Abstract
Background:
Assessing for significant proteinuria in pregnancy (SPIP) stands as a key indicator for diagnosing preeclampsia. However, the initial method typically employed for this assessment, the urine dipstick test, often yields inaccurate results. While a 24-h urine collection is considered the most reliable test, its implementation can lead to delays in diagnosis, potentially affecting both maternal and fetal well-being. The urine protein–creatinine (P/Cr) ratio can be used as an alternative to 24-h urine protein analysis, but its diagnostic accuracy has remained uncertain. There is a need to compare the diagnostic accuracy of urine P/Cr ratio and dipstick urinalysis for SPIP, especially in resource-poor settings.
Objectives:
To determine and compare the diagnostic accuracy of urine P/Cr ratio and dipstick urinalysis in a spot urine specimen for the diagnosis of SPIP among women evaluated for preeclampsia using 24-h urine protein excretions as a gold standard.
Design:
This is a comparative cross-sectional study.
Methods:
The study involved 82 singleton pregnant women evaluated for preeclampsia from 20 weeks of gestation who underwent dipstick and P/Cr ratio tests in the same urine sample. Women at risk of preeclampsia were given a specimen container for the collection of urine samples on an outpatient basis. Participants were trained and told to collect the urine sample 24 h prior to their next antenatal appointment. However, those on admission and evaluated for preeclampsia had their 24-h urine collected in the hospital. The outcome measures included sensitivity, specificity, positive predictive value, negative predictive value, likelihood ratio and accuracy for the two tests. Significant proteinuria was defined as a P/Cr ratio >0.27 or ⩾2+ of proteinuria on the dipstick test. Preeclampsia was confirmed in women with both high blood pressure and SPIP.
Results:
The mean age of participants was 28.65 ± 5.76 years. Comparatively, the diagnostic accuracy (91.46% (95% CI = 83.29–96.59) vs 59.76% (95% CI = 48.34–70.44), p = 0.001), sensitivity (94.74% vs 70.00%, p = 0.021), specificity (84.00% vs 43.75%, p = 0.001), negative predictive value (87.50% vs 48.28%, p = 0.003) and positive predictive value (93.10% vs 66.04%, p = 0.001), respectively, were higher for the spot urine P/Cr ratio than dipstick test. In addition, the positive likelihood ratio and the negative likelihood ratio for spot urine P/Cr ratio versus dipstick test were (1.93 vs 1.24) and (0.07 vs 0.69), respectively.
Conclusion:
The spot urine P/Cr has superior diagnostic accuracy in the determination of significant proteinuria in pregnant women being evaluated for preeclampsia than the widely used dipstick test. A more robust multicenter study is needed to compare the diagnostic accuracy of spot urine PCR with the standard 24-h urine protein in low-income settings.
Plain language summary
• Preclampsia is one of the medical disorders that can complicate pregnancy, and it is a major cause of feto-maternal morbidity and mortality during pregnancy.
• Preeclampsia is the development of elevated blood pressure beyond the normal value during the second half of pregnancy with a significant amount of protein in the urine.
• The detection of a significant amount of protein in the urine is one of the criteria for the diagnosis of preeclampsia; however, there are limitations with the various methods of urine protein detection and estimation.
• The study was conducted among women being evaluated for preeclampsia. We compared the diagnostic accuracy of the urine protein-creatinine ratio and dipstick urine test for the diagnosis of significant proteinuria in pregnancy using 24-hour urine protein excretions as a gold standard.
• Our findings showed that the spot urine protein-creatinine ratio has superior diagnostic accuracy when compared to the regular dipstick urine tests for the detection of significant protein in the urine among pregnant women being evaluated for preeclampsia.
Keywords
Introduction
The examination of urine for the presence of protein constitutes an important part of detecting people with preeclampsia. 1 Its accurate determination is therefore of utmost importance in the diagnosis of preeclampsia, and prompt interventions will contribute significantly to reducing the adverse effects of this condition in pregnancy.
