Abstract
Background
This study explored the feasibility, acceptability, and Māori cultural responsivity of study methods and components of an intervention to support clinicians to resume implementation of Parent–Child Interaction Therapy (PCIT), an evidence-based treatment for disruptive behavior in young children.
Method
This pragmatic, parallel-arm, randomized, controlled pilot trial ran for a 6-month period and included PCIT-trained clinicians who were not delivering, or only rarely using PCIT in their work. Re-implementation strategies were systematically developed and theory-driven and included a mobile co-worker, a portable time-out space, audio-visual equipment, weekly consultation groups, and 2-day targeted PCIT refresher training.
Results
Pre-specified progression criteria included enrolling 20 clinicians, a maximum of 20% attrition, and a monthly survey response rate of at least 80%. Fourteen clinicians enrolled in the trial, there was no attrition, and an 89.8% average survey response rate was achieved. Secondary outcomes included clinician ratings of the usefulness and acceptability of intervention components. The time-out cubicle was considered relatively less useful and acceptable, while the refresher training and manuals were preferred. Study methods and intervention components were considered acceptable by the small proportion of Māori participants. Clinician self-reported Capability, Opportunity, and Motivation to implement PCIT fluctuated monthly across both groups. Pre-/post-changes in each domain within the Theoretical Domains Framework generally showed similar improvement from baseline to follow-up in both groups. There was no observable difference in PCIT adoption in either group.
Conclusion
To the best of our knowledge, this is the first study to pragmatically attempt to re-implement a parent training intervention in a community setting, several years after clinicians’ initial training in the approach (here, an average of 5.36 years). Low attrition and high survey response rates highlighted the feasibility of the data collection methodology. Important opportunities to improve the design of an adequately powered definitive trial are highlighted to minimize future resource waste.
Plain Language Summary
Several effective therapies have been developed to treat children's challenging behavior. A common approach to implementing these therapies in community settings is to train clinicians and provide treatment manuals, consultation with a trainer, and other resources to support the delivery of the therapy. Often, these supports are only available for a limited time. In this study, we recruited clinicians who had been trained in Parent–Child Interaction Therapy several years previously but had not (or had only rarely) been using it in their work. We wanted to explore the concept of re-implementation, or “trying again” after the initial implementation efforts were unsuccessful. It was a deliberately small study, as we wanted to test whether the way we ran the trial, and the supports we provided, would be practical and acceptable. We found that while clinicians enjoyed participating, a future larger trial would need to focus more on recruitment of clinicians, and several of the supports would also need to be adapted.
Keywords
Background
Parent–Child Interaction Therapy (PCIT) is a well-established intervention for conduct problems in children aged 2.5–7 years (Thomas & Zimmer-Gembeck, 2007; Valero Aguayo et al., 2021). PCIT is delivered by way of live coaching of a parent with their child, via a discrete earpiece, from a clinician typically located behind a one-way mirror (Curtin & Thompson, 2023). It has two phases: child-directed interaction (CDI) to enhance the parent–child relationship, and parent-directed interaction (PDI), which includes teaching parents how to utilize time-out with children (Eyberg & Funderburk, 2011; McNeil & Hembree-Kigin, 2011).
To deliver PCIT, master’s-level clinicians receive an initial 40-hr PCIT training, followed by fortnightly 1-hr group consultation for a year (PCIT International Inc., 2018). To become accredited, the clinician, typically an allied health professional, must then complete two cases with fidelity endorsed by an accredited trainer (PCIT International Inc., 2018). In Aotearoa/New Zealand (hereafter, Aotearoa), approximately 135 clinicians had received initial PCIT training at the time of this study, with 16 clinicians accredited, including four trainers (PCIT International website, accessed April 19, 2023). Self-report surveys of PCIT-trained clinicians in Aotearoa suggest that as few as 24% of PCIT-trained clinicians deliver PCIT, often without being accredited, typically seeing only 1–2 families per week for PCIT, commonly alongside a co-worker (Woodfield, Cargo, Barnett, et al., 2020; Woodfield, Cargo, Merry, et al., 2021).
This “train and hope” approach is widespread in the implementation of evidence-based interventions in community settings (Herschell et al., 2010). This is despite increasing awareness that whilst training is necessary to enhance clinician knowledge and skills, it is not always sufficient to result in improved and sustained treatment delivery (Williams & Beidas, 2019). One-size-fits-all training also continues to occur despite the now widespread understanding that implementation context exerts a powerful influence on how, and whether, implementation occurs (Williams & Beidas, 2019). Successful implementation relies upon understanding not only barriers and facilitators to implementation, but also how these barriers act to influence clinician implementation behavior (Lewis et al., 2018; Peters-Corbett et al., 2024).
