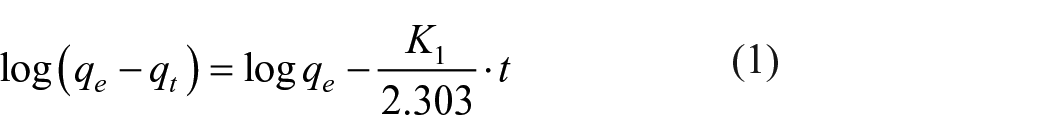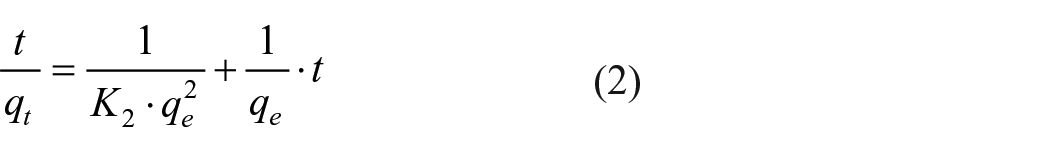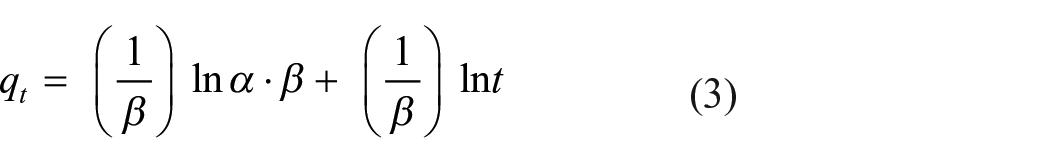Abstract
Among the different photocatalysts, TiO2 (Eg = 3.1 eV, zero charge point (pHpzc = 6.3), and surface = 55 m2/g) is currently the most efficient and the most studied semiconductor due to its strong photocatalytic activity, non-toxicity, and chemical stability. The elimination of DR-80 on TiO2 is studied by adsorption in batch mode and by application of heterogeneous photocatalysis onto TiO2 under UV irradiation. The effects of contact time (0–60 min), initial pH (3–11), dose of the adsorbent (0.5–3 g L−1), and DR-80 concentration (40–60 mg L−1) on the adsorption of DR-80 by TiO2 are studied for optimization of these parameters. The kinetic parameters, rate constants, and equilibrium adsorption capacities are calculated and discussed for each applied theoretical model. The adsorption of DR-80 is well described by the pseudo-first-order kinetic model. The fitting of the adsorption isotherms shows that the models of Langmuir and Temkin offering a better fit and an adsorption 64.102 mg/g at 25 °C of DR-80 are eliminated. The results showed that the photocatalytic efficiency strongly depends on the pH while the initial rate of photodegradation is proportional to the catalyst dose, and becomes almost constant above a threshold value. It was found that the photodegradation is favored at low DR-80 concentrations in accordance with the Langmuir–Hinshelwood model with the constants Kad = 6.5274 L/mg and KL–H = 0.17818 mg L−1 min. However, the adsorption is improved for high DR-80 concentrations. It is found that the degradation depends on both the temperature and the pH with a high elimination rate at high temperature. The photocatalyst TiO2 has a better activity for the degradation of DR-80, compared to some commercial catalysts that have been described in the literature.

