Abstract
This study mainly focused on an orthogonal optimization design of low-density and high-strength ceramic proppants prepared by low-grade bauxite and feldspar that met the standard requirements. The orthogonal experimental design of L25 (53) was employed to study the significance sequence of three factors, including milling time of bauxite, milling time of feldspar, and sintering temperature. The results show that the particle size of feldspar is the most important factor for the performance of the proppants. The longer the milling time of feldspar is, the finer the particle size of feldspar is, and the easier it is to form liquid phase at a lower temperature. For the breakage ratio, the optimal experimental combination is bauxite milled for 4 h, feldspar milled for 8 h, and sintering temperature of 1280°C. The proppants prepared by the optimal combination have bulk density of 1.48 g cm−3, apparent density of 2.7 g cm−3, breakage ratio of 4.07% under 52 MPa closed pressure, and acid solubility of 2.15%.
Introduction
Ceramic proppants are of extreme importance in the hydraulic fracturing process, an effective method for extracting natural oil and gas, shale gas, and coalbed methane resources, as they provide long-term conductivity of the fracture. 1–4 The desirable ceramic proppants typically have high sphericity and roundness, low density, and high strength, because these proppants can improve service years of oil and gas well and increase its production rate. 5– 7 However, most commercially available proppants are sintered between 1300°C and 1600°C, which undoubtedly increases the mining cost.
One alternative to decrease the sintering temperature of ceramic proppants is the addition of sintering aid, including feldspar, 8 dolomite, 9 purple sands, 10 calcium oxide, and manganese dioxide. 11 But these aids rarely reduce the sintering temperature to below 1300°C. Another way is to add as much solid waste as possible, such as fly ash, 12– 14 coal gangue, 4,15 waste ceramic sands, 16 and magnesium slag. 17 The addition of solid waste can not only reduce the sintering temperature but also reduce the preparation cost, but it inevitably reduces the crush resistance of proppants. Therefore, it is particularly important to find a way that can be prepared at lower temperature without reducing the proppants’ performance.
Ma et al. reported that adding 4% feldspar could reduce the sintering temperature of proppants by 100°C, still higher than 1300°C. 8 Feldspar is one of the most common aluminosilicate minerals with relatively low melting point (about 1100°C) and wide melting range. 18 In addition, Mocciaro et al. studied the effect of raw material milling on ceramic proppants properties and found that raw materials with smaller size could improve the crush resistance of proppants. 19 Combined with our previous research results, it can be inspired that the raw materials can be ball milled to further reduce the sintering temperature without affecting the proppants’ crush resistance.
In the present work, low-grade bauxite (containing aluminium oxide (Al2O3) of 60%) and feldspar were used as raw materials to prepare the ceramic proppants at low temperature. Based on the previous research results, 20 the optimal content of feldspar was 15 wt%. The aim of this article was to optimize the design of low temperature preparation of low-density and high-strength proppants based on orthogonal experiments. After the raw materials were ball milled for different time, bulk density, apparent density, breakage ratio, and acid solubility of the proppants were thoroughly investigated. The phase composition and microstructure of the optimized proppants were analyzed.
Experimental
Materials preparation
Ceramic proppants were prepared by pelleting, drying, screening, and sintering with low-grade bauxite and feldspar as raw materials, both of which were produced from the Yangquan Changqing Company in Shanxi Province, China. The average size of the raw materials was 0.05 mm (300 mesh), and the chemical compositions are presented in Table 1. Firstly, bauxite and feldspar were respectively milled for different time (0, 2, 4, 6 and 8 h) by an omnidirectional planetary ball mill (QXQM-6, Changsha Tianchuang Powder Technology Co. Ltd, China). Then the mixture of 85 wt% bauxite and 15 wt% feldspar was moved to a strong mixing machine (R02, Eirich Co. Ltd, Germany) to form spherical particles with the appropriate amount of water. Secondly, the spherical particles were sieved to remain the spheres with 0.5–0.9 mm in size and dried at 90°C for 6 h. Thirdly, the dried spheres were sintered in a multifunction sintering furnace (KBF1700, Nanjing Boyuntong Instrument Technology Co. Ltd, China) in air atmosphere at a temperature range of 1220–1300°C for 2 h with a heating rate of 5°C min−1 and a furnace cooling. Finally, the cooled proppants were passed through a set of sieves of 20/40 meshes.