Tangible and intangible resources are typically directed disproportionately toward these initial training efforts, and relatively few resources are available to support sustained delivery (Clarke & Barwick, 2021; Hall et al., 2022). Considering whether, why, and how clinicians continue to deliver evidence-based interventions post-training is important. In fact, ensuring the sustained delivery of evidence-based interventions by trained clinicians following initial implementation efforts (typically, training) has been described as one of the most pressing translational research challenges of the present day (Nathan et al., 2023). Sustained delivery is also important to ensure the effective use of initial implementation resources and to maximize the impact of the intervention on public health outcomes (Nathan et al., 2023).
Re-Implementation
Even where implementation is not sustained, that is, has stalled or languished, re-implementation may be possible. Re-implementation has been conceptually described but not yet empirically tested in relation to PCIT. The direct cost of PCIT training for one clinician is over $NZD2500, along with indirect costs associated with attending 5 days of initial training and subsequent consultation. As outlined earlier, few PCIT-trained clinicians regularly deliver PCIT in Aotearoa (Woodfield et al., 2020). Investigating the potential of a re-implementation intervention to optimize the initial implementation investment is important.
Conceptually, re-implementation has been described as the process of “resuming implementation, implementing again, and/or implementing differently” Woodfield et al., (2021). Hennein et al. (2021) also similarly described re-implementation simply as “implementation, interrupted.” Moyal-Smith et al. (2023) carried out a scoping review of published literature where re-implementation had been described conceptually or in practice. They proposed a taxonomy and a definition, namely “the systematic process of reintroducing an intervention in the same environment, often with some degree of modification to either the intervention itself or the implementation strategies used to promote uptake.” They proposed three circumstances under which re-implementation may occur: (1) re-implementation following failed implementation, (2) re-implementation due to a flawed intervention, and (3) re-implementation of a discontinued intervention (Moyal-Smith et al., 2023).
Several existing implementation and sustainment frameworks do include provision for subsequent refinement or adaptation of implementation strategies after the initial implementation of an intervention, for example, RE-AIM (Glasgow et al., 1999). However, Moyal-Smith et al. (2023) suggested that the “dynamism that is described in theory has not yet been translated into clear empirical implications for implementation practice.” In practice, it is our impression that this iteration of implementation strategies rarely occurs. If iteration does occur, it typically happens relatively soon after initial implementation, and often in the same context within which the initial implementation occurred. In our program of research, it is common for several years to have passed since initial implementation, and for clinicians to be employed within different service contexts and settings Woodfield et al., (2020). There is a clear need for empirical investigation of a PCIT re-implementation intervention within a practice context, both to optimize existing investment in clinician training and to contribute toward further elucidating the conceptual parameters of re-implementation.
Re-Implementation Intervention Development
We have previously undertaken a behavioral diagnosis process (Michie et al., 2014) to understand the influences on PCIT-trained clinician implementation behavior in Aotearoa. This process, including the selection and prioritization of the implementation intervention components and related material being tested in this trial, is described in detail elsewhere Woodfield, Phillips, et al. (2023). In summary, we utilized the well-established Theoretical Domains Framework (TDF), a synthesis of behavior change theories grouped into 14 domains which characterize the social, environmental, cognitive, and affective influences on behavior (Atkins et al., 2017; Lorencatto, 2022). In our research, clinician implementation behavior appeared to be particularly influenced by two of the 14 domains:
Environmental Context and Resources, specifically lacking suitable PCIT equipment, with (lack of) access to a well-equipped clinic room appearing to influence implementation behavior in several ways. Social/Professional Role and Identity, with beliefs relating to a perception that colleagues view time-out as harmful to children, and clinicians feeling obligated yet isolated in their advocacy for PCIT delivery. There were also concerns that U.S.-developed PCIT is not suitable for non-Māori clinicians to deliver to Indigenous Māori families in Aotearoa.
We then used the Theory and Techniques Tool to identify behavior change techniques, possible intervention components, and their hypothesized mechanisms of action (Carey et al., 2019; Connell et al., 2019; Johnston et al., 2021). Each of these is described in detail in Woodfield, Phillips, et al. (2023). By way of example, we identified that lacking necessary equipment (Environmental Context and Resources) likely limited Physical Opportunity (COM-B component; Atkins et al., 2017), and recommended behavior change techniques from the Theory and Techniques Tool included (a) restructuring the physical environment, (b) adding objects to the environment, and/or (c) prompts/cues. As such, we included a toy pack and access to audio-visual equipment. A second example related to clinicians’ concern that colleagues view time-out as harmful (Social/Professional Role and Identity), which likely influenced their Reflective Motivation (COM-B component), and recommended behavior change techniques included (a) credible source or (b) social support. In response, we incorporated a cross-agency weekly consultation group and a senior Māori trainer and consultation facilitator. The final composition of our re-implementation intervention, described in more detail below, was based on several factors, including the aforementioned behavioral diagnosis process, empirical evidence associated with the effects of these intervention components in other contexts, and available resources Woodfield, Cargo, et al. (2023).
