Abstract
Background
Multidisciplinary diabetes management has been designed to reduce the risk of diabetes-associated complications. Nevertheless, it remains underexplored whether multidisciplinary management may provide the added benefits of lowering cancer risk. This study aims to compare differences in clinical profiles and cancer incidence between patients receiving multidisciplinary and standard management in primary care.
Methods
This retrospective cohort study identified patients who attended general outpatient clinics between year 1997 and 2021 without cancer history using territory-wide electronic health records of Hong Kong. Patients were followed up until December 31st, 2021. Cox regression weighted with inverse probability of treatment was applied to compare the risk of site-specific cancers (colon and rectum, liver, pancreas, stomach, bladder, kidney, and lung) between patients with usual diabetes care (n=25,301), multidisciplinary diabetes management (n=66,382), and those without diabetes (n=54,376).
Results
The risk of developing liver and pancreatic cancers appeared to be attenuated in patients who received multidisciplinary diabetes care, when compared to those with usual care alone (aHRs for liver: 1.90, 95%CI 1.26-2.86 vs 2.26, 95%CI 1.47-3.46; pancreas: 3.47, 95%CI 1.41-8.53 vs 5.57, 95%CI 2.15-14.45), with reference to non-diabetes. Patients with usual diabetes care showed a higher risk of gastric cancer in unweighted model, but the association disappeared in weighted model. Overall, patients who received multidisciplinary diabetes management tended to be younger with less comorbidities.
Conclusion
The risk of liver and pancreatic cancers appeared to be mitigated in patients receiving multidisciplinary diabetes care. Further studies are warranted to evaluate whether multidisciplinary management may provide additional benefits of cancer prevention.
Introduction
Diabetes is one of the leading causes of premature mortality and disability worldwide. 1 It has risen to the 5th leading cause of disability-adjusted life-years (DALYs) in 2023 from the 20th leading cause in 1990. 1 Currently, it is estimated that 10.5% of the adult population are living with diabetes. 2
Type 2 diabetes and cardiovascular disease share many overlapping risk factors, including obesity, dyslipidemia, hyperglycemia, and hypertension. 3 Patients with type 2 diabetes are at risk of developing macrovascular (coronary heart disease, cerebrovascular disease, and peripheral arterial disease) and microvascular (nephropathy, neuropathy, and retinopathy) complications. Nevertheless, it has been recognized that physician care alone may not be sufficient to fulfil the multidimensional care needs of patients, 4 who often live with multiple comorbid conditions and take multiple medications, 5 and hence multidisciplinary diabetes care (beyond standard medical consultations and prescriptions) has been designed to optimize cardiovascular health in order to minimize the risk of vascular complications of patients. 4 Accumulating evidence has shown that multidisciplinary diabetes care (for example, lifestyle modification 6 and medication adherence) 4 may help improve cardiovascular risk profile, 4 and lower the risk of microvascular and macrovascular complications and mortality, 7 when compared to usual diabetes care alone (standard medical consultations and prescriptions).
Nevertheless, beyond the imminent hazards of vascular complications, previous studies have demonstrated that diabetes is linked to an elevated risk of several malignancies, 8 including the liver, 9 pancreas, 10 colon and rectum, 11 and bladder. 12 While emerging evidence supports the benefits of multidisciplinary diabetes care against vascular complications, less evidence is available to support the potential cancer prevention effects from multidisciplinary care. Given (i) the common set of risk factors between diabetes and cancer; and (ii) and the potential improved cardiovascular risk profile 8 as a result of more comprehensive management, it remains less clear whether patients who have type 2 diabetes and receive multidisciplinary diabetes care may differ in site-specific cancer incidence from their standard care counterparts.
This study seeks to compare the clinical profiles and cancer incidence among patients who receive usual diabetes care, multidisciplinary diabetes care, and those who do not have diabetes in primary care.
