Abstract
Background:
Levonorgestrel-releasing intrauterine systems (LNG-IUS) are considered off-label and investigational in the United States for treating non-atypical endometrial hyperplasia (NAEH), atypical endometrial hyperplasia (AEH), and early-stage endometrial cancer (EC), though evidence suggests potential benefit.
Objectives:
To summarize the available evidence regarding the use of LNG-IUS, with or without other therapies, for the treatment of NAEH, AEH, and early-stage (FIGO I/IA) EC.
Design:
Systematic literature review and meta-analyses.
Data sources:
MEDLINE, Embase, Cochrane Central Register of Controlled Trials (CENTRAL), and Cochrane Database of Systematic Reviews (CDSR) were searched from database inception through May 2022.
Methods:
Data from studies reporting complete outcome definitions and study populations that were aligned with the 2014 World Health Organization (WHO) endometrial hyperplasia classification criteria or were defined as FIGO grade 1 stage I/IA EC were extracted and assessed in random-effects meta-analyses. A risk of bias assessment was completed using recommended study-design-specific tools.
Results:
1085 unique records were reviewed; 80 publications were included, with data from 30 assessed in meta-analyses: NAEH, n = 14; AEH, n = 21; EC, n = 5. Complete response (CR) rates at 12 months were high: 86.3% (95% confidence interval (CI): 78.50%–91.55%; n = 342) of patients with NAEH, 83.9% (95% CI: 71.95%–91.34%; n = 196) of patients with AEH, and 51.1% (95% CI: 16.30%–84.90%; n = 20) of patients with EC. LNG-IUS was associated with significantly greater odds of CR at 12 months compared to oral progestins in NAEH and AEH populations, with odds ratios ranging from 2.16 (95% CI: 1.38–3.36) to 4.56 (95% CI: 2.57–8.09). No comparative data were available for EC. Safety information was consistent with that of approved LNG-IUS indications. Risk of bias varied across study designs.
Conclusion:
The findings suggest that NAEH, AEH, and early-stage (FIGO I/IA) EC can be effectively treated with LNG-IUS, with the evidence being strongest for NAEH and weakest for EC. Greater availability of LNG-IUS treatment could provide meaningful benefit to these patients.
Trial registration:
Not applicable.
Plain language summary
This comprehensive review of published scientific studies looked at whether a type of hormonal device called a levonorgestrel-releasing intrauterine system (LNG-IUS) is effective and safe for treating abnormally thickened uterine lining and early uterine cancer. The specific uterine conditions of interest included non-atypical endometrial hyperplasia (NAEH), atypical endometrial hyperplasia (AEH), and early-stage endometrial cancer (EC). Researchers reviewed studies published up to May 2022 and identified 80 relevant articles, focusing on 30 of them for detailed analysis.
A high percentage of patients had positive outcomes. In 86.3% of patients with NAEH, 83.9% of patients with AEH, and 51.1% of patients with EC, there was no evidence of disease after 12 months of treatment. The LNG-IUS was found to be more effective than oral hormone medications for treating NAEH and AEH. However, there was not enough information available to make direct comparisons for patients with EC. The safety of LNG-IUS treatment was similar to what has already been established for its approved uses.
Overall, the published literature suggests that LNG-IUS is an effective treatment option for NAEH, AEH, and EC, with the strongest evidence for NAEH and the weakest evidence for EC. Greater access to this treatment could offer significant benefits to patients with these uterine conditions.
Introduction
Endometrial hyperplasia (EH) occurs when the endometrium (lining of the uterus) becomes unusually thick because of hyperplasia (increased cellular proliferation). 1 EH classification is based on the type of cell changes in the uterine lining and has been described with varying terminology. In 2014, the World Health Organization (WHO) Classification for Female Genital Tract Tumors endorsed a two-tiered schema: non-atypical endometrial hyperplasia (NAEH) and atypical endometrial hyperplasia (AEH). 2 NAEH is also known as benign EH, while AEH is also known as endometrial or endometrioid intraepithelial neoplasia. 2 Older criteria, such as the 1994 WHO classification, which classified hyperplasia as simple or complex, with or without atypia, cannot always be aligned with the 2014 WHO classification.
