Abstract
Purpose
This article reports the development of the NOMAD intervention, a model of multidisciplinary team conferences designed to improve care coordination and treatment for patients with multimorbidity and diabetes. Managing multiple chronic conditions increases care complexity, and multidisciplinary approaches may enhance care coherence, optimise treatment, and improve quality of life.
Method
Guided by the Medical Research Council framework for developing complex interventions, we established a working group, formulated a programme theory, and conducted a preliminary test with qualitative interviews of patients and physicians. The intervention targets patients with diabetes and concurrent chronic conditions affecting the heart, lungs, or kidneys.
Results
The NOMAD intervention was developed as a structured model of regular, in-person multidisciplinary team conferences involving specialists in endocrinology, cardiology, respiratory medicine, nephrology, and pharmacology. A key component was the integration of patient-reported outcomes to enhance patient-centred care, highlighting the aspect of quality of life.
Conclusion
This development process provides a foundation for feasibility testing and future evaluation of NOMAD’s impact in a randomised trial. Our experiences may also inform researchers and healthcare providers developing similar multidisciplinary interventions for complex patient populations.
Keywords
Introduction
Multimorbidity increases complexity in care and treatment. An increasing number of conditions are associated with an increasing number of care providers, number of transitions between care providers and number of hospital trajectories. 1 Rising multimorbidity prevalence represents massive challenges. 2 Patients with multimorbidity experience a significant workload organising care, attending appointments and complying with treatment regimens and lifestyle changes. 3 Problems arise as many diseases share pathophysiological pathways and interact with each other while care providers focus on single-disease treatment. 4
The definition of multimorbidity is still not fully agreed upon. A review of reviews found three main approaches to defining the term: a simple count of diagnoses suitable for epidemiological purposes; a more clinical approach where, for instance, the severity of disease and risk factors are taken into account; and finally, an approach where all biopsychosocial factors are considered together with somatic risk factors, social network and burden of diseases. 5 However, a common definition is the presence of two or more chronic diseases. 6
Studies investigating which diseases tend to co-occur have found that clusters containing diabetes and cardiovascular diseases (CVD) are prominent. 7 Diabetes, a highly prevalent disease, is one of four main non-communicable diseases globally (CVD, cancer, diabetes and chronic respiratory diseases), resides among the top-ten causes of death and has increasing mortality rates.8–11 Type 2 diabetes is associated with co-occurrence of CVD, kidney diseases and mental health issues as well as more underreported comorbidities such as osteoarthritis and respiratory diseases. 12
A multidisciplinary approach has been suggested to address the challenges of multimorbidity care. 13 The multidisciplinary team (MDT) is a well-established approach in oncology and has spread to other fields of medicine in recent years. 14 Nonetheless, the evidence base for how to develop, implement, and evaluate non-oncology multidisciplinary interventions for people with multimorbidity is limited. 15
Therefore, we aimed to develop a complex intervention (Network of Doctors for Multimorbidity and Diabetes – the NOMAD intervention) to address these challenges in multimorbidity care. Our goal was to increase quality and length of life for people with multimorbidity and diabetes through increasing coordination in care and facilitate skills development across specialities. We wanted to use the MDT approach to achieve this.
This article reports the development of the NOMAD intervention and presents the final model.
Method
Framework and theory
Complex multimorbidity necessitates complex and multidisciplinary interventions focusing on care coordination and patient-centredness.16,17 Introducing change (a complex intervention) in large organisations requires evidence-based planning, pragmatic consideration of context and stakeholder involvement, 18 along with attention to existing practice, theory and frameworks. 19
We used the Medical Research Council (MRC) framework for developing and evaluating complex interventions as a guide for the development process. 19 It provides a non-linear approach to intervention development, characterised as dynamic and iterative. In addition, Guidance for developing and evaluating complex interventions by O’Cathien et al. inspired us as well. 20 The reporting of the NOMAD development followed the GUIDance for the rEporting of intervention Development – GUIDED – recommendations. 21 Leaning on the Template for Intervention Description and Replication (TIDieR) checklist, we briefly report the final intervention. 22
The development process consisted of four phases (see Figure 1): 1) Establishing a working group and planning the development. 2) Testing an early intervention model and preliminarily evaluating it, which led to refining and adjusting the intervention. 3) Establishing a programme theory exploring mechanisms of change and hereby creating a detailed logic model. In this phase, we included a journal audit to elucidate characteristics of the patients and MDT discussions, and to inform the design of the feasibility test in phase four. 4) Setting up and conducting a mixed-methods feasibility study of the intervention.
