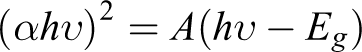Abstract
The exploration of highly efficient visible-light-driven photocatalysts for dye degradation has received great concerns in wastewater treatment. Here, molybdenum disulfide (MoS2) and cadmium sulfide–manganese (CdS-Mn) were sequentially assembled onto titanium dioxide (TiO2) nanotube by a simple hydrothermal method coupled with successive ionic layer adsorption and reaction. A zinc sulfide (ZnS) layer was introduced as a potential barrier for performance improvement; the resultant material exhibits prominent visible-light-induced photocatalytic activity in degrading methyl orange (MO) and 9-anthracenecarboxylic acids, which is 3.16-fold, 2.00-fold, and 1.69-fold or 2.86-fold, 1.56-fold, and 1.47-fold of TiO2, MoS2/TiO2, and CdS-Mn/TiO2 systems, respectively. Furthermore, the synthesized ZnS/CdS-Mn/MoS2/TiO2 composite also possesses a high hydrogen production rate of 408.27 mmol/cm2/h out of water under visible light illumination, which is about 30.08 times greater than that of pure TiO2 and 5.18-fold and 2.52-fold of MoS2/TiO2 and CdS-Mn/TiO2, respectively. The enhanced photocatalyst performances are mainly attributed to the synergetic effects of CdS-Mn, MoS2, and TiO2, forming a Z-scheme system in the CdS-Mn/MoS2/TiO2 electrode, which not only accelerates the interfacial charge transfer efficiency but also preserves the strong redox ability of the photogenerated electrons and holes. In addition, the prepared photoelectrode is highly stable and completely recyclable over hydrogen evolution reaction and organic degradation.
Introduction
With ever-increasing severe fossil resource depletion and environmental pollution, it is becoming urgent need to develop sustainable clean energy resources to address energy and environmental issues. Since the discovery 1 of photoelectrochemical splitting of water on titanium dioxide (TiO2) electrodes, semiconductor-based photoelectrolysis of water to hydrogen and oxygen has attracted more and more public attention. 2 –7 Solar photocatalysis technology, as an eco-friendly and economic technology, can split water to produce H2 and degrade organic pollutants, thereby remediating environment and producing green chemical energy (e.g. H2 gas) at the same time. 8 –10 Photocatalysis employing TiO2-based materials has experienced tremendous research activity in H2 generation, organic pollutants degradation, CO2 reduction, and water and air remediation by exploiting the energy from natural sunlight or artificial illumination. 11 –17 This is due to its low cost, nontoxicity, superb stability, and easy availability. 18,19 But considering that TiO2 is only ultraviolet (UV)-excited due to its broad bandgap (3.0–3.2 eV), its practical application and the efficient utilization of solar energy have been constrained. 20 Therefore, great efforts have been dedicated to extending its energy spectrum from the UV (approximately 5%), that is only a small portion of the sunlight spectrum, to the visible spectral region (approximately 45%). 21 It is highly imperative to explore novel and efficient visible-light-driven photocatalysts. 22 Li et al. 23 illustrated that Pt at TiO2 could accomplish high efficiency of H2 production with simultaneous photodegradation of eosine Y and methylene blue. Zhou et al. 24 prepared TiO2/molybdenum disulfide (MoS2) that exhibits high H2 production rate and shows high performance in photocatalytic degradation of organic dyes. Zhang et al. 25 synthesized TiO2/zirconium dioxide for both photocatalytic degradation and H2 evolution from water splitting. Zong et al. 26 proved that cadmium sulfide (CdS)/MoS2 possesses high visible-light-driven H2 evolution rate that is 36 times of CdS. Santra and Kamat 27 fabricated manganese (Mn)-CdS/TiO2 that exhibits significant enhancement in power conversion efficiencies as compared to undoped CdS. Zhang et al. 28 reported that MoS2/ZnIn2S4/reduced graphene oxide (RGO) could also realize H2 production coupling with pollutant degradation [Rhodamine B (RhB), Eosin Y (EY), Fulvic acid (FA), Methylene Blue (MB), and P-nitrophenol (PNP)].