In the United States (US), EH incidence is 133–208 cases per 100,000 woman-years. Incidence rates of the main EH subtypes are 121 per 100,000 woman-years for NAEH and 16.8 per 100,000 woman-years for AEH.3,4 EH is both a precursor and concurrent marker of endometrial cancer (EC), the sixth most common cancer among women and the most common gynecologic malignancy worldwide. EC develops in 1.6%–10% of women with untreated NAEH and 15%–40% of women with untreated AEH.5–10
At this time, there are no drug therapies approved in the US to treat EH. According to the American College of Obstetricians and Gynecologists (ACOG), EH can often be managed with the off-label use of progestins. 1 Since the majority of NAEH cases are successfully managed medically, hysterectomy is not considered first-line treatment, and surgery is reserved for specific circumstances. 11 For the treatment of AEH, total hysterectomy with bilateral salpingo-oophorectomy is recommended for perimenopausal and postmenopausal women, while ovarian preservation should be discussed with premenopausal women. 11 For individuals who are not surgical candidates or desire preservation of fertility, progestin treatment provides an alternative option. 12 The primary treatment for EC is usually total hysterectomy with bilateral salpingo-oophorectomy, although European guidelines recognize hysteroscopic tumor resection followed by oral or intrauterine progestins as a fertility-sparing option. 13 Evidence suggests that continuous progestin-based therapy has been successfully used in patients with International Federation of Gynecology and Obstetrics (FIGO) grade 1, stage IA (noninvasive) disease who wish to preserve their fertility.14–18
Current US guidelines acknowledge the possible benefit of intrauterine progestin treatment.1,19 However, the use of levonorgestrel-releasing intrauterine systems (LNG-IUS) for the treatment of NAEH, AEH, and early-stage (FIGO I/IA) EC is considered off-label and investigational. However, multiple countries have approved the 52 mg LNG-IUS Mirena as protection from EH during estrogen replacement therapy. In the US, Mirena is currently approved for the prevention of pregnancy for up to 8 years and for the treatment of heavy menstrual bleeding for up to 5 years for women who choose to use LNG-IUS as their method of contraception. 20
There is a growing number of reports describing the use of 52 mg LNG-IUS as a treatment option for women with endometrial conditions. Recently published literature reviews examine results from a subset of specific study designs (RCTs, comparative studies), report data only for patients with EC, or focus on specific country populations.21–24 This systematic literature review (SLR) and meta-analysis was conducted to assess the efficacy, effectiveness, and safety of LNG-IUS with or without other therapies for the treatment of EH (of any type) and grade 1 early-stage (FIGO I/IA) EC across a broad range of interventional and real-world studies including RCTs, single-arm trials, non-interventional/observational studies, and case series. This article is a revised and expanded version of an abstract and associated poster entitled “LNG-IUS for the treatment of EH and early-stage endometrial cancer: A systematic review and meta-analysis,” which was presented at ACOG Annual Clinical and Scientific Meeting, San Francisco, CA, May 17–19, 2024, and is available online. 25
Methods
This study was an SLR to assess the efficacy, effectiveness, and safety of LNG-IUS with or without other therapies for the treatment of EH (of any type) and grade 1 early-stage (FIGO I/IA) EC. Meta-analyses were conducted when possible based on outcome data availability. The SLR search was conducted in MEDLINE, Embase, Cochrane Central Register of Controlled Trials (CENTRAL), and Cochrane Database of Systematic Reviews (CDSR) from database inception through 25 May 2022. Search terms included combinations of free text, Emtree terms, and Medical Subject Headings (MeSH) (Supplemental Appendix A).
Other sources, including recent congress proceedings (2019–2022), the European Union Electronic Register of Post-Authorization Studies (EU PAS Register), and ClinicalTrials.gov, were searched. Reference lists of identified articles, systematic reviews, and clinical trials were also reviewed. Only articles and abstracts of original research published in the English language were included. Citations retrieved by the literature searches were downloaded into reference management software (EndNote X20; Clarivate Analytics, Philadelphia, PA, USA), duplicates were removed, and the remaining entries were uploaded into DistillerSR software (Evidence Partners, Ottawa, ON, USA) for literature screening. The PICOS/PECOS (population, intervention/exposure, comparison, outcomes, study design) framework was used to assess studies for inclusion (Supplemental Appendix B). The PICOS framework was used for interventional studies since the treatments assessed can be described as interventions. The PECOS framework was used for observational studies since treatments assessed can be described as exposures. The article selection process was performed in two phases: title/abstract screening then full-text screening. Titles/abstracts and full-text publications were independently screened by two researchers to determine eligibility according to the PICOS/PECOS criteria. During full-text screening, each study was assigned a single reason for exclusion. For screening disagreements, a final determination was made by a third senior, independent reviewer.
For additional articles identified via targeted searches of online trial registries, one researcher screened the titles or abstracts online and downloaded those that were potentially relevant for the review. Reference sections of selected articles were also reviewed for relevant publications not previously identified. This SLR was not registered on the International Prospective Register of Systematic Reviews (PROSPERO). However, the SLR was conducted following guidance from the Centre for Research and Dissemination Guidance for Reviews and the Cochrane Handbook for Systematic Reviews of Interventions. 26 The Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines were followed, and the PRISMA checklist is included in the Supplemental Appendix. 27
Data were extracted from full-text publications and abstracts into predefined Microsoft Excel tables by one researcher, with validation of all data extraction conducted by a second researcher (Supplemental Appendix C). Any disagreements during data extraction were resolved by consensus.
All publications included in the SLR were reviewed and categorized into four levels of evidence based on their outcome definitions and their ability to align with the 2014 WHO EH classification (Supplemental Appendix D). Level 1/2 studies, defined as those reporting clear and comparable clinical response definitions among patient populations that could be clearly aligned with the 2014 WHO EH classification criteria or were defined as FIGO grade 1, stage I/IA EC, were considered for meta-analyses. The purpose of these four levels of evidence was to categorize studies based on population descriptions for clearer synthesis. This framework was not designed to rate the strength of the evidence.
The studies included in the SLR describe their populations using the terms “female” and “women.” The authors have not further defined these terms for this SLR since they are used in alignment with the original studies.