Traditionally, significant proteinuria (0.3 g/24 h) has been diagnosed through the collection of a 24-h urine sample, and this remains the widely recognized gold standard for the diagnosis of significant proteinuria. 2 However, this process poses several challenges that can impact the accuracy of the test results. These challenges include the need for refrigeration of urine, which is time-consuming for both women and caregivers, as well as being cumbersome and potentially misleading if not collected correctly. 3 Additionally, there is the risk of delivery occurring before the completion of urine collection, leading to delays in diagnosis and treatment initiation, prolonged hospital stays and increased anxiety for the patient.
Due to the above shortfalls in 24-h urine collection, other methods are being proposed, which include the use of a urine dipstick, urine collection over shorter periods (2, 4, 8 or 12 h) and urine protein–creatinine (P/Cr) ratio.
The dipstick is cheap, does not require much technical input and gives an instant result; however, studies show that it has low sensitivity and specificity when compared with the gold standard test. 4 The inaccuracies in dipstick analysis may be attributable to whether the pregnant woman is dehydrated or not and day-to-night changes in protein excretion. A urine dipstick is used to evaluate abnormal changes in a urine sample. 5 It is commonly used for screening for significant proteinuria in pregnancy (SPIP); however, the accuracy of the results has been a source of concern to many obstetricians, as both false-positive and false-negative results can occur. 6 With all these limitations, the urine dipstick test is still the mainstay of screening for proteinuria in obstetric practice in Nigeria and sub-Saharan African countries.
The protein–creatinine (Pr/Cr) ratio is superior to 24-h urine protein measurement, as stated by the UK Chronic Kidney Disease guideline. 7 Creatinine is constantly excreted by the kidneys for an individual (11.0–25.0 mg/kg/day), and its production is a function of the muscle mass. 8 The spot urine Pr/Cr ratio is accepted for evaluating daily protein loss in non-pregnant individuals. 9 A number of international bodies agree that the P/Cr test should be done in place of 24-h urine collection, and 30 mg/mmol (0.265 mg/mg) has been recommended as the cut-off.10,11 The day-to-night changes in the specific gravity of urine lead to different concentrations of urine proteins during the day. When this protein concentration is divided by the urine creatinine content, which depends on the glomerular filtration rate, it will result in a steady ratio all through the day. Therefore, it is accepted as a better test for proteinuria. Geenen et al., in a systematic review and meta-analysis to determine the diagnostic test accuracy of protein–creatinine ratio (PCR) and albumin–creatinine ratio for the diagnosis of significant proteinuria in pregnant women with hypertension, concluded that PCR has high accuracy compared to gold standard 24-h urine collection. 12
Medical disorders in pregnancy associated with significant proteinuria may increase adverse outcomes for the pregnant woman and her baby; therefore, the importance of an accurate assessment of SPIP cannot be overemphasized. Significant proteinuria in pregnancy may be seen in conditions such as preeclampsia, chronic hypertension and renal disease.
Preeclampsia remains a significant cause of maternal and perinatal morbidity and mortality.1,13,14 The diagnosis is made in pregnant women who were previously normal but developed raised blood pressure and significant proteinuria at or after 20 weeks. It affects multiple organs in the body, and it is associated with decreased blood supply to the kidneys and injury to the glomerulus, with its attendant loss of proteins in urine.
A lack of vascular endothelial growth factor (VEGF) specific to glomerular foot processes (podocytes) is linked to kidney impairment in preeclampsia. fms-like tyrosine kinase 1 (sFlt-1), a soluble VEGF receptor, is expressed at higher levels in hypoxic placentas in patients with preclampsia (PE). In the end, sFlt-1 disrupts the filtration barrier and causes proteinuria by preventing VEGF from attaching to its receptor on endothelial cells and podocytes, leading to damage to the podocytes and glomeruloendotheliosis. 15
Most of the previous studies on this have used only subjects in Western or Asian countries. There is a paucity of studies in our environment on the comparison of the P/Cr ratio and the urine dipstick test for significant proteinuria in preeclamptic women. Therefore, this research is undertaken to determine and compare the P/Cr ratio and dipstick test in a spot urine sample for accuracy in detecting the presence of SPIP against 24-h urine protein estimation for the diagnosis of preeclampsia.
Methods
Study design
This is a comparative cross-sectional study. The reporting of this study conforms to the Standard for Reporting Diagnostic Accuracy (STARD) statement. 16
Study settings
This study was conducted at the obstetrics and gynaecology unit of Nnamdi Azikiwe University Teaching Hospital (NAUTH) in Nnewi, Nigeria, between March and September 2020.
