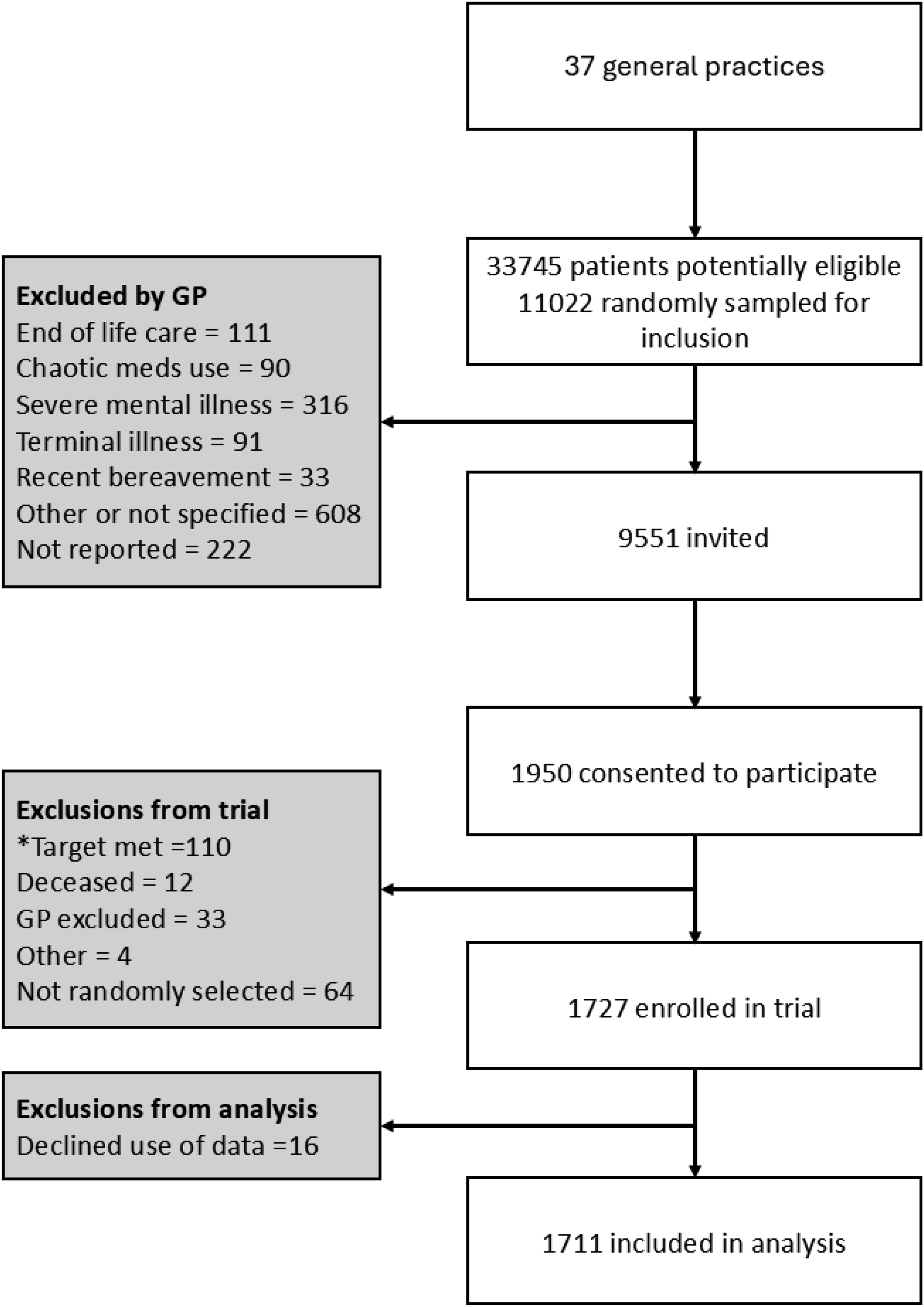


Participant characteristics are shown in Table 1. The mean age was 72 years, 51% were male, and 97% were of white ethnicity. Most participants were retired (71%); had no educational qualifications (26%) or school age qualifications (46%); and lived with others (74%). Almost all participants had multimorbidity (≥2 long-term conditions; 95%). The most common long-term conditions were hypertension (71%), anxiety/depression (48%) and painful condition (44%).