Chemical compositions of the raw materials (wt%).

Al2O3: aluminium oxide; SiO2: silicon dioxide; Fe2O3: ferric oxide; TiO2: titanium dioxide; CaO: calcium oxide; MgO: magnesium oxide; K2O: potassium oxide; Na2O: sodium oxide; LOI: limiting oxygen index.
Characterization
The particle size distribution of the raw materials milled was determined by laser particle size analyzer (LS 13320; Beckman Coulter, Brea, California, USA). The phase compositions of the proppants were characterized by powder X-ray diffraction (XRD, X’Pert PRO; Philips Co. Ltd, Holland) utilizing Ni-filtered copper K α radiation with a scanning speed of 0.02° step−1. The microstructure of the samples was examined by field-emission scanning electron microscope (FESEM, S-4800; Hitachi, Japan). Bulk density and apparent density of the proppants were measured by a calibrated density bottle. Breakage ratio (under 52 MPa closed pressure) was calculated by the formula: η = ω c/ω 0 × 100%, where ω c referred to the weight of the crushed proppants after passing through a 20/40 mesh sieve after testing and ω 0 was the weight of the proppants before crushing.
The test of acid solubility was determined according to the standards. 21 First, 12-3 hydrochloric–hydrofluoric acid solution (i.e. 12 wt% HCl + 3 wt% HF) was prepared. Second, 5 g of proppants was dried to a constant weight at 105°C and cooled in a dryer with a mass recorded as m s (g). And then the proppants were placed in a large beaker with a 100 ml acid solution, covered, and placed in a bath at 66°C for 30 min without stirring. Third, the funnel and filter paper were dried to a constant mass at 105°C, marked as m f (g). Fourth, vacuum filtering and cleaning of the samples with distilled water until the pH value was neutral was carried out. The filter and the remaining samples were dried to a constant mass at 105°C, marked as m fs (g). The acid solubility was calculated by the following formula: S = (m s + m f − m fs)/m s × 100%, where S was acid solubility, expressed as a percentage.
Orthogonal experimental design
Orthogonal experiment method is a kind of designing method to study many factors and levels. 22 Orthogonal experimental design has been demonstrated to be a highly efficient, fast, and economical experimental design method in evaluating the effects of various factors on performance. 23 It conducts tests by selecting a suitable number of representative test cases from many test data, which have evenly dispersed, neat comparable characteristics. 24 The design of the orthogonal experiment is based on the orthogonal table. The factors in orthogonal test are the parameters that influence the performance of ceramic proppants, while the level in orthogonal test means the maximum number of the values that can be taken on by any single factor. By understanding the importance of the various factors and the interactions among them, the major advantage of the method was to reduce the number of tests required but still provide the best level of combination of factors. 23 Consequently, orthogonal experiment was designed to optimize the preparation of low-density and high-strength proppants involving multivariate and multilevel factors.
This article mainly studied the effects of the following three factors: (a) milling time of bauxite, (b) milling time of feldspar, and (c) sintering temperature on the bulk density, apparent density, breakage ratio, and acid solubility of the proppants. Each factor had five levels as presented in Table 2. The standard orthogonal experimental design table L25 (5 3 ) was adopted and a total of 25 samples were tested. The orthogonal experimental design table and tested results are displayed in Table 3.
The orthogonal experimental factors and levels.

The orthogonal experimental design table.