For photocatalytic water splitting to H2 and organic pollutants degradation, the photocatalyst can be excited by solar energy to generate electron–hole (e−-h+) pairs. Then, the electrons can be captured by H+ to generate H2, and the holes can oxidize organic pollutants. 29,30 Therefore, it is crucial to improve the separation and transportation efficiency of photogenerated carriers for better photocatalytic performance. Nowadays, scholars and researchers have exhibited interest in MoS2 as cocatalysts for photocatalytic application due to its low-cost, earth abundant, variable bandgap, and platinum-like electrochemical activities. 31 –36 In our previous research, 13 Mn-CdS and zinc sulfide (ZnS) quantum dots (QDs) have been proved to be powerful strategy to extend the lifetime of charge carriers for performance improvement. To sum up, combining TiO2 with MoS2, CdS-Mn, and ZnS semiconductors could largely improve the photocatalytic activity of the co-sensitized electrode, as the energy levels of MoS2 (bandgap of 1.2–1.9 eV), CdS-Mn (approximately 2.4 eV), and TiO2 (3.0–3.2 eV) are well suitable to one another, which benefits the separation of e−-h+ pairs.
In this work, we developed a visible-light-driven Z-scheme CdS-Mn/MoS2/ TiO2 ternary heterojunction to realize the enhanced photocatalytic activity and proposed an appropriate mechanism (Figure 1). The as-prepared sample showed the best photocatalytic performance, two stable organic contaminant methyl orange (MO) and 9-anthracenecarboxylic acids (9-AC) could be removed in very short time intervals, and the H2 production rate on it achieved 408.27 mmol/cm2/h. The MO and 9-AC degradation efficiency and H2 production rate of it were 3.16, 2.86, and 30.08 times over pure TiO2 because of its improved charge separation and transportation. Hence, it provided new insights to the fabrication of low-cost materials as highly efficient cocatalysts in photocatalytic application.

Postulate diagram for both photocatalytic degradation and photocatalytic H2 evolution with ZnS/CdS-Mn/MoS2/TiO2 nanocomposites. ZnS: zinc sulfide; CdS-Mn: cadmium sulfide–manganese; MoS2: molybdenum disulfide; TiO2: titanium dioxide.
Experiment
Materials and methods
Titanium foil (250-mm-thick, 99.8%) was purchased from Aldrich (Milwaukee, Wisconsin, USA). Sodium fluoride, sodium hydrogen sulfate, sodium sulfite, sodium sulfide (Na2S), cadmium nitrate (Cd(NO3)2), zinc nitrate (Zn(NO3)2), manganese(II) acetate (Mn(CH3COO)2), MO, 9-AC, sodium molybdate (Na2MoO4·2H2O), and thioacetamide (C2H5NS) of analytical grade were purchased from commercial sources and used as received. Double-distilled water was used throughout this experiment. All experiments, excluding those with special annotations, are conducted at room temperature.
The formation process of CdS-Mn/MoS2/TiO2 heterostructures was described as follows. 13 First, 30-mg Na2MoO4·2H2O and 60-mg C2H5NS were dissolved in 20-mL deionized water to form a transparent solution. Then, the prepared TiO2 nanotube arrays (NTAs) by anodic oxidation process was directly immersed into the above solution. The solution was transferred to a Teflon-lined stainless steel autoclave and subsequently heated in an electric oven at 200°C for 24 h. Having been washed several times with high purity water, the obtained MoS2/TiO2 was dried at 50°C for 12 h. The synthetic MoS2/TiO2 was then successively immersed into two different solutions for 1 min each. First, in ethanol solution: 0.05 and 0.0375 mol/L for Cd(NO3)2 and Mn(CH3COO)2, respectively, as cation source. This allowed co-adsorption of Mn2+ and Cd2+ ions. And then in 0.05 mol/L Na2S in methanol/water (7:3/v:v). Following each immersion, the composite was rinsed for 2 min or longer with pure ethanol and methanol, respectively, to remove excess precursors and dried before the next dipping. This immersion cycle was repeated five times for the CdS-Mn layer. 27 The sensitized photoelectrode was designated as CdS-Mn/MoS2/TiO2 and finally capped with two-cycle ZnS layer by the similar procedure as described above except cation source is 0.1 mol/L Zn(NO3)2 in ethanol. The resulting ZnS/CdS-Mn/MoS2/TiO2 heterostructure was heated under nitrogen atmosphere at 300°C for 2 h.