A risk of bias assessment was completed by one researcher for the level 1/2 studies per the recommendations provided by the Centre for Research and Dissemination Guidance for Reviews and the Cochrane Handbook for Systematic Reviews of Interventions (CHSRI, version 5.1.0). 26 The Cochrane Collaboration risk of bias tool was used to assess randomized controlled trials (RCTs), 28 while the Risk of Bias in Non-randomized Studies of Interventions (ROBINS-I) tool was applied to non-randomized comparative studies and to single-arm interventional trials. 29 The Newcastle–Ottawa Scale was used for prospective cohort studies, 30 the adapted Motheral et al. checklist was used for retrospective cohort and registry studies, 31 and the Joanna Briggs Institute (JBI) checklist was used for cross-sectional studies 32 and case series. 33
For the pooled analysis of LNG-IUS efficacy/effectiveness, a random-effects meta-analysis of proportions was conducted. The logit transformation was applied to the proportions prior to pooling (with its back-transformation used prior to plotting). In accordance with current recommendations,34,35 the restricted maximum likelihood (REML) estimator was used to estimate between-study variance. 36 For treatment comparison of LNG-IUS versus oral progestins (OPs), three approaches were used to estimate odds ratios (ORs) for CR: mega-study (i.e., ORs calculated as though all patients were part of a single large study), standard random-effects meta-regression (i.e., models incorporating a random intercept per study estimate), and modified random-effects meta-regression (i.e., models incorporating a random intercept per study). The approaches represent a sliding scale of independence between measures, with the mega-study being the least conservative and the standard random-effects meta-regression being the most conservative. As no individual approach was pre-specified as being the “primary analysis” method a priori, all results are presented equally. The same three approaches were considered for LNG-IUS + OP combination therapies versus LNG-IUS monotherapies. The presence of statistically significant heterogeneity around point estimates was determined via Cochran’s Q-test and its associated p value, 37 while the proportion of total heterogeneity attributable to between-study heterogeneity was summarized using the I2 statistic.38,39. Due to limited reporting, inconsistent outcome definitions, and variation in time points at which outcomes were assessed, meta-analyses were not feasible for recurrence, hysterectomy, pregnancy, and safety outcomes. Descriptive statistics are reported for these outcomes.
All analyses were performed in R version 4.2.2. 40 Meta-analysis models were fit using version 6.1.0 of the meta package, 41 with meta-regression models fit using version 3.8.1 of the metafor package. 36
Results
In total, 1079 unique records were identified for title/abstract screening. Of these, 259 publications underwent full-text screening, and 74 met the predefined PICOS/PECOS eligibility criteria. An additional 5 publications were added by reviewing reference lists, and 1 article was identified through ClinicalTrials.gov searches. A total of 80 publications reporting on 69 unique studies were identified for data extraction (Table 1 and Figure 1; Supplemental Appendices E and F).
Included studies, by indication and study type.

AEH, atypical endometrial hyperplasia; EH, endometrial hyperplasia; EC, endometrial cancer; LNG-IUS, levonorgestrel-releasing intrauterine system; NAEH, non-atypical endometrial hyperplasia; RCT, randomized controlled trial; SLR, systematic literature review; US, United States.
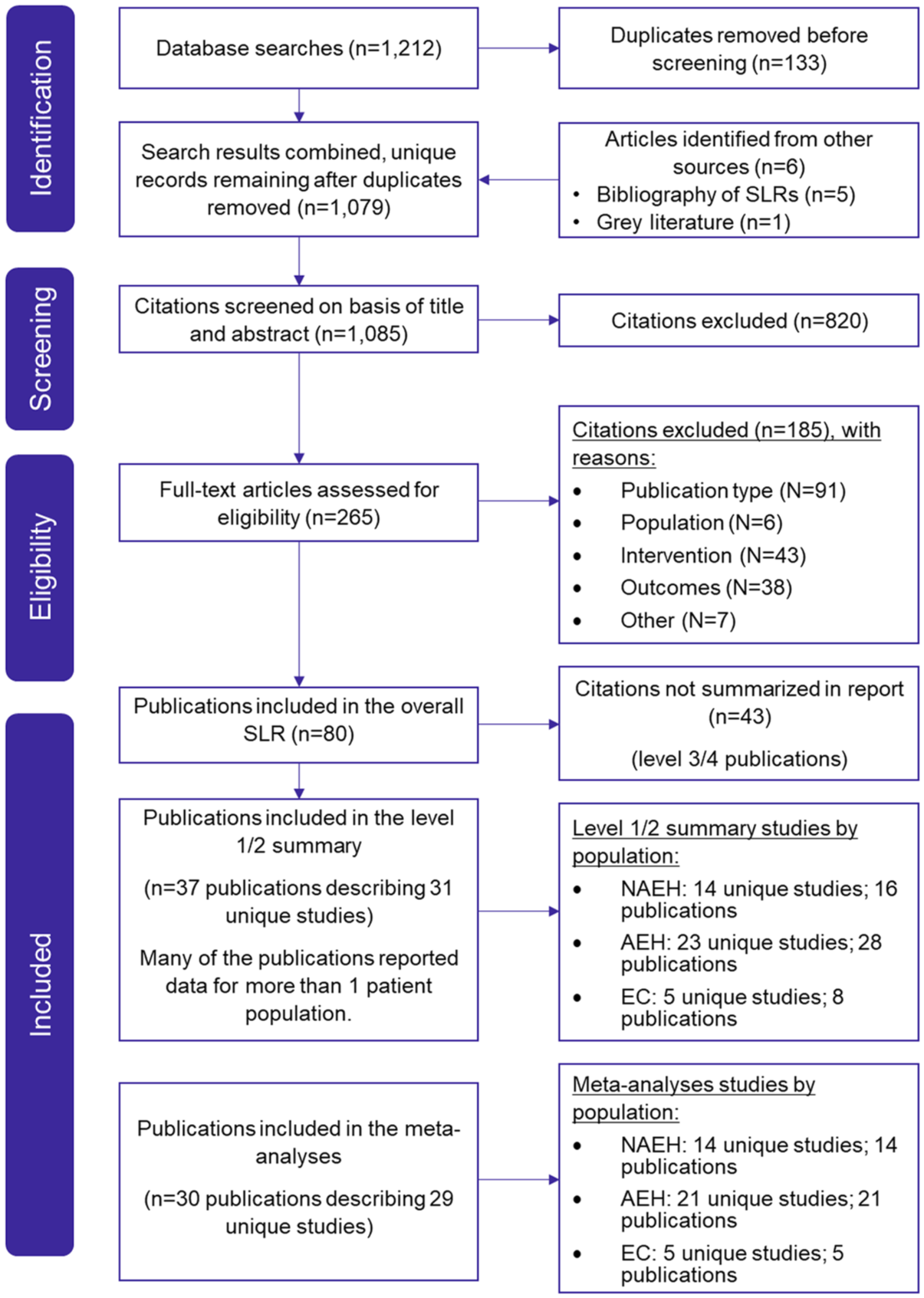
PRISMA flow of literature diagram.