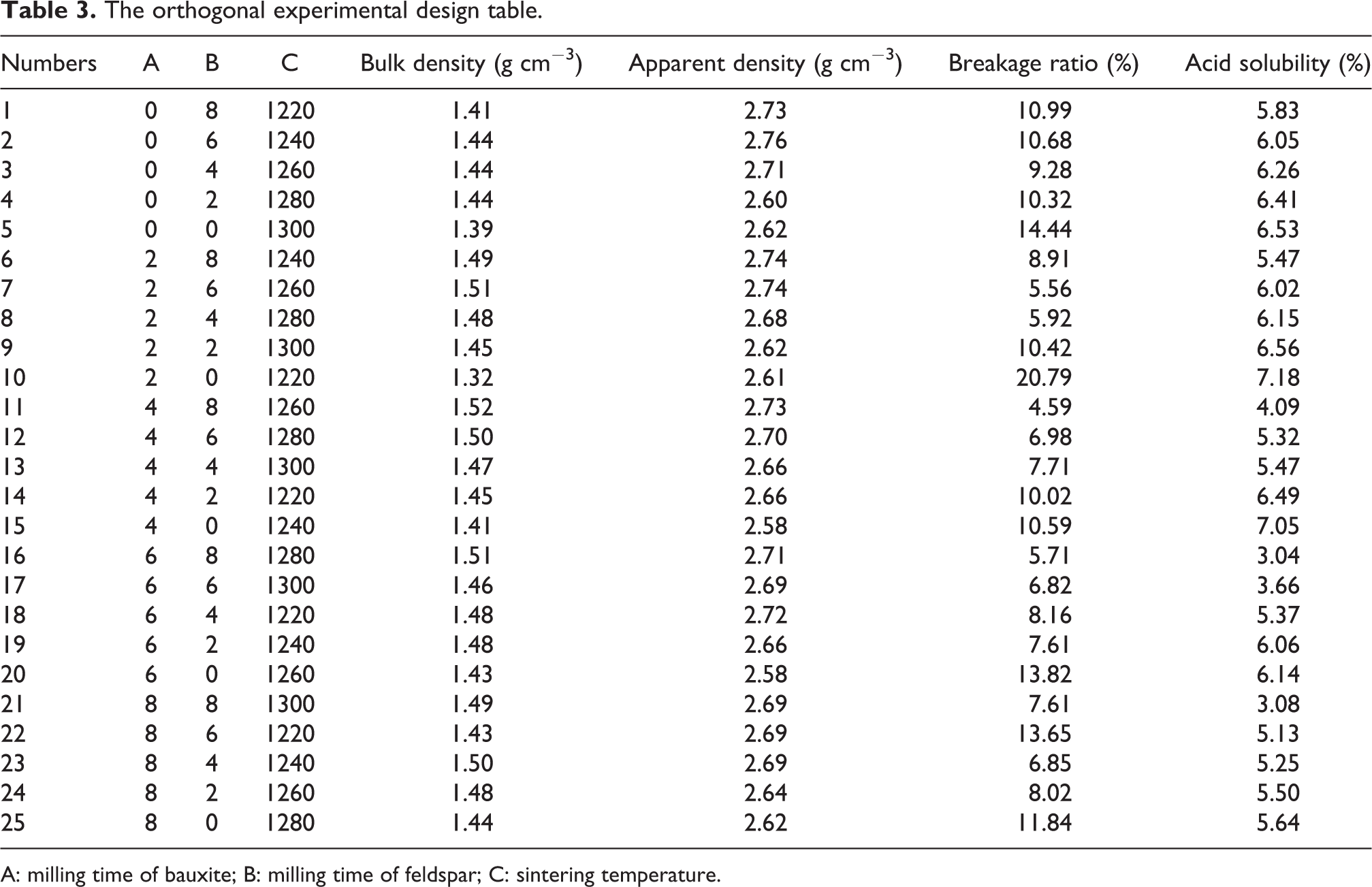
A: milling time of bauxite; B: milling time of feldspar; C: sintering temperature.
Results and discussion
Figure 1 shows the distribution of particle size of the raw materials milled for different time. From Figure 1, the particle size of bauxite and feldspar without milling is mainly concentrated in 0–60 µm. In addition, with the prolongation of ball milling time, the peak values of particle size shift to the left, indicating that the particle size becomes smaller. The average particle size of bauxite and feldspar without ball milling is 18.95 µm and 21.23 µm, respectively, while that of bauxite and feldspar milled for 2 h is obviously reduced to 13.66 µm and 14.79 µm, respectively. By prolongating the milling time of bauxite, the peaks of particle diameter of about 7.5 µm clearly increase, while the peaks of particle diameter of about 30 µm greatly decrease. For feldspar, the peaks of particle diameter of about 6.3 µm gradually increase, while the peaks of particle diameter of about 25 µm sharply decrease with the prolongation of ball milling time. This also shows that ball milling can make the particle size smaller. Then, ball milling the bauxite and feldspar is continued for 6 and 8 h, and the average particle size is 6.33 µm and 7.24 µm and 5.38 µm and 6.82 µm, respectively.