Study population
The study participants were pregnant women at risk and evaluated for preeclampsia that presented to the antenatal clinic and labour ward unit of NAUTH, Nnewi, Nigeria.
Inclusion criteria
Those included in the study were all pregnant women with raised blood pressure diagnosed from 20 weeks of the index pregnancy, pregnant women with ⩾1+ of proteinuria on routine urinalysis from 20 weeks gestational age and pregnant or intrapartum women with a history of convulsion.
Exclusion criteria
The exclusion criteria were as follows: women diagnosed with hypertension and/or renal disease before pregnancy; pregnancy less than 20 weeks of gestation; pregnant women with a history of recurrent infection of the urinary tract; participants with concurrent clinical and laboratory evidence (bacteriuria) of urinary tract infection, molar pregnancy, or multiple pregnancies; and women who delivered before the completion of 24-h urine collection.
Sample size calculation
The minimum sample size required for the study was calculated using Fisher’s formula.
n = Z2pq ÷ d2 or n = Z2 × P(1 − P)/e2, where n = sample size, Z = standard normal deviation at 95% confidence interval, which is 1.96, d = degree of precision set at 0.05 and P = proportion of the target population. In this case, p = 5% (0.05), which is the prevalence rate of preeclampsia worldwide according to Osungbade and Ige, 17 and Q = alternate proportion (1 − P). With a 10% attrition rate, the minimum sample size for the study was 81 participants.
Participant recruitment
Documented, informed consent was obtained from all participating women. Women at risk of preeclampsia were given a specimen container for the collection of urine samples on an outpatient basis. They were told to collect the urine sample 24 h prior to their antenatal visit. However, those admitted to the hospital and evaluated for preeclampsia had their 24-h urine collection done on an inpatient basis.
Study procedure
Mid-stream clean-catch urine sample collection
A container tagged with the patient’s full name and identification number was used to collect the specimen. Thorough washing of the hands with soap and water was done, and the hands were dried with a paper towel. The cap of the specimen container was removed, and care was taken to avoid touching the inside of the cap or the inside of the container. The cap was then placed on the table with the inside of the cap facing up. A sterile wipe was opened, and the patient was taught (by a female research assistant) to separate the labial folds covering the urinary opening with fingers and clean inside using one sterile wipe, moving from the front to the back. One side was cleaned and discarded, and then, with a new sterile wipe, the centre was cleaned and also discarded. The other side was finally cleaned with a new sterile wipe. The participant urinated into the toilet while still parting the urinary opening. The specimen container was introduced into the urine stream until it was half-filled with a mid-stream specimen. The cap was placed back on the container without touching the inside of the cap or the inside of the container.
Twenty-four-hour urine sample collection
Every drop of urine during the 24-h period was collected, and it was started in the morning after emptying the bladder upon waking up from sleep. The time of starting was recorded. Urine was collected into the sample container and stored at room temperature. Urine was also collected at the time of defecation; care was taken to avoid fecal contamination. It was concluded the next morning after collecting the first urine.
Urine dipstick test
A midstream specimen of urine was provided by the participant in a 10-ml sterile container. The test was conducted immediately by dipping the strip into the urine sample, ensuring that the coloured reagent part was completely immersed in urine. The test strip was placed flat on a paper towel and read after 60 s by holding the strip against the container to compare the standardized chart with the sample strip. The result was recorded, and the strip was discarded. The dipstick test was performed by the researcher at any time of the day that the participant was seen, using N-Multistix SG-L, manufactured by Siemens Health Company Ltd., Tokyo, Japan. This Multistix container was colour-coded such that 1+ protein = 30 mg per 100 ml and 2+ protein = 100 mg per 100 ml. The research assistants were blinded to the clinical status of the participants.
