Abstract
Nanodiamonds (NDs) have the characteristics of both diamonds and nanomaterials. However, it is difficult to disperse NDs, and this is why there is less research regarding NDs in the field of aluminum matrix composites. In the present work, NDs were modified via surface oxidation, and ND/Al matrix composites were successfully prepared via mechanical ball milling and vacuum sintering. The effects of different volume fraction of NDs (1%, 3%, 5%, 7%) after surface oxidation on the ND/Al matrix composite were analyzed using a metallographic microscope, scanning electron microscope, infrared spectrometer, X-ray diffractometer, microhardness tester, and universal testing machine. The results show that the optimal temperature of surface oxidation treatment is 673 K, which effectively purifies the surface of ND and introduces appropriate C=O functional groups. NDs are uniformly distributed in the aluminum matrix, and no harmful Al4C3 phase is formed. With an increase in the volume fraction of NDs, the grain size of the matrix first decreases and then increases, and the ultimate compressive strength first increases and then decreases. The volume fraction of ND with better comprehensive performance is 3% and the yield strength increased by 19%.
Keywords
Introduction
Particle-reinforced aluminum matrix composites have become an indispensable high-performance material in transportation, the electronic industry, military defense, aerospace, and other fields because they are low cost and have good mechanical properties, low density, and easy preparation and processing. 1– 4 Finding new particle-reinforced materials is important for developing high-end aluminum matrix composites. Nanodiamonds (NDs) have the characteristics of both nanomaterials and diamonds. In particular, the many excellent properties of NDs include high modulus, high hardness, high thermal conductivity, low friction coefficient, wear resistance, and good chemical stability, and these make NDs a good reinforcement material for aluminum matrix composites. 5– 7
NDs have the general properties of nanomaterials and easily agglomerates. Mechanical dispersion is the most traditional and simple physical dispersion. The most common forms of mechanical dispersion are grinding, ball milling, and high-speed agitation. 8– 10 ND is a lumpy aggregation, and mechanical ball milling dispersion can cause it to be evenly dispersed in the matrix and to be fully refined. The surface modification of NDs is also needed for specific application purposes. Researchers have used carboxylation, hydrogenation, ammoniation, hydroxylation, and surface oxidation to form some chemical functional groups on the surface of NDs. Pedroso-Santana et al. 11 used bead-assisted sonic disintegration and carboxylation procedures. In an aqueous suspension, the aggregate size of diamond is reduced from greater than 1 µm to approximately 100 nm. Tsubota et al. 12 introduced a C=C bond, which is a chemically active bond, to the diamond surface via chemical reaction of benzoyl peroxide on the surface of hydrogenated diamond with linoleic acid. The diamond surface modified with the C = C bond is used as a substrate for surface-modified diamond. Delavar and Shojaei 13 used thermal oxidation at 420°C for different lengths of time (1.5 and 4.5 h) under an atmosphere of air to prepare NDs with variable carboxyl functional group content. Studies have shown that NDs with higher carboxyl functionality can provide higher mechanical properties. Changing, adjusting, and designing the chemical activity of the ND surface can reduce the agglomeration of NDs and improve its dispersion ability. Hsu et al. 14 poured NDs into a mixed solution of H2SO4 and HNO3, which was stirred at 100°C for 72 h and centrifuged. The precipitated particles were washed with deionized water until the pH was neutral, and then the surface of NDs was grafted with carboxyl groups. Martin et al. 15 reported the successful oxidation of diamonds with FeSO4 reagent and discussed its functional effects, such as the removal of amorphous carbon from a diamond surface via oxidation to form CO2 and the formation of hydroxylated diamond (DND-OH) via reaction of the generated –OH group with the diamond surface. Dispersant dispersion is usually achieved by changing the surface charge of nanoparticles. A small amount of dispersant, which is also known as surfactant (surface active agent (SAA)), can significantly change the surface properties of nanoparticles. 16– 18 In general, most of these methods have been combined to improve the dispersion of NDs in the matrix. Li et al. 19 modified NDs and found that they can be stably stored in acetone for 3 months. First, NDs were oxidized at 420°C for 1.5 h, and then NDs and the surfactant sodium octadecyl benzene sulfonate were wet milled together to modify the NDs. Shenderova et al. 20 first removed the impurities from the surface of NDs using a mixed acid solution. They then oxidized the surface of the NDs in air at a suitable temperature and boiled the NDs in chromic acid for 2 h. Finally, the NDs were stably dispersed in an aqueous solution. The particle size of the NDs was about 60 nm.
The depolymerization of NDs is a problem that must be addressed in the development and application of this material. In this study, we modified the surface of NDs via surface oxidation. It is economical and easy to operate. NDs after surface oxidation treatment were used to reinforce aluminum matrix composites, which were prepared via vacuum sintering. The microstructure and mechanical properties of the ND/Al composites were studied. With the rise of nanomaterials in the 21st century, researchers began to study ND/Al composite materials in recent years. It is hoped that this study will provide a reference for future research.
Materials and methods
In this study, 1070 aluminum powder (with a particle size less than 20 µm) and ND powder (with an average particle size of 10 nm) were used. First, raw NDs were modified via surface oxidation. For the surface oxidation treatment, ND raw powder was put into a box resistance furnace and treated it in an atmosphere of air at 673 K and 773 K for 1 h each.
The ND particles were then mixed with pure aluminum powder at 250 r min−1 for 5 h, and the mixed powder with a uniform distribution of ND particles was obtained. The values of the volume fraction of NDs in the mixed powder were 1 vol%, 3 vol%, 5 vol%, and 7 vol%. The mixed powder was pressed into blocks, which were vacuum sintered in an OTF-1200X tube furnace (Hefei Kejing Material Technology Co. LTD, China) at 823 K. The sintering process is shown in Figure 1.