Introduction
The 20th century is characterized by considerable technological progress, accompanied by an unprecedented demographic boom. These two factors have caused world water consumption to have increased from 400 to 7000 billion, of which 32% constitutes the needs of the industry. In addition to a considerable demand for water, progress and population growth are the sources of domestic discharges and industrial as well as agricultural pollution. This further exacerbate the problems of obtaining water for consumption by humans, by degradation of the quality of surface and ground waters and their impact on human health and the natural environment. The scientific community has become aware of the gravity of the situation resulting in strict regulations in terms of the development of processes that consume less water, with recycling and modernization of operating systems. Among numerous toxic chemicals, dyes, organics, and pharmaceuticals are very concerning. Industrial effluents released from the textiles, paint, paper, varnishes, plastics, pulp, cosmetics, and tanneries are among the main causes of water contamination because of their high coloration. The suggested treatments for the removal of colors and dyes from wastewaters are filtration, deposition, coagulation, membrane separation processes, oxidation, and adsorption. 1 In these methods, adsorption has proven to be effective and remains suitable for removing dyes. 2 It is a technique used for the water purification and involves in the accumulation of particles or molecular species of gas, liquid, or dissolved solids on the surface of the adsorbent. It occurs between two phases, such as gas–liquid, liquid–liquid, liquid–solid, or gas–solid interfaces. 3 The process is widely used for the water treatment due to the environmental requirements.4–7 In this respect, activated carbon is mostly employed as an adsorbent, but remains relatively expensive. However, photocatalysis has received significant attention as an effective and sustainable strategy for the removal of organics, dyes, and carcinogenic pollutants which are hazardous to human health and the environment. TiO2, In2O3, NiO, and ZnO are among the semiconductors that have attracted a great interest due to their physical and chemical stabilities, low toxicity, and high photodegradation ability. TiO2 has significant merit for solar-powered vitality exchanging and photodegradation of toxic substances. Its chemical inertness and non-toxicity have likewise made it a popular photocatalyst.8–12 It has a large band gap (Eg = ~3.2 eV), but 5% of the adsorbed solar radiation is efficiently converted into useful forms. Many techniques exist for the elimination of dyes including adsorption,13–17 photochemical and biological degradation, coagulation, chemical oxidation, reverse osmosis, and flotation. The anionic azo dyes with nitrogen–nitrogen double bonds are the largest and most versatile class of organic compounds. 18 However, their oxidation is difficult due to their aromatic and mesomeric structure and poor biodegradability. Direct Red 80 (DR-80) is a typical water-soluble anionic azo dye, commonly present in effluents from the textile, food, pharmaceutical, printing, and paper-manufacturing industries. Due to its toxicity and persistence, these discharges can cause a serious risk to the physicochemical properties of fresh water and aquatic life and suitable technologies are required for their elimination. Therefore, the main objective of the present work is to investigate the potential of TiO2 as a photocatalyst material for the photodegradation of DR-80 from aqueous solutions. The effects of the pH, initial DR-80 concentration, contact time, and TiO2 dose that influence the degradation process of DR-80 on TiO2 are optimized.
Results and discussion
Adsorption of DR-80 onto TiO2
Effect of the initial concentration and contact time
The adsorption capacity of DR-80 increases with time, reaching a maximum after 40 min of contact time and tends toward a constant value indicating that no further ions of DR-80 are removed from the solution. The equilibrium time is reached after 40 min, but for practical reasons, the adsorption experiments can last up to 50 min. We observed an increase of DR-80 adsorption from 38.5 mg/g for 40 mg L−1 of DR-80 to 57.80 mg/g for 60 mg L−1 of DR-80 (Figure 1). Therefore, we can deduce that the adsorption of DR-80 on TiO2 occurs in three steps:
Relatively rapid adsorption of DR-80 in solution due to the availability of free sites on the surface of the adsorbent particles, which reflects the linear increase in the adsorption capacity over time. This step lasts 15 min under the intended conditions of use.
Reduction in the adsorption rate resulting in a small increase in the adsorption capacity due to a decrease in the amount of DR-80 in solution and the number of available sites. This step lasts from 15 to 40 min.
Stabilization of the adsorption capacity is observed after 40 min; this is probably due to the almost total occupancy of the available adsorption sites.