Photoelectrochemical measurements and structural characterization
A standardized configuration with a three-electrode mechanism was used, and the surface photocurrent (SPC) and electrochemical impedance spectra (EIS) were obtained at room temperature with an electrochemical analyzer (CHI-660C, Shanghai Chenhua Instrument Co. Ltd, China). The samples were applied as the functional electrode, one platinum foil as the counter electrode, and a saturated calomel electrode as the reference electrode. 37 –39 The incident light from a 300-W xenon arc lamp was filtered to match the AM 1.5G spectrum with an intensity of 98 mW/cm2 as measured with a radiometer (OPHIR, Littleton, Colorado, USA). Scanning electron microscope (SEM; JSM-6700F (Japanese electronics)) and high-resolution transmission electron microscopy (HRTEM; JEM 2100, JEOL (Japanese electronics)) were used to characterize the morphology and dimension of the products. Energy dispersive X-ray (EDX) spectrometer fitted to electron microscope was used for elemental analysis. Light absorption properties were examined using ultraviolet–visible (UV-vis) diffuse reflectance spectra (UV-2450, Shimadzu, Hangzhou Ruiz Technology Co., Ltd.) within a wavelength range of 200–800 nm. Photoluminescence (PL) spectra were recorded at room temperature using Hitachi (Tianmei (China) Scientific Instruments Co., Ltd.) F-4600 fluorescence spectrophotometer at an excitation wavelength of 270 nm. X-ray diffraction (XRD) patterns were gathered for identification of crystal structures of the samples with an X-ray diffractometer (XRD, M21X, MAC Science Ltd., Japan) employing Cu Ka radiation (ë = 1.54060 Å).
Photocatalytic degradation of MO and 9-AC
The photocatalytic degradation experiments were carried out in a cubic quartz reactor containing MO or 9-AC aqueous solution (0.1 L, 20 mg/L) with the as-fabricated nanocomposite catalyst under constant stirring. An adsorption–desorption equilibrium must be established in darkness for 0.5 h prior to irradiation. A 300 W of xenon arc lamp (98 mW/cm2, 15 cm away from the photocatalytic reactor) was used to illuminate the photoelectrode at room temperature. The concentration change during the degradation procedure was monitored by a UV-vis spectrophotometer (CARY 300 Conc, Agilent technology co., Ltd.) based on its absorption peak at 465 nm for MO or 253 nm for 9-AC, respectively. After measurement, the test sample of 300 µL taken from the solution was immediately put back to the reaction cell to keep the volume constant. Finally, the degraded amount was measured by plotting C t/C o versus time.
Photocatalytic hydrogen evolution test
The experiments of H2 production were performed in a homemade photocatalytic reactor linked to a closed gas-circulating and evacuation mechanism with the as-prepared photoelectrode in methanol/water (1:9/v:v) reactant solution of 100 mL. The reactant solution was thoroughly degassed under flowing high-purity N2 for 40 min prior to irradiation and magnetically stirred during the entire experiment. The evolved H2 was analyzed automatically every 30 min through a gas chromatographer (Agilent Technologies, 6890 N, Network GC System). For stability test, the H2 production experiments of ZnS/CdS-Mn/MoS2/TiO2 were examined by every 4 h and repeated five times.