23
This article describes the first three phases, which occurred in a cyclic, iterative manner rather than a chronological order. NOMAD development process – The three initial phases are reported in this article (blue boxes numbered 1–3). The finished model of the NOMAD is described in a written manual (blue box numbered 5). Green boxes indicate next steps after development. The circular and two-way arrows indicate the iterative nature of the development process. NOMAD: Network of doctors for multimorbidity and diabetes.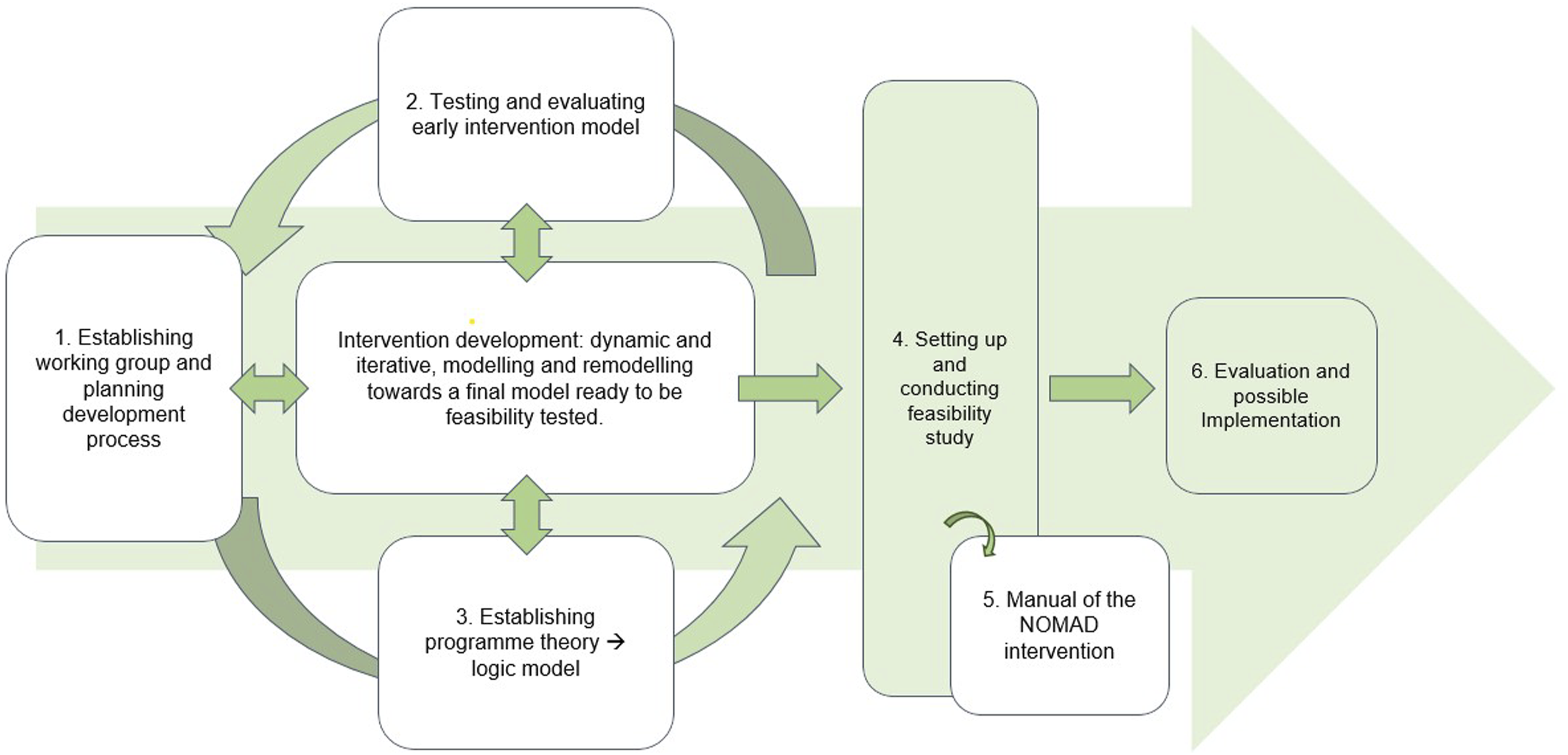
Setting
The intervention development was set at the Steno Diabetes Centre Odense (SDCO), Odense University Hospital (OUH), Odense, Denmark’s third-largest city. Denmark has free-of-charge, tax-funded healthcare for all citizens.