Spot urine protein estimation
The estimation of protein and creatinine content in the urine sample was done using the colorimetric method. The sample was run as soon as it was collected. Boric acid was added as a preservative. In situations where the test cannot be done within 2 h after voiding, the sample is refrigerated immediately and then allowed to return to room temperature before the test is performed. Microbial multiplication in unpreserved urine at room temperature may lead to protein loss. Samples were stored at 0–8°C for a week or at 20°C for a month without significant effect on test results. 18
Proteinuria measurement
Proteinuria was measured by the quantitative method 19 (the sulphur salicylic acid method). 1 ml of urine was added to a test tube, and 3 ml of 3% sulfuric acid was added to the same test tube. The mixture was allowed to incubate at room temperature for 10 min, and the absorbance of the test and the standard were read at 490 nm using a spectrophotometer. The acidification of urine causes the precipitation of protein in the sample (seen as increasing turbidity), which can be subjectively quantified visually or more precisely using photometry.
Creatinine measurement
Creatinine was measured by the quantitative method 20 (JAFFE method). The principle of the test was the peroxidase-like activity of diisopropylbenzene dihydroperoxide and 3,3,3,5-tetramethylbenzidine. It gave rise to a colour change from orange to green to blue.
Spot urinary protein/creatinine ratio
The protein concentration was divided by the creatinine concentration in mg per 100 ml.
Definition of terms
Significant proteinuria in pregnancy
Significant proteinuria is a protein concentration greater than or equal to 300 mg in a 24-h urine specimen, greater than or equal to 2++ on a dipstick in a random mid-stream urine sample or a P/Cr (mg/mg) threshold of >0.27.
Hypertension
Hypertension was defined as the presence of systolic blood pressure ⩾140 mmHg and/or diastolic blood pressure ⩾90 mmHg on two occasions at least 4 h apart. Women who have hypertension and SPIP occurring after 20 weeks of gestation are classified as having preeclampsia.
Outcome measures
The outcome measures were the sensitivity, specificity, positive predictive value, negative predictive value, likelihood ratio, and accuracy of the urine dipstick method and spot urine P/Cr in the diagnosis of SPIP in women evaluated for preeclampsia.
Statistical analysis
The data were checked for completeness and tabulated. Analysis was done using the Statistical Package for Social Sciences (SPSS) version 23 (IBM Corp., Chicago, IL, USA). Data were presented in tables and charts, while continuous data were presented in mean and standard deviation. As shown in previous studies, sensitivity, specificity, positive predictive value, negative predictive value, likelihood ratios and accuracy for the P/Cr ratio versus dipstick test were calculated.21–29 A scatter plot diagram was also done. The p-value ⩽ 0.05 was taken to be significant at a 95% confidence interval.
Results
The study was done over 7 months from March 2020 to September 2020. In all, 97 pregnant women with new-onset elevated blood pressure diagnosed in the second half of the index pregnancy were assessed for eligibility. Eleven of the subjects were excluded. Seven out of the 11 had chronic hypertension, while the remaining four developed hypertension before 20 weeks of pregnancy. Out of the 86 women who were recruited into the study, 82 (95.3%) women completed the study and were analysed, while four women delivered before the completion of 24-h urine collection and were further excluded. The flow chart for this study is shown in Figure 1.

Flow chart of the study.
Table 1 shows the participants’ sociodemographic and clinical characteristics. In all, 78 (95.1%) of the participants were married, while 4.9% were single. The mean age and gestational age of participants were 28.65 ± 5.76 years and 22.76 ± 1.72 weeks, respectively, while the mean systolic and diastolic blood pressure were 167.07 ± 24.06 mmHg and 101.85 ± 14.44 mmHg. In terms of gravidity, primigravida was the most common (29.3%).
Participants’ sociodemographic and clinical characteristics.

The overall performance of the spot P/Cr ratio versus the dipstick test using 24-h urine protein as the gold standard is shown in Table 2. With reference to the positive and negative 24-h urine protein estimation methods, the sensitivity and specificity of the spot P/Cr ratio are 94.74% and 84.0%, while the sensitivity and specificity of the dipstick test are 70.0% and 43.75%, respectively.
Performance of the spot urine protein to creatinine ratio test versus the dipstick test in pregnant women using 24-h urine protein as the gold standard.