Methods
Study design and study population
This is a retrospective cohort study using territory-wide electronic health records of Hong Kong. The Hospital Authority (HA) is a statutory body providing public healthcare services and managing a clinical data repository that stores records on patient demographics, disease diagnoses, prescription records, laboratory measurements, inpatient admissions, outpatient attendances, radiology reports, and clinical notes. Data used in this study were linked to death records from the Immigration Department. Disease diagnoses were coded according to the International Classification of Disease 10th revision (ICD-10), or the International Classification of Primary Care 2nd edition (ICPC-2). Individual-level data across datasets were linked via pseudonymous identifiers. Data were accessed via HA Data Collaboration Lab.
Multidisciplinary diabetes care
Patients who (i) received a diagnosis of type 2 diabetes; (ii) were able to perform their daily living activities independently; and (iii) attended follow-up medical consultations at general outpatient clinics (GOPCs) regularly would be potentially referred for multidisciplinary diabetes care. 13 Patients who received multidisciplinary diabetes care were offered non-pharmaceutical interventions (such as diet advice, smoking counselling and cessation, patient empowerment programme, and patient support hotlines) in addition to usual care (medical consultations and mainly pharmaceutical interventions) after evaluation of their metabolic risk profiles by trained nursing staff. On the other hand, patients with usual diabetes care only were offered follow-up medical consultations with doctors regularly at two-to-four-month intervals. Doctors would monitor their medical conditions, formulate appropriate treatment plan, and refer patients to specialists or allied health professionals if needed.7,14 Historically, since year 2000, the territory-wide multidisciplinary care model based on metabolic risk assessment has been implemented in 18 hospital-based diabetes centres, and scaled up to all 73 public GOPCs in year 2009. 14
Patients
Patients who received public healthcare services between year 1997 and 2021 with complete laboratory records (fasting glucose, low-density lipoprotein cholesterol, high-density lipoprotein cholesterol, triglycerides, and alanine aminotransferase) were first identified. Prescription records on anti-diabetic drugs were extracted to determine diabetes status of patients. Patients who received anti-diabetic drugs during study period were identified as diabetes patients, whereas those who did not receive anti-diabetic drugs during study period were identified as non-diabetes patients. The index dates for diabetes patients and non-diabetes patients were initiation of anti-diabetic drugs and earliest record of selected laboratory measurements respectively. The earliest index date was January 1st, 2000. Those with less than two records on any of the selected laboratory measurements within one year of the baseline were excluded. Records of identified diabetes patients were then matched to the records of individuals who received a clinical diagnosis of type 2 diabetes and participated in multidisciplinary diabetes management in GOPCs within one year of diabetes onset. As a result, 74,547 diabetes patients who received multidisciplinary care, 36,803 diabetes patients who did not receive multidisciplinary care, and 67,777 non-diabetes patients were initially identified. For diabetes patients with usual care and non-diabetes patients, those who did not attend any GOPCs within two years of the baseline were excluded. In addition, non-diabetes patients who had participated in metabolic assessment but never received any anti-diabetic drugs were excluded. For all groups, patients who had a prior history of cancer were further excluded. To exclude potential cases of type 1 diabetes, patients who received diabetes diagnosis below 30 years old were excluded. For patients with usual diabetes care, those who were diagnosed with diabetes below 60 years old
15
and received insulin treatment within one year of diabetes onset (but did not take other anti-diabatic drugs during study period) were excluded. Non-diabetes patients younger than 30 years old at baseline were also excluded. Patients were followed up until a cancer diagnosis, death, or December 31st, 2021, whichever occurred earlier. To minimize reverse causality, those with less than six months of follow-up were excluded.