OUH has a catchment area of approximately 275,000 people. 24 The Steno Diabetes centres provide diabetes treatment to the public, operated and funded by the regional governments, and conduct diabetes-related research funded by the Novo Nordisk Foundation. SDCO coordinated the development process by providing staff support, technical equipment, physical premises and overall project management. The project period was 2019–2025.
Target population
We defined the intervention receivers to be people with diabetes and concurring chronic conditions. We established three referral criteria to identify patients who would likely benefit from the NOMAD: 1) a diabetes diagnosis, 2) concurring chronic conditions affecting the heart, lungs or kidneys and 3) the presence of complexities or challenges in their care or treatment plan, including polypharmacy. Physicians and hospital administrative workers were the intervention providers. Doctors from either hospital departments or general practice (GP) in a geographically limited area (the island of Funen and adjacent islands) could refer patients to the NOMAD.
Establishing the team
During meetings in 2019–2020, we established a working group with representatives from various hospital departments. This marked the start of the development process. The participating medical specialities were chosen based on the project description, internal audit revealing overlapping diseases across specialities, and factors like local resources and workforce capacities. The group’s initial tasks included defining and planning the development process, assigning tasks and drafting a clinical initiative description. Financial considerations and budgeting were also essential. To prevent conflicting priorities, we decided to reimburse departments contributing workers/doctors to ensure they had dedicated time to the MDT.
Early testing
Distribution of interview participants, their position, interview type and medium of communication.

Programme theory and logic model
Following the MRC framework, we collated existing evidence early in development. Two reviewers (DPH, ZE) analysed literature on MDT in chronic, non-malignant disease, noting its heterogeneity and lack of consistent terminology. 25 They found that while MDT studies altered treatment, they showed no impact on clinical outcomes like quality of life or lab values. 25 Building on this, we applied programme theory to create a logic model illustrating the intervention’s hypothesised change mechanisms. 26 Two workshops and an individual assignment refined this model. Additionally, a role-play workshop with actors tested these mechanisms, revealing key communication and relational dynamics in MDT conferences, with and without patient involvement.
Patient-centred approach
To secure patient-centred focus and enable patient influence on NOMAD discussions, we tested two approaches for patient involvement in the conference setting: 1) patient attending the conference and 2) patient completion of a patient-reported outcome (PRO) questionnaire before the conference without attending. The PRO questionnaire brought information about the patient’s quality of life, symptom burden and other aspects of multimorbidity and diabetes into the NOMAD discussion. After testing the two approaches, the working group reached a consensus on whether to include patients in the NOMAD conference.
We reviewed the literature (led by IRS) to identify relevant, validated questionnaires for the PRO, including how to assess the PRO. Additionally, NOMAD clinicians shared their views on valuable patient-reported information to enhance the discussion. We chose a digital questionnaire solution through an official hospital electronic service (an application called ‘Mit Sygehus‘, which translates to ‘My Hospital’). Through this solution, the PRO was included in electronic patient records and thus accessible to all NOMAD members. If the patient was unable to use the digital solution, a healthcare professional filled out the questionnaire by telephone interview. Because the NOMAD sought to enhance care coordination, we established a follow-up session by telephone six months after the NOMAD conference.
User involvement
In our setting, we defined users as patients and healthcare professionals (ie the NOMAD physicians. To ensure relevancy and increase chances of successful implementation we involved both users and stakeholders (eg hospital decision-makers) in the development process.19,27 We involved users on different levels: patients were involved in their own treatment, and patients along with healthcare professionals and decision-makers were involved in the development and evaluation of the NOMAD. For instance, three patient representatives helped form the PRO questionnaire layout and supplementary texts. To initiate and facilitate cross-sectoral collaboration with GP on Funen, we consulted a GP as advisory expert. We established a user panel of patients, relatives and healthcare professionals from both hospital and primary care to incorporate their perspectives, particularly regarding the evaluation of the NOMAD intervention and its possible implementation in clinical practice. We adopted a dynamic approach resembling the Plan–Do–Study–Act (PDSA) cycle, 28 continuously reflecting on the development process to identify uncertainties and engaging relevant users to address issues as they emerged. Regular working group meetings, held in parallel with the intervention (i.e., real-life MDT meetings in their current form), enabled iterative refinement: we acted by conducting MDT meetings, studied outcomes in the working group, planned changes (e.g., extending case discussion time from 20 to 30 minutes), and did by implementing adjustments accordingly.
