Abstract
Objective:
The shear bond strength (SBS) of composite resin cement and color change to enamel bleached with two different concentrations of hydrogen peroxide (HP) bleaching agents in combination with two different laser applications was evaluated.
Materials and Methods:
The Er: YAG laser (λ = 2940 nm) and diode laser (λ = 940 nm) were used with the bleaching agent consisting of 40% HP opalescence (Opalescence Xtra Boost, Ulgen, South Jordan, UT, USA) and 35% HP whiteness (Whiteness HP Blue, FGM Dental Products, Joinville, SC, Brazil). Seventy-two human extracted teeth were randomly divided into two groups: For the 40% HP treatment, 36 specimens were divided into three subgroups: (1) HP agent with no laser treatment, (2) HP agent treated with Er: YAG laser, and (3) HP agent treated with diode laser. The same protocol was performed for the 35% HP. The color change was analyzed using the Commission Internationale de l’ Eclairage (CIE) L*a*b* system and surface roughness was analyzed by an atomic force microscope. The specimens were bonded with resin cement and a shear bond test was performed at 0.5 mm/min. The failure surfaces were evaluated using scanning electron microscope analysis.
Results:
ΔE value of the 40% HP + Er: YAG group was significantly higher than the other groups while diode groups showed the lower SBS values than the control groups (p < 0.05).
Conclusions:
The Er: YAG laser with 40% HP may be effective for the brighter teeth and the use of Er: YAG laser produced higher adhesion between enamel and resin cement than diode laser.
Introduction
During recent years, vital tooth bleaching has gained interest for treatment discolored teeth in esthetic dentistry. 1 There are two types of methods that are used for this application: in-office bleaching and at-home bleaching. Concentrations of carbamide peroxide ranging from 10% to 20% are the most commonly used tooth bleaching technique for at-home bleaching. As an efficient technique, in-office bleaching has been used to achieve immediate whitening results more than accomplished at-home bleaching. 2 The mechanism of the in-office bleaching process is based on hydrogen peroxide (HP) with high concentrations. 3,4 HP is a very unsteady molecule and readily decomposed; as a result, oxygen-free radicals are released. These free radicals penetrate directly through the organic matrix of the tooth surface and interact with organic colored molecules owing to redox reactions. 5 –7 Bleaching treatment is mostly preferred before adhesive restorations are applied. 8 Several studies have shown that HP-based bleaching agents adversely affect the bond strength of resin to enamel and dentin when restorations are applied directly after bleaching treatment. 9,10 It has been reported that higher concentrations of peroxide lead to changes on surface morphology involved in demineralization of hydroxyapatite, which may continue for 1 day to 3 weeks after bleaching. 11
There are various sources of irradiation used to increase the effectiveness of in-office bleaching. 12,13 To accelerate the activation of the bleaching agent, lasers can be used, such as Argon (488/514 nm), CO2 (10.6 µm), Er: YAG (2940 nm), diode (810–980 nm), and Nd: YAG (1064 nm). 6,14 The bleaching agent absorbs laser energy and this results in accelerated oxidation–reduction reaction of the peroxide transfer. 15 The Er: YAG and diode lasers are most commonly used as laser-based photoactivation technique. 16 Er: YAG laser has a high attractiveness for collagen and hydroxyapatite including water in the dental hard structures due to its high absorption in water, causing to reduce thermal effects on surrounding tissues. 17 Diode laser wavelengths are highly absorbed by pigmented tissue and 980-nm wavelength diode laser is the parameter with the highest water absorption. 18
It is known that their use of which result in morphological changes in enamel and dentin, such as the formation of rough surfaces, craters, and cracks. 8,19 –22 When laser energy is used for bleaching, it can neutralize the effect of residual free radicals, which result in improving the bond strength of resin-based materials to enamel and dentin. Therefore, the aim of this study was to investigate the effect of Er: YAG and diode laser on bond strength between resin cement and bleached enamel. The first hypothesis was that different concentration of in-office bleaching agents with Er: YAG and diode laser would affect color change on the enamel. The second hypothesis was that laser treatments would not influence the bond strength values between resin cement and bleached enamel.
Materials and methods
Ethical approval was obtained from Selcuk University for this study (meeting number 2018/3; March 22, 2018); 72 human extracted maxillary central incisors of similar size were used without any caries or restorations. The teeth were immersed in 0.2% thymol solution at 4°C immediately after extraction. All the specimens were evaluated using a stereomicroscope (Olympus, SZ-PT, Japan) at 50× magnification to detect the dentin exposure. A spectrophotometer (VITA Easyshade, Germany) was used to record color variables L*, a*, and b* in the CIE L*a*b*system. The teeth were rinsed with water, dried, and then was measured the color of each specimen using a digital spectrophotometer (baseline measurement
Two different laser systems were used in the study (Table 1). The specimens were allocated to two groups (n = 36), according to which bleaching agent was applied (Table 2).