Particle size distribution of (a) bauxite and (b) feldspar.
In the method, 25 samples of testing were performed, with three repetitions for each test. The test results are presented in Table 3. It can be seen that the bulk density and apparent density of all samples meet the requirements of low-density proppants. The acid solubility also basically meets the standard requirements (≤7%), except for samples 10 and 15. However, the breakage ratio of nearly half of the samples do not meet the standard requirements (≤9%), and the breakage ratio is the key to the proppant performance index. Therefore, a further orthogonal analysis was required.
Range analysis is a statistical method to determine the factors’ sensitivity to the experimental result according to the orthogonal experiment. 25 Range analysis results of bulk density, apparent density, breakage ratio, and acid solubility of the orthogonal experiment are displayed in Table 4. In Table 4, Ki is defined as the sum of the values of according five i levels in the specific factor j (j = A, B, C), and ki refers to the average value of the experimental results which contain the factor j with i level. Range (R) is defined as the distance between the extreme values of the data. The greater the range is, the more sensitive the factor is. 24 In Table 4, the degree of effect of the factors on bulk density and breakage ratio is the same, B > C > A, while that on apparent density and acid solubility is the same, B > A > C. Indicating that factor B plays a significant role for the performance of the proppants, while factor A has less influence on the crush resistance and factor C has less influence on the acid solubility. This also proves that milling additive (feldspar) can effectively improve the performance of proppants. In addition, according to the value of range R, the primary and secondary influencing factors can be seen. The larger the R value is, the more significant the effect is. 23 In Table 4, the R values corresponding to factors A, B, and C of breakage ratio are the largest, which are 3.164, 6.734, and 4.568, respectively. This is consistent with the previous analysis (Table 3), and the breakage ratio is the key to optimization. The optimal level combination can select a better level of the average index according to the value of k. 26 The lower the breakage ratio of proppants is, the stronger the crushing resistance is. Based on the k value (Table 4), the optimal combination of the proppants is A3B1C4.
The range analysis of bulk density, apparent density, breakage ratio, and acid solubility.
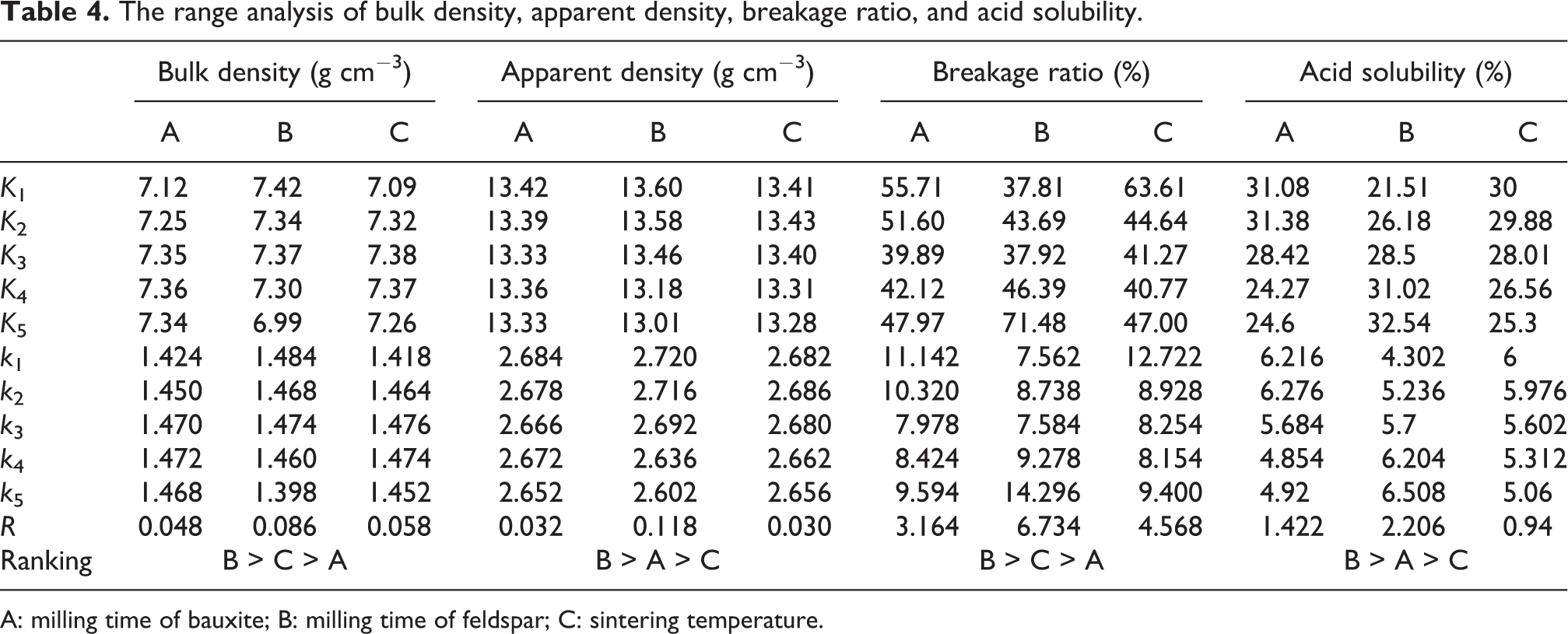
A: milling time of bauxite; B: milling time of feldspar; C: sintering temperature.