Results
We present the results of the development process as the three development phases and finally the NOMAD intervention.
Phase 1: Working group and roles
The working group consisted of physicians, researchers and hospital staff members from five specialities at Odense University Hospital – cardiology, respiratory medicine, nephrology, pharmacology and endocrinology. The group appointed a team leader, a clinical leader and a project manager. Whereas the team leader oversaw the intervention development process, the clinical leader was in charge of conducting the NOMAD conferences. The project manager coordinated all efforts related to both the development process and the clinical NOMAD conferences. Two or three physicians from each speciality were included in the NOMAD clinical team, ie the team performing all the NOMAD conferences.
Phase 2: Early testing interview results
All interviewees had some degree of experience with the NOMAD intervention, either as a physician participating in the NOMAD discussion, as a patient being discussed, as a health secretary organising the NOMAD referrals or as a GP receiving a NOMAD conclusion note.
Among NOMAD intervention providers, responses ranged from very positive to critical. Two key themes emerged 1 : identifying the right patient for NOMAD and 2 clarifying physicians’ responsibilities within NOMAD. Despite criteria on multimorbidity challenges, the ‘right patient’ for NOMAD remained unclear to many physicians, who questioned whom it could truly benefit. Scepticism also surfaced: ‘This is a big setup – I wonder if the benefits outweigh the costs…’ One hospital physician described NOMAD as ‘a necessary initiative,’ emphasising, ‘We work in hospitals where care is increasingly specialised, and the different specialities no longer have a broad approach. The multimorbid patient needs more coordination.’ Echoing this, another physician asked, ‘Who should be in charge of the patient? This is not clear, and we spend a lot of time discussing it.’ Finally, they agreed that GPs should be invited as NOMAD and be able to both refer patients to the conference and participate in the discussion.
From a patient point of view, NOMAD was experienced as positive. One patient described the importance of being seen as a whole person. Diseases cannot be seen and handled separately, as they influence each other and often require prioritisation. Similarly, the collaboration with the GP was seen as beneficial, as the patient expresses, ‘she is just a regular doctor.’
Overall, patients found it challenging to gain an overview of their treatment pathways. Two patients described how they sometimes felt caught between different departments and their GP, feeling it was a struggle. One patient explained that it was difficult to comprehend and separate different treatment pathways, which led to confusion about who they had spoken with and what they discussed. The view of the GP’s role varied among patients; for some, the GP was the one who tied everything together. ‘Yes, it would be an advantage if the GP participated on the meeting [NOMAD conference] and followed up with me after.’ While for others, the GP did not take on this role.
Phase 3: Logic model
The logic model shows that we hypothesise that the NOMAD intervention – given the right resources and input – can result in patient-centred care, produce advantages like feeling seen and heard, reduce inappropriate medication, improve clinician overview of treatment and increase care coordination, ultimately leading to fewer hospitalisations, increased quality of life and reduced mortality. Figure 2 shows a summarised version of the logic model (a complete version of the logic model is available upon request). Logic model – From left to right: Resources that form the base of the intervention (blue circle). The NOMAD intervention before, during and after the NOMAD conference (green boxes). The results of the patient-centred approach are divided into short-term, middle-term and long-term perspectives for healthcare professionals and patients, respectively.
PRO questionnaire and patient participation
The development process resulted in a questionnaire consisting of 59 questions. The questions came from nine different generic, validated questionnaires concerning health-related quality of life, physical and mental well-being, diabetes distress, symptom burden, treatment burden and feeling of involvement (details previously published here. 23 All questions were in Danish. We applied validated score algorithms where applicable, and set up a system of colour codes indicating severity of PRO answers to make assessment easier. When setting up the digital solution for questionnaire delivery, we followed available local guidelines and regulations, such as completing an extensive technical verification test and risk assessment for implementing a medical device (available upon request). The questionnaire also contained a free text question allowing the patient to pinpoint their personal concerns, viewpoints or ideas for NOMAD discussion as well as a question asking if they wanted to be physically or virtually present.