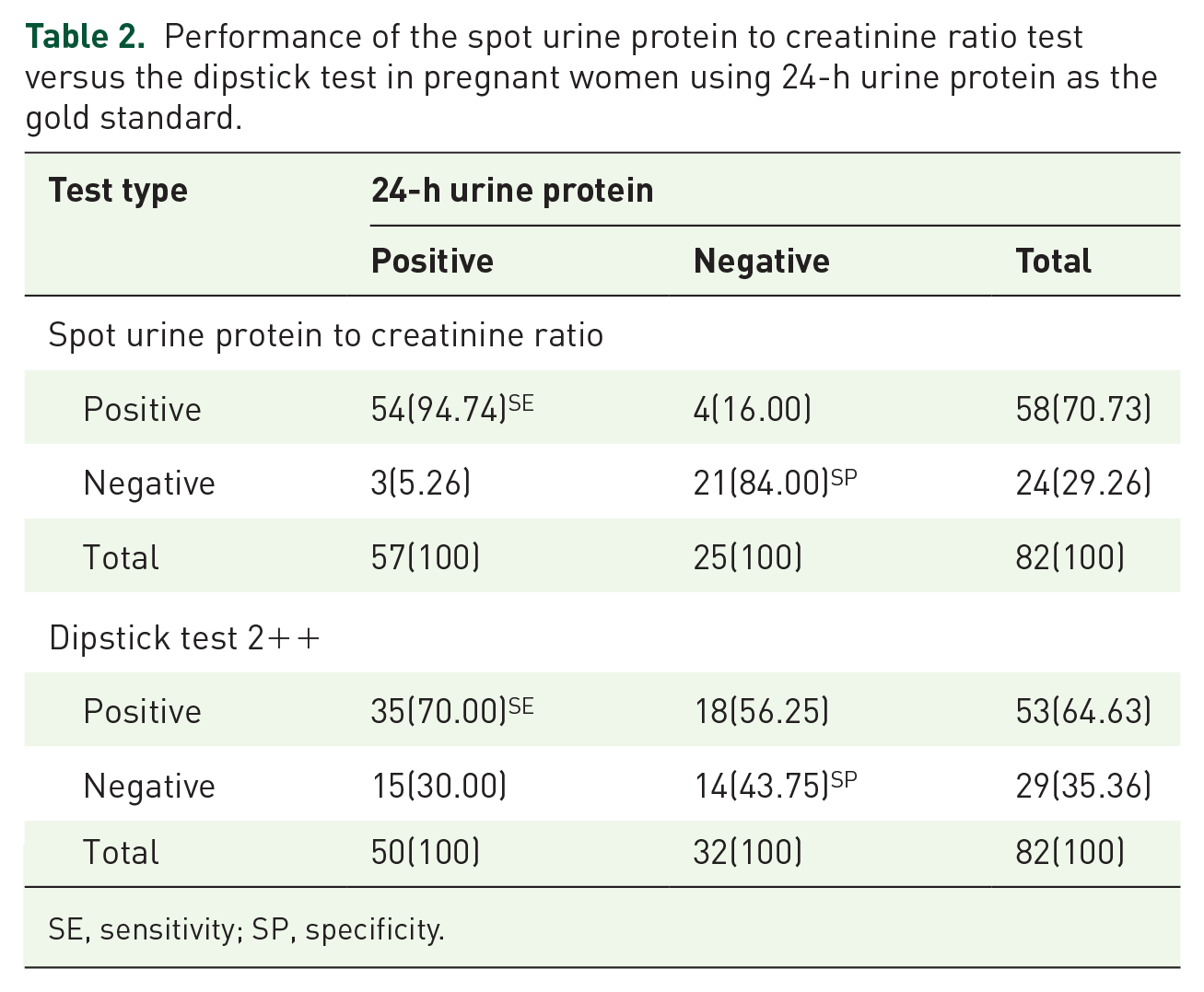
SE, sensitivity; SP, specificity.