The primary aim of the present study was to assess the feasibility of recruitment processes and trial procedures, with the intention of conducting a definitive re-implementation trial in future. Secondary aims included determining the acceptability of trial processes to participating clinicians. Within this, we were interested in identifying any specific Māori cultural factors that may influence recruitment, data collection methods, or sampling frameworks, and determining the acceptability of the re-implementation intervention for Māori participants. Secondary aims also included assessing the acceptability of the re-implementation package to clinician participants; piloting the use of a series of online self-report surveys as a data collection method for assessing the effects of the re-implementation intervention on clinician Capability, Opportunity, and Motivation (Michie et al., 2011); and exploring potential effects of the PCIT re-implementation intervention on clinician adoption of PCIT.
Method
Study Design
The study was a pragmatic, randomized, controlled, parallel-arm pilot trial of a re-implementation intervention.
Participants and Setting
Clinicians were eligible to participate if they had completed a recognized 40-hr initial training in PCIT, were registered to practice in Aotearoa (regardless of PCIT accreditation status), and were based in the Tāmaki Makaurau/Auckland region. Our preliminary research suggested that few, if any, clinicians saw a full caseload of PCIT clients; however if this were the case they remained eligible to participate, as we were interested in whether the provision of additional supports might influence the nature and quality of implementation. According to a database of trained clinicians compiled for previous studies, there were approximately 70 clinicians eligible to participate in the trial at the time.
Cultural Context
It is important to acknowledge the cultural context for this study. Māori are the Indigenous people of Aotearoa, and Te Tiriti o Waitangi (The Treaty of Waitangi) guarantees partnership, participation, and protection to Māori. This is an essential consideration, given the persistent inequitable health outcomes experienced by Māori (Waitangi Tribunal, 2023) and the overrepresentation of Māori in child welfare data (Cram et al., 2015). There are increasingly calls for Nā Māori, Mā Māori, and Tā Māori (by Maori, for Māori, and with Māori) program delivery (Kapa-Kingi, 2019). Māori clinicians comprise at least 10% of all PCIT trainees (Woodfield et al., 2020).
Procedure
Approval for the study was obtained from the Auckland Health Research Ethics Committee (Ref: AH24443). In July 2022, eligible PCIT-trained clinicians were emailed an invitation to participate in a virtual information session. A reminder email was sent 2 weeks later. Clinicians expressing interest in participating (regardless of whether they attended the information session) were emailed a full Participant Information Sheet and Consent Form. Those who consented to participate were invited to complete a baseline Qualtrics survey, immediately prior to attending a complimentary in-person PCIT refresher training on two successive Saturdays in September 2022.
Random allocation to intervention and control groups occurred immediately following the second refresher training day. The study statistician generated a random allocation sequence (using a simple 1:1 randomization with no restrictions). Participants were advised by email and asked not to share the email or discuss its contents with others. Research team members were blinded, except for the first author, who was directly involved in delivery of the re-implementation intervention.
Intervention and Control Conditions
The components of the intervention are described in detail according to the TIDieR checklist (Hoffmann et al., 2014) in the trial protocol Woodfield, Cargo, et al. (2023). In summary, Two-day in-person Access to a Dropbox containing a Complimentary pack of 25 copies of the Optional complimentary weekly 1-hr A complimentary pack of suitable
In addition to this, the Access to a complimentary Access to Access to an
The implementation intervention components were transportable and carefully chosen to be nested within a clinician's existing service delivery context across a large geographical area. Intervention components were available immediately following the refresher training for the 6-month trial period. Of note, in designing the control condition, we intended to provide a more active condition than (re)training as usual, given that participating clinicians had already received training in PCIT. We were mindful of enhancing the content to make participation worthwhile for clinicians and to justify the costs associated with replicating the training (Woodfield, Cargo et al., 2023).
Outcomes and Measures
Primary outcomes of the pilot study were
Feasibility of recruitment processes and trial procedures
Measure: Number of clinicians who enrolled in the trial and the number of clinicians who completed the trial, respectively, along with the feasibility of achieving a response rate to monthly surveys of greater than 80%. We pre-specified trial progression criteria that included enrolling 20 clinicians and a maximum of 20% attrition (Woodfield, Cargo, et al., (2023)).
Secondary outcomes are detailed in the trial protocol (Woodfield, Cargo, et al., 2023) and are summarized as:
Acceptability of intervention components.
Measure: Frequency count of weekly bookings for: (1) the audio-visual equipment, (2) the time-out cubicle, (3) the mobile co-worker, and (4) attendance rates at group consultation sessions. Measures also included participant rating of the usefulness of these components, and qualitative comments regarding their acceptability and usefulness in an end-of-trial survey. Māori cultural responsivity in relation to trial procedures and intervention components
Measure: Items within an end-of-trial survey. Potential efficacy with a view to determining an effect size for power calculation for a future definitive trial.