Effect of the initial concentration (Co) and contact time of DR-80 adsorption onto TiO2.
The DR-80 ions are adsorbed initially on the external surface area of TiO2 which makes the adsorption easy and fast. When the external surface is saturated, the DR-80 ions enter the TiO2 pores and absorb on the internal surface of the particles; this phenomenon takes a relatively longer time. This may be attributed to an increase of the driving force due to the concentration gradient with increasing Co in order to overcome the mass transfer resistance of DR-80 ions between the aqueous and solid phases. Therefore, a higher initial DR-80 concentration (Co) increases the adsorption capacity; the same result was obtained by others.19-21
Effect of the TiO2 dose on the dye adsorption
Figure 2 shows the variation in the quantity of adsorbed pollutant DR-80 as well as its photocatalysis as a function of the dose of TiO2. When the TiO2 dose is increased from 0.5 to 3 g L−1, the adsorbed quantity decreases from 52.5 to 42 mg/g. Consequently, an increase of the catalyst dose hinders the adsorption process, due to electrostatic repulsions between the charges of the same signs for the pollutant DR-80 and TiO2. In the case of photocatalysis, there is a sudden drop in the adsorbed quantity from 48 to 32.5 mg/g for the same dose range.
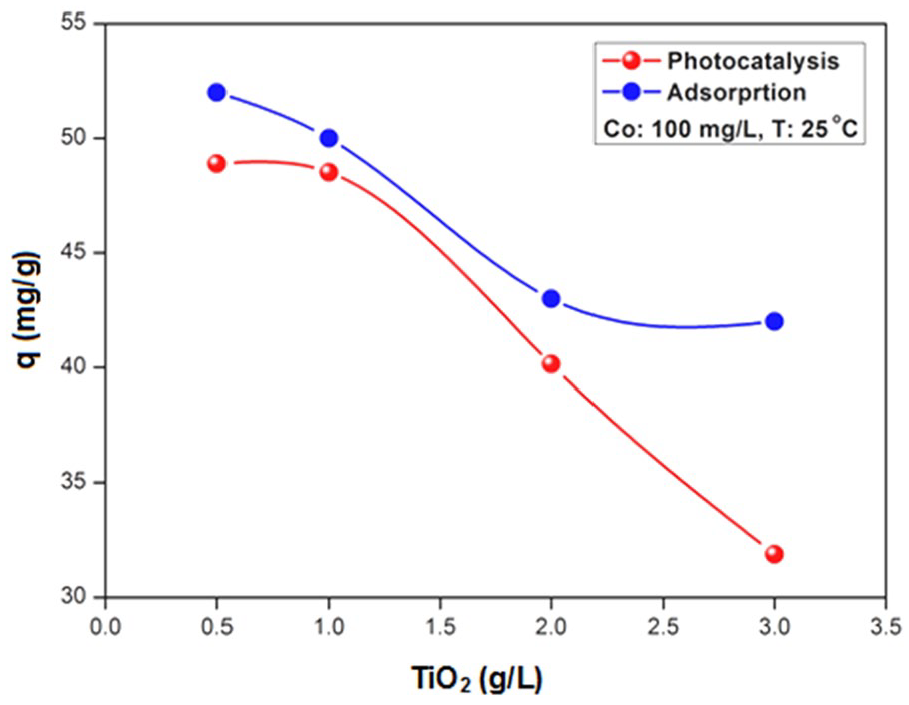
Effect of theTiO2 dose on the adsorption and photocatalysis of DR-80.
Furthermore, Figure 2 clearly reveals an optimal semiconductor dose of 1 g L−1, beyond which there is a decrease in the adsorption capacity. This observation can be explained by the following: raising the dose of the catalyst up to a critical value promotes the aggregation of particles, which results in a decrease in the number of active sites. Under our experimental conditions, the optimal value, beyond which there is the appearance of agglomeration of semiconductor particles occurs, is equal to 0.5 g L−1.
Effect of initial pH
The influence of pH on the adsorption has been studied in the range 3–12 by adjusting pH of the solutions to the desired value, by adding HNO3 or NaOH, while keeping constant the dye concentration, the temperature at 25 °C, and the stirring speed. The evolution of the equilibrium absorption rates (Figure 3) indicates that the pH has a significant influence on the DR-80 adsorption. Textile wastewater usually has a wide pH range. This parameter plays an important role in the characteristics of textile industry pollutant releases and the generation of hydroxyl radicals. In the presence of water, the TiO2 particles are covered by OH− species which take away the protons as illustrated in Figure 3, and effect on the adsorption, and in this way have an influence on the photocatalytic degradation.