Table 3 shows the diagnostic accuracy of the spot P/Cr ratio test versus the dipstick test in pregnant women using 24-h urine protein as the gold standard. The sensitivity (94.74% (95% CI = 85.38–98.90) vs 70.0% (95% CI = 55.39–83.14), p = 0.021) and the specificity (84.0% (95% CI = 63.92–95.46) vs 43.75% (95% CI = 26.36–62.34), p = 0.001) of spot P/Cr ratio were significantly higher than the dipstick test. Similarly, the diagnostic accuracy of the spot P/Cr ratio test was significantly higher than that of the dipstick urine test (91.46% (95% CI = 83.29–96.59) vs 59.76% (95% CI = 48.34–70.44), p = 0.001). In addition, the positive predictive value (93.10% (95% CI = 84.59–97.08) vs 66.04% (95% CI = 57.68–73.50), p = 0.001) and the negative predictive value (87.50% (95% CI = 69.66–95.52) vs 48.28% (95% CI = 34.38–62.45), p = 0.003), respectively, of the spot P/Cr ratio test were significantly higher than that of dipstick urine test. While positive likelihood ratio of the spot P/Cr ratio test was higher than that of dipstick urine test (1.93 (95% CI = 1.32–2.80) vs 1.24 (95% CI = 0.87–1.78); however, the negative likelihood ratio of the spot P/Cr ratio test was lower than that of dipstick urine test (0.07 (95% CI = 0.02–0.30) vs 0.69 (95% CI = 0.38–1.22). This is shown in Table 3.
Diagnostic accuracy of spot urine protein to creatinine ratio test versus the dipstick test in pregnant women using 24-h urine protein as the gold standard.
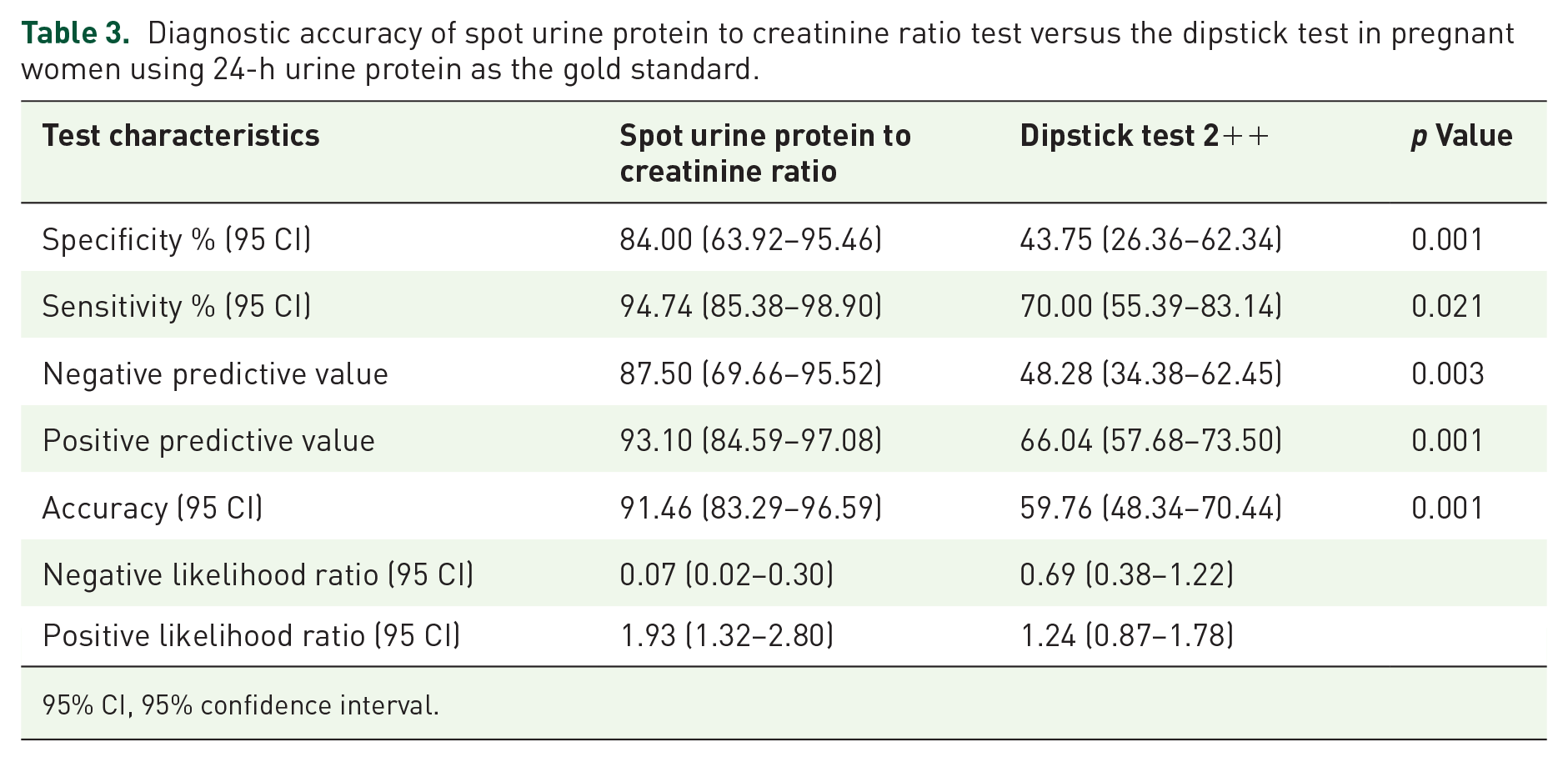
95% CI, 95% confidence interval.