Results and discussion
Characterization of the ZnS/CdS-Mn/MoS2/TiO2
The morphologies of the as-prepared samples were investigated by SEM and transmission electron microscopy (TEM). The MoS2 sample can be synthesized through the hydrothermal method. As shown in Figure 2(a), the TiO2 NTAs are covered with two-dimensional (2-D) MoS2 nanosheets which are as thin as cicada’s wings and are distributed mainly on the top surface of the NTAs (marked with arrows in blue and circles in yellow). The 2-D structure of MoS2 can be better observed by TEM (Figure 2(b)). CdS-Mn and ZnS nanoparticles (NPs) were in turn deposited on the MoS2/TiO2 NTAs by successive ionic layer adsorption and reaction (SILAR) approach. NPs shown in Figure 2(c) and (d) homogeneously covered the surface of MoS2/TiO2 NTAs, resulting in minimizing the surface energy. 38 Significantly, the obtained samples still retain the appearance of TiO2 NTAs template and the structural integrity. No obvious blocking of the entrances is observed and the porosity in the structure is more beneficial for the adsorption process. 38

(a) Scanning electron microscopic images of MoS2/TiO2 NTAs (MoS2 marked with circle in yellow), and (b) transmission electron microscopic images of the MoS2. Scanning electron microscopic images of (c) CdS-Mn/MoS2/TiO2 NTAs and (d) ZnS/CdS-Mn/MoS2/TiO2 NTAs. MoS2: molybdenum disulfide; TiO2: titanium dioxide; NTAs: nanotube arrays; CdS-Mn: cadmium sulfide–manganese; ZnS: zinc sulfide.
From the HRTEM image of the composite (Figure 3(a)), distinct lattice fringes of TiO2, MoS2, and CdS can be observed with the fringe spaces ∼0.2407, ∼0.2346, ∼0.2536, and ∼0.2852 nm confirming to (111, 004) plane of TiO2, (002) plane of MoS2, and (200) plane of CdS, respectively, 13 which indicate that MoS2, CdS, and TiO2 coexist in the composites. The EDX patterns of ZnS/CdS-Mn/MoS2/TiO2 NTAs samples confirm the successful attachment of the MoS2, CdS-Mn, and ZnS (Figure 3(b)). The XRD measurements were carried out to elucidate the nanocrystalline material structure as depicted in Figure 3(c), which further indicates the presence of MoS2, TiO2, and CdS. Therefore, it can be demonstrated that composites were obtained successfully.

(a) High-resolution transmission electron microscopic images of ZnS/CdS-Mn/MoS2/TiO2 NTAs. The corresponding (b) EDX analysis and (c) XRD patterns. ZnS: zinc sulfide; CdS-Mn: cadmium sulfide–manganese; MoS2: molybdenum disulfide; TiO2: titanium dioxide; NTAs: nanotube arrays; EDX: energy dispersive X-ray.
Optical absorption, as an important factor for photocatalytic activity, was investigated by an UV-vis spectrophotometer, and the results were listed in Figure 4(a). The pure TiO2 sample exhibits an optical absorption response between 400 nm and 800 nm, while it can be seen that the ZnS/CdS-Mn/MoS2/TiO2 nanocomposite displays the highest intensity among as-prepared materials, suggesting the improved light harvesting ability ascribed to the synergetic contact of ZnS, CdS-Mn, MoS2, and TiO2.
39
The bandgap energy could be measured by a plot of (αhυ)2 versus (hυ) using the equation
21
(a) DRS spectra of sample 0: TiO2, 1: ZnS/TiO2, 2: MoS2/TiO2, 3: CdS-Mn/TiO2, 4: ZnS/CdS-Mn/TiO2, and 5: ZnS/CdS-Mn/MoS2/TiO2 composite. (b) Plots of (αhυ) versus photon energy (hυ) for estimated optical bandgap of TiO2 and ZnS/CdS-Mn/MoS2/TiO2. (c) The corresponding PL spectra. DRS: diffuse reflectance spectra; TiO2: titanium dioxide; ZnS: zinc sulfide; MoS2: molybdenum disulfide; CdS-Mn: cadmium sulfide–manganese; PL: photoluminescence.