Laser parameters.

Distributions of the experimental groups.

HP: hydrogen peroxide.
Each group was allocated to three subgroups (n = 12), according to the activation with two different lasers or no activation. Subgroup C: Bleaching gel was applied on the enamel surface of the tooth without photoactivation. Subgroup D: Bleaching agent was applied and for photoactivation, the infrared diode laser (BIOLASE Technology, Inc., 4 Cromwell, Irvine, California, USA) was used with the output power of 7 W, 2.8 cm2 spot size, continuous mode, and scanning the surface for 20 s using whitening handpiece. Total energy delivered: 50 J/cm2. Subgroup E: Bleaching gel was applied and for photoactivation, the Er: YAG laser (Lightwalker AT; Fotona, Ljubljana, Slovenia) was used with an energy of 40 mJ, frequency of 10 Hz, output power of 0.4 W, pulse duration of 1 µs, 0.29 cm2 spot size in noncontact mode, and scanning the surface for 20 s using handpiece (HP R17, Fotona). Total energy delivered: 27.58 J/cm2.
The bleaching agent including 40% HP (Opalescence Xtra Boost Ultradent) and 35% HP (Whiteness HP Blue, FGM Dental Products) was applied to enamel surfaces. The bleaching procedure was repeated three times for 10 min made at each session with 1-min interval between sessions. For laser-treated samples, photoactivation was performed for 20 s after the bleaching agent was applied. Then, the surface of the specimen was washed with deionized water. After the final shade evaluation, one specimen from each group was selected for the evaluation of surface roughness with an atomic force microscope (AFM). For each sample, micrographs of different regions were taken using an AFM (NTEGRA Solaris NTMDT, Russia) measuring 25 × 25 µm2.
The crowns were sectioned along the cementoenamel junction using a low-speed diamond saw to separate the crowns and roots. The buccal enamel surfaces of the crowns were then embedded in autopolymerizing acyrlic resin cylinders (Meliodent, Heracus/Kulzer, Hanau, Germany). Then, a self-adhesive resin cement (Panavia SA, Kuraray Noritake Dental, Okayama, Japan) was applied to the bonding surface using a teflon matrix (internal diameter = 3 mm, height = 4 mm) and light curing was performed on four sides for 20 s each side. The specimens were stored in deionized water at 37°C for 24 h prior to aging and shear bond strength (SBS) test. The SBS was measured using a universal testing machine (TSTM 02500, Elisa Ltd, Istanbul, Turkey) at a crosshead speed of 0.5 mm/min until bonding failure occurs. SBS values (MPa) were calculated as the ratio of the fracture load and bonding area. One specimen for each group was selected and then morphological alterations in the enamel of the failed buccal surfaces were investigated by scanning electron microscope (SEM; Jeol, 6400 JSM, Tokyo, Japan). Photomicrographs were taken at 1000× magnifications of representative regions.
Statistical analysis
Data were analyzed with two-way analysis of variance (ANOVA) and one-way ANOVA, followed by multiple comparisons using Tamhane’s T2 test for ΔE values (p < 0.05). The data of shear bond test were analyzed with ANOVA followed by Tukey’s test (p < 0.05).
Results
Table 3 presents the mean ΔE values with standard deviations of all groups. There was a statistically significant difference among the groups (p < 0.05). The Opalescence-Er:YAG (OE) group had significantly higher ΔE values than other groups (p < 0.05). There were no statistically significant differences between the remaining groups (p > 0.05). Color change was higher in all groups when bleaching agent was activated by a laser source according to with no-photoactivated groups. The morphological alterations of enamel surface for six groups were obtained by AFM and SEM. The all-laser-irradiated groups with obvious morphological changes showed after treatment and it was rather irregular and rough; however, control groups were rather smooth (Figure 1(a) to (f)). In control groups of smooth enamel surface with some debris, no morphological differences were found in relation to the bleaching treatment. In diode groups, a characteristic fish scale appearance of the enamel with some debris is apparent, mild alterations on surface smoothness and slightly increased porosity occurred and debris on the surface. Er: YAG groups showed slight alterations, microporous surface, and intermittent depressions of various depths (Figure 2(a) to (f)).
ΔE values and standard deviations of groups.