Effect of intervention on adoption: Clinician adoption of PCIT was assessed by self-report of the number of unique families seen for PCIT in the most recent representative 1-week period at 6 months post-randomization, compared to baseline. Effect of intervention on clinician Capability, Opportunity, and Motivation: Measures included clinician self-reported Capability, Opportunity, and Motivation to use PCIT (Michie et al., 2014), assessed by Likert scale items within a monthly self-report survey based on the measure described by Keyworth et al. (2020). A previously validated 61-item TDF measure (Seward et al., 2017) was also nested within the baseline survey and end-of-trial survey.
An end-of-trial survey also included contextual information, such as what (if any) additional training clinicians engaged in during the trial period, and whether there were any changes to their employment context during the trial.
Analytical Considerations
We determined that, given our interest in pragmatic feasibility and the relatively small sample size typically inherent in pilot trials, we would predominantly summarize participant characteristics and outcome measures using descriptive statistics. We had planned to carry out some exploratory analyses relating to clinician adoption of PCIT, by way of a two-sample t-test. We pre-determined that we would note, although not statistically account for, observed differences between groups at baseline. The qualitative component of the survey was intentionally modest, as we planned to conduct (and have subsequently carried out) semi-structured interviews with clinician participants. Data from interviews will be analyzed and reported elsewhere. Given the small number of participants in each arm of this trial and the modest amount of qualitative survey data, we have simply presented these responses verbatim.
Results
Feasibility of Recruitment Processes and Trial Procedures
Regarding the feasibility of recruitment processes, nine clinicians attended a pre-trial online information session, and eight of these clinicians went on to enroll in the trial, along with an additional six clinicians who did not attend an information session. As such, a total of 14 clinicians enrolled in the trial and there was no attrition; that is, no participants withdrew from the trial. The 2-day PCIT refresher training was attended by 11 clinicians (78.6%) on Day 1 and eight clinicians (57.1%) on Day 2. Participant flow during the study is described in Figure 1, according to the CONSORT 2010 extension to randomized pilot and feasibility trials (Eldridge et al., 2016).

CONSORT Flow Diagram.
Participant characteristics are described in Table 1. In summary, participating clinicians exclusively identified as female (100%), a majority identified as New Zealand European (50.0%), had completed their initial 5-day PCIT training an average of 5.36 years previously, were predominantly psychologists (41.2%), with 6–10 years of professional experience (28.6%), working in private practice (27.8%), and/or child protection services (27.8%). There were no clear imbalances between groups.
Participant Characteristics.
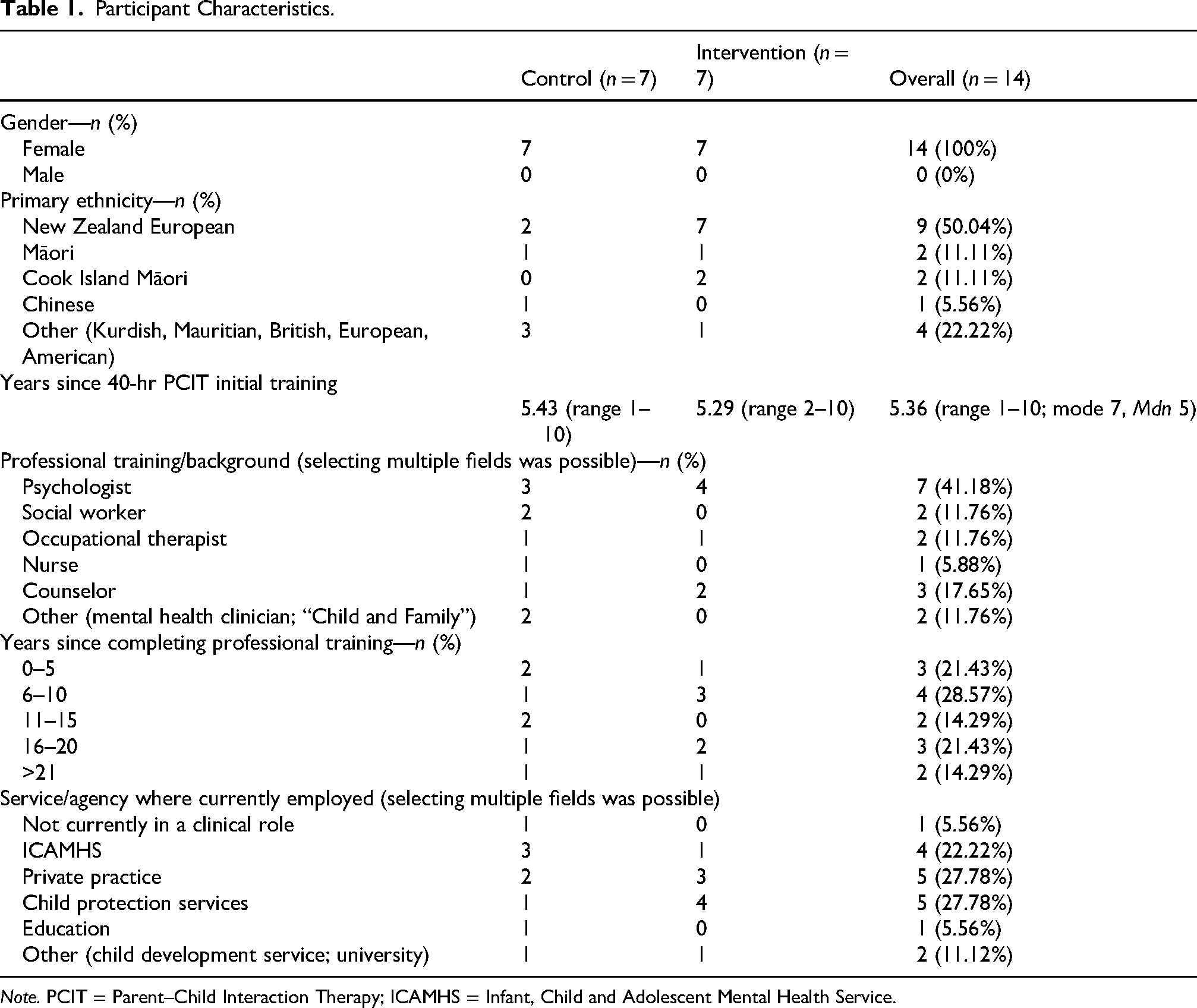
Note. PCIT = Parent–Child Interaction Therapy; ICAMHS = Infant, Child and Adolescent Mental Health Service.
In the end-of-trial survey, no participating clinicians reported changes to the agency they worked for during the trial. Half reported having participated in additional training during the 6-month trial period. This included unspecified Māori language courses, a child and adolescent research conference, an infant feeding scale training, developmental trauma training, a sand-tray workshop, risk assessment, and training relating to substance use disorders in addition to training relating to well-articulated paradigms such as ADOS-2 training (Lord et al., 2000), emotion-focused therapy (Greenberg, 2004), Lego-based therapy (Baron-Cohen et al., 2014), and dialectical behavior therapy (Linehan, 1987) training.