Figure 2 shows the scatter plot diagram showing the relationship between the dipstick test and the protein-to-creatinine ratio in a spot urine sample for the determination of SPIP. There was no relationship between the dipstick test and the PCR ratio in a spot urine sample for the determination of SPIP in women evaluated for preeclampsia.

Scatter plot diagram showing the relationship between the dipstick test and the protein-to-creatinine ratio in a spot urine sample for determination of SPIP.
Discussion
The motivation for this study was that accurate examination of urine for the presence of protein constitutes a very important aspect of care to detect people with preeclampsia and its complications. Prompt and accurate diagnosis may contribute significantly to reducing the adverse effects of this condition in pregnancy. The principal finding of the present study was that the spot P/Cr ratio has higher diagnostic accuracy, sensitivity, specificity, negative predictive value, positive predictive value and positive likelihood ratio than the dipstick test. However, the negative likelihood ratio for the dipstick test was expectedly higher than the spot P/Cr ratio. The spot P/Cr ratio showed a better correlation with 24-h urine protein measurement than the dipstick test for the detection of significant proteinuria in pregnancy.
Previous studies have reported varying degrees of accuracy and sensitivities for the dipstick urinalysis. Ebeigbe et al. 30 in their study that compared the accuracy of the spot P/Cr ratio with that of the dipstick test reported a similar accuracy to our finding for the spot P/Cr ratio of 89.5% and dipstick test accuracy of 55.8%. However, they reported a higher false-positive rate and false-negative rate for the dipstick test when compared to the protein-to-creatinine ratio test. The findings from this study show that the use of dipstick tests for the determination of SPIP is associated with a high potential to overdiagnose because of the high false-positive rate, which increases the tendency to embark on unnecessary interventions in the false-positive cases as well as missing out on those with severe disease due to lower sensitivity. This will further increase the adverse maternal and fetal outcomes associated with preeclampsia in those with false-negative results due to delays in instituting appropriate management protocols. Unnecessary interventions such as caesarean sections or induction of labour may be instituted, leading to preterm deliveries and their attendant complications, such as respiratory distress syndrome, hypothermia and an increased rate of admission into the neonatal intensive care unit, as well as surgical and anaesthetic complications.
The present study also showed that the spot P/Cr ratio had a higher sensitivity and specificity value when compared to the dipstick test. These results are comparable to those reported by Ebeigbe et al., who showed a sensitivity of 91.2% and a specificity of 88.5% for the spot P/Cr ratio. Ebeigbe et al. 30 also reported a similar sensitivity of 73.5% and a similar specificity of 44.3% for the dipstick tests. Additionally, Stefańska et al., 31 in their study that compared the dipstick test and spot P/Cr ratio to 24-h urine protein, reported sensitivity and specificity of 89% (95% CI: 75, 97) and 100% (95% CI: 87, 100), respectively, for the protein to creatinine ratio test. These findings are similar to those of the index study. Katarzyna et al. also found a similar correlation between the spot P/Cr ratio and 24-h urine protein estimation. Although the authors Dwyer et al. 4 employed a cut-off of 0.28 for the spot P/Cr ratio, they also reported a comparable sensitivity of 66% (95% CI: 52) and specificity of 95% (95% CI: 86, 99), and a dipstick test sensitivity of 41% (95% CI: 28, 55) and specificity of 100% (95% CI: 93, 100). As revealed in previous studies, the present study shows that the spot P/Cr ratio has more reliable positive and negative test results than the dipstick test. Hence, it is a much better test for the detection of significant proteinuria in preeclamptic women. Teeuw et al., in a systematic review to evaluate the diagnostic accuracy of urine dipstick tests for proteinuria in pregnant women suspected of preeclampsia, concluded urine dipsticks performed poorly at excluding preeclampsia in hypertensive pregnant women. They recommended that the development of accurate and low-cost bedside proteinuria tests is needed. 32
The present study also demonstrated that there was no relationship between the spot P/Cr ratio and the dipstick test. This could be the result of some mechanisms. For instance, the production of creatinine is constant for every individual, and it depends on the muscle mass of that individual, unlike the excretion of protein in the urine, which depends on the degree of endothelial damage to the kidneys. However, in a prospective observational study done by Baba et al., 33 they determined the P/Cr ratio in a spot urine sample after a dipstick test. They noted that the frequency of positive dipstick test results increased with increasing creatinine concentrations, while the frequency of SPIP was lower in urine samples with higher creatinine concentrations. This clearly indicates that false-positive dipstick results were likely to occur in concentrated urine samples with a higher creatinine concentration for the prediction of SPIP.