Eight patients participated in their NOMAD discussion: six participated physically and two virtually alongside their GP. On three occasions, relatives also participated. The working group decided that the current NOMAD setup was not appropriately geared to accommodate patient participation. They therefore recommended PRO questionnaires along with six-month individual follow up as sole elements to ensure patient-centredness.
The final model: The NOMAD intervention
The development process resulted in the Network of Doctors for Multimorbidity and Diabetes – the NOMAD intervention. The NOMAD is regularly scheduled MDT-meetings that occur every two weeks, is an in-person meeting at Odense University Hospital, lasts for two hours and can discuss up to four patients per meeting ( = 30 minutes per patient case). A NOMAD intervention timeline is visualised in Figure 3. Patients who meet the referral criteria can be referred either from GP or from hospital doctor. If GP refers the patient, then they participate in the meeting online. The NOMAD consists of a fixed panel of medical experts, representing endocrinology, cardiology, respiratory medicine, nephrology and clinical pharmacology. When relevant, the NOMAD can invite other experts, such as palliative specialists. The referred patient completes a PRO questionnaire before the NOMAD conference. The NOMAD members read the patient’s health records thoroughly, including the PROs, and assess treatment from their area of expertise. The pharmacologist conducts a comprehensive medication review to assess appropriateness, safety and compliance. The NOMAD team agrees on a coordinated plan for treatment, care and follow up, including who is in charge. Six months after the NOMAD conference, the patient completes the PRO questionnaire again. A healthcare professional then follows up by reviewing the electronic medical record to assess whether the NOMAD recommendations have been addressed, and the patient is contacted by telephone. If significant issues are identified, the patient may be re-referred for a new NOMAD conference. Please refer to the published protocol for a more detailed description of the follow up procedure.
23
A manual of the intervention is available in Danish upon request. NOMAD intervention timeline from referral to end of follow-up after six months.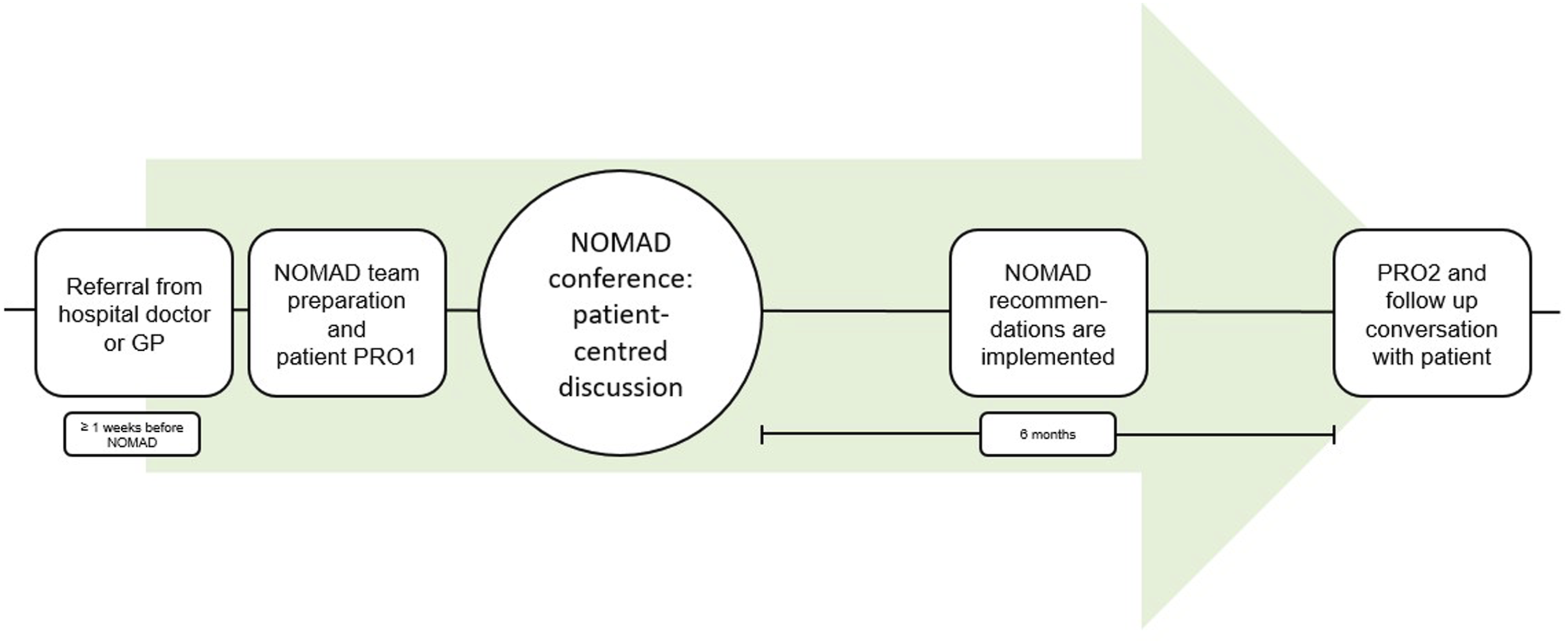
We produced a pamphlet aimed at both patients and physicians (in Danish, available upon request) aiming to summarise what the NOMAD is, the target group and how to refer a patient. We distributed the pamphlet in hospital departments and to the GPs all over Funen. To increase awareness about the NOMAD, we gave presentations at hospital departments’ morning meetings, in GP offices and at regional GP/healthcare worker conferences. Additionally, we provided written material for hospital departments, such as instructional guides about the intervention.
Discussion