Effect of the pH on the adsorption of DR-80.
The adsorption capacity on TiO2 increases significantly with decreasing pH and predominates at low pHs. Indeed, in acidic medium, electrostatic attractions exist between the TiO2 positively charged surface and the negatively charged anionic form of DR-80. In addition, the phenomenon of particle agglomeration decreases. Conversely, in basic medium, the adsorption capacity decreases because of electrostatic repulsion with TiO2 and DR-80, both negatively charged. For pHs higher than the pKa of the dye, we have the ionized form of DR-80, resulting in an electrostatic repulsion between the catalyst, represented by TiO− and the anionic form of the dye. In addition, the weak adsorption of dyes at basic pH is accentuated by the competitive adsorption of hydroxy ions which occupy the active sites; the same result has been obtained elsewhere.22,23
Adsorption kinetics
Kinetic studies are important since they describe the uptake rate of adsorbate, the residual time of the whole process, several models have been proposed for this purpose. In this work, the kinetic data of DR-80 adsorption are examined using pseudo-first-order, 24 pseudo-second-order, 25 Elovich, 26 and intra-particle diffusion 27 models expressed, respectively, by the equations
where qt (mg/g) is the amount of DR-80 adsorbed on TiO2 at the time t (min); K1 (min−1) and K2 (g/mg min) are the pseudo-first-order and pseudo-second-order kinetic constants, respectively. The slope and intercept of the plots ln(qe − qt) versus t and t/qe versus t were used to determine the constants K1 and K2, qe·α (mg/g min) is the initial adsorption rate, and β (mg/g) relates the degree of surface coverage and the activation energy involved in the chemisorption. Kin is the intra-particle diffusion rate constant (mg/g min1/2), qt is the amount of DR-80 adsorbed at time t, and C (mg/g) is the intercept. The plot of qt versus t1/2 enables the determination of both Kin and C. The constants of all the models deduced after modeling are given in Table 1.
Pseudo first-order, pseudo second-order, Elovich and intraparticles diffusion model constants and correlation coefficients for DR-80 adsorption onto TiO2.

Adsorption equilibrium isotherms
To assess the performance of the adsorbent, different isotherms exist, among which the Langmuir, 28 Freundlich, 29 Temkin, 30 and Elovich 31 are presented in Figure 4. Besides, the isotherm models are applied using the optimized of the parameters




where Ce is the equilibrium concentration (mg L−1), qmax is the monolayer adsorption capacity (mg/g), and KL is the constant related to the free adsorption energy (L/mg). The constant KF characterizes the adsorption capacity of the adsorbent (L/g) and n is an empirical constant related to the magnitude of the adsorption driving force. The plot lnqe versus lnCe permits the determination of the constant KF and n. Therefore, the plot of qe versus lnCe enables determination of the constants AT and BT. KE (L/mg) is the Elovich constant at equilibrium, qmax (mg/g) is the maximum adsorption capacity, qe (mg/g) is the adsorption capacity at equilibrium, and Ce (g L−1) is the concentration of the adsorbate at equilibrium. KE and qe are calculated from the plot ln(qe/Ce) versus qe. The constants of the different models deduced after modeling are given in Table 2.
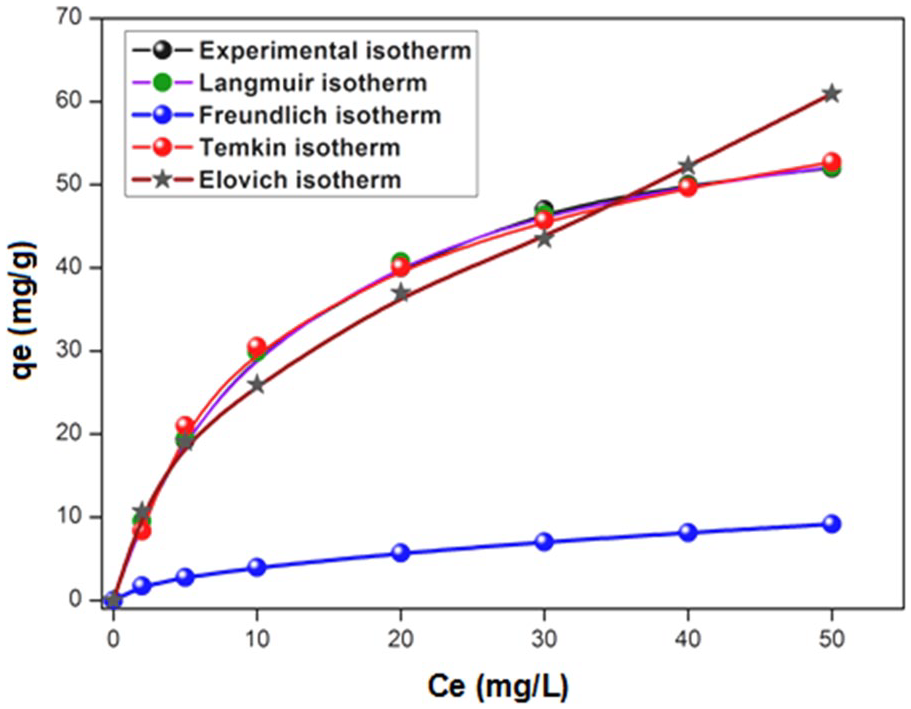
Adsorption isotherms of the different models under the optimum conditions.
Adsorption isotherm coefficients of different models.