16
For patients who developed cancer during follow-up, those who were (i) diagnosed with cancer at other non-target sites prior to diagnosis of cancer at studied sites; (ii) concurrently diagnosed with more than one studied site-specific cancers; or (iii) diagnosed with cancer at non-target sites only were further excluded. Consequently, a total of 25,301 patients with usual diabetes care, 66,382 patients with multidisciplinary diabetes care, and 54,376 patients without diabetes were finally included. Figure 1 shows the flowchart of patient selection. Flowchart of patient selection. Abbreviation: DM, Diabetes Mellitus.
Outcomes
The outcomes of interest were diagnosis of cancers of the colon and rectum (ICD-10: C18-21), liver (ICD-10: C22), pancreas (ICD-10: C25), stomach (ICD-10: C16), bladder (ICD-10: C67), kidney (ICD-10: C64-66, C68), and lung cancers (ICD-10: C33-34) during follow-up. These site-specific cancers are associated with obesity 17 or diabetes,8–12 or of high cancer incidence. 18 Due to the earlier age of onset of breast cancer and the apparent inverse association with diabetes for prostate cancer in most observational studies, 8 breast and prostate were not chosen as studied cancer sites since these two hormone-related cancers may exhibit patterns distinct from other site-specific cancers.
Covariates
Covariates included demographics (sex and age at baseline), behavioral factor (smoking), disease history, medication use, and laboratory measurements. Smoking was extracted from clinical notes, and categorized as ever versus never smoker. Disease history included possible risk factors for cancer (liver cirrhosis, 19 chronic viral hepatitis, 19 fatty liver, 19 chronic obstructive pulmonary disease [COPD], 20 pneumonia, 21 tuberculosis, 20 haematuria, 22 and cystitis), 22 and common comorbidities (ischemic heart disease, cerebrovascular disease, and heart failure). Disease diagnoses were determined from both inpatient and outpatient diagnosis codes. The presence of fatty liver was determined from both diagnosis codes and radiology reports (ultrasonography, computed tomography, and magnetic resonance imaging). Medication use included anti-diabetic drugs (metformin, sulfonylurea, insulin, dipeptidyl peptidase-4 inhibitors, acarbose, meglitinide, glitazone, sodium-glucose cotransporter-2 inhibitors, and glucagon-like peptide-1 receptor agonists), aspirin, non-steroidal anti-inflammatory drugs, anti-coagulants, anti-platelets, statins, anti-hypertensive drugs (angiotensin receptor blockers, alpha-blockers, angiotensin-converting enzyme inhibitors, beta-blockers, calcium channel blockers, and diuretics), and acid-suppressant drugs (H2-receptor antagonists and proton pump inhibitors). Medication use was defined as whether a patient had been prescribed a drug at baseline. Laboratory measurements included fasting glucose, low-density lipoprotein cholesterol, high-density lipoprotein cholesterol, triglycerides, and alanine aminotransferase. Laboratory measurements were taken as the mean value of at least two records closest to the baseline within one year.
Data analysis
Baseline characteristics of patients were presented in mean with standard deviation or median with interquartile range for continuous variables, and count with proportion for categorical variables. Comparison of baseline characteristics between patients with usual diabetes care and multidisciplinary diabetes care was conducted using logistic regression to adjust for demographics, behaviors, disease history, medication use, and laboratory measurements. The risk of site-specific cancers across three groups was estimated and compared using unweighted and weighted Cox proportional hazards regression separately. In the weighted models, weighting was assigned using inverse probability of treatment, where a synthetic population was created to balance measured baseline covariates between treatment groups. The probability of treatment of each individual was estimated using logistic regression and the weight assigned to each individual was the inverse of the probability of treatment an individual actually received. Baseline covariates used for weighting were age, sex, use of anti-diabetic drugs (metformin, sulfonylurea, insulin, and dipeptidyl peptidase-4 inhibitors), and fasting glucose. The estimates were reported in adjusted hazard ratio (aHR) with 95% confidence interval (CI). The cumulative site-specific cancer incidence (liver and pancreas) across three groups during follow-up was graphically examined. Statistical significance was set at p<0.05, two-sided.