We developed a concept of MDT conferences aimed at people with multimorbidity and diabetes – the NOMAD intervention. Guided by recognised guidelines for complex intervention development,19,20,27 the final model resulted in 1) a clinical NOMAD team, 2) workflow processes related to preparing, performing and follow-up of NOMAD conferences and 3) written material for patients and physicians, respectively. Overall, these results constitute a care approach for people with multimorbidity and diabetes, ready for feasibility testing.
Other programs aim to improve diabetes care by targeting primary care providers, addressing health disparities, and improving patient outcomes. One example is the Echo Diabetes Action Network (EDAN), 29 which takes a more generic approach by using anonymised cases and meeting in global learning collaborative hubs. The focus is on professional learning and peer support rather than on individual patients.
In contrast, the NOMAD intervention is based at a single hospital and involves specialists from five disciplines. It focuses on referred patients, aiming to improve treatment and care pathways for each individual case.
The establishment of an interprofessional collaboration, such as the NOMAD working group and clinical team, necessitates the engagement of motivated individuals. Creativity researcher T. Amabile argues that creating new ideas, methods or concepts – eg a complex intervention like the NOMAD – relies on three components: expertise, creative thinking and motivation. 30 Establishing a team with the desired level of ingenuity requires diversity in expertise and creative thinking among team members that can complement each other and transform into useful results. 30 However, this emphasis on individual motivation may pose challenges to the generalisability of our process, as identifying and engaging suitable team members can be subjective and influenced by various local/structural factors. This ambiguity may not be readily apparent to those attempting to replicate our approach.
The clinical NOMAD team composition aligns well with the most prevalent diseases in the background population. 31 Cardiovascular, pulmonary and nephrological diseases all display higher prevalence among people with diabetes compared to background population, and – depending on definition – most of these patients are subject to polypharmacy.12,32,33 Also in a global perspective, diabetes (including kidney disease), cardiovascular and chronic respiratory diseases constitute the main categories of non-communicable diseases together with cancer and mental health disorders. 34 Thus, a shortcoming in our team composition is the lack of mental health expertise. Numerous studies highlight co-occurrence of somatic and psychiatric disease as a barrier for coherent and optimal care and point out a potential to improve outcomes for these patients.35–37 Held together with the fact that multimorbidity is associated with increased prevalence of mental health disorders and co-occurrence of the two increases risk of functional decline,38,39 lacking mental health expertise represents a limitation in our setup.