R2: determination coefficient; ΔQ: Temkin energy.
Photodegradation of DR-80 in the presence TiO2/UV
As will be evident from the recent literature, there has been substantial and rapid progress made on water purification on semiconducting oxides. In this contribution, we report the photodegradation of DR-80 onto TiO2 under UV irradiation. The principle of the heterogeneous photocatalysis (Figure 5) is based on the activation of the semiconductor by energetic photons (hν ⩾ Eg), with Eg being the forbidden band. Electron (e−)/hole (h+) pairs are formed, which result from the passage of electrons from the valence band (VB) to the conduction band (CB). The electrons reduce oxygen adsorbed onto the surface of TiO2 into O2•− while the holes react with superficial OH− ions to form highly oxidizing hydroxyl radicals (HO•) that are responsible for the degradation of pollutants. The advanced oxidation process (AOP) is widely used in the mineralization of organic molecules and involves active radicals, for example, among which HO•, owing to its high potential (2.3 VSHE). When a photocatalyst is exposed to solar radiation, several active species are generated, namely: holes (h+), electrons (e−), hydroxyl radicals (HO•), and the superoxide radicals (O2•−). The photodegradation occurs by involving one or more active species; the overall mechanism on TiO2 is as follows:
1. On the TiO2 surface, creation of an e−/ h+ pair

2. Reduction reaction

3. Oxidation reaction

4. Thus, the radicals HO• react with the organic compounds following diffusion-controlled kinetic
32
The photocatalytic efficiency generally decreases with augmenting initial concentration Co. A high concentration Co leads to an increasing number of DR-80 molecules adsorbed on the surface, thus reducing the penetration of photons, and this decreases the conversion efficiency and the (e−/h+) pairs become insufficient.
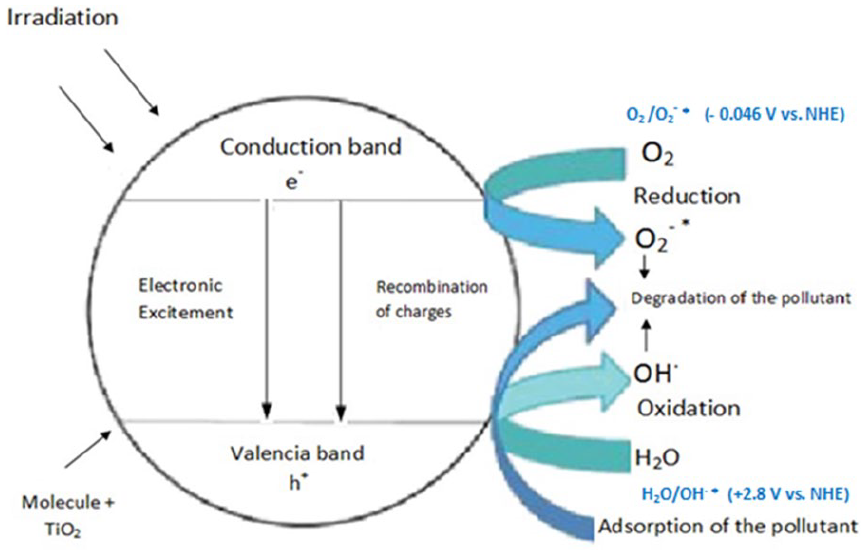
Principle of photocatalysis under irradiation.
Heterogeneous photodegradation of DR-80 by the TiO2/UV system
The photocatalytic degradation of DR-80 was carried out in a static reactor used also for direct photolysis under polychromatic external light. Suspensions containing TiO2 (1 g L−1) and DR-80 (concentration: 60 mg L−1), were first brought into contact for 40 min, to reach equilibrium and were then irradiated.
Effect of the TiO2 dose on the degradation kinetics
The effect of the TiO2 dose on the photodegradation was carried out for an initial DR-80 concentration of 100 mg L−1 and TiO2 dose between 0.5 and 3 g L−1. As expected, the rate is rapid on increasing of the photocatalyst dose up to a concentration of 1 g L−1; beyond which only a little improvement is observed (Figure 2). This threshold value is attributed to the almost total absorption of UV photons by TiO2 particles located in the area close to the light source. The adsorption rate progresses slightly with the TiO2 dose; this can be explained, on the one hand, by the decrease of the pH medium, which increases the band bending at the interface TiO2/electrolyte to the optimal value of 0.4 V. This modifies the surface state of the semi-conductor and alters the adsorption process. On the other hand, this thermodynamically favorable influence is compensated for the fact that increasing the TiO2 amount can promote the aggregation, thus reducing the number of active sites.
Influence of pH
The pH of the solution greatly affects the TiO2 surface charge and the size of the aggregates; the pH for which the surface charge of the oxide is zero (point of zero charge: pHpzc) is found to be 6.3. Before and after this pH, the surface of the oxide is charged positively and negatively 33