where α, h, υ, Eg , and A are absorption coefficient, Planck’s constant, light frequency, bandgap energy, and a constant, respectively. Different materials have different Eg , the smaller the material Eg , the greater light wavelength is required. Meanwhile, the lower energy that is needed, the more easily an electron is transited, and the higher photocatalytic activity is obtained. So, according to the plot of (αhυ)2 versus (hυ) in Figure 4(b), extrapolation of the linear part until its intersection with the hυ axis gives the values of Eg , the Eg of TiO2 (0) is determined to be 3.0 eV which is in accordance with the published reports. 37,40,41 The Eg of the as-prepared ZnS/CdS-Mn/MoS2/TiO2 (5) was found to be 1.72 eV. The Eg of (1–4) is 1.28 eV, 1.62 eV, 1.37 eV, and 1.49 eV, respectively (Table 1).
The bandgap and photocatalytic property of samples.

MO: methyl orange; 9-AC: 9-anthracenecarboxylic acids.
The above findings justified that introduction of semiconductor nanomaterials with narrow bandgap as photosensitizers can broaden the TiO2 absorption spectrum to the visible region. The superior light absorption leads to the generation of superfluous photoinduced e−-h+ pairs, consequently resulting in an enhanced photocatalytic performance. PL emission is an effective way to observe the separation of photogenerated carriers. Generally, PL emission results from the recombination of photogenerated e−-h+ pairs. Therefore, a lower PL intensity indicates a higher e−-h+ pair separation efficiency. 30 From Figure 4(c), the PL intensity of ZnS/CdS-Mn/MoS2/TiO2 decreases obviously compared with other samples, suggesting that the as-prepared nanocomposite can well inhibit the recombination of photogenerated carriers.
Photoelectrochemical measurements
The SPC response of as-synthesized ZnS/CdS-Mn/MoS2/TiO2 enhanced dramatically more so than bare TiO2, and TiO2 shows a faint photocurrent response, consistent with its broad-bandgap characteristics (Figure 5(a)). As displayed in Figure 5(a), the photocurrent generated by the pure TiO2, ZnS/TiO2, MoS2/TiO2, CdS-Mn/TiO2, ZnS/CdS-Mn/TiO2, and ZnS/CdS-Mn/MoS2/TiO2 electrodes are 0.943, 1.318, 1.751, 2.407, 3.251, and 4.152 mA/cm2, respectively. The photocurrent of ZnS/CdS-Mn/MoS2/TiO2 is about 4.40-fold that of TiO2 electrode. The enhancement of visible-light-induced photocurrent for the composites demonstrates more efficient separation and less recombination of photoinduced e−-h+ pairs at its interface. Meanwhile, the electrochemical impedance spectroscopy (EIS) analysis showed a similar result. Figure 5(b) illustrates the EIS changes of the current samples. The diameter of the semicircular of the Nyquist plots symbolizes the surface charge transfer resistance of the sample. Lower resistance and smaller radius are preferred for charge transfer. 41 The diameter of the composite is smallest, which reveals that the composite expresses a more efficient charge separation and electron transfer ability. The results are in accordance with the SPC testing. An equivalent circuit containing a constant phase element, a series resistance Rs and the interface resistance Re of the electrode/electrolyte was presented through software (ZSimpWin) analysis (inset in Figure 5(b)).

(a) Surface photocurrent curves and (b) EIS Nyquist plots of sample 0: TiO2, 1: ZnS/TiO2, 2: MoS2/TiO2, 3: CdS-Mn/TiO2, 4: ZnS/CdS-Mn/TiO2, and 5: ZnS/CdS-Mn/ MoS2/TiO2, respectively. TiO2: titanium dioxide; ZnS: zinc sulfide; MoS2: molybdenum disulfide; CdS-Mn: cadmium sulfide–manganese.