Thirteen of the 69 unique studies included in the SLR were conducted in the US, while the remaining 56 studies were conducted in other countries or did not report a location. Forty-seven studies (54 publications) reported EH treatment with LNG-IUS alone, while 11 studies (13 publications) reported EH treatment with LNG-IUS in combination with another therapy (LNG-IUS combination therapy). Of the included studies assessing EC, 25 studies (30 publications) reported EC treatment with LNG-IUS alone, and 21 studies (27 publications) reported LNG-IUS combination therapy (Table 1).
Thirty-one unique studies (37 publications) were identified as level 1/2 studies based on their ability to align with the 2014 WHO EH classification criteria and the reporting of clear and comparable response definitions. Fourteen studies (16 publications), including 6 RCTs, 6 prospective cohort studies, and 2 retrospective cohort studies, reported on patients with NAEH (Tables 1 and 2; Supplemental Appendix G). All 14 of the studies on patients with NAEH examined LNG-IUS treatment alone and were included in the meta-analyses.
NAEH level 1/2 studies.
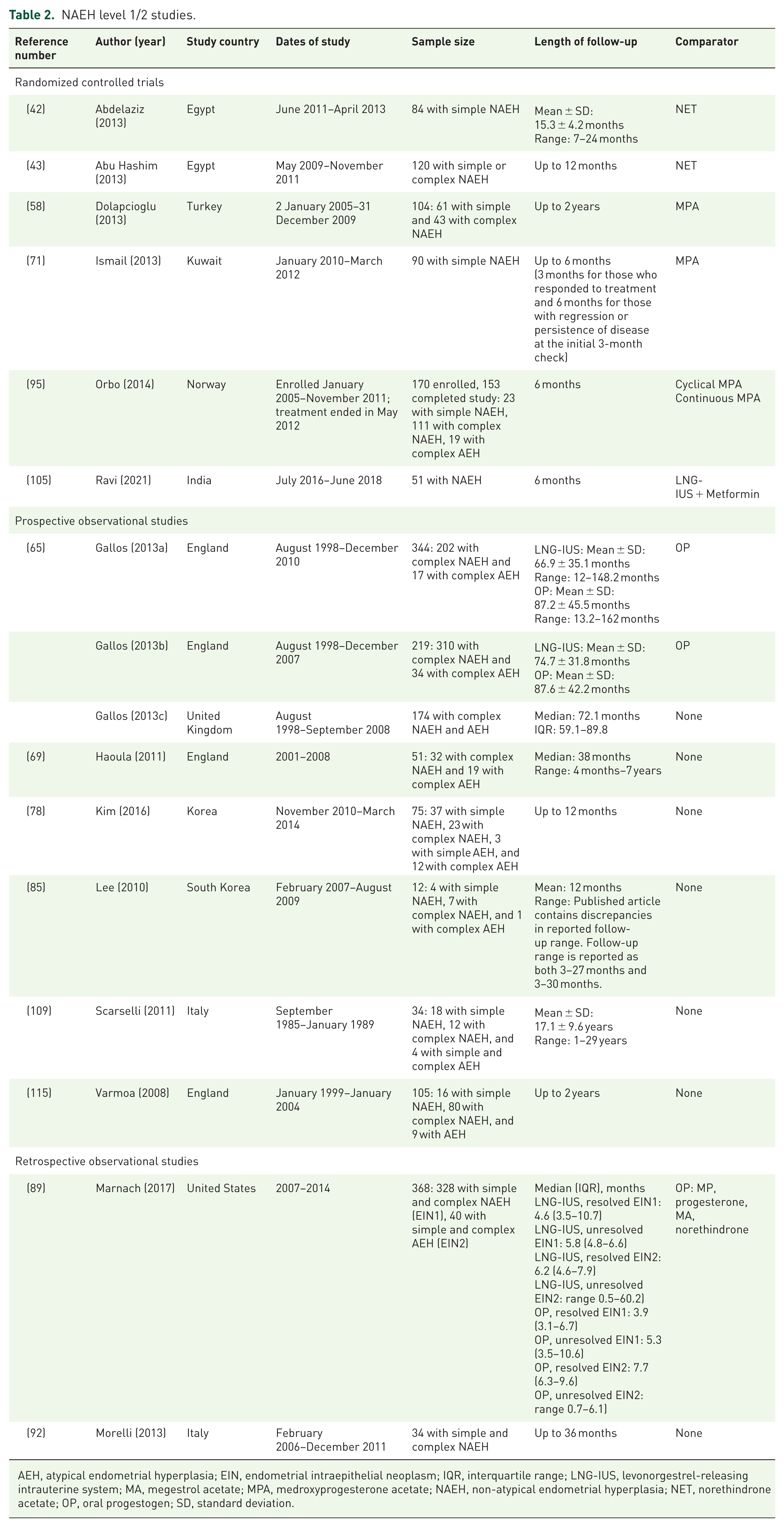
AEH, atypical endometrial hyperplasia; EIN, endometrial intraepithelial neoplasm; IQR, interquartile range; LNG-IUS, levonorgestrel-releasing intrauterine system; MA, megestrol acetate; MPA, medroxyprogesterone acetate; NAEH, non-atypical endometrial hyperplasia; NET, norethindrone acetate; OP, oral progestogen; SD, standard deviation.