Under these conditions, the photocatalytic degradation of ionized organic compounds is significantly affected by the pH resulting in repulsive interactions between the ionized pollutant and the surface charge of the photocatalyst, thus reducing the probability of encountering the photocatalyst. The pH is a parameter that determines the surface properties of solids and the state of the pollutant as a function of its pKa and characterizes the water to be treated. In general, when a compound is partially ionized or is carrying charged functions, it is necessary to consider the electrostatic interactions that occur with TiO2. Indeed, according to the zero charge point (pHpzc), the surface charge of the solid depends on the pH. Thus, for TiO2, the surface is positively charged below pHpzc (6.3), and negatively charged above pHpzc.
The TiO2 surface is positively charged in acidic solution related to the fixation of protons and negatively in basic medium. The surface charge influences the dye adsorption and, therefore, can promote or limit the adsorption.
The influence of pH on the kinetics of degradation of DR-80 by photocatalysis was studied in the pH range (3–12) for a DR-80 concentration of 100 mg L−1 and TiO2 at 1 g L−1. The evolution of the levels (Figure 6) indicates that the pH plays a crucial role in the oxidation of DR-80. Its effect is directly linked to the electrical state of the surface of the catalyst and to the DR-80 adsorption. In general, when a compound is partially ionized or carries charged functions, it is necessary to consider the electrostatic interactions that occur with TiO2. In the pH range (3–6.5), DR-80 is soluble in cationic form, and TiO2 is the positively charged form which favors repulsions, consequently reducing the rate of degradation. However, in the pH range (6.5–12), the pollutant DR-80 is soluble in its anionic form while TiO2 is positively charged. The existence of these attraction forces leads to an increase in the kinetics of degradation of this substrate in this medium

Influence of the pH on degradation kinetics.
Influence of the initial concentration (Co)
Figure 7 illustrates the effect of the initial concentrations (Co) of DR-80 (40–100 mg L−1) in the presence of TiO2 at 1 g L−1 at free pH. The rate remains broadly unchanged from one concentration to another; we observe an exponential decay with kinetics of apparent order equal to 1 in all cases. As expected, the curves clearly show that the discoloration of the solution takes a longer time as long as the initial DR-80 concentration is high. As summarized in Table 3, this observation results in the continuous decrease of the rate constant (K) and by the continuous increase of the half-lifetime (t1/2). Previous studies showed that the pollutant degradation by heterogeneous photocatalysis follows the Langmuir–Hinshelwood (L–H) model, where the degradation rate is proportional to the fraction of the catalyst surface covered by the substrate molecules. The L–H model is given by the following equation