Contrary to predictive test values, likelihood ratios are less subject to variations in the presence of a disease in a given population. A likelihood ratio greater than 1 indicates that the test result is associated with the presence of the disease, whereas a likelihood ratio less than 1 indicates that the test result is associated with the absence of the disease. The further likelihood ratios are from 1, the stronger the evidence for the presence or absence of the disease. Likelihood ratios above 10 and below 0.1 are considered to provide strong evidence to rule in or rule out diagnosis, respectively, in most circumstances. 34 In this study, the positive likelihood ratio was 1.93 versus 1.24 and the negative likelihood ratio was 0.07 versus 0.69 for the spot P/Cr ratio and dipstick test, respectively. This finding is interesting because the positive likelihood ratio of the spot P/Cr ratio was greater than 1, which indicated that the spot urine to creatinine ratio was a reliable test for identifying the presence of significant proteinuria. Similarly, the negative likelihood ratio of 0.07 for the spot P/Cr ratio provided strong evidence that the spot P/Cr ratio is very reliable for ruling out significant proteinuria. These findings agree with the findings of the Dwyer et al. study. 4
In this study, the sub-group analysis of women at pre-term gestation compared to term gestation showed that the majority of the participants had preterm delivery. This shows that a greater burden of the disease, in terms of fetal morbidity and mortality, results from complications of preterm birth.
The clinical implications of the findings could be very fascinating; for instance, the dipstick test could be described as cheap, easy to perform, does not require technical expertise and gives an instant result. On the other hand, P/Cr ratio tests are more expensive and require technical expertise and trained laboratory personnel when compared to dipstick urinalysis. The P/Cr ratio test result could be available within 30 min (though longer than the dipstick test time), but it is still timely to make a definitive diagnosis and institute appropriate treatment. Though the P/Cr ratio test may be more expensive, its benefits in terms of sensitivity, specificity and accuracy far outweigh the drawback of cost because the consequences of misdiagnosis and unnecessary interventions are far-reaching.
The strength of this study lies in the fact that our study provided updated information on the accuracy of the spot P/Cr ratio test compared to the urine dipstick test for evaluating significant proteinuria. However, the limitation of the study was that we could not determine the accuracy of the test with respect to the severity of preeclampsia. Also, the study is a single-centre study; hence, it is difficult to generalize our findings; hence, a multicentre, large sample size study is necessary to strengthen or refute our findings. In future studies, factors such as differences in the detection thresholds, variability in test conditions or patient heterogeneity should be evaluated to determine its significant contribution in the interpretation of findings.
Conclusion
In this study, the spot P/Cr ratio, a hospital-based test, has superior sensitivity, specificity and accuracy in the determination of significant proteinuria in pregnant women being evaluated for preeclampsia than the widely used protein dipstick tests. There was no correlation between the protein dipstick test and the spot P/Cr ratio test for the detection of significant proteinuria in pregnancy. While the dipstick test may at best be used as a screening tool because of the cost/convenience, there may be a need for universal adoption of the spot P/Cr ratio test for the confirmation of significant proteinuria in pregnancy in women evaluated for preeclampsia in this setting.