Half the participants were prescribed eight or more medicines. 8% of participants were unsure about what some of their prescription medicines were for and 1% were unsure about what all their medicines were for. Most (68%) had high adherence to medications and only 5% had low adherence. Almost two thirds (61%) had two or more PIPs. The number of consultations in the last 12 months ranged from 0 (7%), to 20 or more (4%), with most having 1-9 consultations (69%). Just over a half of participants had no treatment burden (14%) or low burden (38%) and 23% reported high treatment burden.
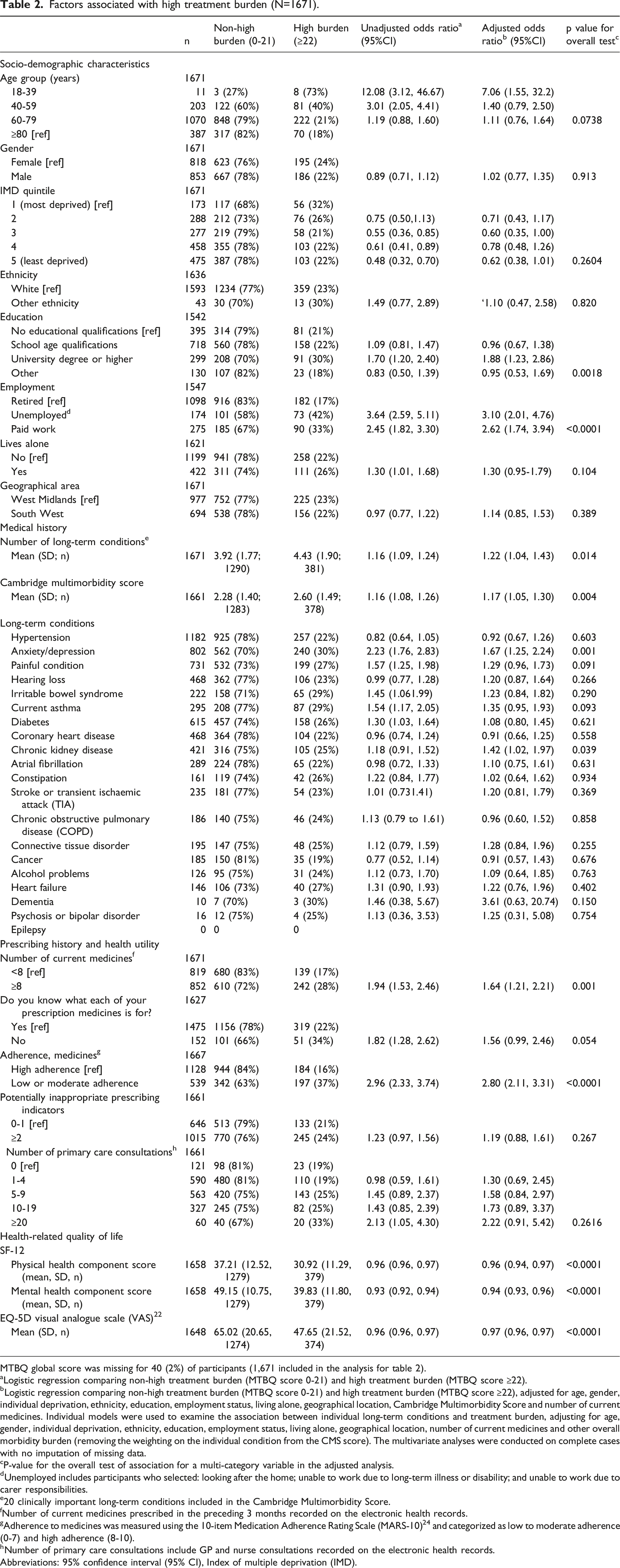
Factors associated with high treatment burden (N=1671).