Composite material sintering process.
The metallographic structure of the ND/Al composite was observed using a 4XC-MS inverted metallurgical microscope (Shun Yu Optical Technology Co. LTD, China). The corrosive agent was a preformed Keller reagent. The obtained specimens were cut and polished and then immersed in Keller reagent for 20 s. The phase analyses of ND powder and ND/Al composites were carried out using a Hitachi SU8220 scanning electron microscope (SEM) (Hitachi, Japan), energy dispersive spectrometer (EDS), and X-ray diffraction (D/MAX 2500V). The surface functional groups of the original diamond particles and of the surface-modified ND particles were analyzed using a Nicolet 6700 infrared spectrometer (Nicolet, US). The mechanical properties of ND/Al composites were characterized using an automatic electronic densitometer (GH-128E), microhardness tester (HVT-1000), and a universal testing machine (Instron 8801 (Instron, UK)).
Results and discussion
ND surface oxidation
As seen in Figure 2, there is an obvious (002)g graphite diffraction peak near 2θ = 20°–30°. Compared to the unmodified ND of line 1 in the figure, after surface oxidation treatment at different temperatures, the peak is slightly higher than the background at 2θ = 20°–30°. However, there is no sharp (002)g graphite diffraction peak and only an obvious diamond diffraction peak, which indicates that the surface oxidation treatment can effectively remove the graphite phase. Figure 3 is the infrared spectrum of ND. The absorption peaks of the hydroxyl O-H group are at 1631.16 cm−1 and 3434.82 cm−1. There is no C=O group on the surface of untreated ND particles. After the surface oxidation treatment, the absorption peak at 1785.10 cm−1 is obviously enhanced. The surface oxidation modification introduces the C=O functional group, and with an increase in the temperature of surface oxidation treatment, the amount of the C=O functional group also increased. However, when the surface oxidation temperature was 773 K, the absorption peak of the hydroxyl O-H group at 3434.82 cm−1 was enhanced, and the weight loss of ND powder after the surface oxidation treatment was significantly higher than that at 673 K.

XRD patterns of raw NDs and treated ND particles. XRD: X-ray diffraction; ND: nanodiamond.

FTIR spectra of original NDs and treated ND particles. FTIR: Fourier transform infrared; ND: nanodiamond.
Figure 4 shows the metallographic structure of ND/Al composites. The grain boundary of the composites that were prepared via vacuum sintering are clear and uniform, and this shows that mechanical ball milling has a better effect on the mixed material. Also, the mixed powder can be evenly distributed, and this results in fewer defects such as holes in the matrix. The surface oxidation modification of NDs removes impurities from the surface of particles and facilitates the insertion of NDs particles into the gap of Al particles. With an increase in the volume fraction of NDs in the composites, the agglomeration of ND particles becomes serious, and defects such as holes and loosening of the matrix increase. When the volume fraction of NDs reaches 7 vol%, the number of pores increased obviously, and the partial grain boundary was elongated because of the segregation of NDs, which affects the comprehensive properties of the composites. The best microstructure of ND/Al composites is the 3 vol% ND/Al composite. The EDS mapping of the 3 vol% ND/Al composite is shown in Figure 5. According to the distribution of C, the ND particles are evenly distributed in the aluminum matrix. Figure 6 is an SEM image of ND/Al mixed powder after mechanical milling for 5 h. After milling, the larger particles are Al powder, the particles have a smooth surface and a round shape, and the particle size was about 2 µm. The ND particles are evenly distributed on the surface of Al particles, and this shows that ball milling was effective at mixing ND particles and aluminum powder. Also, after repeated crushing and impacting during mechanical milling, the NDs are embedded in the gap between the two Al particles. The results show that after surface oxidation modification of NDs, the ball milling process refines the grain size of the matrix and evenly mixes the reinforcement particles with the matrix powder, and this improves the dispersion strengthening effect.