The proppants with the optimal combination were prepared by 85 wt% of bauxite milled for 4 h and 15 wt% of feldspar milled for 8 h at 1280°C. The bulk density and apparent density are 1.48 g cm−3 and 2.7 g cm−3 respectively, the breakage ratio under 52 MPa closed pressure is 4.07%, and the acid solubility is 2.15%. According to the standard requirements, 21 the bulk density of proppants is not greater than 1.65 g cm−3, the apparent density is not greater than 3.0 g cm−3, the breakage ratio under 52 MPa closed pressure should be not greater than 9%, and the acid solubility should be not greater than 7%. Obviously, the optimized proppants meet the requirements.
XRD pattern and SEM images of the proppants with the optimal combination are shown in Figures 2 and 3. From the pattern (Figure 2), the main reflection can be indexed to corundum (Al2O3) (JCPDS: 74-1081), and secondary reflection can be indexed to mullite (Al4.59Si1.41O9.7) (JCPDS: 79-1455). The diffraction peaks of corundum are strong and sharp, indicating the formed corundum has better crystallinity. With the addition of corundum and mullite, some reflection is detected, attributing to albite. This also shows that feldspar remains in the preparation process.

XRD pattern of the proppants with the optimal combination. XRD: X-ray diffraction.

SEM images of the proppants with the optimal combination: (a) low magnification and (b) high magnification. SEM: scanning electron microscope.
Figure 3(a) displays low-magnification SEM image of the surface of the proppants. In Figure 3(a), the proppants present a fully glazed surface, as well as some irregular pores. Combined with the literature, 27 the glazed surface is mainly related to the addition of feldspar. Feldspar with low melting point easily forms glassy phase at low temperature and makes the proppants glazed compacts owing to its high viscosity. Figure 3(b) presents high-magnification SEM image of the proppants and shows that corundum is in the lamellar or granular form, while mullite is in the form of whisker. Mullite whiskers are interspersed in the corundum grains and fill the gaps between the grains, which can improve the properties of the proppants.
Conclusions
In this study, an orthogonal experiment was designed with the help of three-factor, five-level orthogonal table to optimize the preparation of low-density and high-strength proppants. The optimal combination was displayed by the orthogonal range analysis. The factor of milling time of feldspar has the greatest influence on the performance of proppants. The optimal combination is A3B1C4, meaning bauxite milled for 4 h, feldspar milled for 8 h, and sintering temperature of 1280°C. Bulk density is 1.48 g cm−3, breakage ratio under 52 MPa closed pressure is 4.07%, and acid solubility is 2.15% after the proppants are prepared by the optimal combination. The study provides a new and feasible way to improve the performance and preparation cost of proppants in the future.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Shanxi Province Science Foundation in China (201901D111036) and the Key Research and Development (R&D) Projects of Shanxi Province (201803D31029) and (201903D121101) from China.