Photocatalytic degradation of MO and 9-AC
MO is a typical azo dye and is known to be carcinogenic and mutagenic. 40 9-AC is one of the derivants of persistent organic pollutants. 42 Thus, complete degradation of MO and 9-AC in wastewater is necessary. In this work, photocatalytic behavior of as-synthesized nanocomposite for MO and 9-AC degradation was investigated, respectively, and the results are shown in Figure 6(a) and (b). An adsorption–desorption equilibrium must be assured prior to irradiation. The change in MO or 9-AC concentration during the degradation process was characterized by its characteristic absorbance at 465 nm or 253 nm, respectively. The characteristic absorbance of MO or 9-AC decreases rapidly as the time prolonged and the solution eventually turned colorless. It only takes about 100 min for ZnS/CdS-Mn/MoS2/TiO2 to degrade MO and about 35 min to completely degrade 9-AC.

(a) and (b) UV-vis determination of photocatalytic degradation of MO and 9-AC using ZnS/CdS-Mn/MoS2/TiO2 NTAs as the catalyst under AM 1.5G illumination. (c) and (d) Visible-light photocatalytic degradation activity of various samples and reference samples for MO and 9-AC, respectively. (e) and (f) corresponding degradation efficiency of different photoelectrodes. UV-vis: ultraviolet–visible; MO: methyl orange; 9-AC: 9-anthracenecarboxylic acids; ZnS: zinc sulfide; CdS-Mn: cadmium sulfide–manganese; MoS2: molybdenum disulfide; TiO2: titanium dioxide; NTAs: nanotube arrays.
In order to compare the performance, pure TiO2, ZnS/TiO2, MoS2/TiO2, CdS-Mn/TiO2, ZnS/CdS-Mn/TiO2, and ZnS/CdS-Mn/ MoS2/TiO2 are explored as photocatalysts under the same experimental conditions. As illustrated in Figure 6(c) and (d), the degradation rate is as follows: pure TiO2 < ZnS/TiO2 < MoS2/TiO2 < CdS-Mn/TiO2 < ZnS/CdS-Mn/TiO2 < ZnS/CdS-Mn/MoS2/TiO2. ZnS/CdS-Mn/MoS2/TiO2 has the best photocatalytic performance among all the six samples, obtaining 98% and 100% degradation efficiencies of MO and 9-AC, respectively. Whereas only 31%, 44%, 49%, 58%, and 66% or 35%, 50%, 64%, 68%, and 74% degradation efficiencies of MO and 9-AC for the other photocatalysts are achieved, respectively (Figure 6(e) and (f)). The direct photolytic degradation without catalyst shows a little degradation efficiency of 17% (MO) and 18% (9-AC), while the removal efficiency with ZnS/CdS-Mn/MoS2/TiO2 NTAs as catalyst in dark is 5% (MO) and 5.2% (9-AC).
Photocatalytic H2 evolution from water splitting
Figure 7 represents the hydrogen evolution from the water under visible light irradiation. The ZnS/CdS-Mn/MoS2/TiO2 catalysts show significantly more enhancement in photoactivity as compared to pure TiO2, ZnS/TiO2, MoS2/TiO2, CdS-Mn/TiO2, and ZnS/CdS-Mn/TiO2. Typically, ZnS/CdS-Mn/MoS2/TiO2 gives an H2 production activity of 408.27 mmol/cm2/h, which is about 30.08-fold (13.57 mmol/cm2/h), 9.20-fold (44.35 mmol/cm2/h), 5.18-fold (78.76 mmol/cm2/h), 2.52-fold (162.00 mmol/cm2/h), and 1.68-fold (241.74 mmol/cm2/h) that of the pure TiO2, ZnS/TiO2, MoS2/TiO2, CdS-Mn/TiO2, and ZnS/CdS-Mn/TiO2 samples, respectively. The stability of the ZnS/CdS-Mn/MoS2/TiO2 for H2 production was tested over five cycles. After the fifth cycle, there were no obvious declines in evolution rates, suggesting excellent recycling stability of the as-fabricated nanocomposite for hydrogen evolution from water.