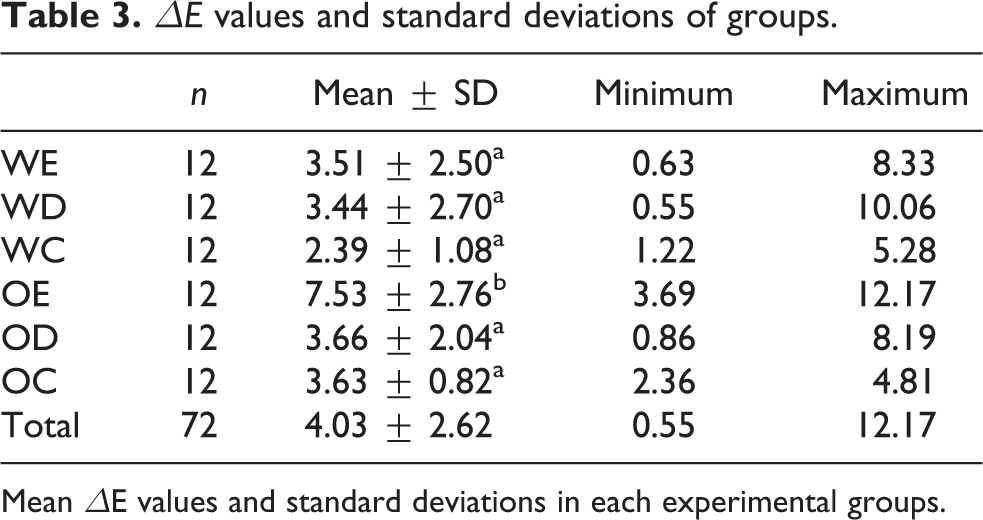
Mean ΔE values and standard deviations in each experimental groups.

Observations of the AFM images of an enamel specimen on bleached enamel with (a)–(c) 40% HP and (d)–(f) 35% HP bleaching gel for without laser activation, diode laser, and Er: YAG laser. HP: hydrogen peroxide; AFM: atomic force microscope.

SEM micrographs of enamel surfaces, specimens were treated with (a)–(c) 40% HP and (d)–(f) 35% HP for control, diode laser, and Er: YAG laser. HP: hydrogen peroxide; SEM: scanning electron microscope.
Mean SBS values and their standard deviations are illustrated in Table 4. The laser-treated groups produced a decrease in the SBS of enamel compared to the control groups. Diode laser-treated groups showed lower SBS values compared to the control groups (p < 0.05). The Whiteness-Control (WC) group showed highest SBS value (28.29 ± 3.86 MPa), followed by the Opalescence-Control (OC) group (26.7 ± 3.38 MPa). There was no significant difference between the two groups (p > 0.05).
Shear bond strength values of all groups (MPa).

Mean shear bond strengths (Mpa) and standard deviations in each experimental groups.
Discussion
This study evaluated the spectrophotometric, morphological analysis and bond strength of the bleaching results on enamel between Er: YAG and diode lasers. Bleaching agents containing HP have been used in the in-office bleaching clinically to lighten teeth. 24 The whitening process of the in-office bleaching involves in using high concentration HP that readily releases oxygen-free radicals and perhydroxyl anions that penetrate through dental structures, leading to bleaching of the colored pigments impregnated in the enamel. 25,26 To accelerate catalyzing HP decomposition, the use of energy sources, such as LED, halogen, heat, and lasers, is widely preferred.
In the literature, there is no consensus about which light sources are useful in improving color change. Wetter et al. 26 compared no activation group, diode laser, and LED activation groups with bleaching gel and they found that diode laser showed a significantly better result than LED and bleaching gel only. 27 On the other hand, it has been reported that there is no significant result between LED and diode laser photoactivation in bleaching efficacy. 28
In this study, a mean total color difference value greater than 3.3 was obtained for each group except without laser activation 35% HP. It is well-known that the color change values less than 3.3 can be considered clinically insignificant. 29 Specimens in the Er: YAG laser-activated groups showed the better mean total color difference value but statistically significant difference was obtained when using the Er: YAG laser with only 40% HP gel. Therefore, the first hypothesis of this work was accepted. This result could be explained by the higher HP concentration of the bleaching agent. The previous study has shown a similar result comparing photoactivation with Er: YAG and KTP laser and they found that the Er: YAG group with 35% HP gel had a significant difference in color change, although there was no significant difference in color change when used 6% HP gel. 23 Our result indicated that better color change values were obtained for the diode laser group when compared without photoactivation, although there was no statistical difference in both the groups.