MTBQ global score was missing for 40 (2%) of participants (1,671 included in the analysis for table 2).
aLogistic regression comparing non-high treatment burden (MTBQ score 0-21) and high treatment burden (MTBQ score ≥22).
bLogistic regression comparing non-high treatment burden (MTBQ score 0-21) and high treatment burden (MTBQ score ≥22), adjusted for age, gender, individual deprivation, ethnicity, education, employment status, living alone, geographical location, Cambridge Multimorbidity Score and number of current medicines. Individual models were used to examine the association between individual long-term conditions and treatment burden, adjusting for age, gender, individual deprivation, ethnicity, education, employment status, living alone, geographical location, number of current medicines and other overall morbidity burden (removing the weighting on the individual condition from the CMS score). The multivariate analyses were conducted on complete cases with no imputation of missing data.
cP-value for the overall test of association for a multi-category variable in the adjusted analysis.
dUnemployed includes participants who selected: looking after the home; unable to work due to long-term illness or disability; and unable to work due to carer responsibilities.
e20 clinically important long-term conditions included in the Cambridge Multimorbidity Score.
fNumber of current medicines prescribed in the preceding 3 months recorded on the electronic health records.
gAdherence to medicines was measured using the 10-item Medication Adherence Rating Scale (MARS-10) 24 and categorized as low to moderate adherence (0-7) and high adherence (8-10).
hNumber of primary care consultations include GP and nurse consultations recorded on the electronic health records.
Abbreviations: 95% confidence interval (95% CI), Index of multiple deprivation (IMD).
Following adjustment using multivariable analysis, the magnitude and direction of most associations was generally maintained. Of note, there was particularly strong evidence (p<0.0001) for a positive association between high treatment burden and being unemployed or in paid work compared to being retired (adjusted odds ratio (aOR) 3.10, 95% confidence interval (CI) 2.01 to 4.76; aOR 2.62, 95%CI 1.74 to 3.94 respectively); low/moderate adherence to medication (aOR 2.80, 95%CI 2.11 to 3.31); and poor HRQoL (aOR 0.97, 95%CI 0.96 to 0.97 for a one point increase in VAS; aOR 0.96, 95%CI 0.96 for a one point increase in PCS; aOR 0.94, 95% CI 0.93-0.96 for a one point increase in MCS). Positive associations were also found between high treatment burden and university or higher education (aOR 1.88, 95% CI 1.23 to 2.86, p=0.018); greater levels of polypharmacy (≥8 medicines; aOR 1.64; 95%CI 1.21-2.21, p=0.001); multimorbidity (mean CMS; aOR 1.17; 95% CI 1.05 to 1.30, p value=0.004); anxiety/depression (aOR 1.67; 95% CI 1.25-2.24, p=0.001); and chronic kidney disease (aOR 1.42, 95% CI 1.02 to 1.97, p=0.039). Individuals aged 18-39 years were more likely to report high treatment burden compared to those aged ≥80 years (aOR 7.06, 95% CI 1.55 to 32.3), although overall there was no evidence (p=0.0738) for an association between age and high treatment burden.
Analyses of high treatment burden in relation to the interaction between polypharmacy a and age, multimorbidity, knowledge of medicines and adherence.

aPolypharmacy defined as ≥8 prescribed medicines in the last 3 months.
bLogistic regression comparing non-high treatment burden (MTBQ score 0-21) and high treatment burden (MTBQ score ≥22), adjusted for age, gender, individual deprivation, ethnicity, education, employment status, living alone, geographical location, Cambridge Multimorbidity Score (except in the CMS score analysis) and GP practice (random effect).
cCambridge multimorbidity score calculated using weighted scores for each of the long-term conditions. 25 .
dAdherence to medicines was measured using the 10-item Medication Adherence Rating Scale (MARS-10) 24 and categorized as low to moderate adherence (0-7) and high adherence (8-10).
The multivariate analyses were conducted on complete cases with no imputation of missing data.