Results
Baseline characteristics of patients according to diabetes status and management type.

IQR, interquartile range; SD, standard deviation.
Baseline clinical profile
Compared with diabetes patients with usual care, diabetes patients with multidisciplinary care tended to be younger (mean: 59.61 vs 69.29 years, p<0.001), and were less likely to have cerebrovascular disease (2.17 vs 6.78%, p<0.001), heart failure (1.74 vs 11.09%, p<0.001), COPD (0.46 vs 2.39%, p<0.001), pneumonia (0.14 vs 0.67%, p=0.002) and insulin treatment (11.11 vs 28.79%, p<0.001). They were also less likely to receive H2-receptor antagonists (57.34 vs 71.78%, p<0.001) and proton pump inhibitors (20.61 vs 45.45%, p<0.001). In addition, they tended to have lower ALT (35.37 vs 38.17 U/L, p<0.001), HDL cholesterol (1.23 vs 1.25 mmol/L, p<0.001), but a higher level of fasting glucose (8.43 vs 7.42 mmol/L, p<0.001). However, there was no significant difference in smoking habits, liver cirrhosis, chronic hepatitis B status, history of haematuria, or tuberculosis, after adjusting for demographics, smoking, disease history, medication use, and laboratory measurements (Table 1).
Cancer incidence
Cancer incidence across different organ sites according to diabetes status and management type.

aHR, adjusted hazard ratio; CI, confidence interval. Models were adjusted for age, sex, smoking, disease history (liver cirrhosis, chronic hepatitis B/C, fatty liver, chronic obstructive pulmonary disease, pneumonia, tuberculosis, haematuria, cystitis, ischemic heart disease, cerebrovascular disease, and heart failure), medication use (anti-diabetic drugs, aspirin, non-steroidal anti-inflammatory drugs, anti-coagulants, anti-platelets, statins, anti-hypertensive drugs, and acid-suppressant drugs), and laboratory measurements (fasting glucose, low-density lipoprotein cholesterol, high-density lipoprotein cholesterol, triglycerides, and alanine aminotransferase). aHR in bold indicates statistical significance at an alpha level of 0.05.
aWeighted using inverse probability of treatment.