Metallographic structure of ND/Al composite after surface oxidation treatment: (a) 1 vol% NDs, (b) 3 vol% NDs, (c) 5 vol% NDs, and (d) 7 vol% NDs. ND: nanodiamond.

EDS mapping of 3 vol% ND/Al composite: (a) Corresponding distribution of Al (b) and C (c) in Figure 5(a). EDS: energy dispersive spectrometer; ND: nanodiamond.

SEM of ball milled mixed powder with NDs and pure aluminum powder. SEM: scanning electron microscope; ND: nanodiamond.
XRD analysis results of ND/Al composites that had different volume fractions of NDS are shown in Figure 7(a). Regardless of how the volume fraction of ND was changed, the 2θ angles of the obvious diffraction peaks are 38.38°, 44.64°, 65.04°, 78.17°, and 82.41° in the ND/Al composites, and these correspond to the diffraction peaks of Al. (According to PDF#85-1327, the 2θ angles are 38.47°, 44.71°, 65.10°, 78.23°, and 82.44°.) The XRD patterns of the ND/Al composites deviate 0.1° to the left, which is within the allowable range of system error. Yang et al. 21 and others have reported similar findings. Li and Bowen 22 detected the diffraction peaks of the Al4C3 phase in a graphene/Al composite, and Che et al. 23 detected the diffraction peaks of the Al4C3 phase in Al/diamond composites. Combined with the above literatures, we can conclude that Al4C3 is not produced in this experiment, on account of no diffraction peak of the hard and brittle intermediate phase Al4C3 in Figure 7(a). According to the Scherrer formula, the average crystallite size of the ND/Al composites is shown in Figure 7(b). With an increase in the volume fraction of NDs, the crystallite size first decreases and then increases. To a certain extent, the addition of NDs refines the matrix grains and plays a role in fine grain strengthening. When the volume fraction of NDs was increased to 7 vol%, the crystallite size of the ND/Al composites increased to 47.849 nm. This indicates that the agglomeration of ND particles increases the grain size of the matrix after sintering. Meanwhile, the mechanical properties of the ND/Al composites will be decreased.