AbbreviationsConfidence interval (CI), Cambridge Multimorbidity Score (CMS).
Discussion
This study of mostly older adults experiencing potentially problematic polypharmacy found that almost a quarter of participants reported high treatment burden. Several sociodemographic factors (e.g. being unemployed or in paid work compared to being retired, and university education), the presence of multimorbidity and several specific health problems (e.g. anxiety/depression), and various aspects of medication use (e.g. increasing numbers of medications, poor knowledge of medicines, low/moderate medication adherence) were independently associated with high treatment burden. The strength of association between medication count and high treatment burden was greatest in younger individuals, and those with multimorbidity, poor medication literacy and poor medication adherence.
Comparison with existing literature
Our findings align well with the wider literature. We have expanded on the current body of knowledge by further exploring the association between treatment burden and knowledge of medicines; the association between treatment burden and multimorbidity; and the interaction between higher levels of polypharmacy, treatment burden and other factors. To interpret the study findings, we have drawn on the cumulative complexity model. 9 This conceptualises treatment burden as the balance between workload and capacity.
In line with other studies, we found an association between younger age and high treatment burden.16,27 Furthermore, the association between medication count and high treatment burden was most marked in younger individuals. One explanation for this is that younger people may be less willing to accept the need for multiple medicines and may expect to maintain their health with less effort. As a result, they may have different expectations of how much work should be required to manage their well-being compared to older people, who may be more accustomed to ongoing medical treatments. A further explanation may be younger patients having less capacity to manage the work of looking after their health due to competing priorities, such as work and caring for children and parents. This hypothesis is supported by our finding that people who were retired were less likely to report high burden of treatment.
To our knowledge, this is the first study to investigate the relationship between treatment burden and knowledge of medicines. We found that individuals who were uncertain about their medicines reported higher treatment burden. This aligns with findings elsewhere in the literature of an association between high treatment burden and low health literacy more generally.8,17 Furthermore, as hypothesised, we found that the strength of association between medication count and treatment burden was greatest in those individuals with poor medication literacy. This may reflect individuals with poor knowledge about their medicines being less able to manage the work of being a patient. This is also supported by the finding that poor medication adherence also modifies the association between treatment burden and medication count, with poor adherence being indicative of excessive patient workload.
We found, as reported elsewhere, a positive association between number of long-term conditions and high treatment burden.6–8,17,28 However, simple counts fail to capture differences between specific long-term conditions. Our finding that a multimorbidity score (CMS) weighted according to its different component conditions is strongly associated with high treatment burden is therefore an important, albeit expected, new observation, with multiple health problems both increasing the individual’s workload and impairing their physical or mental capacity to cope. 25 As hypothesised, we found that the strength of association between medication count and treatment burden was greatest in individuals with high multimorbidity scores.
We also found strong evidence of an association between high treatment burden and anxiety/depression. This has been reported in other studies and may reflect that having a mental health condition impacts on an individual’s capacity to manage the work of looking after their health.6,7,29,30 Related to this, high treatment burden was also associated with low physical and mental health related quality of life, in keeping with existing literature.6,7,28 This highlights the negative impact high treatment burden can have on an individual’s wellbeing and the importance of novel interventions to assess, monitor and reduce treatment burden.
In this study, we found an association between being highly educated (University degree or higher compared with no educational qualifications) and high treatment burden. This aligns with the findings of a population health survey in Denmark 31 but differs from studies in the United States.29,32 One possible explanation is that highly educated patients may have higher expectations of themselves and the healthcare system.