Cumulative liver cancer incidence during follow-up by diabetes status and management type.
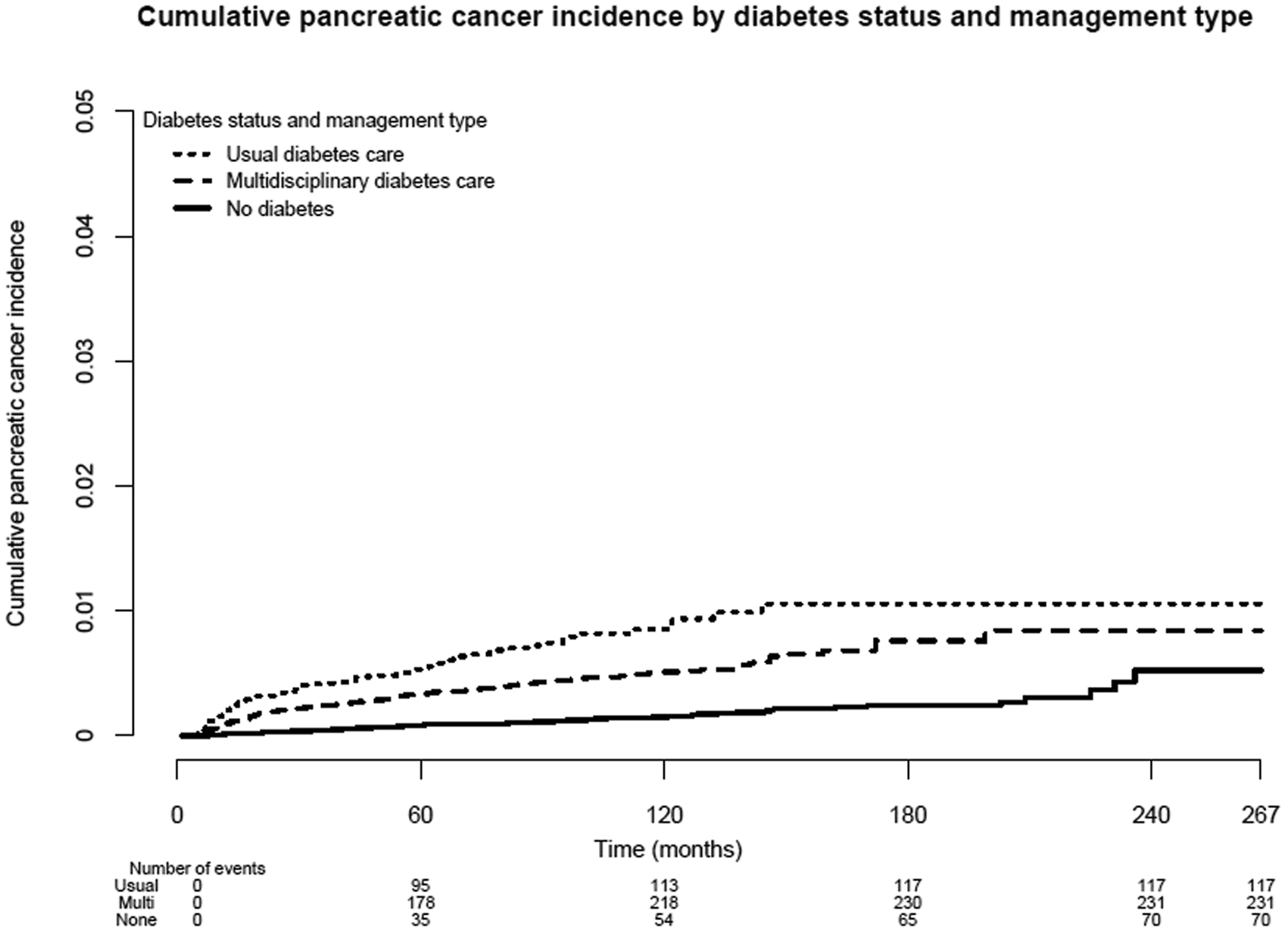
Cumulative pancreatic cancer incidence during follow-up by diabetes status and management type.
Discussion
The current study compared the clinical profiles and site-specific cancer incidence among patients who received usual diabetes care, multidisciplinary diabetes care, and those without diabetes in primary care. Overall, the risk of developing liver and pancreatic cancers was attenuated in patients receiving multidisciplinary diabetes care, when compared to those with usual diabetes care alone. While patients with usual diabetes care showed an increased risk of developing gastric cancer when comparing to those without diabetes in unweighted model, the association disappeared in weighted model, where the intrinsic differences in baseline characteristics were handled using inverse probability of treatment weighting. In addition, patients receiving multidisciplinary diabetes care seemed to be younger and have less comorbid conditions when compared to those receiving standard diabetes care alone.
While accumulating evidence has supported the risk reduction in suboptimal cardiovascular profile, 4 vascular complications 7 and mortality 7 in multidisciplinary diabetes care, findings of the present study suggest the potential additional risk reduction in several diabetes-associated cancers, namely liver and pancreatic cancers, even adjusting for the differences in baseline profile. Prior research suggests not only the multifactorial nature of diabetes 4 but also the co-occurrence of multimorbidities and polypharmacy with diabetes, 5 rendering challenges in physician care alone.4,5 The observed attenuated risk could be due to several possibilities: (i) differences in referral patterns resulting in more favorable clinical outcomes in patients receiving multidisciplinary diabetes care; or (ii) healthier lifestyle patterns and improved cardiovascular health supported by multidisciplinary care favorably modifying cancer outcomes in patients with diabetes.