Near-infrared lasers, such as diode lasers, are poorly absorbed by dental hard substance. 30 Diode laser wavelength (940 nm) is well absorbed in pigmented tissue, such as blood, while Er: YAG laser wavelength (2940 nm) is well absorbed in water. 31 It can be recommended as a suitable laser for the use of an aqueous bleaching gel because of its high absorption in water. Gutknecht et al. stated that the temperature increase during the bleaching of the laser occurs only in the upper layer within the gel, but the diode laser transfers energy from the gel to the tooth. 32 Overall, we believe that the Er: YAG laser wavelength is completely absorbed in the gel only, while diode laser wavelength is comparatively weaker absorbed in the gel.
In this study, it was purposed to compare the ultrastructural changes in enamel caused by bleaching agents of different concentrations of activation methods with SEM images. In the literature, contradictory results have been reported for the effect of bleaching on the structure of the enamel surface. Previous studies reported that HP-based bleaching agents adversely affect the bond strength of resin to enamel after immediate bleaching procedure. 9,33 The change of organic matrix of enamel after bleaching decreases the bond strength with resin-based materials. 34,35 In addition, it is known that the use of lasers may affect bonding by causing changes in dental hard tissue morphology after bleaching. In the present study, bond strength on enamel after postbleaching with two agents and two different lasers was compared. According to shear test, the lowest bond strength values were observed in the diode laser-irradiated groups. This result is probably due to poor penetration of the resin to the diode-irradiated enamel surface. Mirhashemi et al. compared the bond strength of a resin-based composite adhesive to enamel after bleaching with or without diode laser treatment and they found that diode laser reduced the bond strength of the resin onto bleached enamel surfaces. 36 This result was consistent with our findings. When the surface morphology of the diode laser group is evaluated that were observed with some debris, increased porosity, and enamel irregularities than the other groups. This result was consistent with the previous study, which found surface modification after the bleaching was detected by SEM. 37
Several morphological alterations could be examined on enamel surface after in-office bleaching, such as increased porosity, characterized erosion, and pore with loss of prismatic components. 38 There was no difference in bond strength between the control groups and highest bond strength values were obtained in this study. Therefore, the second hypothesis was rejected. The uniformity of the aprismatic surface layer can be observed by SEM images, although the samples of the control group bleaching agents cause slight erosive effects on the enamel and the surface is not completely smooth. Previous study reported that bleaching agents can cause structural changes on the enamel surface by SEM analysis. 39 The dissociation of the organic content of enamel also causes erosive effect between the enamel prisms. 22 Nevertheless, Mahringer et al. stated that the main morphological change resulting from the bleaching process occurred during the first hour of bleaching and there was no significant change after 6 h of postbleaching. 40 This reduction could be related to high concentration of residual oxygen released from the bleaching agents that inhibit resin polymerization and infiltration into the bleached enamel. This result was possibly related to remineralization of enamel. Coceska et al. demonstrated that during tooth bleaching with HP, significant loss of Na and Mg occurred; when HP was activated with a laser, significant loss of Ca and P occurred in the enamel surface. 41 In addition, they stated that these structural changes, which indicate loss of mineralized tissue, can be reversed with remineralizing toothpaste.
Several studies have documented that the effect of Er: YAG laser on the SBS of resin to enamel and result of these studies commonly indicated that Er: YAG laser treatment adversely affected the bonding to resin materials. 42 –44 Regarding the result of the shear test, the all Er: YAG laser-irradiated groups exhibited lower bond strength than the control groups in this study. Slight surface alteration with a porous and irregular structure after the Er: YAG laser irradiation process was detected with SEM. The studies have been reported that Er:YAG laser irradiation cause microexplosions, macroscopic and microscopic roughness during enamel ablation. These changes obstructs the penetration of the resin to the enamel surface by blocking the microspaces on the enamel surface. 45,46
As limitations of this study, the effect of thermal cycling on color change and bond strength was not evaluated. Another limitation is that only two types of lasers were used for photoactivation, and one type of resin material was used to evaluate the bond strength. Further studies are needed to determine whether the laser treatment and wavelength are more effective to improve the bleaching result. In addition, it is necessary to investigate the effect of different types of resin materials in increasing the bond strength with enamel after bleaching, the results of in vitro tests may provide a useful prediction for evaluation of clinical conditions.
Conclusions
Within the limits of this study, it can be concluded that the use of a diode laser is not necessary in improving bleaching results and the use of diode laser in bleaching resulted in a decreased bond strength. The Er: YAG laser-activated 40% HP gel was found to be more effective than the diode laser in bleaching result.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