XRD pattern (a) and crystallite size (b) of ND/Al composites with different volume fractions of NDs. XRD: X-ray diffraction; ND: nanodiamond.
Mechanical properties of ND/Al composites
The effects that different volume fractions of NDs have on the experimental density and relative density of the ND/Al composite are shown in Figure 8. The experimental density and relative density of the composite decreased with an increase in the NDs volume fraction, and the highest relative density of the composite was 92.8%. This is because when pressureless sintering is used, small particles cannot fully fill the gaps of large particles, the gap between powders is large, and the wettability between ND and Al is poor. Thus, the interface between ND and Al was not closely combined after sintering. Also, the number of interface gaps increased so that the density of the composites decreased. When the volume fraction of ND was 7 vol%, the relative density was only 89.68%, and this shows that when the volume fraction was increased, the ND agglomeration seriously increased defects, such as holes and cracks, which has a great influence on the density of the material. Figure 9 shows a histogram of the effect that different volume fractions of NDs had on the hardness of the composite. With an increase in the volume fraction of NDs, the microhardness of the composite enhanced. There have two reasons. The first is that when the volume fraction of NDs are added, the grain boundary fraction per unit area is larger, and the dislocation movement is more prominent. Thus, grain boundary strengthening was achieved to improve the hardness. The second is ND, as a reinforcing phase particle, is added to the soft matrix, which can significantly increase the microhardness of the matrix. When the volume fraction of NDs was 7 vol%, the maximum microhardness was 60.1 HV, which is 69.3% higher than that of pure aluminum.
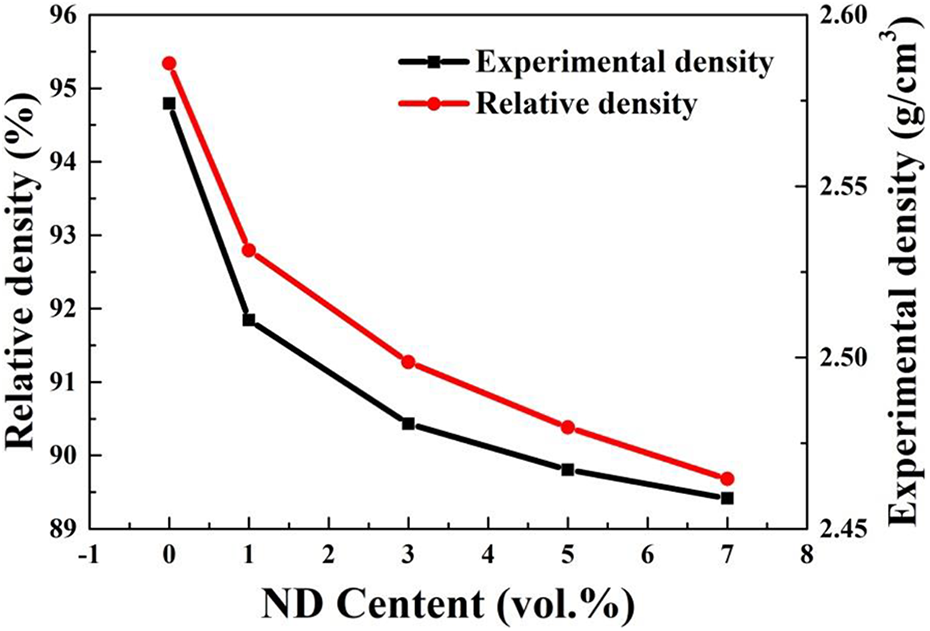
Experimental density and relative density curves of ND/Al composites. ND: nanodiamond.

Influence of the volume fractions of NDs on hardness of composites. ND: nanodiamond.
The compression test results of the composites are shown in Figure 10. The ultimate compressive strength first increased and then decreased with an increase in the volume fraction of NDs, and the elongation decreased with an increase in the volume fraction of NDs. When the volume fraction of NDs was 3 vol%, the ultimate compressive strength was 175.74 MPa, which is 19.0% higher than that of pure aluminum (147.73 MPa). When the volume fraction of NDs was more than 5 vol%, the ultimate compressive strength was lower than that of pure aluminum. This is because the wettability of NDs and the Al matrix is very poor, and the agglomeration phenomenon of NDs is serious, which makes the material performance worse or even worse than that of the base metal.

Curves of compressive strength and elongation of composite with different volume fractions of NDs. ND: nanodiamond.
Conclusions
The surface-oxidized ND-reinforced Al matrix composites were prepared via vacuum sintering, and the microstructure and mechanical properties were studied. The following conclusions are drawn:
NDs are uniformly distributed in the Al matrix, and the harmful Al4C3 phase is not present.
The better temperature for the surface oxidation treatment is 673 K, which effectively purifies the surface of NDs and introduces a proper amount of the C=O functional group, which has better dispersion and is more economical.
With an increase in the volume fraction of NDs, the grain size of the matrix first decreased and then increased, and the ultimate compressive strength first increased and then decreased. The volume fraction of NDs increased to 3 vol%, and the yield strength increased by 19%. The increasing in strength results from the uniform distribution of fine particles and NDs in the matrix phase.
Footnotes
Authors’ contributions
All authors contributed equally and significantly in writing this article. All authors read and approved the final manuscript.
Acknowledgments
The authors thank Dr Shanshan Hu, the associate professor of College of Mechanical Engineering, Guangxi University, and Dr Jianmin Zeng, the professor of the Center Ecological Collaborative Innovation for Aluminum Industry in Guangxi University, for providing the help for this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by National Natural Science Foundation of China [Grant No. 51965005], Guangxi Science and Technology Major Project [Grant Nos. AA17204036-1 and AA17202008-1], the Natural Science Foundation of Guangxi [Grant No. 2018GXNSFAA281258], and the Open foundation of Guangxi Key Laboratory of Processing for Non-ferrous Metals and Featured Materials, Guangxi University [Grant No. 2019GXYSOF02].