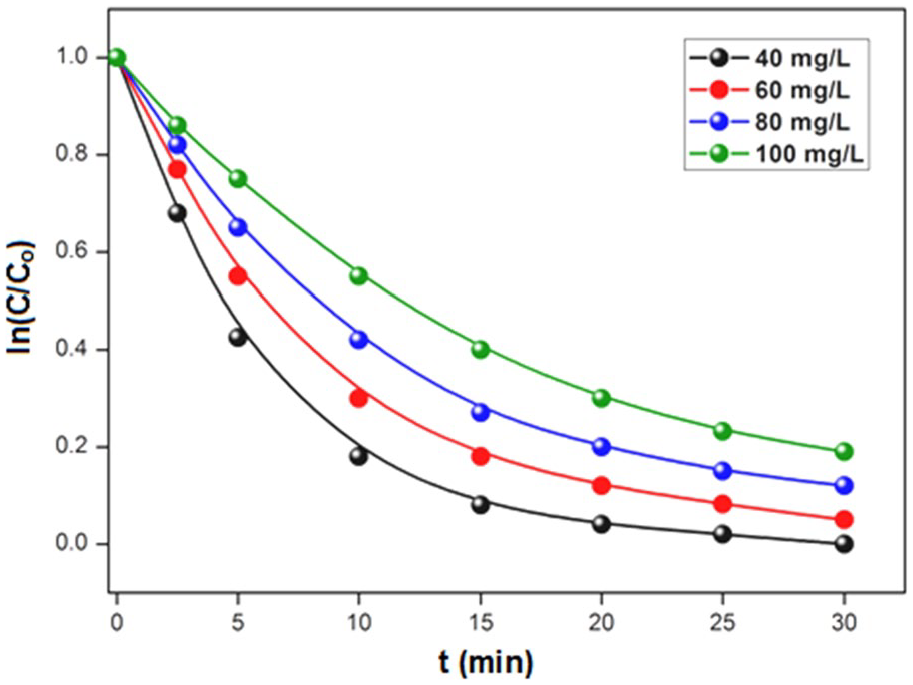
Influence of the initial concentration of DR-80 on the degradation kinetics.
Determination of the photodegradation coefficients.

For low concentrations (C < 10−3 M), the term K·C (where K is the constant and C is the concentration) can be ignored below 1. Therefore, the reaction follows a pseudo-first-order kinetic

Integration of this equation gives

The initial rate (ro) is given by

where V is the photodegradation rate (mg L−1 min), Vo is the initial rate of photodegradation (mg L−1 min), C is the pollutant concentration at time t (mg L−1), Kad is the constant of the adsorption equilibrium (mg L−1 min), KL–H is the L–H kinetic constant (L/mg), and Kapp (Kad·KL–H) is the apparent rate constant (min−1) which is dependent on Co. The plot of ln(Co/C) versus time (t) for different DR-80 concentrations (Co) and various catalyst amounts is given in Figure 8. The photocatalytic degradation follows perfectly pseudo-first-order kinetics for Co values of 10 and 15 mg L−1; the constants Kapp (Table 3) indicate that the rate of discoloration increases with raising Co, which corresponds to the L–H adsorption model. A linear expression can be occasionally obtained from the equation by plotting Kapp−1 against Co
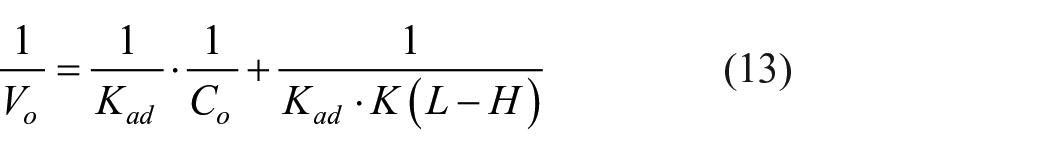

Application of the Langmuir–Hinshelwood model for the degradation kinetics.
The plot of 1/Vo against 1/Co gives linear relationship (Figure 9). From the slope (1/Kad) and the intercept (1/Kad·KL–H), the constants Kad and KL–H for the photocatalytic degradation of DR-80 are, respectively, 1.992 mg L−1 min and 0.2906 L/mg.