Regarding the feasibility of monthly surveys as a data collection method, response rates to surveys were baseline survey (100%), Month 1 (85.7%), Month 2 (100%), Month 3 (92.9%), Month 4 (78.6%), Month 5 (71.4%), and end-of-trial survey (100%) for an average survey response rate of 89.8%. Qualitative comments from participants in the end-of-trial survey suggested that monthly surveys were acceptable as a data collection method, with participants describing the surveys as “easy to complete” and “to the point.” Several participants noted that the monthly surveys were “motivating … brought my focus back to what I wanted to achieve through the study,” or that the surveys “did make me stop and think and reflect about PCIT and whether I had scope to implement it currently.” They were also described as “a good way to keep [clinicians] engaged.”
Regarding the feasibility of randomization, a test of blinding was carried out, and of the 13 clinicians who responded to this item, all correctly identified which group they had been allocated to. Qualitative comments suggested that clinicians understood and accepted that a control group was “necessary” but suggested it was “nigh on impossible” to “keep resources confidential.” One participant described “guilt” at having been allocated to the intervention group and not using the resources available.
Acceptability of Intervention Components
No bookings were received for the audio-visual equipment, the time-out cubicle, or the mobile co-worker during the trial.
Mean ratings of acceptability and usefulness from two sets of 7-point Likert scales in the end-of-trial survey are presented in Figures 2 and 3. In summary, the time-out cubicle was considered least acceptable by both the control and intervention groups and was also rated as least useful by both groups. The refresher training and resources were considered most acceptable to both groups, and the refresher training was rated most useful by both groups. Verbatim qualitative comments relating to acceptability and usefulness drawn from the end-of-trial survey are presented in Table 2.

Mean

Mean
Verbatim Qualitative Comments Relating to Acceptability and Usefulness of Intervention Components, From End-of-Trial Survey.

Note. PCIT = Parent–Child Interaction Therapy.
Twenty-one weekly hour-long online consultation groups were held during the 6-month trial, allowing for a brief hiatus over the Christmas period. The overall mean number of clinicians attending per week was 2.48 (range 0–7). Further information relating to attendance rates is presented in Table 3.
Attendance at Weekly Group Consultation Sessions.

Māori Cultural Responsivity
Within the end-of-trial survey, four participants indicated that they identified as Māori and responded to the relevant survey items, indicating that
Potential Efficacy
Effect of intervention on adoption: PCIT sessions are typically held weekly. Hence, we measured clinician adoption by asking clinicians to self-report the number of unique families to whom PCIT was delivered in the most recent representative 1-week period. We assessed clinician adoption at baseline and at the end of the trial (6 months post-randomization). As is apparent in Table 4, there was no observable difference between the mean number of clients provided with PCIT in the most recent representative week between baseline and end-of-trial.