Strengths and limitations
High quality data, collected using robust processes as part of a large multi-centre trial, are an important strength of this study. We were able to combine survey data with data from the EHR and there were minimal missing data. Rather than relying on self-report of long-term conditions, we captured these data from the EHR. Long-term conditions were based on a list validated for primary care research purposes (CMS), and multimorbidity was quantified using a validated measure. 25 Underpinning our work is the choice of a validated patient-reported measure of treatment burden (MTBQ).6,20 The MTBQ has been translated, culturally adapted and validated in multiple languages, and has been shown to be valid in different cultural contexts.7,33–35
A limitation of the study is that 97% of participants reported their ethnicity as white, with low representation from global majority ethnicities may limit the generalisability of the findings. Non-English speaking patients were excluded from the trial. This group would be an interesting group to investigate as it is likely they experience high treatment burden, not least due to difficulties navigating UK healthcare services. In keeping with other studies, few participants lived in more deprived areas, further limiting generalisability. These issues highlight the importance of striving for more inclusive research and capturing a wide range of patient voices. Adherence was assessed self-reported without objective validation (e.g. pill counts), possibly leading to an overestimation. Despite adjusting for many covariates, residual confounding cannot be excluded. Findings must also be interpreted in the context of a population recruited as part of a clinical trial and, as this was an observational study, causality cannot be inferred.
Implications for research, practice and policy
Treatment burden is not currently considered by policy makers when reviewing and developing models of care for adults with multimorbidity. We believe our work adds to a growing body of literature calling for recognition and reduction of treatment burden in patients with multiple long-term conditions and polypharmacy. Policies and interventions aimed at optimising medication use should include assessment and management of treatment burden. Our work points to certain groups of patients to target for support with high burden of treatment, including those who are younger, prescribed greater numbers of medicines, have poor knowledge of their medicines, have poor medication adherence, and are diagnosed with anxiety/depression. The findings also support the need to educate patients about their medicines, which may not only improve adherence but also potentially reduce treatment burden. Finally, our findings support the use of the Cambridge Multimorbidity Score to identify patients at risk of high burden of treatment.
Conclusion
This study provides clear evidence for the association between high treatment burden and a number of important sociodemographic and clinical factors, including several aspects of medication use. It is apparent that the strength of association between treatment burden and number of medications is modified by various factors related to patients’ capacity to manage their health. Interventions to reduce treatment burden should target individuals with polypharmacy, multimorbidity, mental health conditions and work and caring commitments. Supporting patients to understand what their medicines are for may reduce treatment burden.
Supplemental Material
Supplemental Material - Development of the Associations between treatment burden, socio-demographics, clinical factors and use of medicines in adults with polypharmacy: A cross-sectional study
Supplemental Material for Associations between treatment burden, socio-demographics, clinical factors and use of medicines in adults with polypharmacy: A cross-sectional study by Polly Duncan, Chloe Gamlin, Deborah McCahon, Daisy Gaunt, Beenish Ashfaq, Rupert A. Payne in Journal of Multimorbidity and Comorbidity.
Footnotes
Acknowledgements
The study was hosted by NHS Bristol, North Somerset and South Gloucestershire Integrated Care Board. Trial delivery was facilitated by the NIHR Clinical Research Network and the Bristol Trials Centre. We would like to thank the patients and practices who participated in the study, members of the study patient and public involvement advisory group, and members of the independent trial steering committee and data monitoring committee.
Ethical considerations
The IMPPP trial protocol and associated study materials were approved by the Wales REC 6, NHS Research Ethics Committee (REC reference 19/WA/0090), and Health Research Authority. Written informed consent was obtained from all participants in the study. The informed consent process included the option for sharing anonymous data for subsequent research. Favourable opinion for the current study was received from the University of Bristol, Health Science Student Research Ethics Committee (Ref: 18975).
Consent to participate
Patients were invited by post. Those who wished to participate completed a consent form and baseline survey and returned both via post to the research team. Only participants who consented to their data being used for further research were included in the current analysis.
Consent for publication
Participants consented to study findings being published.
Funding
The IMPPP trial was funded by the NIHR Health and Social Care Delivery Research (HSDR) programme (ref. 16/118/14). The views expressed are those of the authors and not necessarily those of the NIHR or the Department of Health and Social Care.
Declaration of conflicting interests
PD developed and validated the original Multimorbidity Treatment Burden Questionnaire (MTBQ). RP developed and validated the Cambridge Multimorbidity Scale.
Data Availability Statement
STROBE statement
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