It has been recognized that diabetes often coexists with a wide range of long-term conditions across various organ systems beyond vascular complications, for example, arthritis, 23 infectious diseases,23,24 and mental illnesses.23,25 Several multifactorial diseases including heart failure, chronic kidney disease, COPD, and metabolic-associated fatty liver disease, which may share common underlying mechanisms linked to cancer, 26 are frequently present in patients with diabetes. In addition, prior research 27 suggests that the presence of chronic kidney disease in patients with diabetes could be an indicator of the presence of other vascular complications, since different tissue-specific complications share the same underlying pathogenetic mechanisms driven by chronic hyperglycemia. These suggest a degree of interconnectedness across different comorbid conditions in diabetes, and the possibility of managing cardiovascular health reducing the risk of other conditions, including cancer.
The present study adds to the literature by exploring the potential benefits of multidisciplinary diabetes care beyond improving cardiovascular profile and reducing the risk of vascular complications. Findings of the study suggest that multidisciplinary diabetes care may also lower the risk of diabetes-associated cancers, including liver and pancreatic cancers. Furthermore, since multidisciplinary diabetes care is designed to address the multidimensional care needs of patients due to the common co-occurrence of multimorbidities and polypharmacy, 4 beyond reducing the imminent risk of vascular complications, it may potentially mitigate the risk of other conditions frequently present in diabetes. Additionally, cardiovascular disease and cancer have been historically recognized as distinct entities and their potential linkage has been understudied.28,29 A recent meta-analysis 28 has shown that patients with cardiovascular disease have a higher risk of developing overall cancer and pancreatic cancer when compared to those without cardiovascular disease. Given the shared risk factors between cardiovascular disease, diabetes, and cancer, their interrelationships may warrant further investigation.
Several limitations may exist in the present study. First, the observed risk reduction in patients receiving multidisciplinary diabetes care could be due to inherent differences in clinical characteristics, referral patterns, or patient preferences. Nevertheless, these differences may not totally explain the differential associations between diabetes and cancer across different organ sites. In addition, results between no weighting and weighting with inverse probability of treatment remained consistent. Second, blood glucose or HbA1c records were not checked to exclude the possibility of undiagnosed diabetes for non-diabetes status due to infeasibility. Third, some potential confounders such as alcohol use or adiposity indicators were not available. Fourth, time-varying exposure to covariates was not captured. Further studies with longer follow-up are warranted to explore whether multidisciplinary diabetes management may not only alleviate the risk of vascular complications but also diabetes-associated cancers and potentially other comorbid conditions. Future research is needed to evaluate generalizability of the findings to other populations due to differences in healthcare systems and cultural beliefs. 30
Conclusion
The risk of developing liver and pancreatic cancers appeared to be attenuated in patients receiving multidisciplinary diabetes care when compared to those receiving standard diabetes care. Future research is warranted to explore whether more comprehensive diabetes management may not only help lower the risk of vascular complications but also diabetes-associated cancers, and potentially other common comorbid conditions in patients with diabetes.
Footnotes
Acknowledgements
None.
Ethical considerations
Ethics approval for secondary data analysis was provided by the Chinese University of Hong Kong - Survey and Behavioural Research Ethics Committee (reference number: SBRE-22-0386).
Consent to participate
Patient consent to participate was waived since individuals were not identifiable in this study.
Consent for publication
Patient consent for publication was waived since individuals were not identifiable in this study.
Author contributions
Conceptualization & Methodology, STYY, CTH, and EKY; Data Curation & Formal Analysis, STYY; Writing – Original Draft Preparation, STYY; Writing – Review & Editing, CTH, EYML, AL, and EKY; Supervision, CTH and EKY; Funding acquisition, AL.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Hong Kong SAR government - Strategic Public Policy Research Funding Scheme (S2019.A4.015.19S).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data is not available for sharing due to access restriction.