Twenty-three studies (28 publications), including 2 RCTs, 1 single-arm trial, 8 prospective cohort studies, 11 retrospective cohort studies, and 1 case series, reported on patients with AEH were identified (Tables 1 and 3; Supplemental Appendix G). Two studies (1 RCT and 1 prospective cohort) assessed LNG-IUS in combination with other treatments; the LNG-IUS combination therapy treatment arms included LNG-IUS + metformin, LNG-IUS + weight loss, and LNG-IUS + dilatation and curettage (D&C) at 2 time points (at 0 and 6 months), LNG-IUS + D&C at 3 time points (at 0, 3, and 6 months), and LNG-IUS + gonadotropin-releasing hormone agonist (GnRHa) + D&C at 3 time points (at 0, 3, and 6 months). Twenty-one of these 23 studies were included in the meta-analyses; 2 studies were not included due to a lack of extractable outcome data or insufficient data to determine outcome time points (Table 1).
AEH level 1/2 studies.

This study is described as both a retrospective study and a retrospective case series.
Two D&C procedures at 0 and 6 months.
Three D&C procedures at 0, 3, and 6 months.
AEH, atypical endometrial hyperplasia; D&C, dilation and curettage; EC, endometrial cancer; EH, endometrial hyperplasia; EIN, endometrial intraepithelial neoplasm; GnRHa, gonadotropin-releasing hormone agonist; IPTW, inverse probability of treatment weighting; IQR, interquartile range; LNG-IUD, levonorgestrel-releasing intrauterine device; LNG-IUS, levonorgestrel-releasing intrauterine system; MA, megestrol acetate; MPA, medroxyprogesterone acetate; NA, not applicable; NAEH, non-atypical endometrial hyperplasia; OP, oral progestogen; SD, standard deviation; Tx, treatment.
Five studies (eight publications), including one RCT, one prospective cohort study, and three retrospective cohort studies, reported on patients with EC (Tables 1 and 4; Supplemental Appendix G). Two retrospective cohort studies reported on patients treated with LNG-IUS alone, while three studies (one RCT, one prospective cohort, one retrospective cohort) reported on LNG-IUS combination therapy treatment arms, including LNG-IUS + medroxyprogesterone acetate (MPA), LNG-IUS + GnRHa, LNG-IUS + megestrol acetate (MA), LNG-IUS + MA + metformin, LNG-IUS + metformin, and LNG-IUS + weight loss. All five studies were included in the meta-analyses. Studies were included in multiple population categories (NAEH, AEH, EC) if they reported stratified subgroup data for each population.
EC level 1/2 studies.

Considered related publications due to possible study population overlap.
This study is described as both a retrospective study and as a retrospective case series.
EC, endometrial cancer; EH, endometrial hyperplasia; GnRHa, gonadotropin-releasing hormone agonist; LNG-IUD, levonorgestrel-releasing intrauterine device; LNG-IUS, levonorgestrel-releasing intrauterine system; MA, megestrol acetate; MPA, medroxyprogesterone acetate; SD, standard deviation.
Efficacy/effectiveness
Non-atypical endometrial hyperplasia
CR rates were consistently high across all 14 level 1/2 studies reporting on LNG-IUS treatment for NAEH. CR rates ranged from 64.3% to 100%42,43,58,65,69,71,78,85,89,92,95,105,109,115 at the longest treatment duration time point (3–36 months) and/or longest post-treatment follow-up time point (up to 29 years), with 12 of 14 studies reporting CR rates of ⩾80% (Table 5; Supplemental Appendix H).43,58,65,69,78,85,89,92,95,105,109,115 The percentage of LNG-IUS patients lost to follow-up was reported in three studies, with one study reporting much higher lost to follow-up rates (0%–5.1% vs 41.3%).43,78,105
CR in patients with NAEH, AEH, and EC.

CR at the longest treatment duration time point and/or longest post-treatment follow-up time point
LNG-IUS combination therapies included LNG-IUS + weight loss, LNG-IUS + MET, LNG-IUS + 2 D&Cs, LNG-IUS + 3 D&Cs, LNG-IUS + GnRHa + 3 D&Cs.
LNG-IUS combination therapies included LNG-IUS + MPA (2 studies), LNG-IUS + MA, LNG-IUS + MA + MET, LNG-IUS + GnRHa, LNG-IUS + MET, LNG-IUS + weight loss.