Stable hydrogen evolution from water under visible light irradiation using 0–5: TiO2, ZnS/TiO2, MoS2/TiO2, CdS-Mn/TiO2, ZnS/CdS-Mn/TiO2, and ZnS/CdS-Mn/MoS2/TiO2, respectively. TiO2: titanium dioxide; ZnS: zinc sulfide; MoS2: molybdenum disulfide; CdS-Mn: cadmium sulfide–manganese.
Possible photocatalytic reaction mechanism
On the basis of the above findings and related studies, the possible mechanism of the photocatalytic degradation and photocatalytic H2 evolution from water splitting with ZnS/CdS-Mn/MoS2/TiO2 nanocomposites is put forward and illustrated in Figure 1.
According to the literature, 25,43,44 the minimum values of the conduction band (CB) of TiO2, CdS, and MoS2 are −0.51, −0.52, and −0.05 eV, and the maximum values of the valence band (VB) are 2.70, 1.88, and 1.70 eV, respectively. The semiconductor CB position is more negative than the hydrogen electrode reaction potential (0 eV), and the location of VB is positive as compared to that of the potential oxygen electrode (1.23 eV) in the reaction, so that the photogenerated holes can effectively oxidize water. In ZnS/CdS-Mn/MoS2/TiO2 samples, when it was illuminated by visible light, the holes in TiO2 or CdS-Mn would shift into the VB of MoS2, and the electrons on the CdS may inject in either the Fermi level of Mn or transfer into the CB of MoS2. Since the bottom of the CB edge of TiO2 is higher than MoS2, electrons on the CB of TiO2 go into the CB of MoS2.
In the photocatalytic reaction process, ZnS/CdS-Mn/MoS2/TiO2 is excited by the light irradiation, resulting in the production of e−-h+ pairs. The electrons can be better combined with the O2 in the solution to generate the superoxide radical anion
The photocatalytic mechanism of water splitting coupling with organic pollutants degradation was proposed as follows 10





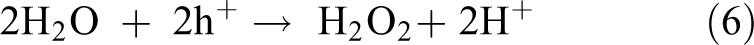






Conclusions
To summarize, the ternary photocatalyst (CdS-Mn/MoS2/TiO2) has been synthesized by a simple hydrothermal method coupled with SILAR. The ZnS/CdS-Mn/MoS2/TiO2 showed the best photocatalytic performance for MO dye and 9-AC degradation under sunlight irradiation with the removal rate of 98% (MO) in 100 min and 100% (9-AC) in 35 min. It is worth noting that the H2 production rate on it reached 408.27 mmol/cm2/h, which is significantly higher than that of TiO2, MoS2/TiO2, ZnS/TiO2, MoS2/TiO2, CdS-Mn/TiO2, and ZnS/CdS-Mn/TiO2. The ZnS/CdS-Mn/MoS2/TiO2 photocatalyst still showed good stability and photocatalytic activity in fifth cycling experiment. The enhanced photocatalytic activity is ascribed to the synergistic effect of an increased lifetime of photoexcited charge carriers in the Z-scheme nanocomposite. The possible separation and retention of the photoinduced e−-h+ pairs were efficiently improved and lead to higher oxidation or reduction capability. Our study can supply a novel sight for exploring newly low-cost materials as highly efficient cocatalysts in photocatalytic application for addressing the increasing energy shortage and environmental pollution.
Footnotes
Acknowledgments
The authors would like to thank the editor and reviewers for helpful comments and suggestions.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the National Natural Science Foundation of China (grant nos 21502051 and 21205039), the Natural Science Foundation of Hunan Province (2016JJ6101), Sponsored by Program for Science and Technology Innovation Talents of Hunan Province (2017TP1021; kc1704007), and Dr Start-up Foundation (grant no. 15BSQD14).