Effect of intervention on clinician COM-B and TDF domains: Clinicians rated their Capability, Opportunity, and Motivation to deliver PCIT on a 10-point Likert scale in each monthly survey, for example, “I currently have the physical (in my body) and/or psychological capability to use PCIT,” from strongly disagree (0) to strongly agree (10). Mean monthly ratings are presented in Figures 4–6.
All participants responded to all 7-point Likert scale items on the 61-item TDF measure (Seward et al., 2017) nested within the baseline survey and end-of-trial survey. A mean of the mean ratings (summary mean, or pooled mean) for each item within the 14 domains is presented in Figures 7 and 8. Supplementary Table 6 provides the pre-intervention and post-intervention summary means.



Mean Monthly Self-Rated Capability, Opportunity, and Motivation to Deliver PCIT

Pre-Intervention and Post-Intervention TDF Domains, Pooled Means for Control Group.

Pre-Intervention and Post-Intervention TDF Domains, Pooled Means for Intervention Group.
Adoption: Mean Number of Families Seen for PCIT in the Most Recent Representative Week.
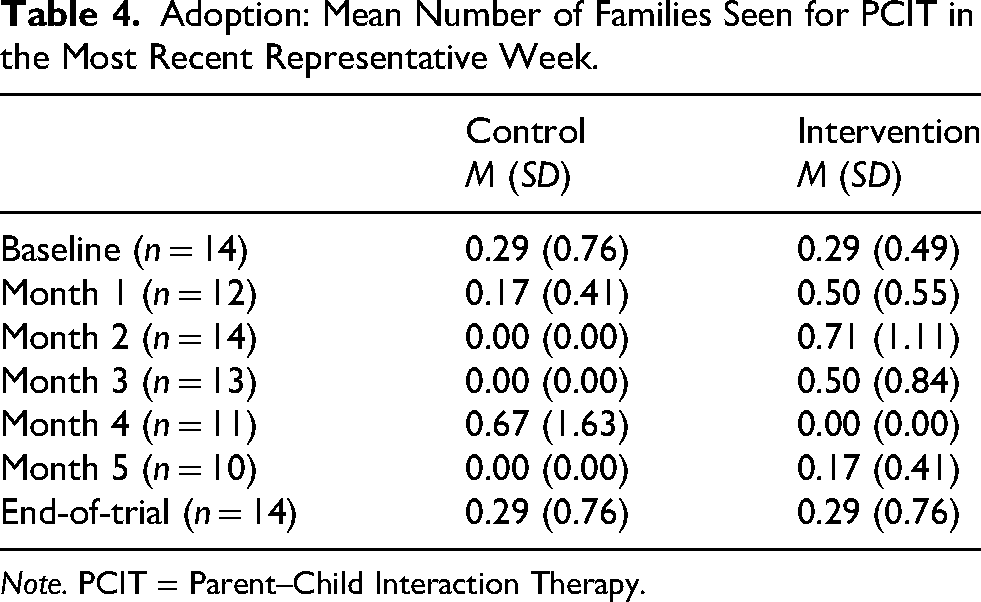
Note. PCIT = Parent–Child Interaction Therapy.
Deviations From Protocol
Whilst we had intended to carry out some exploratory analyses alongside descriptive statistics for clinician adoption of PCIT by way of a two-sample t-test, given that there was no observable difference it did not seem warranted.
Allocation to groups had been proposed to occur by way of distribution of sealed opaque envelopes at the end of the second day of in-person training. However, only approximately half of enrolled clinicians attended the second day of training (refer above), so the study statistician used a random number generator within Stata to allocate participants to groups via unique anonymized study identifiers, and the first author sent an individualized confidential email to each participant following the second day of training.
Discussion
To the best of our knowledge, this is the first study to pragmatically attempt to resurrect the implementation of a parent training intervention in a community setting, several years after the clinician's initial training (in this study, an average of 5.36 years). We had pre-determined that it would be feasible to progress to a fully powered randomised controlled trial in future if at least 20 clinicians enrolled in the trial, and there was a maximum of 20% attrition (Woodfield, Cargo, et al., 2023). While the lack of attrition from the pilot was encouraging, recruiting only 14 clinicians (of a possible 70), and recruiting only a small number of Māori clinicians suggests that more active and/or targeted recruitment efforts may be required in a future trial. Our average survey response rate of 89.8% indicated that online surveys were an acceptable and feasible data collection method.