AE, atypical endometrial hyperplasia; CI, confidence interval; CR, complete response; D&C, dilation and curettage; EC, endometrial cancer; LNG-IUS, levonorgestrel-releasing intrauterine system; MA, megestrol acetate; MET, metformin; MPA, medroxyprogesterone acetate; NA, not assessed; NAEH, non-atypical endometrial hyperplasia; OP, oral progestin.
Meta-analyses were used to calculate point estimates at multiple time points; point estimates for the percentage of patients with NAEH achieving CR were 87.7% (95% confidence interval (CI): 69.54%–95.67%; n = 311) at 6 months and 86.3% (95% CI: 78.50%–91.55%; n = 342) at 12 months.
Seven studies reporting on LNG-IUS treatment for NAEH also included OP treatment arms. CR with OP treatment ranged from 36.7% to 100%, with only three of seven studies reporting CR rates of ⩾80%. The meta-analyses found that LNG-IUS treatment was associated with significantly greater odds of CR at 12 months compared to OPs, with ORs of 2.16 (95% CI: 1.38–3.36) in a meta-regression model incorporating a random intercept per study. ORs were even higher when using a meta-regression model incorporating a random intercept per estimate (2.71 (95% CI: 1.18–6.19)) and when using the mega-study approach (2.53 (95% CI: 1.72–3.73)).
Overall, CR rates for patients with NAEH treated with LNG-IUS were generally high (⩾80%), and patients treated with LNG-IUS were 2–3 times more likely to have CR at 12 months compared to patients treated with OPs.
Atypical endometrial hyperplasia
CR rates were similarly high for patients with AEH. Twenty-two of 23 level 1/2 studies on LNG-IUS treatment of AEH assessed CR, and nearly 70% (15 of 22 studies) reported CR rates of ⩾80% for AEH treated with LNG-IUS alone at the longest treatment duration time point (6–24 months) and/or longest post-treatment follow-up time point (up to 29 years) (Table 5; Supplemental Appendix H). The overall CR range reported was 37.5%–100%. Loss to follow-up was reported in three studies in women with AEH treated with LNG-IUS; proportions lost to follow-up were 3.5%, 118 14.5%, 67 and 41.3%. 78
When assessed via meta-analyses, point estimates for the percentage of patients with AEH achieving CR with LNG-IUS alone were 81.1% (95% CI: 65.03%–90.84%; n = 173) at 6 months and 83.9% (95% CI: 71.95%–91.34%; n = 196) at 12 months.
Two studies on patients with AEH included LNG-IUS combination therapy treatment arms, and the CR rates ranged from 75% to 100%.73,94 However, meta-analyses were not possible for LNG-IUS combination therapies due to the low number of studies assessing each treatment combination.
In comparison, CR rates for patients with AEH treated with OPs were reported in nine observational studies and ranged from 9% to 88%,47,57,65,88–90,94,95,119 and only two studies reported a CR rate ⩾80% at the longest post-treatment follow-up timepoint.94,95 In addition, the meta-analysis found that LNG-IUS treatment was associated with significantly greater odds of CR at 12 months compared to OPs, with an OR of 2.42 (95% CI: 1.24–4.71) estimated using a meta-regression model with a random intercept per study. Estimates were higher when using a meta-regression model incorporating a random intercept per estimate (3.25 (95% CI: 1.38–7.67)) and when using the mega-study approach (4.56 (95% CI: 2.57–8.09)).
Overall, CR rates for patients with AEH treated with LNG-IUS were generally high (⩾80%), and patients treated with LNG-IUS were 2–5 times more likely to have CR at 12 months compared to patients treated with OPs.
Grade 1 early-stage (FIGO I/IA) EC
Across the five level 1/2 studies that assessed grade 1 FIGO stage I/IA EC, CR rates ranged from 29% to 78.6%67,100,103,104 and 32% to 87.5%73,77,79,100 for LNG-IUS alone and LNG-IUS combination therapy, respectively (Table 5; Supplemental Appendix H). Percentages of patients lost to follow-up were reported in three studies (four publications) and ranged from 0% to 20.5%.
From the meta-analyses, point estimates of the percentage of patients with EC achieving CR with LNG-IUS alone were 67.6% (95% CI: 29.88%–91.10%; n = 48) at 6 months and 51.1% (95% CI: 16.30%–84.90%; n = 20) at 12 months. Meta-analyses were not possible for LNG-IUS combination therapies due to the small number of studies with each treatment combination.
Recurrence or relapse rates were inconsistently reported. Across studies of NAEH, four reported recurrence rates after LNG-IUS, with ranges from 0% to 25%.58,66,85,115 Recurrence rates for patients with NAEH treated with OPs, reported in two of the same studies,58,66 were higher at 12%–46%. One additional study reported recurrence, but the data were not stratified by population. 109 The proportion of LNG-IUS patients who underwent hysterectomy was reported in four studies and ranged from 0% to 22.0%.43,58,69,105 The proportion of patients treated with OPs who underwent hysterectomy was reported in two studies and ranged from 19.2% to 57.4%.43,58 Five additional studies (seven publications) reported hysterectomy data that were not stratified by both population and treatment.42,64–66,89,109,115
Across studies of AEH, the rate of recurrence with LNG-IUS was reported in seven studies (eight publications) and ranged from 3% to 36%.64,67,87,101,104,115,117 Only one study reported recurrence in patients with AEH treated with OPs (50%). 65 Two additional studies reported recurrence, but the data were not stratified by both population and treatment type.94,109 Seven studies reported the proportion of patients who underwent hysterectomy after LNG-IUS treatment, with a range of 1.8% to 50%.44,67,69,88,101,106,117 Six other studies reported hysterectomy, but synthesis was limited due to variation in reporting. 101
In studies assessing EC, the rate of recurrence after LNG-IUS treatment alone varied considerably between the two studies (three publications) that reported this outcome, from 0% to 50%.67,103,104 Only one study reported a recurrence rate of 12.5% after LNG-IUS treatment in combination with OPs. 79 The proportion of patients who had undergone hysterectomies was only reported in two studies. In Giampaolino et al., 21.4% of patients treated with LNG-IUS alone had undergone a hysterectomy after 24 months. 67 In Pino et al., hysterectomy rates were 40%, 33.3%, and 58.1% for LNG-IUS treatment with OPs, with OPs + metformin, and with GnRHas, respectively. 100
It should be noted that across all studies reporting recurrence and hysterectomy, there was considerable variation in the duration of treatment and length of follow-up. In addition, whether patients were still on treatment when recurrence occurred and the reasons for hysterectomy were not consistently reported.