The decision to compensate departments lending work force to the project arose from an a priori consideration of intervention sustainability and potential implementation in clinical practice.19,27,40 Arguably, a financial incentive to provide work force to the NOMAD could shift team member’s intrinsic motivation to partake in a meaningful clinical intervention to a more extrinsic motivation: ‘I have to be here because my employer says so’ or ‘this is good business for my department’ (possible opinions, not quotes). On the other hand, simply compensating departments for the pre-agreed amount of time the physicians dedicate to NOMAD, without offering additional payment, should not incentivise participation financially. Instead, it serves as a means of enabling their involvement. The financial compensation over such a long development and feasibility period (five years) from project funds enable departments to participate throughout the project period, and sustained participation will hopefully promote intervention normalisation and embedding in daily practice,41,42 ultimately increasing the chance of implementation once the project period (and funds) end.
Articulating the mechanisms of change increased comprehensibility of how our intervention would facilitate change ie increased quality of life and length of life. This marked a turning point in the development process: after this point, the working group shared a common understanding of the NOMAD’s potential to fulfil its purpose. 41 We therefore emphasise this as a rewarding step in the development process that – in our experience – enhanced onboarding among team members. The logic model uncovered relevant aspects that we, for instance, used to select appropriate PRO questions. Our selection of PRO questions resonates well with other studies concerning which variables are relevant to focus on in multimorbidity care. 43
The terms person-centred and patient-centred are closely related and oftentimes used interchangeably.44,45 A review of reviews suggest that a person-centred approach concerns the goal of a meaningful life, whereas a patient-centred approach concerns the goal of a functional life. 46 We decided to use the latter when describing our intervention and its purposes. By patient-centred, we understand a care approach where the patient is respected as a person, and their standpoint is taken into account when making decisions about care and treatment. 44
Early testing interviews revealed that patients wanted to participate in their own NOMAD discussion, aligning with findings from another feasibility study. 47 However, when offered the opportunity, only a few accepted. This discrepancy between stated and actual preference, coupled with the observation that patient presence decreased discussion effectiveness, led us to remove this option. This may reduce the level of patient-centredness in the NOMAD discussions. If reintroduced, we will prioritise a patient-friendly venue, provide dedicated support throughout the process (eg a healthcare professional accompanying the patient) and align expectations to ensure continuity and understanding for the patient. 48
Implications
Throughout the process, we prioritised generalisability. Therefore, the NOMAD can be adapted to other settings with complex patient care, while considering local circumstances as outlined in the MRC framework. 19
We believe the NOMAD will benefit both intervention recipients and providers. Patients may experience improved care coordination, leading to a better quality of life. Formal knowledge sharing, interaction among professionals and collaboration across hospital departments and health sectors can enhance patient care, underscoring the vital role of the GPs as core generalists in the healthcare system. Future studies should explore the benefits of knowledge sharing across disciplines and sectors when investigating the effects of complex interventions for people with multimorbidity.
Conclusion
We developed a complex intervention for people with multimorbidity and diabetes – the NOMAD – that we expect will increase coordination in care and treatment and facilitate competence development across specialities. The NOMAD is tested for feasibility between 2023 and 2025 to determine if the design is appropriate for a randomised study of effect. 23 This article reports the development process, aiming to inform other developers embarking on similar endeavours.
Footnotes
Acknowledgments
We are very grateful to all the patients, GPs and secretaries who have participated, provided feedback and otherwise contributed to this work. Special thanks to Camilla Hansen and Ewa Romanczuk for their relentless dedication and great effort in organising this project.
Ethical considerations
As this study focused on the development process of the NOMAD and contained no intervention on human subjects, no ethical approval was necessary. The department of quality studies at Odense University Hospital approved the journal audit.
Consent to participate
All interview-participants returned written, informed consent to participate.
Consent for publication
Informed consent for publication was provided by the interview-participants.
Author contributions
The NNF provided an initial project description (available upon request) and had no role in the development process beyond that. The researchers were at liberty to amend the initial project description, as they felt appropriate. All authors contributed in the development of idea and NOMAD concept including written material. ADOZ was project leader. JH, ADOZ, PD, PBJ, SLJ, SN, VP, MKP, MTR, IDR and DPH constituted the clinical team. SJB collected data and wrote the first draft. MJR supervised the qualitative elements. MBH and SJB developed the PRO-questionnaire. All authors critically reviewed and accepted the manuscript.
Funding
The Novo Nordisk Foundation (Steno MDT), the Region of Southern Denmark and Steno Diabetes Centre Odense funded this work. Region Syddanmark, J.nr.: 23/7880, A1913. Steno Diabetes Center Odense, A5903.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
As mentioned in the text, any additional documents can be made available on request.