Drawing conclusions around the feasibility of progressing requires some nuance. We acknowledge that the criteria we have used to decide whether it is feasible to progress are somewhat blunt, and it may have been useful to incorporate more subtle considerations. For example, Bugge et al. (2013) proposed an algorithm to aid decision-making around progressing pilot and feasibility trials, and Murray et al. (2010) described a tool to improve trial design and procedures, with implementation in mind. Adopting a wider lens may also be important, for example, considering progression criteria in harmony with each other, rather than in isolation, and incorporating consideration of other contextual factors external to the trial (Mellor et al., 2023). Pearson et al. (2020) propose the notion of a traffic light system, rather than strict cut-offs or thresholds for progression criteria. Under such a system, an “orange” designation may be appropriate here.
Trial Recruitment
Our ability to understand factors contributing to clinicians’ decision not to enroll was limited, as essentially all clinicians who attended an information session or contacted the research team went on to enroll. The small number of Māori participants limited our ability to understand the acceptability of recruitment and trial processes to Māori clinicians.
Re-Implementation Intervention Components
Overall, we observed a dissonance between ratings of usefulness and actual use. Intervention components were generally rated as acceptable and perceived as useful. The acceptability of this collection of implementation intervention components to clinicians may have been influenced by the supports that clinicians are familiar with receiving in Aotearoa, namely training and ongoing consultation. The time-out cubicle, AV equipment, and co-worker were not utilized at all by those clinicians to whom they were available. It is possible that the refresher training and the toy package (available to both groups) were sufficient to result in the pre-post differences observed in the TDF domains, and that the additional supports did not add value. Also, with reference to the COM-B model (Michie et al., 2014), whilst provision of supports in this study perhaps addressed a lack of physical opportunity, it may be that further consideration ought to be given to enhancing social opportunity in future studies (we did not differentiate between the two in our COM-B measure). Finally, judging by the relatively low acceptability and low (relative to other components) perceived usefulness of the time-out cubicle, this intervention component likely requires modification or substitution in a future trial.
Both groups were able to access consultation sessions, and supervision or consultation is an implementation strategy in itself (Bearman, 2021), one that ought to be measured carefully, and typically involves discrete techniques that are associated with improved implementation outcomes, namely active learning and audit and feedback (Choy-Brown et al., 2023). Supervision and consultation may improve implementation outcomes by way of enhancing reflective motivation, psychological capability, or particularly in the case of group supervision, social opportunity (Woodfield, Phillips et al., 2023). A future, larger trial may involve offering consultation or supervision solely to the intervention group.
Measures and Outcomes
While adoption was a secondary outcome in this pilot study, it appears that adoption measures may require further consideration in a future trial. A more detailed assessment of whether and how participating clinicians use PCIT in their work at baseline would have been informative, including to provide data on more subtle shifts in adoption practices. In hindsight, it would also have been useful to measure the total number of clients seen for PCIT across the trial period for each group as a measure of adoption. It would also be useful to assess whether a PCIT case was co-worked by another participating clinician—that is, possibly “counted twice.” It will be important for any future measure of adoption to be better able to discern relatively slight changes. Relating to other outcomes, it would be useful in future to assess fidelity to the re-implementation intervention itself, for example, attendance at refresher training and uptake of tangible materials.
Monthly self-reported Capability, Opportunity, and Motivation to deliver PCIT were informative, including that the COM domain scores were higher in the control group, in many instances, than the intervention group, particularly from Month 3 onwards. It is possible that by this point clinicians in the intervention group had decided or realized that they would be unlikely to utilize the implementation supports before the end of the trial. Of note, seasonal patterns will need to be controlled for in a future trial, as the extended Christmas break that occurs in Aotearoa (occurring prior to the Month 4 survey) appeared to influence these.
Limitations
In this study, the focus of implementation supports was exclusively at the individual clinician level and within existing service delivery parameters, which represents a limitation. Incorporating patient-, service-, or system-level implementation strategies is likely to be necessary in future, including investigating how determinants across these levels interact to influence implementation outcomes across different phases of implementation (Powell et al., 2020). The assumption that individual clinicians have the discretion to select which treatment(s) to offer is fraught, as organizational and policy context can powerfully influence which treatments are funded or endorsed. Future re-implementation efforts ought to adopt a multi-level, comprehensive approach.
Relatedly, future strategies are likely to be more effective if tailored to the context within which the clinician is delivering care. For example, clinicians within public services may already have access to a trained co-worker, while the addition of a co-worker may be an effective intervention for a clinician in an independent private practice.
Māori Cultural Responsivity
PCIT is an imported manualized program developed in the United States. Recent Māori-led research has investigated the acceptability of PCIT to whānau (families) (Cargo, 2020). To enhance implementation in Aotearoa, understanding the acceptability of both PCIT itself, and any implementation or re-implementation strategies to Māori clinicians is also important, and this study presented an opportunity to do so. However, with one exception, we are a tauiwi/non-Māori group of researchers. Kaupapa Māori research ought to ideally be conducted “by Māori, for Māori, with Māori.” As such, while it was important to canvas the acceptability of research processes and re-implementation supports to Māori participants in this trial, it was also important to demonstrate cultural humility by not presuming to lead this work. Hence, cultural responsivity was not a primary outcome in this study, and we are limited in our ability to describe required cultural adaptations to future research processes or re-implementation supports.