Overall, studies for patients with grade 1 FIGO stage I/IA EC treated with LNG-IUS are limited. However, available data suggest that LNG-IUS treatment can induce regression in these patients.
Pregnancy in women seeking to conceive
No level 1/2 studies of patients with NAEH assessed conception rates and pregnancy outcomes in women seeking to conceive.
The rates of pregnancy in patients with AEH who attempted to conceive following LNG-IUS treatment were reported in two observational studies and ranged from 40% to 55%.67,87 The percentage of pregnancies resulting in live births was consistently high across both AEH studies, ranging from 83% to 100%.67,87 Three additional studies reported rates of pregnancy in patients who attempted to conceive (20%–54%), but this outcome was not stratified by treatment type or patient population.94,97,117 Pregnancies were reported by patients who used assisted reproductive technology and by patients who conceived naturally.67,87 However, synthesis of these outcomes was limited due to variation in frequency and methods of reporting.
The rate of pregnancy in patients with EC who attempted to conceive after treatment with LNG-IUS was reported in two observational studies. In both studies, patients received treatment with LNG-IUS in combination with OPs, GnRHas, or metformin, and pregnancy rates ranged from 33.3% to 57.1%.79,100 Pregnancies were reported by patients who underwent in vitro fertilization and by patients who conceived naturally.79,100 The occurrence of at least 1 miscarriage was reported in each study, although the exact method of reporting varied.79,100
Pregnancy is possible after prior LNG-IUS treatment for endometrial conditions, and most pregnancies result in successful delivery and live birth. However, data are limited since the desire for conception was not consistently reported.
Safety
Overall, adverse events (AEs) reported for all three populations (NAEH, AEH, and EC) treated with LNG-IUS were generally similar to AEs reported by patients treated with LNG-IUS for approved indications and those included in the Mirena prescribing information. 20 A listing of AEs reported in the studies is shown in Supplemental Appendix I.
Discontinuation of LNG-IUS monotherapy or combination treatment was reported in five studies (n = 343) including NAEH patients (2.9%–8.3%), six studies (n = 568) including AEH patients (2.9%–20%), and two studies (n = 179) including EC patients (0%–10.6%). Reasons for treatment discontinuation included spotting, irregular bleeding, cramping, LNG-IUS expulsion, decision to pursue surgery, and a malpositioned LNG-IUS inserted in the cervix.
Rates of LNG-IUS expulsion for patients treated with LNG-IUS alone were generally similar to those reported in the clinical trials of Mirena (<5% in four out of the five publications (n = 347) reporting expulsion rates). One single study reported >5% of women expelling the device (rate of 12%). 44 It concerns a study in 36 women with AEH/EC who were postmenopausal and unfit for surgery. 44 Although no data were provided for the specific group presenting device expulsion, the publication reported that the enrolled women had a mean BMI of 51 kg/m2 (range: 22–84). This implies a high proportion of obese or morbidly obese patients, a factor that could have contributed to the increased expulsion rate observed in this study.
One additional study (n = 165) reported an LNG-IUS expulsion rate of 1.8%, but this outcome was reported for all population subgroups (AEH and EC) and treatment arms (LNG-IUS, LNG-IUS + metformin, and LNG-IUS + weight loss) combined. 73 No studies reported device expulsion for EC patient subgroups.
Across studies of NAEH, AEH, and EC patients treated with LNG-IUS, no unexpected safety issues were identified, and the safety profile for these populations is comparable to LNG-IUS treatment for other approved indications.
Discussion
Efficacy/effectiveness
This systematic review and meta-analysis shows that LNG-IUS was associated with high rates of EH regression in patients with NAEH and AEH: for the treatment of NAEH, analysis of 14 studies demonstrated a CR rate of 87.7% (95% CI: 69.54%–95.67%; n = 311) at 6 months and 86.3% (95% CI: 78.50%–91.55%; n = 342) at 12 months, whereas for treatment of AEH, analysis of 23 studies demonstrated a CR rate of 81.1% (95% CI: 65.03%–90.84%; n = 173) at 6 months and 83.9% (95% CI: 71.95%–91.34%; n = 196) at 12 months. Although more limited, the available evidence indicates that LNG-IUS can also induce regression in up to 50% of patients with EC.