Determination of adsorption and photocatalytic constants.
Conclusion
The AOP involves the active species generated during water purification. These species like HO• induce highly oxidizing properties used for the destruction of organic wastes. Qualitative analysis performed immediately after the discharge also revealed the presence in solution of mineral ions derived from the degradation of the parent molecule.
The first part of this study has shown that TiO2 can successfully be employed as an effective adsorbent for the removal of DR-80 from aqueous solution. The Temkin and Langmuir isotherm models provided a better fit for the equilibrium adsorption data, with a maximum adsorption capacity of 64.102 mg/g at 25 °C being calculated.
The adsorption of DR-80 ions by TiO2 follows a pseudo-first-order kinetic model, which relies on the assumption that chemisorption is the rate-limiting step. In chemisorption, the DR-80 ions are attached to the adsorbent surface by forming a chemical bond and tend to find sites that maximize their coordination number with the surface. The adsorption kinetics of the DR-80 dye on TiO2 shows that the equilibrium time is reached after 40 min, in good correlation with the pseudo-first-order kinetic model.
The value of qmax is in good agreement with that of recent previous work, suggesting that DR-80 can be easily adsorbed on TiO2. The elimination of DR-80 from an aqueous solution, an organic compound widely used in the textile industry, was also used in the photocatalytic degradation.
The slight degradation of DR-80 by direct photolysis (at free pH) was observed. The photocatalytic degradation was affected by the initial DR-80 concentration according to the L–H kinetic model. The parameters influencing the degradation kinetics revealed that the pH and catalyst dose gave the best result under optimal conditions (pH: 5, TiO2 dose: 1 g L−1, low concentration, and high light flux). Photocatalysis as used for water purification has mainly developed around TiO2 thanks to the considerable advantages that it presents, including the stability, low-cost, non-toxicity, and high photocatalytic performance under UV light.
Materials and methods
UV-Vis absorption spectrum
The spectrum of DR-80 shows the existence of two bands in the UV region (221 and 280 nm), resulting from the transition π → π*, due respectively to the presence of benzene and naphthalene. The bands at 372 and 531 nm relating to the transition n → π* are due to the presence of chromophore groups (N=N). 34 An acidic or basic pH does not affect the absorption spectrum of this dye.
Preparation of the adsorbate
The Solophenyl Red 3BL (DR-80; molecular weight C45H26N10Na6O21S6 and 1373.05 g/mol) was purchased from Merck Company (India); the chemical and physical proprieties of DR-80 are reported in Table 4. A stock solution was prepared by dissolving 1 g of solid DR-80 in 1 L of double distilled water. The working initial solutions (40–100 mg L−1) were prepared daily by dilution to give the desired concentrations. All other chemicals used were of analytical grade.
General characteristics of DR-80.

Characteristics of titanium oxide
TiO2 is currently one of the most efficient semiconductors because of its high photoactivity, non-toxicity, and stability. It is used in the manufacture of paints (55%–60%), plastics (15%–20%), and paper (15%). It is also employed in the pigmentation of printing inks, rubber, textiles, synthetic fibers, ceramics, white cements, and cosmetics. 35 The advantages of applying TiO2 as a catalyst are as follows: its biological and chemical inertness, low cost, reusability, and non-resistance to mass transfer. It can be used under standard conditions (atmospheric pressure, oxygen being used as an oxidant) and can lead to the mineralization of organic carbon into CO2 under UV light. The most attractive form is P25 Degussa (80% anatase and 20% rutile); its properties are grouped in Table 5. The intimate contact between these two phases can explain its significant photocatalytic activity.
Characteristics of TiO2 (P25 Degussa).

Batch adsorption studies
The effects of the influencing parameters such as the initial DR-80 concentration (Co: 40–60 mg L−1) and pH (3–12) on the adsorptive removal of DR-80 were studied in a batch mode for a specific contact time (0–60 min); the pH was adjusted with HCl or NaOH (0.1 mol L−1). For the kinetic studies, the desired quantity of TiO2 was contacted with 30 mL of DR-80 solution in the Erlenmeyer flask. Then, the flask was placed on a rotary shaker at 250 r/min and samples were taken at regular time intervals and vigorously centrifuged (3000 r/min for 10 min) to separate the solid particles. The remaining DR-80 concentration in the supernatant was titrated using a Perkin Elmer UV-Vis spectrophotometer (Model 550S at the maximum wavelength λmax = 531 nm). The amount of DR-80 ions adsorbed by activated carbon qt (mg/g) was calculated using the following equation

where Co is the initial DR-80 concentration, Ct is the DR-80 concentrations (mg L−1) at time (t), V is the volume of solution (L), and m is the mass of the TiO2 (g).
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.