In planning future research, alongside considering research team composition, an appropriate methodological approach may be to design for equal explanatory power. Equal explanatory power means that research has either prioritised Māori participation in quantitative research or is constructed so that the Māori sample is equally powered to answer the research question in simple and/or complex analyses … It is not “over-sampling” Māori, rather it is appropriate sampling and respect for the Māori “voice”. Constructing a sample with equal power to answer the research question for Māori as well as non-Māori will provide multiple opportunities for dissemination. (Reid et al., 2017)
Future Research
Whilst we intentionally attempted randomization at the clinician level, cluster randomization appears to be necessary in the future, given the results of the test of blinding and the potential for intervention contagion. However, cluster randomization would require an even larger sample size, and the sample size required to detect the anticipated slight changes in adoption is already likely to be substantial. A multi-center, international trial is likely to be necessary.
Insights from our previous research suggested that lacking required equipment was a barrier to clinician use of PCIT, and provision of equipment might remedy this. However, clearly it is not this simple. Qualitative responses to survey items were in keeping with our earlier work and confirmed several possible hypotheses that future research ought to test. PCIT-trained clinicians may perceive that their professional colleagues do not endorse their use of PCIT due to concerns about time-out causing harm or exacerbating existing trauma for children (Woodfield et al., 2021; Woodfield, Cargo, et al., 2023). It is also possible that clinician knowledge that time-out is both effective and safe for parents to use, is overpowered by the emotional valence associated with teaching and coaching parents to use the technique. Therapist anxious distress and associated avoidance of intra-intervention components such as time-out is understudied as an influence on clinician decision-making and implementation of evidence-based treatments (Klein et al., 2024).
It is possible that the validity of identified implementation determinants was undermined by weaknesses in methods used to assess them. Methods for assessing influences on clinician implementation behavior have predominantly relied upon clinician self-report (Martinez et al., 2014), and this study is no exception. Self-report data may be undermined by recall issues or social desirability, leniency, or self-serving cognitive biases, including attributing implementation failures to external influences rather than internal factors (Atkins et al., 2017; Landsverk et al., 2023; Martinez et al., 2014). Data from self-report surveys may reflect participants’ cognitions about influences on their behavior, rather than actual causes (Francis et al., 2012). Validity may be enhanced by incorporating multiple perspectives and data sources (e.g., direct observation, policy documents) alongside self-report (Atkins et al., 2017). Technological advances may enable novel methods such as biometric data from wearable devices to detect changes in clinician heart rate, which may indicate stress.
In conclusion, low attrition from the trial and high survey response rates suggested good engagement from, and acceptability to, participating clinicians. However, we suggest that—rather than progress this trial in its current form—consideration is given to adapting aspects of the methodology, particularly recruitment methods. Some intervention components (i.e., the time-out cubicle and the nature of the control condition) also appear to require further consideration and/or modification. It may be prudent to carry out another pilot study to further shape trial design and intervention components prior to proceeding to a definitive trial. The outcomes of this pilot are informative for researchers and other stakeholders who have an interest in re-implementing or resurrecting stalled implementation of parent training in community settings. We trust that sharing these data will facilitate judicious use of limited research and practice resources in future.
Supplemental Material
sj-docx-1-irp-10.1177_26334895251363418 - Supplemental material for Re-Implementation of Parent–Child Interaction Therapy (PCIT) in the Community: Findings From a Pilot Randomized Controlled Trial
Supplemental material, sj-docx-1-irp-10.1177_26334895251363418 for Re-Implementation of Parent–Child Interaction Therapy (PCIT) in the Community: Findings From a Pilot Randomized Controlled Trial by Melanie J Woodfield, Sarah Fortune, Tania Cargo, Sally Merry and Sarah E Hetrick in Implementation Research and Practice
Footnotes
Acknowledgements
The authors wish to acknowledge Dr Gemma Tricklebank (Ngāti Mahuta, Ngāti Maniapoto) for her expertise and support in facilitating consultation groups and delivering training for participating clinicians.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research was supported by a Clinical Research Training Fellowship for Dr Melanie Woodfield from the Health Research Council (HRC) of New Zealand. At the time of the study, Professor Sally Merry held the Cure Kids Duke Family Chair in Child and Adolescent Mental Health. Associate Professor Sarah Hetrick held an Auckland Medical Research Foundation (AMRF) Douglas Goodfellow Repatriation Fellowship and was a Cure Kids Research Fellow. Dr Sarah Fortune has received project funding from Cure Kids, the Hugo Trust, the Health Research Council of New Zealand, and the Oakley Mental Health Foundation. Funders were not involved in study design or execution.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