Comparative efficacy/effectiveness
Compared with OPs in the meta-analyses, LNG-IUS was associated with significantly increased odds of CR at 12 months in both NAEH and AEH populations. Other time points were not assessed due to a lack of data, but these results confirm that LNG-IUS is particularly suited to provide long-term treatment of EH due to its mode of administration, which provides continuous release of progestin in the uterine cavity. 20
Pregnancy and fertility
Successful pregnancies were reported after LNG-IUS treatment, although desire for conception was not reported consistently across the three populations (NAEH, AEH, EC), and the majority of pregnancies reported resulted in successful delivery and live birth. Pregnancy rates reported in women who attempted to conceive were lower than those expected in women of fertile age, 122 but this is considered a reflection of the underlying condition rather than an effect of prior treatment with LNG-IUS, as women using LNG-IUS for contraception return quickly to their normal fertility after removal. 20
Safety
No new safety concerns (related to LNG-IUS) have been identified in studies of patients with NAEH, AEH, or EC; the safety profile appears consistent with that described for other approved indications.
Findings compared to other published literature
Overall, our findings are consistent with the recently published Cochrane review and meta-analysis by Mittermeier et al., 21 who concluded that there was “moderate-quality” evidence that 3–6 months of treatment with LNG-IUS was more effective than treatment with OPs over short-term follow-up (up to 6 months). Our study adds to the literature on this topic, providing further reassurance that CRs are observed at least 12 months of follow-up with LNG-IUS treatment. In addition, we showed significantly greater odds of CR compared to treatment with OPs. Mittermeier et al. 21 reviewed only RCTs and cross-over trials, while this SLR included RCTs, single-arm trials, non-interventional/observational studies, and case series. RCTs are considered the gold-standard study design for comparing treatments/interventions (e.g., LNG-IUS vs other treatments). However, observational data from real-world settings provide additional evidence regarding effectiveness and safety and a more complete picture of LNG-IUS use in these populations.
Limitations
The studies discussed in this report, defined as level 1/2 evidence, had clear outcome definitions and aligned with the 2014 WHO classification criteria (EH) or FIGO grading (EC). However, there were considerable discrepancies regarding other study elements, including sample size, study power, study region, treatment history, treatment duration, and follow-up length. There were also limitations in the methods for evaluating endometrial response; the majority of studies followed clinical practice, but this does not include independent pathologist review, which was only utilized by a few studies.
Risk of bias varied across study designs. The clinical trials had a low risk of bias across most design elements, although some methodological choices, such as a lack of placebo and a lack of participant blinding, which introduced potential bias, were likely due to the nature of LNG-IUS treatment. Observational studies generally had clear methodologies, but limitations included a lack of information on the reliability and validity of the data sources and treatment groups differing at baseline (Supplemental Appendix J). Gaps in the literature included long-term outcome data, with very few studies reporting defined time points greater than 12 months and consistent reporting of pregnancy and safety outcomes. Sensitivity analyses excluding studies with a high risk of bias were not conducted, as they would have further reduced the number of studies included for each outcome and would therefore likely not be robust.
For the studies included in the meta-analyses, between-study heterogeneity varied between outcomes. Notably, residual between-study heterogeneity was low in meta-regression models (random intercept per study) assessing the comparative effect of LNG-IUS versus OP on CR at 12 months with I2 values of 33.0% for patients with NAEH and 37.9% for patients with AEH. When assuming a greater degree of independence between study estimates (random intercept per estimate), I2 increased to 68.6% for patients with NAEH and 42.2% for patients with AEH. Between-study heterogeneity was substantial when assessing the absolute efficacy of LNG-IUS on CR at 6 months in patients with NAEH (I2 = 83.2%), AEH (I2 = 68.1%), and EC (I2 = 74.0%) and ranged from moderate to substantial at the 12-month time point in patients with NAEH (I2 = 61.0%), AEH (I2 = 54.3%), and EC (I2 = 64.4%).38
Conclusion
Overall, the published literature supports LNG-IUS as an effective treatment option for patients with NAEH, AEH, and appropriately selected patients with grade 1 early-stage (FIGO I/IA) EC. The availability of a non-surgical option may be especially beneficial for patients with these conditions who wish to avoid surgery and/or retain fertility. Because these conditions significantly affect women’s lives, and definitive surgical treatment may not be appropriate, desired, or possible for many, greater availability of LNG-IUS treatment would provide meaningful benefit to these patient populations.
Supplemental Material
sj-docx-1-reh-10.1177_26334941261433726 – Supplemental material for Levonorgestrel-releasing intrauterine system treatment for endometrial hyperplasia and early endometrial cancer: a systematic review
Supplemental material, sj-docx-1-reh-10.1177_26334941261433726 for Levonorgestrel-releasing intrauterine system treatment for endometrial hyperplasia and early endometrial cancer: a systematic review by Kristina Rosa Bolling, Elizabeth Suh-Burgmann, Susan D. Reed, Shannon Westin, Michael Fassett, Yesmean Wahdan, Eeva Lukkari-Lax, Juliane Schoendorf, Nils Schoof, Carsten Moeller, Livia Delgado, Malia Gill, Maria Lorenzi, Benjamin Feakins, Erika Wissinger and Angie Lee in Therapeutic Advances in Reproductive Health
Footnotes
Acknowledgements
The authors acknowledge Kylie Matthews, MS, of Cencora for assistance with manuscript preparation and copyediting. Elizabeth Suh-Burgmann, Susan D. Reed, Shannon Westin, and Michael Fassett had full access to all data in the study and take responsibility for the integrity and accuracy of the data analysis.
Declarations
Supplemental material
Supplemental material for this article is available online.
